Abstract
Carotid artery stenting (CAS) is an alternative strategy to carotid endarterectomy for the prevention of ischemic stroke. Acute carotid stent thrombosis (ACST) is an extremely rare but devastating complication of CAS. Although cases of successful recanalization have been reported, there is still a lack of experience regarding the choice of ACST treatment methods and the timing of such treatments, especially when patients are confirmed CYP2C19*2 heterozygotes. Here, we report a case of successful revascularization after ACST in a patient with CYP2C19*2 heterozygosity. We also review the literature and discuss appropriate treatment strategies for this devastating and rare event.
Keywords
Introduction
Carotid artery stenting (CAS) is an alternative strategy to carotid endarterectomy for the prevention of primary or secondary ischemic stroke in selected patients. 1 Acute carotid stent thrombosis (ACST) is an extremely rare event, with an incidence rate of 0.5% to 0.8%, 2 but it is accompanied by devastating complications. 2 Although cases of successful recanalization have been reported,3,4 there is still a lack of experience regarding the choice and timing of ACST treatment methods. Here, we report the successful revascularization of ACST in a patient with CYP2C19*2 heterozygosity that was confirmed by genetic testing. We also review the literature and discuss appropriate treatment strategies for this rare event.
Case report
The study protocol was approved by the Ethics Committees of the Fifth People’s Hospital of Chengdu, and the patient provided written consent to the publication of this report.
A 69-year-old man was admitted to the Fifth People’s Hospital of Chengdu (Chengdu, China) because of right limb weakness, right central facial paralysis, and hemianalgesia for 2 days. The right limb weakness was aggravated and barylalia was detected for half a day. The patient had a 4-year history of hypertension caused by poor blood pressure control. After hospitalization, magnetic resonance imaging showed multiple infarcts in the internal border zone, in a rosary-like pattern along the left centrum semiovale. Computed tomography (CT) angiography revealed severe stenosis at the beginning of the left internal carotid artery (Figure 1). In addition, transcranial Doppler (TCD) showed increased blood flow in the middle cerebral artery. The patient’s platelet count was 107 × 109/L and his coagulation function was normal. Multi-disciplinary consultation occurred, including experts in cardiology, respiratory medicine, neurosurgery, and anesthesiology. On the basis of the patient’s poor lung function, anesthesiologists indicated that the risk of general anesthesia was relatively high, and left carotid stenting was suggested rather than endarterectomy for secondary prevention. Before the surgery, the patient was administered aspirin (100 mg), clopidogrel (75 mg), and atorvastatin (20 mg) for 5 days. The patient’s National Institute of Health Stroke Scale (NIHSS) score was 5.

Preoperative examination of the patient. (a) Magnetic resonance imaging showed multiple internal border zone infarcts in a rosary-like pattern along the left centrum semiovale. (b, c) Computed tomography angiography showed severe stenosis at the beginning of the left internal carotid artery (LICA). (d) Doppler ultrasonography showed the formation of atherosclerotic plaque in the LICA, and that blood flow velocity was accelerated in the lumen
The CAS was performed under local anesthesia. Heparin was administered intravenously at a dose of 5000 U (100 U/kg) bolus immediately after the femoral artery puncture. Angiography revealed 90% stenosis in the left carotid artery using the North American Symptomatic Carotid Endarterectomy Trial criteria. First, we positioned a distal protection device (SpiderFX; eV3 Inc., Plymouth, MN, USA) to prevent emboli. Next, the stenosis of the left internal carotid artery was predilated with a 5- × 30-mm balloon (Viatrac 14 Plus; Abbott Vascular, Temecula, CA, USA). Finally, a 7- × 40-mm self-expandable carotid stent (Wallstent; Boston Scientific, Marlborough, MA, USA) was placed using a 0.36-mm guidewire and an 8F guide catheter (Boston Scientific). The CAS was performed without any complications (Figure 2), and the patient did not show any symptoms during the CAS procedure.

