Abstract
Background:
Despite continuous advances in management over the last two decades for pulmonary embolism (PE), treating intermediate- to high-risk PE remains challenging. There is a critical need to expand our understanding of the clinical spectrum beyond systolic blood pressure (SBP) to inform the intricate decision-making process for initiating advanced treatment in patients with intermediate- to high-risk PE.
Objectives:
We aimed to characterize the range of SBP values in patients with intermediate- to high-risk PE undergoing advanced therapy across different clinical settings. The secondary objective included the incidence of bleeding complications (intracranial, major, and minor).
Design:
We conducted a systematic review in alignment with PRISMA guidelines. The review involves a comprehensive, structured search of multiple electronic databases.
Data sources and methods:
We created two groups: Group 1, case reports and series, and Group 2, randomized control trials and cohorts with intermediate-risk PE who underwent advanced treatment. In addition, we performed an exploratory analysis in Group 1, created solely for descriptive purposes, to determine the frequency of the previously mentioned impending deterioration factors in the literature and further clarify their potential role in initiating advanced treatment.
Results:
We identified 1871 intermediate-high risk PE patients who received advanced therapy, divided into two groups: Group 1 for case reports and series, and Group 2 for randomized controlled trials and cohorts. In total, 77.1% reported SBP; the weighted average was 125.8 and 129.2 mmHg for Groups 1 and 2, respectively. The most common initial clinical presentations were dyspnea, syncope, and chest pain. In the exploratory analysis, we found that borderline SBP (110–120 mmHg), right ventricular dysfunction, and heart rate (⩾120 bpm) were the most frequent impending clinical deterioration factors.
Conclusion:
This systematic review shows that the SBP range for advanced treatment (129.2 and 125.8 mmHg) aligns with previous clinical models. However, it is important to recognize that while these SBP ranges may suggest practice variation, causality or definitive clinical instability cannot be inferred from aggregate data without patient-level outcomes. Outside of randomized controlled trials, clinical decision-making regarding advanced treatment may not fully align with international recommendations in real-world scenarios.
Trial registration:
Research Registry number: 2065.
Background
Despite the continuous advancements in management over the last two decades in pulmonary embolism (PE), the mortality1,2 and optimal treatment for intermediate- to high-risk PE remain a significant challenge. 3 Current evidence does not establish the efficacy or safety of thrombolysis in patients with SBP >90 mmHg. American4,5 and European 6 current guidelines recommend initial advanced treatment for high-risk or massive PE and for those with clinical instability during the in-hospital first hours or days while on anticoagulants based primarily on <90 mmHg systolic blood pressure (SBP).5,7 In addition, all international guidelines4–6 discourage advanced treatment because of significant bleeding incidence (11.5%) and hemorrhagic stroke (2.0%) for intermediate- to high-risk PE.7,8 Recently, a retrospective cohort study showed that SBP between ⩽110 and ⩽120 mmHg and diastolic BP ⩽65 mmHg had a close relationship with a higher risk of in-hospital death in intermediate- to high-risk PE patients. 9 In addition, a substudy of the RIETE registry showed that SBP <110 mmHg was associated with increased mortality in the same PE population. 10 Barco et al. 11 recently identified a higher-risk population as thrombolysis responders through three clinical indicators of severity at clinical presentation, including an SBP ⩽110 mmHg. Considering this evidence, an open question remains: What is the SBP range in a large population of intermediate-to high-risk PE patients undergoing advanced treatment? Therefore, identifying the SBP range that drives this population to advanced treatment, while considering the potential bleeding risk, 3 remains fundamental in determining a high-risk phenotype.11,12 We thus undertook a systematic review to identify the SBP range in intermediate- to high-risk PE submitted to advanced treatment across different clinical settings, to determine whether initial advanced treatment is performed on a specific clinical phenotype in accordance with international guidelines.
Objectives
We aimed to characterize the range of SBP values among patients with intermediate- to high-risk PE undergoing advanced therapy across different clinical settings. The secondary objective included the incidence of bleeding complications (intracranial, major, and minor).
Data sources and methods
We systematically searched from January 1, 2010, to September 1, 2021. Figure 1 shows the flowchart, including the four phases of PRISMA statements 13 and the number of papers obtained, duplicated, and excluded (Supplemental Table 1). This study was registered post-study at Research Registry (https://www.researchregistry.com/), registration number: 2065. We searched through EBSCO, OpenGrey, PubMed, Wiley, and Web of Science databases. In addition, we conducted a manual search (“snowball sampling method”). 14 We used the following Medical Subject Headings (MESH) terms: (pulmonary embolism) OR (intermediate-high risk) OR (submassive) OR (right ventricular dysfunction) OR (clinical worsening) OR (early clinical worsening) OR (systemic thrombolysis) OR (catheter-directed thrombolysis), OR (ultrasound-assisted catheter-directed thrombolysis).
