Abstract
Carotid blowout syndrome (CBS) is a potentially fatal condition requiring prompt diagnosis and intervention. CBS primarily affects patients with a history of surgery or irradiation for head and neck malignancies. In this report, we describe a case of CBS in which the carotid artery ruptured 2 months after carotid artery stenting in a patient with a previous history of surgery and irradiation for pharyngeal cancer. Although the distance between the orifice of the common carotid artery and the ruptured site was short, the “no-touch” technique—using the Newton-shaped stiff inner catheter (Newton-T®, Medikit) during the guiding catheter navigation—and snare fixation enabled a stable procedure. Two self-expanding covered stents (GORE® VIABAHN®, Gore) covered the ruptured site. Despite adverse conditions, the combination of these techniques led to effective management of impending CBS, a potentially fatal disease. This case report highlights the feasibility of covered stents for CBS and the techniques used for guiding catheter navigation and stabilization throughout the procedure.
Plain language summary
Carotid blowout syndrome (CBS) is a potentially fatal carotid artery rupture. In this report, we experienced a rare complication of CBS 2 months after carotid artery stenting. Considering the anatomic difficulty in navigating the guiding catheter, the “no-touch” technique using the catheter with the dedicated shape and stiffness and snare fixation were employed. Two self-expanding covered stents covered the ruptured site, and the patient recovered without neurological complications. This case report highlights the feasibility of covered stents for CBS and the techniques used for guiding catheter navigation and stabilization throughout the procedure.
Introduction
Carotid blowout syndrome (CBS) is a life-threatening condition of carotid artery rupture that typically occurs as a late complication after surgery or radiotherapy for neck cancer. 1 Conventional treatments, including surgical ligation or embolization of the carotid artery, are sometimes unsuitable due to impaired wound healing or insufficient collateral circulation. We describe a case of impending CBS in which the proximal end of a stent ruptured 2 months after carotid artery stenting (CAS), which was successfully treated with covered stents. Although the distance between the orifice of the common carotid artery (CCA) and the aneurysmal lesion was short, the combination of the “no-touch” technique 2 during guiding catheter (GC) navigation and snare fixation enabled a safe and stable procedure.
Case presentation
A man in his 80s with a permanent tracheal fistula after surgery and radiotherapy for pharyngeal cancer 25 years earlier was brought to our institution, presenting with right-sided hemiparesis. Multiple cortical infarctions on the left hemisphere and irregular plaques were observed in the ipsilateral CCA. The CCA lesion was treated with CAS (CASPER-RX® 10 × 30 mm, Terumo, Tokyo, Japan). Dual antiplatelet therapy (DAPT) with aspirin and clopidogrel was administered pre-procedure and continued post-procedure. Two months after CAS, the patient was hospitalized for sepsis. On admission, a painful pulsatile mass in the left neck was noted along with fever. Computed tomography angiography (CTA) revealed a 63-mm pseudoaneurysm extending to the proximal half of the CCA, including the proximal edge of the stent. The patient presented with impending CBS with oozing blood on the neck surface (Figure 1). Blood culture identified Staphylococcus aureus. Since the patient was in poor condition due to sepsis and the anterior and posterior communicating arteries were not visualized on CTA, we decided to reconstruct the affected CCA with covered stents.
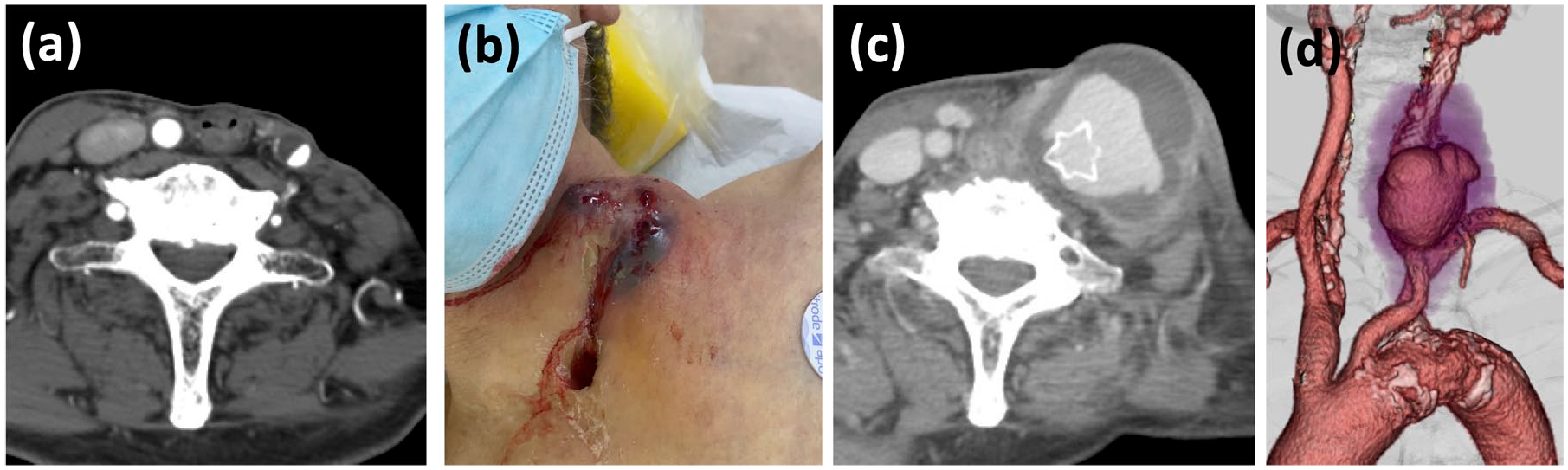
(a) Axial image of the left common carotid artery before carotid artery stenting showing decreased surrounding tissue due to previous surgery. (b) Pulsatile mass in the left neck with oozing blood. (c) Axial and (d) 3D-rendered images from computed tomography angiography showing a pseudoaneurysm at the proximal edge of the stent. Note the opened flare-end of the CASPER-RX at the ruptured site (c).
