Abstract
Stomach cancer is one of the most common cancers worldwide, despite its declining overall incidence. Although there are differences in incidence, etiology and pathological factors, most studies do not separately analyze cardia and noncardia gastric cancer. Surgery is the only potentially curative treatment for advanced, resectable gastric cancer, but locoregional relapse rate is high with a consequently poor prognosis. To improve survival, several preoperative and postoperative treatment strategies have been investigated. Whereas perioperative chemotherapy and postoperative chemoradiation (CRT) are considered standard therapy in the Western world, in Asia postoperative monochemotherapy with S-1 is often used. Several other therapeutic options, although generally not accepted as standard treatment, are postoperative combination chemotherapy, hyperthermic intraperitoneal chemotherapy and preoperative radiotherapy and CRT. Postoperative combination chemotherapy does show a statistically significant but clinically equivocal survival advantage in several meta-analyses. Hyperthermic intraperitoneal chemotherapy is mainly performed in Asia and is associated with a higher postoperative complication rate. Based on the currently available data, the use of postoperative radiotherapy alone and the use of intraoperative radiotherapy should not be advised in the treatment of resectable gastric cancer. Western randomized trials on gastric cancer are often hampered by slow or incomplete accrual. Reduction of toxicity for preoperative and especially postoperative treatment is essential for the ongoing improvement of gastric cancer care.
Introduction
Epidemiology
Gastric cancer is a major problem worldwide: it is the second leading cause of cancer death, affecting approximately 1 million new individuals per year [Kamangar et al. 2006]. Whereas the incidence in males is twice as high as in females, there is also a marked geographic variation. Highest incidence rates occur in North-East Asia (up to 70 per 100,000), Eastern Europe and much of the eastern part of South America, while lowest incidence rates are seen in North America (8 per 100,000), Africa and South and West Asia [Yamaoka et al. 2008]. Stomach cancers can anatomically be classified as noncardia (fundus, corpus and antrum) and cardia cancers, with noncardia cancers constituting the majority of all gastric cancers worldwide. Whereas the incidence of noncardia gastric cancer has declined over the past decades [Kelley and Duggan, 2003; Howson et al. 1986], there has been a rapid increase in the incidence of cardia gastric cancer until the early 1990s, which has not persisted in the current century [Steevens et al. 2010a; Wu et al. 2009; Pohl and Welch, 2005].
Carcinogenesis
Two distinct histologic types of gastric cancer have been defined by Lauren: an intestinal type, which is characterized by irregular tubular structures in areas of mucosal inflammation; and a diffuse type, which can be characterized by discohesive cells and pools of mucus [Lauren, 1965]. Gastric carcinogenesis of the intestinal type is thought to be a multifactorial process involving irritation of the mucosa by environmental factors, acid secretion and bacterial nitrite and N-nitroso compounds production from dietary nitrates. The intestinal type gastric cancer is mostly found in the distal stomach and typically arises through the Correa’s cascade, progressing from the successive steps of normal gastric epithelium infected by
Etiology
The childhood environment is an important factor in the risk of developing gastric cancer [Coggon et al. 1990; Kolonel et al. 1981].
Environmental risk factors for noncardia gastric cancer include
Of all cancers of the stomach, about 10% arise in individuals with a family history of gastric cancer [La Vecchia et al. 1992]. HDGC develops in subjects with a germline mutation in one allele of the E-cadherin gene (CDH1) [Guilford et al. 1999]. During a recent consensus meeting of the International Gastric Cancer Linkage Consortium, updated results on carriers of 58 families with a CDH1 mutation showed a more than 80% lifetime risk of developing diffuse gastric cancer [Fitzgerald et al. 2010]. Familial preponderance has been described in other familial cancer syndromes, such as Lynch syndrome [Gylling et al. 2007], Li–Fraumeni syndrome [Varley et al. 1995], and Peutz–Jeghers syndrome [Giardiello et al. 2000; Boardman et al. 1998] as well. In these families the intestinal type of gastric cancer prevails.
