Abstract
Obscure gastrointestinal bleeding (OGIB) is one of the most challenging disorders faced by gastroenterologists because of its evasive nature and difficulty in identifying the exact source of bleeding. Recent technological advances such as video capsule endoscopy and small bowel deep enteroscopy have revolutionized the diagnosis and management of patients with OGIB. In this paper, we review the various diagnostic and therapeutic options available for the management of patients with OGIB.
Introduction
Obscure gastrointestinal bleeding (OGIB) is defined as bleeding from the gastrointestinal (GI) tract that persists or recurs without an obvious etiology after an initial evaluation using bidirectional endoscopy and imaging with a small bowel radiograph [Pasha et al. 2009]. One important characteristic of OGIB is that it is almost always recurrent. It accounts for approximately 5% of all gastrointestinal bleeding [Raju et al. 2007]. The majority of OGIB cases are caused by lesions in the small bowel, which are difficult to assess with traditional endoscopic and radiological procedures. The remaining undiagnosed cases may be due to lesions overlooked on conventional upper endoscopy and colonoscopy.
Classification of obscure gastrointestinal bleeding
OGIB can be divided broadly into obscure overt and obscure occult bleeding. Overt OGIB is characterized by the presence of clinically perceptible bleeding, that is, melena or hematochezia. Occult OGIB is bleeding that is not apparent to the patient or physician but becomes evident with development of iron deficiency anemia with or without a positive fecal occult blood test.
Etiologies of obscure gastrointestinal bleeding
Causes of OGIB may potentially include any lesion from the oral cavity to the anorectum that can bleed into the GI tract. The most common cause of OGIB is vascular lesion, typically found in the small bowel. Vascular lesions are associated with a variety of conditions including but not limited to renal failure, scleroderma, CREST syndrome, (Calcinosis cutis, Raynaud phenomenon, Esophageal-dysmotility, Sclerodactly and Telengiectasia) radiation injury, collagen vascular disease (such as Ehler Danlos syndrome), and Von Willebrand disease. Other vascular causes include Dieulafoy lesions, gastric antral vascular ectasia, portal hypertensive gastropathy, varices (esophageal, gastric, small bowel and colonic) and hemorrhoids. Ulcerations in the GI tract are also quite common. Hayashi and colleagues demonstrated that up to 12% of patients with OGIB were found to have small bowel ulcerations [Hayashi et al. 2005].
Neoplasms of the small intestine are an important cause of OGIB [Rockey, 2010]. The most common benign tumor causing OGIB is a leiomyoma and the most common malignant tumor causing OGIB is a leiomyosarcoma. Benign tumors tend to bleed more than malignant tumors. Other neoplastic causes include carcinoids, GI stromal tumors, adenocarcinoma, ampullary adenoma, and metastatic lesions such as melanoma.
Inflammatory causes of OGIB include esophagitis, peptic ulcer disease, Cameron erosions, inflammatory bowel disease, Meckel’s diverticulum, nonsteroidal anti-inflammatory drug (NSAID)-related gastropathy and enteropathy. Other rare causes include hemobilia, hemosuccus pancreaticus, hereditary hemorrhagic telangiectasias, Von Willebrand disease, psuedoxanthoma elasticum, amyloidosis, and an aortoenteric fistula. Aortoenteric fistula is a rare but life-threatening cause of OGIB. These fistulae are almost always secondary to previous reconstructive aortic surgery, occurring at a frequency of 0.5 % following such surgeries [Bastounis et al. 1997].
Diagnostic evaluation
The medical history and physical examination of patients with OGIB should focus particularly on signs and symptoms of the medical condition that are likely to be overlooked, including those involving the small bowel. In patients with occult OGIB, it is imperative to exclude any cause of anemia, including and not limited to malabsorption, and to document objective evidence of OGIB. Details pertaining to the clinical presentation (the presence or absence of overt bleeding), the nature of bleeding (hematemesis, hematochezia, or melena), any bleeding diathesis (medication use like NSAIDs), and also any comorbidities (valvular heart disease or vasculitis), along with any pertinent family history or history of radiation exposure might provide important clues to the underlying etiology of OGIB.
Evaluation of OGIB involves two types of investigations: radiological and endoscopic evaluations. Radiological evaluation includes the following studies.