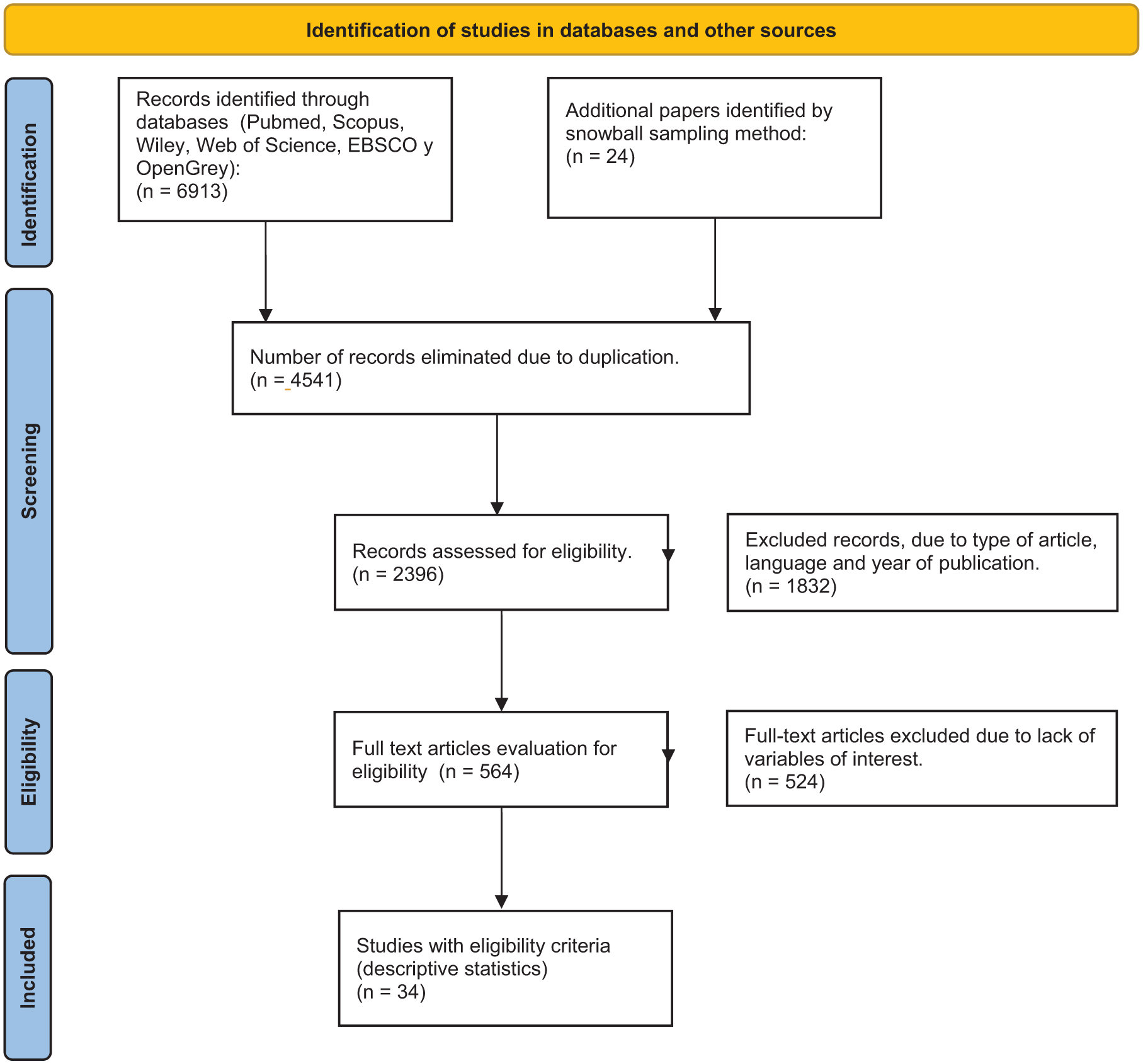
Flowchart for systematic reviews and meta-analysis (PRISMA).