Staging
In the Western world, staging is performed according to the American Joint Committee on Cancer (AJCC) and the International Union Against Cancer (UICC) [UICC, 2009]. The Japanese Gastric Cancer Association has its own staging system of gastric carcinoma [Japanese Gastric Cancer Association, 1998]. Until recently, the Japanese staging of nodal status (N) was based on location of the positive nodes. Nowadays both Japanese and Western systems are based on the number of positive lymph nodes, which seems to be more reproducible, provided that a minimum number of 15 lymph nodes are removed and analyzed [Karpeh et al. 2000].
Tumors of the gastroesophageal junction (GEJ) are often misclassified as either gastric when they should be esophageal, or
Stage grouping for gastric cancer according to the 6th (2002) and 7th (2010) edition of the AJCC staging system [Edge et al. 2010; Greene et al. 2002].

T, tumor classification; N, nodal status; M, metastases status.
Survival
As more than half of the patients in the Western world present with stage III or IV gastric cancer, overall prognosis is poor [Hundahl et al. 2000]. A recent survey shows that 5-year survival in all gastric cancer patients in Europe is only 24.1% [Sant et al. 2009]. Survival for all patients in the US is comparable: in the period 1999–2005, survival was 26.5%. For patients with metastatic disease at initial presentation, 5-year survival is <5% [Horner et al. 2009]. In patients treated with surgery in the US in the period 1985–1996, stage-specific (AJCC 6th edition) 5-year survival was 58% for stage IB, 34% for stage II, 20% for stage IIIA and 8% for stage IIIB [Hundahl et al. 2000]. In contrast, Japan has 5-year survival rates of approximately 60% [Kamangar et al. 2006]. This difference has been addressed to mass screening programs using photofluorography [Hamashima et al. 2008], differences in tumor biology and location with more intestinal subtypes and distal locations, and stage migration due to higher lymph node yield in Japanese series [Bunt et al. 1995]. In a comparative analysis between a US and a Korean center, multivariate analysis applying different patient and tumor characteristics and the number of resected lymph nodes, shows a higher disease-specific survival for Korean patients as compared with US patients (hazard ratio [HR] 1.3), suggesting the possibility of an intrinsic biologic difference between gastric cancer in the US and Korea [Strong et al. 2010].
Recurrence patterns
With increasing cancer stage, the risk of locoregional relapse increases, thus diminishing survival. In a combined analysis of several autopsies series, eventually 80–93% of all patients developed locoregional relapse [Gunderson, 2002]. A retrospective study on 367 patients with clinically complete recurrence data in a single center revealed that 54% of recurrences were locoregional, whereas distant sites were involved in 51%. Of all recurrences, 79% developed within the first 2 years [D’Angelica et al. 2004]. In a single-center study performed during 1949–1971, reoperations as second-look procedures in 107 previously resected gastric cancer patients, both symptomatic and asymptomatic, revealed locoregional failure in 23% as the only site of relapse [Gunderson and Sosin, 1982]. Data from a US randomized trial showed the highest relapse in locoregional sites, even after postoperative chemoradiation had been administered [Macdonald et al. 2001].
Surgical treatment
Resection is a prerequisite for the curative treatment of localized gastric cancer. It can be divided into three major approaches: endoscopic (sub) mucosal resection (EMR) or dissection (ESD), minimally invasive surgery, and open gastrectomy. Endoscopic mucosal resection is only used for the treatment of early gastric cancer (EGC), which is defined as a tumor of the stomach limited to the mucosa or submucosa regardless of lymph node metastases [Kitaoka et al. 1984]. This topic is not further covered in this review.