Technetium 99 m-labeled red blood cell nuclear scan
The role of the nuclear scan, and in particular, technetium 99 m-labeled red blood scan, continues to be limited in patients with OGIB. This study can detect GI bleeding at a rate of more than 0.1–0.4 ml/min. It has been used as a successful tool in diagnosing rapid GI bleed in actively bleeding patients [Raju et al. 2007]. Despite higher sensitivity than angiography, its role in OGIB is limited because of low accuracy in localization [Voeller et al. 1991]. A delay in scanning has been shown to lead to misinterpretation of results, especially because of pooling of blood in dependent sites [Emslie et al. 1996]. A Meckel’s scan is a variant of technetium 99 m-labeled red blood scan, which exhibits a sensitivity of 64–100% in detecting cases of Meckel’s diverticulum [Sfakianakis and Conway, 1981].
Angiography
The data on clinical utility of angiography in the setting of OGIB are limited. Angiography can detect both acutely bleeding and nonbleeding lesions, particularly angiectasias, which often have characteristic angiographic features. It has the potential to detect bleeding at a rate of more than 0.5 ml/min. The diagnostic yield of this test is dependent on the rate of bleeding, with successful localization in up to 50–75% of patients with active bleeding and less than 50% of patients with cessation of bleeding [Lewis and Goldfarb, 2003]. The main benefit of angiography is the ability to perform therapeutic embolization with use of Gel foam and coils [Rahn et al. 1982]. The complications of this procedure are psuedoaneurysm, arterial thrombosis, dissection and bowel infarction. The overall incidence of complications has been reported to be 0.9% of total procedures performed in one study [Silverstein et al. 1981]. The use of anticoagulants such as heparin, antifibrinolytics and urokinase prior to angiography (provocative angiography) has recently been tested. Although the test may be performed safely, overall diagnostic yield still remains low [Bloomfeld et al. 2000].
Computed tomography enterography and computed tomography enteroclysis
Computed tomography enterography (CTE) and CT enteroclysis are dedicated examinations of the small bowel which can detect both vascular lesions and tumors. The technique utilizes luminal distention after ingestion of a large volume of contrast, thereby permitting visualization of mucosal details and vasculature. A recent study in patients with OGIB using multiphase CTE reported a diagnostic yield of 45% [Huprich et al. 2008]. The main limitation of this technique arises because of inadequate bowel distention with oral contrast secondary to bowel obstruction, GI dysmotility, patient intolerance, and in patients with kidney insufficiency due to limitation in using contrast.
Computed tomography angiography
CT angiography (CTA) involves catheterization of the abdominal aorta followed by helical CTA before and after intra-arterial injections of contrast medium. The site of hemorrhage is recognized as a hyperdense area because of extravasation of contrast medium into the intestinal lumen. CTA uses less oral contrast material than CTE and therefore luminal distention is less. CTA is preferred over CTE or CT enteroclysis if an emergent examination is required as in cases with massive GI bleeding or when the patient cannot tolerate oral contrast. In a prospective study of 18 patients with bleeding colonic angiectasias, the sensitivity, specificity, and positive predictive value of helical CTA were 70%, 100%, and 100% respectively compared with the gold standard of colonoscopy and mesenteric angiography [Junquera et al. 2000].
Endoscopic evaluation
All endoscopic evaluations should begin with an upper and lower GI endoscopy. If these are negative, some experts still would consider repeating the above procedures before proceeding with evaluation of the small bowel. Until recently, small bowel evaluation was inaccessible to endoscopic imaging and therapy without surgery. Complete endoscopic imaging of the small intestine has evolved from invasive intraoperative endoscopy (IOE) to a less invasive examination with techniques such as capsule endoscopy and enteroscopy. Recent development of single and double balloon enteroscopy (SBE and DBE) has allowed us to offer endoscopically directed hemostasis without the need for laparotomy.
Traditional endoscopic modalities include the following.