We created two groups: Group 1, case reports and series, and Group 2, randomized control trials and cohorts with intermediate-risk PE who underwent advanced treatment to determine their SBP measurements. To provide a perspective on the use of advanced treatment in a real-world setting and on the generalizability of the study, we included case reports and series in group 1, intended to describe a broader range of clinical variables. Finally, we performed an exploratory analysis to determine whether clinical decision-making for advanced treatment was guided solely by international guideline recommendations (SBP <90 mmHg) or also considered the previously defined factors of impending clinical deterioration. 12 Data were also collected on demographic characteristics, clinical presentation, and imaging studies to confirm the presence of PE, right ventricular dysfunction (RVD), and advanced treatment modalities (Tables 1–4). Data on demographic characteristics, clinical presentations, and imaging studies were also collected to confirm the presence of PE, RVD, and advanced treatment modalities (Tables 1–4). We utilized summary statistics for continuous and categorical variables, and no hypothesis testing was performed. Establishing a dedicated group to review case reports and case series systematically will improve our understanding of key clinical and para-clinical variables and facilitate the adoption of advanced pulmonary arterial reperfusion therapies in real-world settings. International clinical guidelines recommend using these therapies based on specific SBP cut-offs. However, the need for a thorough evaluation of critical factors, including indicators of impending clinical deterioration, cardiac biomarkers, electrocardiographic findings, and cardiovascular imaging, has been emphasized in the treatment of normotensive patients with PE.
Impending clinical deterioration factors reported in randomized controlled and cohort studies included in the systematic review.
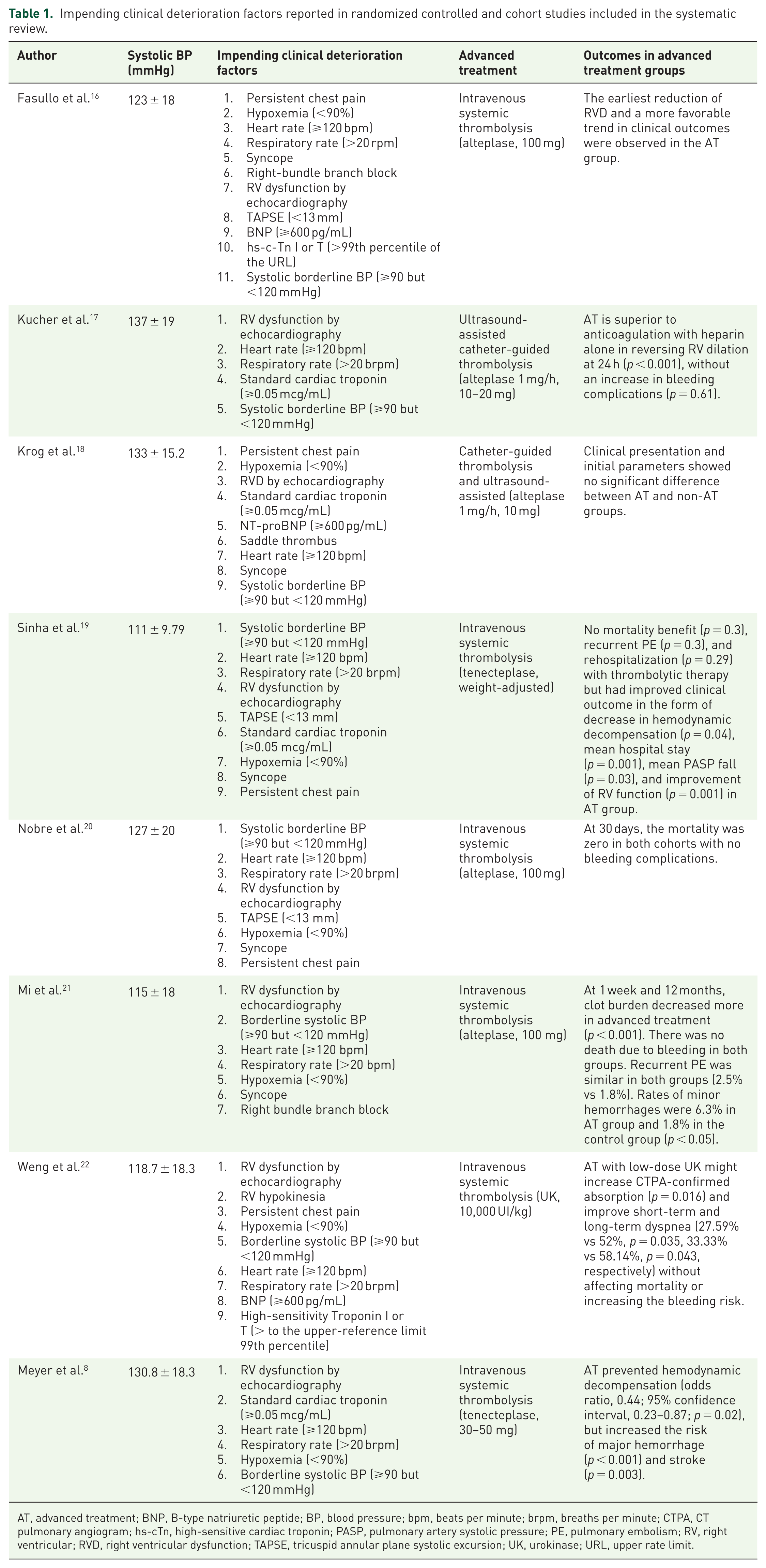
AT, advanced treatment; BNP, B-type natriuretic peptide; BP, blood pressure; bpm, beats per minute; brpm, breaths per minute; CTPA, CT pulmonary angiogram; hs-cTn, high-sensitive cardiac troponin; PASP, pulmonary artery systolic pressure; PE, pulmonary embolism; RV, right ventricular; RVD, right ventricular dysfunction; TAPSE, tricuspid annular plane systolic excursion; UK, urokinase; URL, upper rate limit.