Laparoscopic surgery
Minimal invasive surgery for the treatment of gastric cancer is mainly performed in Korea and Japan, with the majority of patients treated for early and distal gastric cancer. However, with increasing laparoscopic experience and improvement in instrumentation, more extensive procedures and treatment of more advanced gastric cancers is becoming more common. Although laparoscopic gastrectomy has been performed since 1991, only four, mostly single-center, randomized controlled trials comparing the technique with open gastrectomy have been reported [Kim et al. 2010; Hayashi et al. 2005; Huscher et al. 2005; Lee and Han, 2005; Kitano et al. 2002]. Laparoscopic gastrectomy has been discussed in two reviews which indicate oncologic equivalency and safety based on the current small patient numbers [Shehzad et al. 2007; Shiraishi et al. 2006]. Large multicenter randomized controlled trials are necessary to establish the role of laparoscopy in the treatment of gastric cancer.
Extent of gastric resection and margins
Total gastrectomy is the indicated treatment for tumors located in the proximal or middle third of the stomach [Maruyama et al. 1996]. As compared with a total gastrectomy, a proximal gastrectomy for proximal gastric cancer is associated with a markedly higher rate of complications such as anastomotic stenosis and weight loss [An et al. 2008]. For distal gastric cancer, a distal gastrectomy is the recommended therapy provided that an adequate margin can be obtained. Two randomized trials investigated the impact of total
Microscopically positive resection margins (R1) are associated with a significantly worse prognosis as compared with a microscopically radical (R0) resection, especially in patients with early stage disease [Cho et al. 2007; Kim et al. 1999]. An Italian study investigated the minimal margin that should be obtained to ensure radical surgery in T3–4 tumors, and suggested a minimum margin of 6 cm [Bozzetti, 2001]. Data from the Netherlands show that survival in patients with an R1 resection is comparable with patients with positive cytology after abdominal washing [Songun et al. 1996], indicating that intra-operative frozen-section examination is mandatory for potentially curative resections of gastric cancer.
Lymph node dissection
As the primary tumor penetrates more deeply through the wall of the stomach, the risk of lymph node metastases increases. The Japanese Classification of Gastric Carcinoma [Japanese Gastric Cancer Association, 1998] defined 16 different lymph node stations surrounding the stomach (Figure 1), which are divided into three groups, each group further away from the primary tumor site. In a D1 dissection, the stomach (total or distal) plus the perigastric lymph nodes are removed. For a D2 dissection, additional removal of the nodes along the left gastric, the common hepatic, the splenic and the left hepatoduodenal artery is performed as well as some stations that are different for proximal, middle and distal tumors. With a D3 dissection, an even more extended lymphadenectomy is performed, including paraaortic and posterior hepatoduodenal nodes. For adequate staging a minimum of 15 lymph nodes should be evaluated [Karpeh et al. 2000].

Lymph node stations as defined by the Japanese Research Society for Gastric Cancer [Japanese Gastric Cancer Association, 1998], with nodal stations defined for each type of lymph node dissection. Originally published by the American Society of Clinical Oncology [Dikken et al. 2010].
Three prospective randomized trials have been performed that compared D1 with D2 lymph node dissection [Hartgrink et al. 2004a; Cuschieri et al. 1999; Dent et al. 1988]. In an early trial, 43 patients were randomized between a D1 or D2 dissection, and with a median follow up of 3.1 years no differences in survival were detected [Dent et al. 1988]. A British trial that randomized 400 patients for D1 or D2 dissection, showed equal 5-year survival rates (35%
In Japan, a D2 lymph node dissection is seen as standard treatment for curative resections [Nakajima, 2002]. Convinced of the benefits of extended lymph node dissection, Japanese surgeons consider it generally unethical towards patients to run a randomized trial including an arm with a D1 lymph node dissection. A Japanese trial randomizing 523 patients for D2 alone or D2 combined with paraaortic node dissection showed no significant difference in 5-year survival while there was a trend towards more surgery-related complications in the paraaortic group (28%
In conclusion, in Western countries there has been an extensive debate on the role of a D2 lymph node dissection, which can now be considered a recommended type of surgery for advanced gastric cancer, with removal of at least 15 lymph nodes for adequate staging. In Asian countries at least a D2 dissection is performed.