Push enteroscopy
Push enteroscopy is widely used in the diagnosis and management of OGIB. Originally, adult or pediatric colonoscopes were used to examine the proximal small intestine. Subsequently dedicated push enteroscopes were developed with working lengths of 220–250 cm [Ogoshi et al. 1973]. Push enteroscopy allows limited evaluation of the proximal small bowel, approximately 50–100 cm distal to the ligament of Trietz. The diagnostic yield is reported to be between 3% and 70% with the majority being vascular lesions [Chak et al. 1998]. One limitation results from the looping of the enteroscope in the stomach, which leads to an increase in patient discomfort and precludes further advancement into the small bowel. The use of an overtube resolved this problem partially but did not improve the diagnostic yield further [Taylor et al. 2001]. The role of push enteroscopy is largely limited to endoscopic therapy in patients with known proximal small bowel lesions or those who have proximal small bowel lesions on video capsule endoscopy (VCE).
Sonde enteroscopy
This is an endoscopic technique that is dependent on peristaltic propagation of the flexible enteroscope through the small intestine. Sonde enteroscopy allows detection of bleeding lesions which are located beyond the reach of push enteroscopy [Berner et al. 1994]. However, because the procedure is very prolonged in duration and cannot perform any more advanced therapies, this modality is no longer utilized in clinical practice [Waye, 2001].
Intraoperative enteroscopy
IOE involves the endoscopic evaluation of the small bowel by surgical assistance via laparotomy or laparoscopy. It can be performed orally, rectally or via surgical enterotomy. In 1980, Bowden and colleagues performed IOE by passing a fiberoptic colonoscope first orally and then anally while the surgeon manually telescoped the bowel loops over the tip of the endoscope. The terminal ileum can be reached in up to 90% of patients while minimizing the mortality and morbidity associated with enterotomy. The diagnostic yield of this procedure ranges between 58% and 88%. In a recent case series of 25 patients with OGIB, IOE enabled complete evaluation of the small intestine in all patients and resulted in therapeutic intervention in 16 patients [Douard et al. 2000]. Rebleeding has been reported to occur in 12–60% of patients after IOE [Zaman et al. 1999], mainly due to the fact that pathology might be overlooked from limited visibility and the evanescent nature of vascular ectasias. Other complications of IOE include serosal tears, avulsion of mesenteric vessels, prolonged postprocedure ileus, and perforation [Lopez et al. 1996]. Because of high mortality rates reported in older literature, this procedure is reserved for patients with recurrent OGIB requiring multiple transfusions and negative diagnostic and therapeutic yield on other modalities, including VCE and DBE.
Video capsule endoscopy
The present small bowel capsule measures about 11 m × 26 mm in size, containing light-emitting diodes, a lens, a color camera chip, two batteries, a radiofrequency transmitter, and an antenna [Meron, 2000]. The capsule obtains images and transmits the data via radiofrequency to a recording device worn on the patient’s waist. It has the capability to capture images at the rate of 2 frames/s over an 8 h period. Images are transmitted to a recording device and can be downloaded and viewed on a computer station with appropriate software. Software is available to provide localization of the capsule based on signal strength to skin sensors, and other algorithms can aid in the identification of blood or vascular lesions. VCE in patients with ongoing bleeding, low hemoglobin values, and ongoing transfusion requirements are associated with a higher diagnostic yield in finding the source of bleeding [May et al. 2005]. Patients with swallowing disorders, small bowel obstruction, multiple prior abdominal surgeries or patients with suspected strictures are some of the factors which might preclude the use of VCE. Recent studies have suggested that VCE is safe in patients with pacemakers [Payeras et al. 2005] and defibrillators [Leighton et al. 2005], although these patients need to be monitored carefully during the procedure.