Demographic characteristics, comorbidities, risk factors, and clinical presentation in intermediate- to high-risk PE patients under advanced treatment.

Weighted mean from reported data.
Number and percentage of reported data.
COPD, chronic obstructive pulmonary disease; DBP, diastolic blood pressure; PE, pulmonary embolism; SBP, systolic blood pressure; VTE, venous thromboembolism.
Diagnostic imaging, right ventricular performance, and cardiac biomarkers in studies included in the systematic review. a

Frequency and percentages according to the total number of studies included in the systematic review.
PSAP; mean of the values reported in the studies.
Abnormal or elevated values.
BNP, B-type natriuretic peptide; CTPA, computed tomography pulmonary angiogram; hs-cTn, high-sensitivity cardiac troponin; IVC, inferior vena cava; PASP, pulmonary artery systolic pressure; RCT, randomized control trials; RV/LV, right ventricular/left ventricular; RVD, right ventricular dysfunction; TAPSE, tricuspid annular plane systolic excursion; V/Q, ventilation-perfusion.
Advanced treatment modalities, regimens, and adjunctive treatment in studies included in the systematic review. a
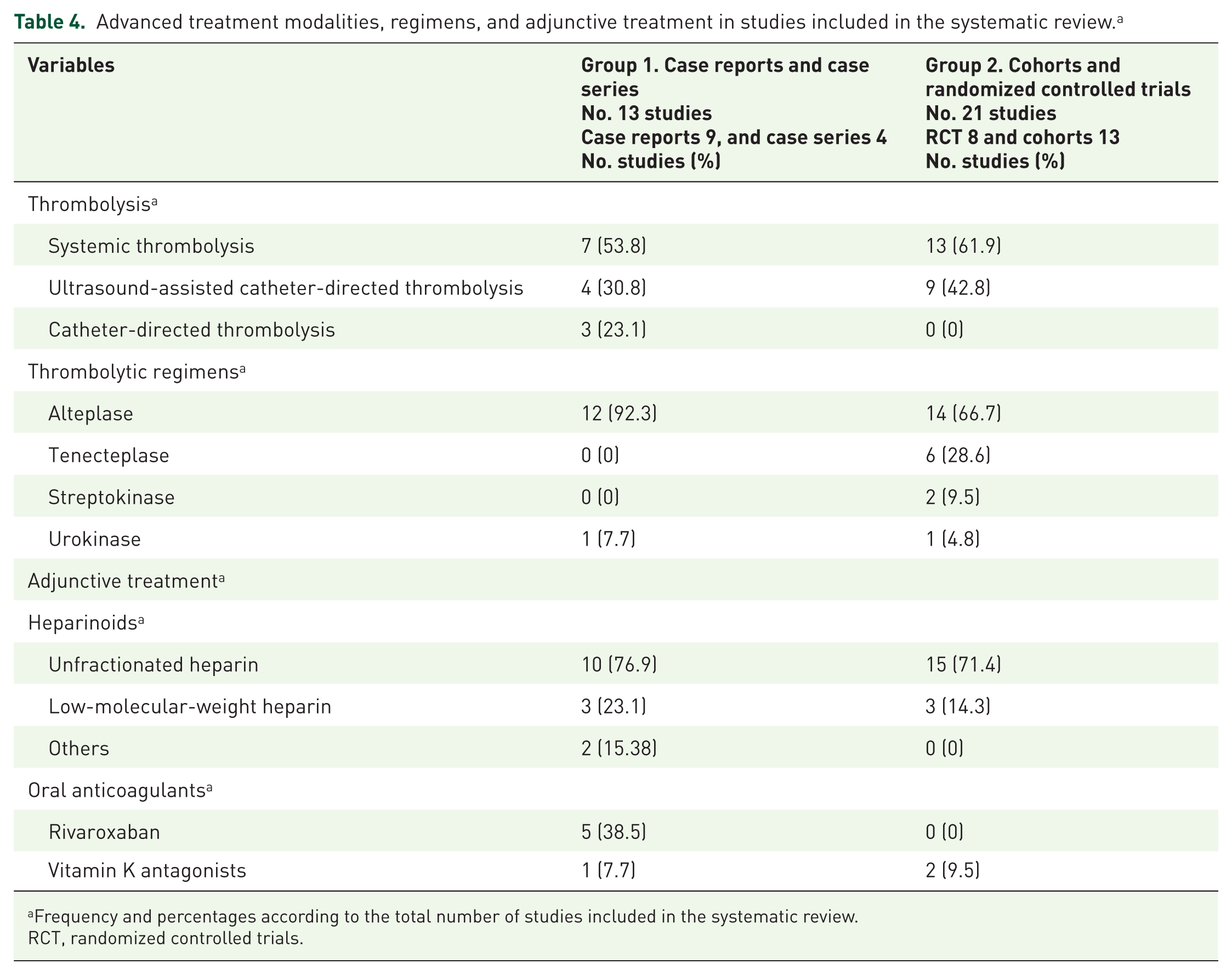
Frequency and percentages according to the total number of studies included in the systematic review.