Accepted adjuvant and neoadjuvant therapies
Because adequate locoregional or systemic control is difficult to obtain with resection alone, surgery can be combined with adjuvant or neoadjuvant treatment. A distinction between accepted and nonstandard adjuvant and neoadjuvant therapies is provided in Table 2. Randomized studies on adjuvant and neoadjuvant treatment of gastric cancer are summarized in Table 3.
Currently available treatment strategies for advanced, resectable gastric cancer.

Randomized studies on preoperative and postoperative therapy in gastric cancer.

Specification of cardia cancer: 1true cardia, 2stomach divided into two parts (upper/lower), 3stomach divided into three parts (upper/middle/lower).
3y: 3-year survival, 5y: 5-year survival.
GC: gastric cancer, GEJ: cancer of the gastroesophageal junction, ALE: adenocarcinoma of the lower esophagus, EBRT: external beam radiotherapy, SURG: surgery, IORT: intraoperative radiotherapy.
Dox: doxorubicin, F: doxyfluridine, L: leucovorin, MMC: mitomycin C, MTX: methotrexate, C: cisplatin, Doc: docetaxel, 5FU: 5-fluorouracil; E: epirubicin wk: weeks, y: years, m: months, NS: nonsignificant.
Postoperative chemotherapy
Adjuvant chemotherapy may eliminate occult residual locoregional or metastatic disease after surgery. More than 30 randomized trials have been performed evaluating adjuvant chemotherapy in gastric cancer over the past two decades. Although the earlier trials were small, during the last decade trials with up to 400 patients have been performed in Southern Europe. Most find a small survival benefit, which is mostly nonsignificant [Cascinu et al. 2007; De Vita et al. 2007; Nitti et al. 2006; Bouche et al. 2005; Bajetta et al. 2002]. Different treatment regimens were tested, including 5-fluorouracil-based chemotherapy with or without anthracyclines, with or without mitomycin C, and platinum with etoposide. Most of these studies are included in several meta-analyses [Sun et al. 2009; Hu et al. 2002; Janunger et al. 2002; Panzini et al. 2002; Mari et al. 2000; Hermans et al. 1993; Earle and Maroun, 1999], which all except for one [Hermans et al. 1993] show a small, significant increase in survival for adjuvant chemotherapy of 3–5% (Table 4). However, the benefit of this increase in daily clinical practice is modest.
Meta-analyses on adjuvant chemotherapy.
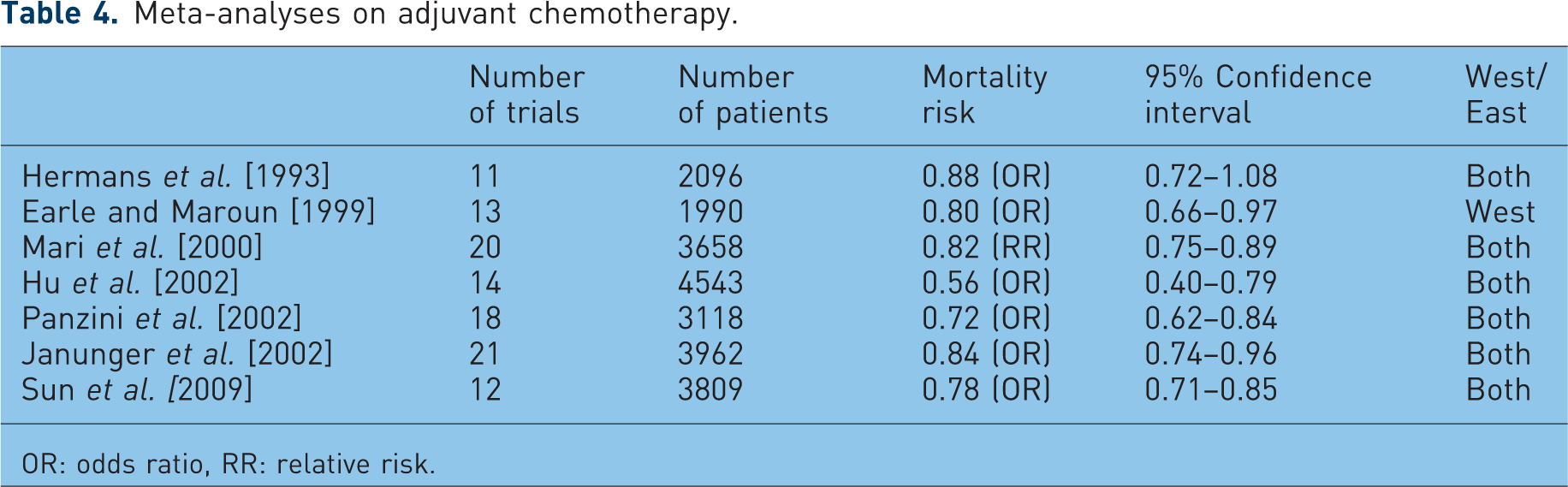
OR: odds ratio, RR: relative risk.