Two meta-analyses have demonstrated that in patients with OGIB, VCE is superior to radiological techniques [small bowel follow through (SBFT) and enteroclysis] and to push enteroscopy. The first meta-analyses of 20 prospective studies totaling 537 patients compared VCE with one or more alternative diagnostic modalities for evaluating the small bowel in patients with OGIB [Triester et al. 2005]. The yields of VCE and push enteroscopy were 56% and 26%, for VCE and barium radiographs the yields were 42% and 6%. The number needed to test with VCE to yield one additional clinically relevant finding over either of the two other diagnostic modalities was 3. In another meta-analysis comprising 289 patients with OGIB, the pooled ‘diagnostic rate difference’ between VCE and other diagnostic tests (push enteroscopy and SBFT) was 36.9% [Marmo et al. 2005]. Published experience shows that VCE identifies the cause of bleeding in the small bowel twice as often as push enteroscopy. The validity of findings made by VCE was confirmed in a prospective study involving 47 patients, comparing VCE with IOE, which is considered to be the gold standard in small bowel visualization. The overall yield of VCE was 74% and the overall yield for both procedures was 76.6%. In 36 patients, bleeding sites were identified by both techniques and only by VCE in two patients and by IOE in one. The sensitivity and specificity of VCE were 95% and 75% respectively and positive and negative predictive values were 95% and 86% respectively [Hartmann et al. 2005]. After VCE directed interventions, 50–66% of patients have been reported to remain transfusion free without recurrent bleeding at follow up, which was over 11.4 months [Estevez et al. 2006]. Two studies with mean follow-up periods of 17 and 19 months reported low rebleeding rates of 11% and 5.6% respectively in patients with negative VCE [Macdonald et al. 2008; Lai et al. 2006]. The major limitations of VCE include the lack of control of its movement and inability to perform therapeutic maneuvers. Second, it is difficult to localize and estimate the size of small bowel lesions precisely with current technology. Also, although infrequent, one of the potential complications includes retention of VCE in the small bowel; this occurs in less than 1% of patients with localized pathology [Barkin and Ross, 1998]. A history of NSAID use may be associated with increased risk of capsule retention [Sears et al. 2004]. Although the majority of patients with capsule retention are asymptomatic, complications such as aspiration, retention in Zenker’s diverticulum, intestinal perforation, and obstruction have been reported [Gonzalez Carro et al. 2005]. Retained capsules are removed by either endoscopy or surgery.
Balloon-assisted enteroscopy
This procedure utilizes the principle of push and pull enteroscopy and there are two types available: DBE and SBE.
Double balloon enteroscopy
DBE, first described in 2001, consists of an enteroscope and an over tube with a balloon at their distal ends. It allows for complete visualization of the small intestine using a 200 cm long enteroscope with an outer diameter of 8.5 mm equipped with a 140 cm long over tube with an outer diameter of 12 mm. Latex balloons at the tip of the enteroscope and the over tube are inflated and deflated with air from a pressure-controlled pump system. By inflating the overtube balloon enough to grip the intestinal wall, the endoscope can be inserted further without forming redundant loops in the small intestine. The over tube can then be inserted while the endoscope balloon is still inflated. A prospective controlled trial [May et al. 2007] of 52 patients with suspected OGIB showed that per oral DBE is significantly superior to push endoscopy with regards to the length of the small bowel visualized (230 cm versus 80 cm) and detection of pathologic lesions (63% versus 44%). In the same study, DBE identified additional lesions in deeper parts of the small bowel in 78% of patients who had positive findings on push endoscopy (Table 2). DBE can be performed by either the per oral or antegrade approach or through the anus or retrograde approach. Per oral examinations achieved a mean distance of 360 ± 177 cm beyond pylorus in four centers in the US multicenter study [Mehdizadeh et al. 2006]. Total enteroscopy, defined as an initial DBE from one approach with India ink tattooing using a sclerotherapy needle at the farthest insertion point followed by a DBE in the opposite direction with visualization of the tattoo mark, is not necessary in the majority of patients. The decision to perform total enteroscopy is usually dependent upon the discretion of the endoscopist, the degree of clinical suspicion for a small bowel lesion, and inability to detect a small bowel lesion using a single approach. The reported success rates of total enteroscopy range from 16% to 86%. A higher success rate is reported in the Asian population compared with the western population, although the reasons for this are unclear [Pasha et al. 2009]. A recent meta-analysis based on 11 studies revealed that VCE and DBE have comparable diagnostic yields in small bowel disease, including OGIB, and pooled overall yield for VCE and DBE were 60% and 57% [Pasha et al. 2008]. Many studies have compared VCE and DBE in patients with OGIB (Table 3). The main advantage of DBE over VCE is the therapeutic capabilities, including biopsies, tattooing, hemostasis, polypectomy, balloon dilatation, and foreign body retrieval. The main limitations of DBE include its invasive nature, prolonged duration, and need for additional personnel. It is becoming increasingly clear that DBE carries a risk of complications, especially during or after therapeutic interventions. The complications most frequently reported to date include pancreatitis, postpolypectomy bleeding and intestinal perforation. A large, multicenter, international complication survey reported a total of 40 complications in 2362 DBE procedures (1.7%), which included bleeding (13 cases), perforation (five cases), and pancreatitis (seven cases). A total of 13 complications were seen in 1728 diagnostic DBEs (0.8%), and 27 during 634 therapeutic procedures (4.3%) [Mensink et al. 2007].