RCT, randomized controlled trials.
Study selection
After the initial database search, we removed duplicate results and filtered by language, study type, and year of publication. Finally, we read the titles and abstracts of the full-text papers in pairs to assess their eligibility, excluding those without relevant variables. Consequently, we included the remaining papers in the systematic review, excluding those published in languages other than Spanish and English. We did not apply any filters or restrictions beyond the publication date and language. We organized the references using the reference management software Zotero (https://www.zotero.org/). Studies were eligible for this analysis if they met all of the following criteria: Patients older than 18 years, symptom onset of the index event within 14 days, PESI score of class III–V or sPESI greater than 1, confirmed PE by imaging (computed tomography pulmonary angiography (CTPA), lung ventilation-perfusion scan, or pulmonary angiography) with RVD diagnosed by echocardiography or CTPA, abnormal biomarker measurements of B-type natriuretic peptide, pro-BNP-NT, standard or high-sensitivity cardiac troponin, SBP greater than 90 mmHg, and advanced treatment. In addition, we excluded trials with massive PE associated with cardiac arrest, hypotension (SBP <90 mmHg), cardiogenic shock, or surgical thrombectomy, and trials without SBP measurements.
Data collection process
We performed the initial search with two sub-investigators. The results of the initial search were then paired to avoid selection bias. In case of confusion or discrepancy regarding the inclusion of a study, the corresponding author issued the final decision. Disagreements were resolved by consensus. All investigators interpreted the data from the included articles to improve data quality. All authors, including clinicians, cardiologists, and medical students, received appropriate training from a clinical cardiologist. Finally, the corresponding author and sub-investigator jointly reviewed the final version. We did not contact the authors to obtain information about patients with incomplete information.
Bias risk in individual studies
We performed paired information extraction to reduce the potential risk of selection bias. We also did a gray literature analysis and manual search to reduce the risk of reporting bias.
Quality criteria
Two sub-investigators performed a quality assessment using the Newcastle–Ottawa Scale with nine domains of cohort and case–control studies. We used the Prevalence Critical Assessment Instrument, a 10-domain instrument for cross-sectional studies.
Exploratory analysis
One unanswered question is whether the physicians in charge follow international recommendations for initiating advanced therapy in this PE subgroup. In addition, identifying a higher-risk phenotype for advanced treatment and considering the potential risk of bleeding remains of fundamental importance. Thus, we undertook an exploratory analysis to determine whether clinical decision-making for advanced treatment follows international guidelines or includes clinical factors indicative of impending clinical deterioration 12 in intermediate- to high-risk PE.
Definitions
We defined advanced treatment as systemic thrombolysis, catheter-directed thrombolysis, and ultrasound-assisted catheter-directed thrombolysis. According to the author’s definition, we used European guidelines for intermediate and intermediate- to high-risk patients, 6 and RVD (echocardiography or CTA). In addition, we defined abnormal B-type natriuretic peptide, NT-proBNP, or standard or high-sensitivity cardiac troponin I or T assays according to cutoff levels in the original studies. In the randomized controlled and cohort studies in the systematic review, we identified variables historically associated with poor outcomes 12 as impending clinical deterioration factors (Table 1). We defined borderline SBP as ⩾90 but <120 mmHg.9,10 We classified bleeding complications according to the ISTH definition 23 : Major bleeding criteria, including fatal, intracranial, intraocular, intraspinal, pericardial, intramuscular with compartment syndrome, intraarticular, or bleeding causing a fall in hemoglobin level of 20 g/L or leading to transfusion of ⩾2 units of whole blood or red cells.