Sakuramoto and colleagues were the first to show a significant benefit in overall survival for postoperative chemotherapy in a large, adequately powered trial performed in an Asian patient population. In this study 1059 patients with stage II/III gastric cancer were randomized following at least D2 and R0 resection between surgery alone or surgery plus S-1 (oral fluoropyrimidine) for 12 months. Compliance after 12 months of chemotherapy was 66%. After 3 years, overall survival (80%
Overall, many early trials showed no or little advantage of postoperative chemotherapy. However, meta-analyses indicate a statistically significant but clinically equivocal survival benefit for adjuvant chemotherapy. Whereas Western trials focus on multidrug regimens, in Japan S-1 is considered to be of superior value. Compliance for postoperative chemotherapy remains a problem: in most Western studies 4–6 months of combination chemotherapy gives compliance rates from 87% to 43%, with hematological and gastrointestinal toxicities as the main reasons for not completing the treatment schedule. None of the randomized trials distinguished between cardia or noncardia cancer.
Postoperative chemoradiotherapy
Radiosensitizing drugs, such as 5-fluorouracil, have been added to radiotherapy with the intent to enhance the cytotoxic effect of radiotherapy on locoregional occult residual disease and to reduce locoregional relapse. Four early randomized trials showed the benefit of 5-fluorouracil-based chemoradiotherapy (CRT) over surgery alone [GITSG, 1990, 1982; Klaassen et al. 1985; Moertel et al. 1984], while another early study was negative[Dent et al. 1979]. However, patient numbers in these studies were small (
The key trial supporting the role of adjuvant CRT was the US Intergroup 0116 trial [Macdonald et al. 2001], in which 556 patients with stage Ib to IV gastric cancer who had received an R0 resection were randomized to no further treatment or postoperative CRT. Adjuvant treatment consisted of one cycle of 5-fluorouracil, leucovorin and 45 Gy of radiation with 7 days of 5-fluorouracil administered in 5 weeks, followed by two more cycles of 5-fluorouracil plus leucovorin. Treatment compliance in the CRT group was 64%; 17% stopped treatment because of mostly hematologic and gastrointestinal side effects. Major reasons for premature discontinuation in the other patients were early disease progression or patient’s request. Overall survival at 5 years was significantly higher in the CRT group (40%
At the same time, an observational study from South Korea compared 446 patients who underwent D2 gastrectomy with 544 patients that underwent D2 gastrectomy followed by CRT per the Intergroup 0116 protocol [Kim et al. 2005]. After a median follow-up of 66 months, there was a significant benefit in survival in the CRT group (57%
In a meta-analysis of postoperative CRT, 5-year overall survival is significantly higher with CRT as compared to surgery alone (odds ratio [OR] 0.45, 95% confidence interval [CI] 0.32–0.64). Despite a higher frequency of severe and life-threatening toxicities in the CRT group, overall compliance for the CRT was 73%. The majority of patients in this analysis are nonetheless derived from the Intergroup trial [Fiorica et al. 2007].