Single balloon enteroscopy
SBE is the most recent balloon-assisted endoscopic technique that has been introduced for the evaluation and management of small bowel disorders. This procedure includes a 200 cm long high-resolution enteroscope with a 2.8 mm working channel and 9.2 mm outer diameter that uses a 140 cm long by 13.2 mm outer diameter flexible over tube. The only silicone balloon at the tip of the over tube can be deflated and inflated by a balloon control module with a pressure range of 6–16 kPa. This pressure allows atraumatic traction on small bowel mucosa. The tip of the endoscope is angled and hooked behind a fold, if possible, to achieve the same stabilizing effect of the endoscope tip balloon used in the DBE system. The over tube balloon is used to pleat the small bowel onto the over tube. With serial inflations and deflations of the over tube balloon and hooking and grasping with the endoscope tip, the endoscope tip can be advanced deep into the small bowel with a push and pull technique [Upchurch and Vargo, 2009]. Ideally, as in DBE, the balloon is not inflated in the area of the proximal duodenum to avoid trauma to the papilla and possibly lessen the risk of pancreatitis.
Few preliminary studies have been done so far, all of which indicate comparable diagnostic yield and therapeutic capabilities with SBE, with less procedure time compared with DBE, along with ease of use. A preliminary report of 78 SBE procedures performed in 41 patients of whom 12 had OGIB found that SBE allowed evaluation of the small bowel in a safe and effective manner, including performance of total enteroscopy. The diagnostic yield was 66% [Tsujikawa et al. 2008]. In another study from a tertiary care center, a total of 161 patients underwent 172 procedures. Antegrade and retrograde approaches were used in 83% and 17% of patients respectively. The average insertion depth using the antegrade approach was 132 cm beyond the ligament of Treitz. The average insertion depth from the retrograde approach was 73 cm above the ileocecal valve. The average procedure time was 40 min overall, 38 min (range 12–90) antegrade and 48 min (range 28–89) retrograde. The diagnostic yield was 58% (99/172); 42% (72/172) were therapeutic cases. No significant complications were reported [Upchurch et al. 2010]. In another study 106 patients with suspected small bowel disease underwent 131 SBE procedures [Ramchandani et al. 2009]. The mean insertion depth was 255.8 ± 84.5 cm beyond the duodenojejunal flexure by the oral route and 163 ± 59.3 cm proximal to the ileocecal valve via the retrograde approach. The mean duration of the procedure for antegrade and retrograde enteroscopy was 65.9 ± 19.5 and 72.3 ± 18.3 min respectively. Total enteroscopy was possible in 25% of cases. The diagnostic yield in patients with OGIB was 60%. In 21% of patients, therapeutic interventions were carried out. No major complications were observed. These initial experiences suggest that SBE has a comparable diagnostic yield to DBE along with similar therapeutic intervention possibilities, including the advantages of a shorter duration of procedure and fewer complications like pancreatitis. However, larger prospective trials are required to compare DBE and SBE before any conclusion can be drawn.
Spiral enteroscopy
Spiral enteroscopy is a new technique of endoscopic evaluation of the small bowel which was first proposed in 2006. Spiral enteroscopy applies the mechanical advantage of a screw to convert rotational force into linear force and pleat the small bowel on the enteroscope. The mechanical advantage of the screw is equal to the circumference over pitch. Therefore, the higher the spiral, the more pulling power, and the smaller the pitch, the greater the mechanical advantage of the spiral. The current device is called the Discovery SB over tube from spirus corporation, has been approved by Food and Drug administration with a CE mark. This over tube has an overall length of 118 cm, the internal diameter is 9.8 mm, the outer diameter is 14.5 mm, the spiral height is 5.5 mm, and the spiral length is 22 cm. The device has variable stiffness and a locking device that fixes the Discovery SB over tube to the endoscope, still allowing rotation of the over tube at the other end. The proximal end of the Discovery SB over tube has two foam handles to assist rotation.