Results
In the identification phase, we collected 6937 papers from PubMed, Scopus, and Web of Science. We also identified 24 articles through snowball sampling. Next, we carefully reviewed the full text for eligibility criteria and selected 34 papers for the systematic review.8,16–18,20–22,24–49
Table 1 shows the research papers included in group 2, their mean SBP measurements, impending clinical deterioration factors, advanced reperfusion therapy employed, and their clinical outcomes. Table 2 shows the main demographic characteristics of Groups 1 and 2. In both groups, the patients were around 60 years old, and cardiovascular comorbidities and risk factors were common in PE patients. Furthermore, dyspnea drove most PE patients’ clinical presentation associated with syncope or chest pain. All patients were clinically stable at presentation. Table 2 also shows the number of patients under advanced treatment; 77.1% of the papers reported SBP, and the weighted average of the total number of patients included was 125.8 and 129.2 mmHg for Groups 1 and 2, respectively. CTPA guided the diagnosis, and echocardiography or CTPA established RVD. The report of biomarker determinations was low (Table 3). The most frequent advanced treatment was systemic thrombolysis with alteplase, followed by ultrasound-assisted catheter-directed thrombolysis. Unfractionated heparin was used as an adjunctive treatment (Table 4). Table 5 shows the principal bleeding complications, including intracranial hemorrhage (Group 2: 18 patients, 1%; Group 1: 1 patient, 3.4%). In group 2, we identified historical clinical variables at risk for poor outcomes, 12 such as RVD, borderline BP, significant tachycardia, respiratory distress, and persistent hypoxemia (Table 1).
Bleeding complications and outcomes in intermediate- to high-risk PE patients under advanced treatment.

Frequency and percentage according to the total number of patients reported in the study.
PE, pulmonary embolism.
Discussion
This systematic review is the first to analyze SBP ranges in large cohorts of patients with intermediate- to high-risk or submassive PE who received advanced treatment. Several notable findings emerged. First, the identified borderline SBP ranges (Supplemental Table 2) are consistent with previous clinical observations associated with poor outcomes.9,10 Second, the narrow SBP range often used to guide clinical decision-making may give the impression of stability that can be misleading, underscoring the need to consider redefining SBP thresholds or establishing hemodynamic instability equivalents that better characterize the intermediate- to high-risk phenotype. Third, outside of randomized controlled trials, clinical decisions regarding advanced treatment may not fully align with international guidelines. Finally, bleeding complications remain a key concern associated with advanced treatment, underscoring the importance of more accurately identifying which submassive PE patients are most likely to benefit from aggressive interventions.
Intermediate- to high-risk patients represent a heterogeneous group with in-hospital mortality rates ranging from 3% to 15%, 50 and optimizing treatment strategies remains challenging. 3 The mechanisms leading to clinical deterioration involve sudden increases in pulmonary vascular resistance and right ventricular (RV) strain, triggered by flow impedance, local hypoxia-induced vasoconstriction, and vasoactive peptide release from platelets and thrombin. 50 This cascade can potentially lead to RV overload, reduced RV stroke volume, increased wall tension, myocardial ischemia or type II infarction, cardiogenic shock, and death.50,51 Early intervention with advanced therapies might help interrupt this hemodynamic cascade, potentially preventing thrombus resistance and improving RV function in submassive PE patients.
SBP measured in the emergency room or ICU is a readily available, reliable parameter for risk stratification and decision-making between advanced treatment and anticoagulation alone in intermediate- to high-risk PE. 9 At presentation, the average SBP was 125.8 mmHg in Group 1 and 129.2 mmHg in Group 2 (Table 2). Although these ranges are similar, they may not reflect meaningful differences. These SBP values suggest clinical stability, which aligns with the design of the PEITHO and ULTIMA studies, intended to evaluate novel therapies in intermediate- to high-risk PE rather than mirror routine clinical decision-making in practice. However, in Group 1, the SBP range suggests that physicians may not have fully aligned with international guidelines and recommendations, as advanced treatment was often initiated before overt hemodynamic instability occurred, despite anticoagulation therapy. However, individual clinical decision-making may vary, and aggregate data alone cannot fully capture the rationale behind these choices.
The change in SBP range from clinical presentation to the point of initiating advanced treatment suggests an early transition from intermediate to high risk. These findings align with previous evidence. Keller et al. 9 found that at admission, SBP ⩽120 mmHg (p = 0.006) or ⩽110 mmHg (p = 0.001), along with diastolic BP ⩽65 mmHg (p = 0.01), was closely associated with a higher risk of in-hospital death in intermediate- to high-risk PE patients. Similarly, a post hoc RIETE substudy 10 involving the same population demonstrated that an SBP <110 mmHg was associated with increased all-cause and PE-specific mortality. These data, along with our findings in Group 1, suggest that compensatory mechanisms, such as tachycardia and increased systemic vascular resistance, may sustain SBP within the 90–120 mmHg range, potentially masking genuine clinical instability in submassive PE patients.