Several phase I/II studies on CRT with new types of chemotherapy have been performed to improve the interaction between chemotherapy and radiotherapy. A study from Germany in which patients were treated with 45 Gy of radiotherapy plus folinic acid, 5-fluorouracil, paclitaxel and cisplatin, showed that this four-drug regimen had an acceptable toxicity profile [Kollmannsberger et al. 2005]. Three studies from the Netherlands demonstrated the feasibility of radiotherapy combined with daily capecitabine and cisplatin [Jansen et al. 2009, 2007a, 2007b]. Radiotherapy fields contained the gastric bed and the anastomosis, with lymph node regions depending on the location of the primary tumor A side study on renal toxicity in 44 patients from these studies showed that there is a progressive relative functional impairment of the left kidney after postoperative CRT for gastric cancer, emphasizing that radiotherapy doses to the kidney should be minimized by using newer techniques such as intensity modulated radiotherapy (IMRT) in order to reduce toxicity while gaining the full benefit of survival of postoperative CRT [Jansen et al. 2007c].
In conclusion, postoperative CRT shows an advantage in survival over surgery alone, but the question remains as to whether this effect persists after an extended lymphadenectomy and radical resection. New treatment regimens on CRT opting for equal or better efficacy and reduced toxicity are currently under investigation.
Perioperative chemotherapy
The most important limitation of postoperative therapy is the impaired patient performance status after a gastrectomy that can hamper or even prevent delivery of the planned adjuvant treatment [Bozzetti et al. 2007]. Part of this is caused by the nutritional status and insufficient nutritional support that is given in this patient group prone to major weight loss [Bozzetti et al. 2007, 2001]. For this reason, the concept of neoadjuvant treatment might be a valuable alternative, while the postoperative therapy still can be administered when tolerated. The main goal of giving neoadjuvant chemotherapy is to treat micrometastatic disease at an early stage and to improve resectability by tumor downsizing and downstaging [Cunningham et al. 2006].
In the beginning of the 1990s the concept of perioperative chemotherapy was tested for its feasibility in a small study, showing a compliance rate of 72% and an acceptable toxicity profile [Ajani et al. 1991]. The MRC Adjuvant Gastric Infusional Chemotherapy (MAGIC) trial, randomized 503 patients with advanced (more than submucosal), resectable adenocarcinoma of the stomach, esophagogastric junction, or lower esophagus for surgery and perioperative chemotherapy
A French prospective trial randomized 224 patients with adenocarcinoma of the stomach (25%), the GEJ (64%) or lower esophagus (11%) between chemotherapy plus surgery (
Only a few studies have been performed on preoperative chemotherapy without postoperative treatment. In a randomized trial from the Netherlands, 59 patients were treated with surgery alone (
Based on these underpowered studies, it is difficult to draw conclusions about the role of preoperative chemotherapy without postoperative therapy.
The choice between established treatment paradigms
Whereas adjuvant chemotherapy with S-1 is an established regimen in Japan, the Western debate currently focuses on the use of postoperative chemoradiation
To compare preoperative with postoperative chemotherapy, a Swiss/Italian study randomized 70 patients for docetaxel, cisplatin and 5-fluorouracil either before or after surgery. This trial closed early because of poor accrual. In the neoadjuvant group, 75% completed the whole treatment schedule, as compared with 34% in the postoperative group (66% started with postoperative chemotherapy). Neoadjuvant chemotherapy could be delivered with a higher dose intensity without decreasing the chances for radical surgery or an increase in perioperative mortality [Biffi et al. 2010].
Based on these results, preoperative chemotherapy should be considered standard treatment in patients with advanced (more than submucosal), resectable gastric cancer. With a significantly higher compliance rate as compared with postoperative therapy, it not only reduces tumor burden, but also increases the chance for an R0 resection. When tolerated, adjuvant therapy should also be administered, but no standard regimen for this has been established. Patients with (distant) micrometastases will benefit more from systemic chemotherapy, but so far there is no adequate diagnostic modality or molecular marker to identify distant micrometastases. A different approach on predicting the efficacy of postoperative chemotherapy is grading histological response in the resection specimen after preoperative chemotherapy. Such a response, however, has not proven to be associated with survival in a US study [Mansour et al. 2007]. Patients at high risk for a local recurrence, for example patients who undergo an R1 resection, may benefit most from postoperative chemoradiation [Dikken et al. 2010], although this has not been addressed in a prospective study yet.