Endovascular procedure 1
Under general anesthesia, a 9-Fr sheath was placed in the groin. Since the pseudoaneurysm was located at a short distance from the CCA orifice, and the patient had a tortuous type III aortic arch according to Madhwal’s definition, 3 along with a steep angle between the aortic arch and the left CCA, difficulty was anticipated in navigating the GC to the appropriate position without shifting the ruptured vessel. Therefore, the “no-touch” technique with a Newton-T® catheter (Medikit, Tokyo, Japan) was employed to navigate a 7-Fr Flexor Shuttle Sheath® (COOK Medical, Bloomington, IN, USA) to the proximal CCA. Briefly, the primary curve of a 4-Fr Newton-shaped stiff inner catheter contacts the lesser curvature of the aortic arch, and this contact, along with the catheter’s stiff properties, buffers the kickback force and enables the GC to advance to the appropriate position without touching the lesion (Figure 2). 2 To prevent the GC from slipping down into the aorta during stent-graft navigation, a snare introduced via the right brachial artery stabilized the GC. Systemic heparinization was initiated at this stage to prevent uncontrollable bleeding from the ruptured vessel. The first self-expanding covered stent (GORE® VIABAHN®, 8 mm × 10 cm, Gore, Flagstaff, AZ, USA) was navigated to the CCA over a 0.014-inch wire and deployed to seal the pseudoaneurysm. To address the potential risk of vessel injury progression from inflammation, a second covered stent (GORE VIABAHN, 8 mm × 5 cm) was deployed distally with adequate overlap. Balloon touch-ups with an 8-mm balloon at the distal and proximal edges ensured proper sealing, resulting in no endoleak (Figure 3). Since the patient was on DAPT after CAS, no additional antithrombotic treatment was administered.

(a) Newton-T catheter. (b–f) Schematic diagram of the procedure. (b) The GC was snared within the right subclavian artery (arrowhead). (c) The GC was then pulled back to the aorta. (d, e) The “no-touch” technique: a 4-Fr Newton-T was engaged in the left common carotid artery. The primary curve of the Newton-T contacted the lesser curvature of the aortic arch (arrow), buffering the kickback force. The GC was advanced to the target position proximal to the aneurysm without touching the lesion. (f) With firm stabilization by the snare, a covered stent was deployed to seal the pseudoaneurysm.

Images from endovascular procedure 1. (a) The GC was secured with a snare (arrowhead). (b, c) The “no-touch” technique: the primary curve of the Newton-T contacted the aortic wall, absorbing kickback force during GC navigation (arrow). The GC was advanced to the CCA without touching the lesion, and a snare stabilized the GC to prevent slipping down during stent delivery. (d–f) Angiographic sequence: initial angiography (d), covered stent deployment (e), and final angiography (f), showing successful treatment of the ruptured vessel with two VIABAHNs with no endoleak.
Clinical course
The patient recovered from sepsis without neurological complications. Sepsis was initially treated empirically with meropenem and vancomycin, and subsequently with cefazolin and levofloxacin based on the sensitivity results, leading to resolution. The patient underwent myocutaneous flap surgery in the following month. A follow-up X-ray at 4 months revealed an opening of the distal flare-end of the CASPER-RX, indicating the vessel wall disruption (Figure 4). The distance between the opened flare-end and the distal tip of the VIABAHN was minimal, necessitating additional treatment with a covered stent extension.

(a) Computed tomography angiography after the first procedure showing vessel reconstruction. (b, c) Skin erosion treated with myocutaneous flap surgery. The protruding flare-end (arrowhead) was removed to prevent damage to the myocutaneous graft. (d) Follow-up X-ray showing the distal flare-end of CASPER-RX opening, indicating vessel disruption (arrow). (e, f) Endovascular procedure 2: (e) the external carotid artery was coiled, and an additional VIABAHN was deployed distally to the internal carotid artery. (f) The carotid artery was successfully reconstructed.