Additional evidence comes from a prospective cohort study of 92 patients with intermediate- to high-risk PE who were referred for catheter-directed thrombolysis. 52 Baseline hemodynamics showed that 40% of patients had a cardiac index ⩽1.8 L/min/m2 with an average SBP of 122.1 ± 21.69 mmHg, indicating a normal SBP range associated with a low cardiac output phenotype in this group. Moreover, the FLASH registry, 53 which included intermediate- to high-risk PE patients undergoing mechanical thrombectomy with hemodynamic assessment, reported a 34.1% prevalence of normotensive shock (SBP >90 mmHg and cardiac index <2.2 L/min/m2). Within this cohort, higher composite shock scores, incorporating biomarkers of RV dysfunction, ischemia, ventricular performance, thrombus burden, potential embolization, and cardiovascular compensation, predicted a 58.3% incidence of normotensive shock (OR 5.84). These findings highlight the phenomenon of false clinical stability and underscore the need to refine shock assessment, perhaps by revalidating risk scores in intermediate- to high-risk or submassive PE populations.
On the other hand, SBP alone has limitations, particularly because it captures only two-thirds of the cardiac cycle, and coronary perfusion occurs predominantly during diastole. In acute PE, diastolic coronary flow can be significantly impaired. 3 Therefore, redefining hypotension to include diastolic BP in the context of intermediate- to high-risk PE is crucial.4,6 Although RVD is a marker of poor prognosis and often drives the clinical decision-making to initiate advanced treatment, current recommendations for high-risk PE are to monitor the patient and initiate advanced pharmacological or mechanical treatment based on SBP behavior (<90 mmHg). 6 However, the FLASH registry has demonstrated that it is possible to identify patients with an SBP >90 mmHg and an abnormal cardiac index (normotensive shock). This phenotype can be determined using serum lactate levels or new echocardiographic approaches. Recent echocardiographic studies reveal that measures such as left ventricular outflow tract velocity time integral (<15 cm), a surrogate for cardiac output, correlate with higher mortality risks and may help predict normotensive shock. 54 In addition, a low RV outflow tract velocity time integral (<14.7 cm), indicating reduced RV stroke volume, has been independently associated with mortality in pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension. 54 Incorporating these echocardiographic parameters into current risk stratification models could improve patient assessment and identify those at higher risk within the intermediate- to high-risk PE groups.
Bleeding complications, particularly intracranial hemorrhage, remain among the most severe adverse events in the acute phase of PE. Concerns over intracranial bleeding significantly contribute to the cautious use of thrombolytic therapy in high-risk PE. 55 The risk of major bleeding, particularly intracranial hemorrhage, appears to outweigh the potential benefits, especially in elderly patients, where advanced age is a strong predictor of bleeding risk. This underscores the need for cautious patient selection, as there is no clear evidence of mortality reduction or long-term benefits, such as reduced chronic thromboembolic disease, in intermediate- to high-risk PE. 56 The incidence of major bleeding with initial anticoagulation ranges from 1.1% to 2.2%, while bleeding associated with thrombolytic agents like alteplase can range from 8% to 21%, with intracranial hemorrhage rates from 0% to 7.4%. 56 Data from the German nationwide inpatient sample (2005–2020) indicate an intracranial hemorrhage incidence of approximately 0.03%, with higher risk factors including age ⩾70 years, severe PE, recent surgery, acute kidney injury, and prior stroke. 55 Recent studies, including our own, have reported intracranial hemorrhage rates around 0.6%, with overall bleeding complications of 19.5%, including minor (6.1%) and major bleeding (5.3%) in non-elderly patients undergoing ultrasound-assisted, catheter-directed thrombolysis. 12 Given the potential impact of bleeding risks on patient outcomes, accurately identifying patients at higher risk of bleeding who might benefit from advanced therapies is essential for optimizing care. The study adds to the literature by highlighting the concept of “false clinical stability” in normotensive patients, urging a redefinition of SBP thresholds and the development of more comprehensive hemodynamic instability equivalents beyond SBP alone. It supports earlier findings that SBP alone has limitations and emphasizes the need to incorporate additional factors (e.g., diastolic BP, cardiac index, RVD) to achieve more accurate risk stratification.