Questions on the use of postoperative chemotherapy or chemoradiation, after preoperative chemotherapy and surgery, are prospectively addressed in the Dutch CRITICS trial (see http://www.critics.nl), in which patients receive three cycles of preoperative ECC (epirubicin, cisplatin, and capecitabine), followed by D1+ surgery (D2 dissection without a splenectomy or pancreatectomy). Postoperative therapy consists of another three cycles of ECC, or CRT with capecitabine and cisplatin without epirubicin.
Nonstandard adjuvant and neoadjuvant therapies
Intraperitoneal chemotherapy
With a curative resection for gastric cancer, positive peritoneal washings occur in 7% of the patients [Ribeiro et al. 2006], whereas more than 50% will develop a peritoneal carcinomatosis at some point during follow up. Risk factors for positive cytology include serosal invasion and lymph node metastases [Bonenkamp et al. 1996]. The concept of intraoperative intraperitoneal chemotherapy (IPC) has been tested in several trials on gastric cancer. IPC can be combined with hyperthermia (HIPC) and can also be administered directly after surgery (early postoperative intraperitoneal chemotherapy [EPIC]).
Most trials on IPC are included in a meta-analysis, which reports on studies where patients received normothermic IPC, HIPC, or EPIC with or without postoperative systemic chemotherapy. Patient numbers of the 10 included, and mostly Asian, studies varied from 67 to 268. This meta-analysis showed a significant improvement in survival with HIPC alone (HR = 0.60, 95% CI 0.43–0.83) and HIPC combined with EPIC (HR = 0.45, 95% CI 0.29–0.68). There was also a trend towards improved survival with IPC, but this was not significant in combination with either EPIC alone or delayed (after recovery from surgery) postoperative intraperitoneal chemotherapy. Intraperitoneal chemotherapy was associated with higher risks of neutropenia and intra-abdominal abscess [Yan et al. 2007].
A more recent large Korean study, that was reported in abstract form only and was not included in the meta-analysis, randomized 640 patients with serosa-positive, but M0 resectable gastric cancer to adjuvant systemic mitomycin C and doxifluridine with or without IPC with cisplatin. With a median follow up of 3.5 years, overall survival was significantly higher in the IPC group (71%
Summarizing, HIPC in Asian trials is associated with a significant benefit in survival, at the cost of an increased postoperative complication rate. Therefore, this treatment modality is used with restraint in Western countries, and is considered an investigational strategy, not intended for standard daily practice.
Postoperative/intraoperative radiotherapy
Several studies investigated the effect of postoperative and intraoperative radiotherapy. A British randomized study with 436 patients found no difference in 5-year survival between surgery alone, surgery plus radiotherapy (45–50 Gy) or surgery plus chemotherapy (mitomycin C, doxorubicin, and 5-fluorouracil) postoperatively. Compliance for the protocol-defined dose in the radiotherapy group was 66%, with poor patient condition and withdrawal of consent as the most important reasons for failure [Hallissey et al. 1994]. A meta-analysis reporting on preoperative and postoperative radiotherapy also revealed no significant difference for postoperative radiation [Valentini et al. 2009].
Intraoperative radiotherapy (IORT) has been tested in several relatively small trials. In an American randomized trial, 41 patients were treated with surgery (control arm: early stages) and postoperative radiotherapy (control arm: advanced stages), or with surgery and IORT (experimental arm: all stages). Locoregional recurrence rates were lower for the IORT group (44%
Based on these underpowered studies, adjuvant radiotherapy as single modality following surgery has no role in routine daily clinical practice. IORT might be further investigated in patients with unfavorable tumor characteristics.