A few studies have been done using the spiral enteroscopy technique (Table 1). Akerman and colleagues [Akerman et al. 2008] performed a prospective study with 75 patients. Two endoscopes were used, the Olympus SIF Q180 manufactured by Olympus America Inc 1 and the Fujinon EN-450T5 manufactured by Fujinon, Inc 2 . The average depth of insertion past the ligament of Treitz was 243 cm for the Olympus enteroscope and 256 cm for the Fujinon enteroscope. The average time to reach the maximum depth was 18.7 min and the total procedure time was 29 min in the Olympus group and 16.2 and 26 min respectively in the Fujinon group. The diagnostic yield was found to be 32% and 22% respectively in the Olympus and the Fujinon groups. The low diagnostic yield was thought to be due to the young age of the cohort and the lack of capsule study prior to the procedure. Morgan and colleagues [Morgan et al. 2010] performed a multicenter prospective trial in 148 patients over 10 centers. Spiral enteroscopy was successful in 96% of patients, including 72% of patients with OGIB. The average depth of insertion was found to be 250 cm with the average time being 45 min for therapeutic procedures and 34 min for nontherapeutic procedures. In another study by Esmail and colleagues [Esmail et al. 2009], 47 patients were studied retrospectively. Fifty-four of 57 procedures were successful. The average depth of insertion was 246 cm, the average total procedure time was 28 minutes and the diagnostic yield was 51%. Few studies have been done to compare DBE with spiral enteroscopy. However, one such case–control study by Schembre and Ross compared DBE with spiral enteroscopy. Thirty-four cases of DBE and 19 cases of spiral enteroscopy were analyzed. The groups had similar demographics, including age, prior VCE findings, and indications. The diagnostic yield was similar in both groups: 70% in DBE and 65% in spiral enteroscopy. The average procedure time was 77 min for DBE and 59 min for spiral enteroscopy. A crossover study was performed in three patients. In one patient spiral enteroscopy exceeded the DBE tattoo site by 100 cm; in one patient spiral enteroscopy reached the same site as DBE; and in one patient DBE was performed successfully after spiral enteroscopy failed.
Major studies done in spiral enteroscopy.
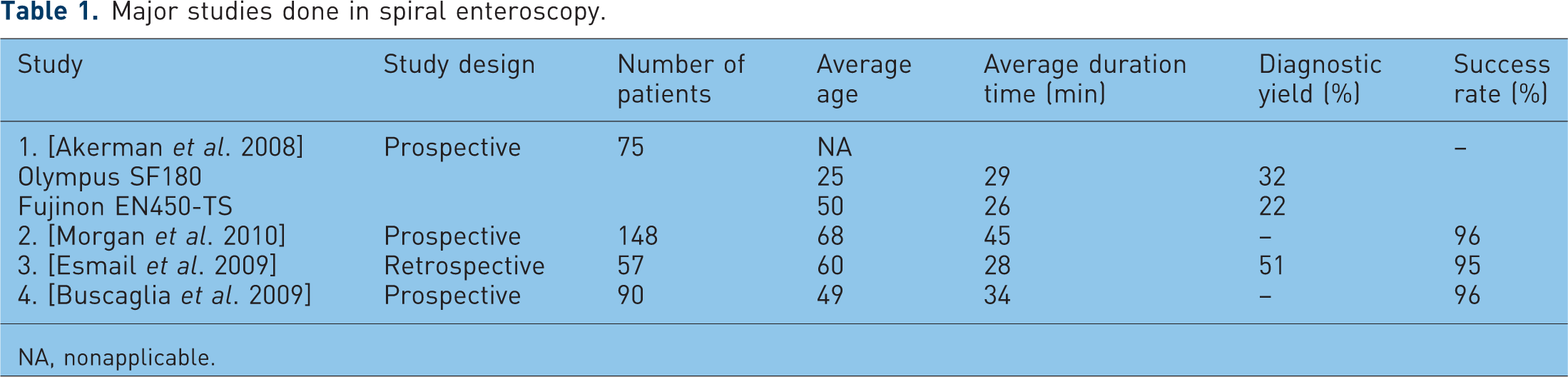
NA, nonapplicable.
Major studies done in double balloon enteroscopy.

NA, nonapplicable.