Angiography before and after carotid artery stenting. (a) Angiography revealed 90% stenosis in the left carotid artery according to the North American Symptomatic Carotid Endarterectomy Trial criteria. (b) Carotid artery stenting was performed without any complications, and stenosis of the left internal carotid artery was fully resolved.
After the surgery, the patient continued to take aspirin (100 mg), clopidogrel (75 mg), and atorvastatin (20 mg). Five days after the surgery, the patient became lethargic, with gaze to the left side, motor aphasia, and right hemiplegia; the patient’s NIHSS score was 18. Thirty minutes later, CT angiography showed acute stent thrombosis of the left internal carotid artery (Figure 3).
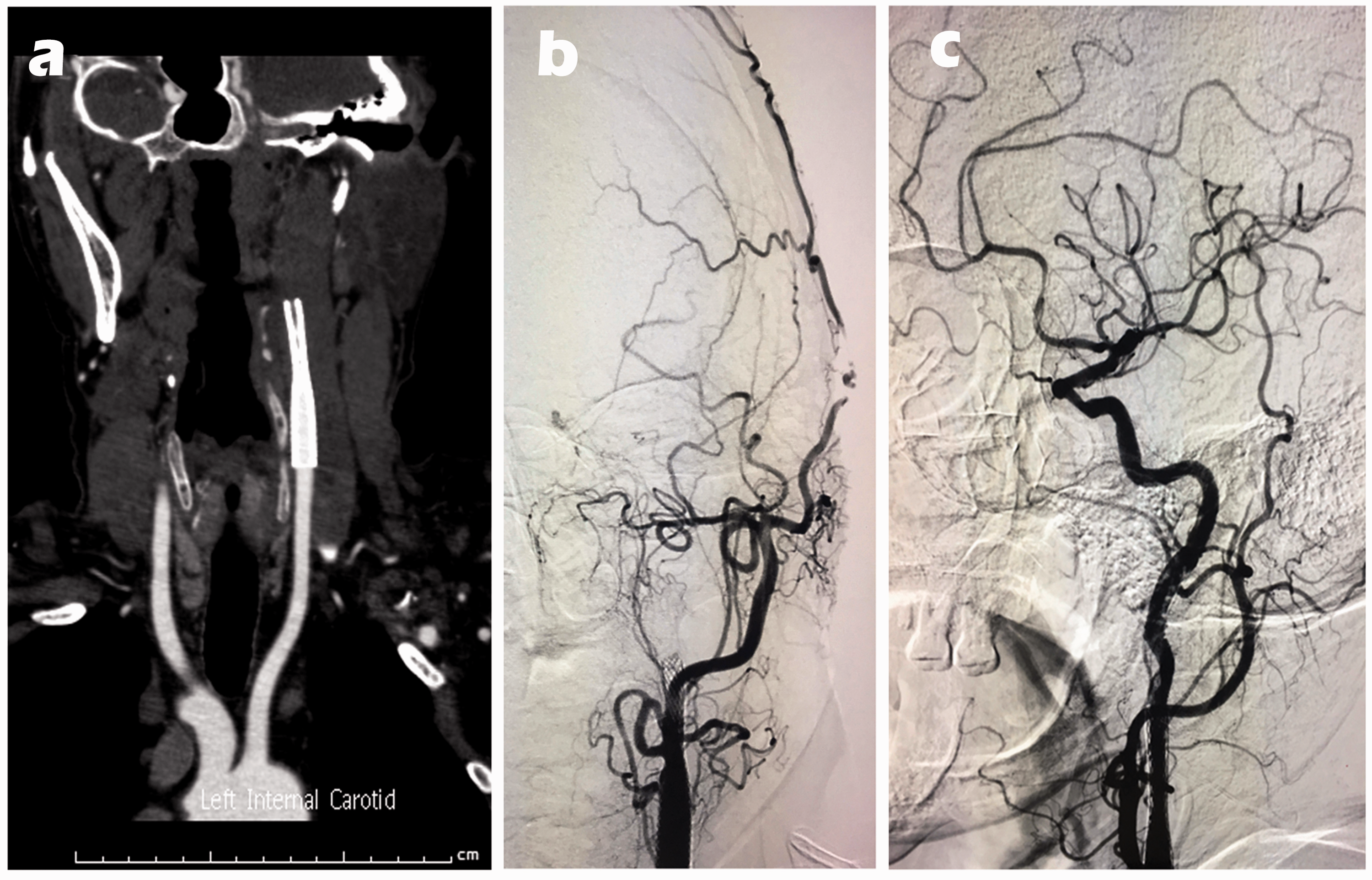
Acute carotid stent thrombosis removal in the patient. (a) At 5 days after surgery, computed tomography angiography showed acute stent thrombosis of the left internal carotid artery. (b) Angiography confirmed acute stent thrombosis of the left internal carotid artery, however, there was no blood flow at the distal end of the stent. (c) Thrombus aspiration was performed on the patient with acute carotid stent thrombosis.
We immediately performed thrombus aspiration via the right percutaneous transfemoral access under local anesthesia. After the 8F guide catheter (Boston Scientific) was placed, angiography showed acute thrombosis at the proximal end of the stent, without forward blood flow. A Synchro2 Microwire (Stryker, Kalamazoo, MI, USA) with a Rebar 18 microcatheter (ev3 Inc.) showed thrombosis in the M1 segment. We preferentially opened the middle cerebral artery using a 4- × 20-mm Solitaire FR stent (ev3 Inc.) with an accompanying thrombus that was approximately 2 to 3 cm long. Next, a 6F Navien (ev3 Inc.) was used to enter the stent thrombus for thrombus aspiration, and a thrombus of approximately 4 cm in length was aspirated (Figure 4). After the angiography, blood flow in the stent was partially restored; however, the forward blood flow remained very slow. A Spider distal protection device (ev3 Inc.) was then positioned, and a 5- × 30-mm balloon (Boston Scientific) was expanded twice by 12 atm. Angiography revealed a marked improvement in anterior blood flow, but there was some thrombus in the stent. Tirofiban (10 mL) was administered through a Rebar 18 microcatheter (ev3 Inc.). After this treatment, angiography revealed the complete disappearance of the thrombus and complete recovery of the forward blood flow (Figure 3). After surgery, the patient’s NIHSS score was 12.