Preoperative radiotherapy
In a Chinese prospective randomized trial, 370 patients with cardia gastric cancer were randomized for surgery alone or preoperative radiotherapy (20 × 2 Gy in 4 weeks) followed by surgery after 2–4 weeks. The 5-year survival rates were 30% for the RT group as compared with 20% for the surgery alone group with a higher R0 resection rate in the RT group and no statistical difference in postoperative mortality and morbidity. Increased pathologic response rate to radiotherapy correlated with increased survival [Zhang et al. 1998].
A Russian study randomized 102 patients with resectable gastric cancer to radiotherapy (5 × 4 Gy in 1 week) plus surgery within 5 days or surgery only. Tolerance of the radiotherapy scheme was acceptable. The difference in 5-year overall survival between the two groups (39%
A meta-analysis based on the abovementioned three trials showed an advantage of neoadjuvant radiotherapy over surgery alone in 3- and 5-year survival (OR 0.57 and 0.62) [Fiorica et al. 2007]. Another meta-analysis on preoperative, intraoperative, and postoperative radiotherapy showed a significant increase in 3- and 5-year survival as well (RR 1.26) with most survival benefit using the preoperative approach [Valentini et al. 2009].
In summary, data on neoadjuvant radiotherapy are still limited, but suggest an advantage in survival over surgery alone. The largest trial has been performed in patients from a high incidence area with exclusively cardia cancer.
Preoperative chemoradiotherapy
Currently, most accruing randomized trials focus on perioperative chemotherapy and postoperative chemo(radio)therapy. However, several phase I/II studies have combined the administration of neoadjuvant chemotherapy with neoadjuvant radiotherapy [Ajani et al, 2006b, 2005, 2004; Allal et al. 2005]. Although results are promising with different chemotherapy schedules all containing 5-fluorouracil and cisplatin, multicenter phase III trials are necessary in order to evaluate whether this treatment strategy can improve survival.
Conclusion and future perspectives
Surgery remains the primary curative treatment for locally advanced gastric cancer. A D2 dissection is the recommended type of surgery in Western countries, while in the East at least a D2 lymph node dissection is performed. Despite the effort to improve surgical quality, locoregional relapse rate remains high with a consequent poor prognosis.
Currently accepted adjuvant and neoadjuvant therapies include adjuvant chemotherapy, postoperative chemoradiation, and perioperative chemotherapy. Adjuvant chemotherapy is mainly given in Japan with S-1, but has not been evaluated in the West because of limited experience with S-1 in Western patients. The Western debate focuses on the use of postoperative chemoradiation
Several currently accruing or yet unpublished trials (Table 5) focus on the choice of the optimal postoperative treatment. In the Dutch CRITICS trial, patients receive three cycles of preoperative chemotherapy (ECC) followed by surgery, after which they receive another three cycles of ECC, or postoperative chemoradiation. The Korean ARTIST trial, which finished accrual, randomized patients who received a D2 dissection between postoperative chemotherapy (cisplatin and capecitabine) and postoperative chemoradiation. No preoperative therapy was administered. Feasibility data of this study were reported at ASCO-GI 2009 showing good toxicity profiles with compliance rates of 75%
Current phase III trials for treatment of gastric cancer.

Explanations of acronyms: ECC: epirubicin, cisplatin, capecitabine; CC: cisplatin, capecitabine; CRT: chemoradiotherapy; GEJ: gastroesophageal junction; 5-FU: 5-fluorouracil; ECF: epirubicin, cisplatin, 5-FU
Western randomized controlled trials on gastric cancer are often hampered by slow or incomplete accrual. Reduction of toxicity for preoperative and especially postoperative treatment and adequate nutritional support is essential for the ongoing improvement of gastric cancer care. Currently accruing Asian trials mainly focus on improved adjuvant chemotherapy with or without immunotherapy.
Most of the studies covered in the current review mention the rate of cardia cancer in the trial population. However, subgroup analyses for cardia
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