Endovascular procedure 2
Under local anesthesia, a 9-Fr sheath was placed in the groin, and a 7-Fr Flexor Shuttle Sheath was navigated to the mid-CCA with support from a 6-Fr inner catheter and a half-stiff guidewire advanced into the external carotid artery. The external carotid artery was obliterated with seven detachable coils using the double-catheter technique to avoid type II endoleak. Then, a GORE VIABAHN 8 mm × 5 cm was deployed distally and touched up with balloons of 8 mm proximally and 5 mm distally. The secured vessel extended to include the internal carotid artery, and no endoleak was observed (Figure 4).
Outcome and follow-up
The patient was transferred to a rehabilitation facility for disuse syndrome and remained free of neurological and vascular complications. However, the patient died of a pressure ulcer infection, unrelated to CBS, 7 months after the first procedure.
Discussion
CBS is a life-threatening condition with approximately 60% mortality and 40% neurological complication rates without proper treatment. 1 Vascular fragility, loss of surrounding tissues, impaired repair function, and susceptibility to infection after surgery and radiotherapy for neck cancer are typical causes of CBS. CBS, as a late complication of CAS, is rarely encountered in clinical practice. We speculate that systemic infection spreading to the stent site and mechanical stress from the stent edge on the movable proximal CCA, along with the aforementioned factors, may have contributed to this failure.
As for treatment options, given the invasiveness of direct vascular surgery and the fragility of surrounding tissues, less invasive endovascular treatments are preferred. 4 Endovascular treatments include embolization of the carotid artery and reconstruction with covered stents.5,6 In cases of intolerance to embolization or hemodynamic instability, stent therapy can be used as the treatment of choice. The periprocedural complication and late rebleeding rates for covered stents have been reported to be 2.5%–11.1% and 31.9%–38.9%, respectively, whereas those of embolization were 10.3%–10.5% and 9.1%–13.2%,4,7 making stent therapy the preferred option for this emergent, life-threatening condition, particularly in cases with poor collateral circulation. In this case, the aplasia of the anterior and posterior communicating arteries on CTA indicated a “very high risk” for carotid embolization based on Lee et al.’s criteria, 8 leading us to select stent therapy.
GORE VIABAHN for vascular repair is a self-expandable stent that adopts an expanded polytetrafluoroethylene membrane with a heparin coating, and is primarily used for peripheral artery injuries. It is now favored in the cervical region with the advantage of a lower profile and less friction, with a technical success rate of 100% and a rebleeding rate of 7% at 12 months. 9
The use of stent-grafts in infected sites raises significant concern. To assess the extent of infection involving adjacent structures, imaging modalities commonly include CTA, with ultrasound and magnetic resonance imaging as additional options depending on clinical context. In our case, ultrasound was unsuitable due to the presence of a pre-procedural oozing pulsatile mass (Figure 1(b)) and post-procedural skin defect (Figure 4(b)). While the pre-procedural infection extent was assessed with CTA, post-procedural infection was primarily evaluated through direct observation of the skin condition. For subsequent antibiotic management, the European Society for Vascular Surgery guidelines on stent-graft infection—which address similar scenarios—recommend long-term administration, typically for 4–6 weeks, or longer when necessary. 10
As for technical issues, navigating the GC without contacting the aneurysm and stabilizing the GC while advancing the stent system are challenging, especially when the aneurysm is located at the proximal CCA. We employed the “no-touch” technique using Newton-T, a stiff Newton-shaped inner catheter, which absorbed kickback force by contacting the lesser curvature of the aortic arch. 2 This approach is particularly useful for tortuous anatomy or proximal CCA lesions. In addition, a snare navigated from the brachial artery fixed the GC, providing firm stabilization for the advancement of the stent system.
Coverage range is a crucial factor in repairing a disrupted vessel. Considering the potential for the inflammatory vessel damage to extend, the secured vessel should extend beyond the apparent ruptured site. In the first procedure of this case, the first covered stent isolated the pseudoaneurysm from the bloodstream, whereas the second stent was placed distally to prevent future vessel disruption. Although the vessel disruption did extend to the distal edge of the CASPER-RX, the distally deployed second VIABAHN prevented late rebleeding.
The optimal peri- and post-procedural antithrombotic management for covered stent treatment remains a topic of debate. Systemic heparinization is typically administered immediately after sheath insertion. However, in this case, it was delayed until the GC was positioned, due to concerns about whether GC navigation could be completed without displacing the ruptured vessel and causing massive, uncontrolled bleeding. For peripheral use of covered stents, DAPT with aspirin and thienopyridine is recommended to prevent in-stent thrombosis. 11 In our case, the patient was already on DAPT per the standard post-procedural regimen for CAS. In treating patients without prior antiplatelet therapy in emergency settings, a loading dose of aspirin and thienopyridine should be considered, based on individual bleeding risk.
Despite several adverse conditions, the combination of devices, techniques, and peri- and post-procedural antithrombotic management resulted in the successful treatment of impending CBS, a potentially fatal disease.
Conclusion
We report a case of impending CBS 2 months after CAS for an irradiated and dissected neck, successfully treated with covered stents. When patients cannot tolerate embolization of the affected arteries, reconstruction with covered stents may be a feasible option for treating CBS. When lesions are located in the low position of CCA, the combination of the “no-touch” technique and snare fixation can be a useful option.