Exploratory analysis
Our findings suggest that clinical decision-making for advanced treatment in normotensive PE patients may be based on precisely defined impending clinical deterioration factors 12 (borderline SBP, RVD by echocardiogram, tachycardia, respiratory distress, and sustained hypoxemia; Supplemental Table 2). Interestingly, the post hoc analysis from the PEITHO trial also identified that borderline BP (⩽110 mmHg), tachypnea (>20 breaths/min), oncologic patients, and chronic heart failure were clinical variables associated with all-cause mortality, hemodynamic collapse, and recurrent PE in this population.11,22 In those with at least two of the above criteria under anticoagulation alone, adverse events occurred in 20%. 22 Furthermore, in a post hoc analysis of two PE registries, the rate of an unfavorable outcome was higher in patients with a heart rate ⩾100 bpm (7.6%) and ⩾110 bpm (8.3%) than those with a heart rate <100 bpm (3.0%). 22 The Cardiovascular and Interventional Radiological Society of Europe’s working group recently proposed several significant clinical, imaging, and laboratory severity indicators for intermediate- to high-risk PE. 16 The factors identified in this study as impending clinical deterioration (Table 1 and Supplemental Table 2) are similar to the severity indicators proposed by the European working group. This exploratory analysis aligns with the European working group proposals, 26 previous findings from our exploratory analyses,10,57 and the FLASH registry score, 53 suggesting that it may be valuable to investigate whether specific phenotypes within submassive or intermediate- to high-risk PE patients are associated with a greater likelihood of benefiting from advanced treatment. This exploratory analysis only generates hypotheses; it is not confirmatory.
Limitations
The present study has the following limitations: First, the review is primarily descriptive and does not compare outcomes between advanced therapy and anticoagulation alone. The main source of patients is randomized controlled trials and cohort studies, so the statistical value added by the reports and case series is unclear. We excluded studies of patients treated with endovascular or surgical thrombectomy, so the results apply only to patients undergoing systemic or catheter-directed thrombolysis. Likewise, we excluded thrombolytic drug combinations and different thrombolysis modalities within the same study to minimize biases and confounders in the quantitative analysis, which does not preclude the potential loss of information. We did not include lactate measurements. Although the primary adverse event of advanced treatment is major bleeding, there is a wide range in bleeding rates due to small-sample studies, various definitions of major bleeding, a heterogeneous patient population, variable adjunctive treatments, and the unknown timing of bleeding complications. 17 In addition, there is an inconsistency in the definitions of major bleeding across different research studies. While patient characteristics and clinical presentation symptoms appear to correlate with adverse outcomes, no outcome studies confirm that patients with any combination of impending clinical deterioration factors, significant clinical, imaging, and laboratory severity indicators, or a FLASH score indicating an early adverse outcome may clinically benefit from upfront reperfusion therapy. 53 Another limitation of the study is that the design did not include an analysis of the relationship between BP readings, mortality, bleeding, and recurrence. Post-study registration may also have introduced bias into our results, and this is another limitation.
Conclusion
This systematic review shows that the SBP range for advanced treatment (129.2-125.8 mmHg) aligns with previous clinical models. However, it is essential to recognize that while these SBP ranges may suggest practice variation, causality or definitive clinical instability cannot be inferred from aggregate data without patient-level outcomes. Outside of randomized controlled trials, clinical decision-making regarding advanced treatment may not fully align with international recommendations in real-world scenarios. In addition, randomized trials targeting patients with SBP 110–120 mmHg are required to determine the net clinical benefit of thrombolysis versus anticoagulation.
Supplemental Material
sj-docx-2-tak-10.1177_17539447251414584 – Supplemental material for Systolic blood pressure range in patients with intermediate- to high-risk pulmonary embolism undergoing advanced treatment: a systematic review
Supplemental material, sj-docx-2-tak-10.1177_17539447251414584 for Systolic blood pressure range in patients with intermediate- to high-risk pulmonary embolism undergoing advanced treatment: a systematic review by Hector Lopez-de la Garza, Carlos Jerjes-Sanchez, Victor E. Lozano-Corres, Christian Camacho Mondragon, Maria Fernanda Reyes-Chavez, Ricardo J. Estrada-Mendizabal, Jose Max Narvaez-Paliza, Victor Baylo Valdez and Cecilia Gocher Janet in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-pdf-1-tak-10.1177_17539447251414584 – Supplemental material for Systolic blood pressure range in patients with intermediate- to high-risk pulmonary embolism undergoing advanced treatment: a systematic review
Supplemental material, sj-pdf-1-tak-10.1177_17539447251414584 for Systolic blood pressure range in patients with intermediate- to high-risk pulmonary embolism undergoing advanced treatment: a systematic review by Hector Lopez-de la Garza, Carlos Jerjes-Sanchez, Victor E. Lozano-Corres, Christian Camacho Mondragon, Maria Fernanda Reyes-Chavez, Ricardo J. Estrada-Mendizabal, Jose Max Narvaez-Paliza, Victor Baylo Valdez and Cecilia Gocher Janet in Therapeutic Advances in Cardiovascular Disease
Footnotes
Acknowledgements
The results presented in this article are the views and conclusions of the authors and do not necessarily represent the views of the institutions to which they are affiliated or any other subject involved in the publication. We do not endorse any product or service that could be referred to in this article.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