Thrombus that was aspirated during surgery.
Postoperative CT showed severe cerebral edema and contrast agent leakage without hemorrhage. At 2 days after the thrombus aspiration, CT angiography revealed complete recanalization of the stent, and showed that most of the contrast agent was absorbed. Ten days after the second surgery, the results of the patient’s CYP2C19 genotype were received. The patient was a CYP2C19*2 heterozygote (CYP2C19*1/*2). The patient was discharged from hospital taking aspirin, clopidogrel (triple dose), and atorvastatin. After 6 months of follow-up, the patient’s NIHSS score was 3 and his modified Rankin scale score was 1. Furthermore, there was no in-stent restenosis observed in the CT angiography at that time (Figure 5).

Radiographic changes in the patient after thrombus aspiration. (a) Postoperative computed tomography (CT) showed contrast agent leakage. (b, c) At 2 days after thrombus aspiration, the CT angiography showed complete recanalization of the stent, and most of the contrast agent had been absorbed. (d) There was no stent restenosis in CT angiography at the 6-month follow-up.
Discussion
It has been reported that the incidence of in-stent restenosis after CAS ranges from 3% to 16.6%, and that the majority of patients are asymptomatic. 5 ACST is extremely rare, but has devastating complications, and occurs within 30 days after CAS. 2 We reviewed articles related to ACST that were published in English, and have listed their details in Table 1.4,6–25
Acute stent thrombosis after carotid artery stenting.

A, asymptomatic; ACST, acute carotid stent thrombosis; F, female; ICA, internal carotid artery; LCCA, left common carotid artery; LICA, left internal carotid artery; M, male; NM, not mentioned; RICA, right internal carotid artery; rt-PA, recombinant tissue plasminogen activator; S, symptomatic; STA-MCA, superficial temporal artery-middle cerebral artery.
There are several causes of ACST, such as inadequate antiplatelet therapy, early discontinuation of antiplatelet therapy, clopidogrel resistance, a hypercoagulable state, soft plaque and its protrusion, local vessel dissection, vasospasm, and intimal injury.18,25,26 A number of reasons, including the cause of CAS (e.g., stent underexpansion, in which the stent does not fully adhere to the blood vessel, 25 or balloon burst 15 ), plaque protrusion, 4 and vasospasm, may lead to immediate ACST. These can be resolved in a timely manner without any damage to the nervous system. In contrast, other reasons, such as inadequate antiplatelet therapy,7,8,12 early discontinuation of antiplatelet therapy, 11 clopidogrel resistance, and a hypercoagulable state, 18 may cause ACST a few days after a CAS procedure. This may eventually cause death or severe paralysis. 7
In our patient, it may be that more than one factor caused ACST. After being affected by ACST, we tested the CYP2C19 status of our patient, which was CYP2C19*1/*2. This is one of the genetic polymorphisms that cause clopidogrel resistance. Although not all patients who are CYP2C19*2 heterozygotes are low responders for clopidogrel, in the absence of a hypercoagulable state and because antiplatelet therapy was adequate in our patient, we speculate that clopidogrel resistance may have been one of the important causes of his ACST. Moreover, we did not perform post-dilation in this patient; thus, stent underexpansion may be another reason for his ACST. 5
We performed thrombus aspiration immediately after the patient was affected by ACST. Because ACST is a rare complication, its optimal treatment remains unknown. The treatment of ACST should be based on a combination of thrombogenesis, time (intraoperative, early, or late after surgery), the severity of neurological deficits, and the area of the infarction. For cases with severe clinical deterioration, ACST treatment should be relatively positive, with a goal of rapid revascularization to avoid serious consequences. 12 The solution for ACST may be multiplex, including drugs,4,6,14 thrombolysis,7,8,15,16,23 thrombolytic therapy combined with anticoagulation or antiplatelet therapy, 4 and surgical therapy including thromboendarterectomy, 11 mechanical thrombolysis, and thrombus aspiration.12,20 The use of these two methods (thromboendarterectomy and endovascular treatment) flexibly has also been reported. 24
Our patient was discharged from hospital when he was taking aspirin, clopidogrel (triple dose), and atorvastatin. Previous studies have reported that tripling the maintenance dose of clopidogrel, to 225 mg daily, in heterozygote carriers of CYP2C19*2 achieves levels of platelet reactivity similar to those seen with the standard 75 mg dose in noncarriers. 27 In addition, at the 6-month follow-up, there was no in-stent restenosis in CT angiography in our patient. This finding further supports the idea that a triple dose of clopidogrel is effective in CYP2C19*2 heterozygotes. Another option to consider may be the use of antiplatelets such as prasugrel and ticagrelor, which are less influenced by CYP2C19, rather than using a triple dose of clopidogrel.
Conclusion
ACST is an extremely rare event, but it can be a fatal complication after CAS. In our patient, it may be that more than one factor caused ACST. The treatment of ACST after CAS must be undertaken immediately to restore blood flow and avoid major adverse neurological events. The present case report helps to illuminate the available treatment strategies, rather than providing general therapeutic recommendations.