In a review of 1750 patients who underwent spiral enteroscopy, six perforations occurred during the procedure and all patients were taken to surgery and did well postoperatively [Akerman and Cantero, 2009]. In short, spiral enteroscopy is a new technique for visualization of the small bowel, which so far has demonstrated diagnostic yield, total time of procedure and depth of insertion that compare favorably with DBE and SBE. It can be performed in postgastric surgery patients, including those who have had the Roux-en-y procedure who require endoscopic retrograde cholangiopancreatography. Retrograde spiral enteroscopy can also be performed; however, more prospective studies are required to compare the competing technologies.
Pharmacological management
Pharmacotherapy of OGIB should be considered when endoscopic therapy, surgical intervention, or angiographic therapy is not practical or is ineffective, or in patients in whom the source of bleeding is unknown or the pathology is too diffuse to be amenable to ablative therapy. Medical management with hormonal therapy (estrogen with or without progesterone), somatostatin analogues, thalidomide, erythropoietin, and Von Willebrand factor have been utilized in the past. The role of hormonal therapy continues to be the most controversial [Grant, 2002], and only one small observational study has reported the beneficial effects of hormonal therapy [Barkin and Ross, 1998]. More recently, a randomized placebo controlled trial included 72 patients with chronic bleeding caused by gastro duodenal, colonic and diffuse angiectasias who were treated with ethinyl estradiol and norethisterone. Patients with hereditary hemorrhagic telangiectasia and cirrhosis were excluded. The primary endpoint was failure to prevent recurrent bleeding, either an acute episode or persistent iron deficiency anemia despite persistent iron therapy. The result did not reveal any beneficial effects, irrespective of the disease site [Junquera et al. 2001]. Treatment failed among 39% of treated patients and 46% of the placebo group, with similar rates of rebleeding and transfusion requirements over a period of 2 years. Rebleeding was most likely to occur among patients with a previous history of multiple bleeds and in patients with chronic renal failure.
Hormonal therapy is thought to lead to improved vascular integrity and decrease vascular angiogenesis by inhibiting endothelial growth factor [Fujita et al. 2000]. Somatostatin and its analogue octreotide have been anecdotally reported to be beneficial in patients with obscure GI bleeding from angiectasias and blue rubber bleb nevus syndrome, possibly due to their inhibitory effect on angiogenesis and splanchnic blood flow [Blich et al. 2003]. Recently, some case reports have shown the beneficial effect of thalidomide and its antiangiogenic effects in patients with OGIB due to angiectasias and hereditary hemorrhagic telangiectasia [Perez-Encinas et al. 2002]. In a recent case report, erythropoietin was believed to stop chronic diffuse hemorrhage from GI mucosa. The exact mechanism is not known; however, it may be related to the complex effect of erythropoietin on the platelet–subendothelial interactions and also on protein C, protein S, and antithrombin III levels [Zaharia-Czeizler, 2001]. Potential therapeutic strategies focusing on replenishing and maintaining the level of reduced high molecular-weight multimer of Von Willebrand factor may offer a therapeutic potential for management of patients with bleeding angiectasias and needs to be studied. Hence, medical management of OGIB should be reserved for patients with a negative comprehensive evaluation and those who fail endoscopic, radiological, and/or surgical management.
Overview of studies on capsule endoscopy (CE) and double balloon enteroscopy (DBE) in obscure gastrointestinal bleeding.

NA, nonapplicable.
Summary
OGIB is defined as persistent or recurrent bleeding from a difficult to identify source. OGIB accounts for approximately 5% of all cases of GI bleeding and is most commonly caused by lesions in the small bowel. OGIB represents one of the most challenging disorders faced by gastroenterologists due to its evasive nature and difficulty in identifying the exact source of the bleeding. Recent technological advances such as capsule endoscopy, balloon-assisted enteroscopy, spiral enteroscopy and CTE have significantly improved our ability to diagnose and manage these patients. These advances and the capacity to successfully perform endoscopic therapeutics have replaced surgical procedures and resulted in a trend towards noninvasive evaluation and endoscopic management of OGIB. In patients with persistent bleeding after endoscopic therapy and those who are poor operative candidates, pharmacotherapy may be tried. Further studies are needed to evaluate the impact of these techniques on utilization of healthcare resources and clinical outcomes.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None declared.
1
Olympus America Inc. 3500 Corporate Parkway, P.O. Box 610, Center Valley, PA 18034-0610
1
Fujinon, Inc.10 High Point Drive Wayne, New Jersey 07470
