Abstract
Infective endocarditis (IE) is an increasingly recognized condition with high morbidity. Patients with atypical symptoms, culture-negative infections, and prosthetic cardiac devices and implants represent challenging populations to evaluate and manage. Recent major society guidelines have recommended the appropriate incorporation of multimodality imaging in the evaluation of these more complex IE cases. This article draws on the available literature regarding the different cardiac imaging modalities and discusses the role of multimodality imaging in IE.
Keywords
Introduction
Infective endocarditis (IE) is a serious condition associated with significant morbidity and high 1-year mortality rates, estimated to be around 30%, despite evolving diagnostic and therapeutic strategies.1,2 The incidence of IE has also been rising since the 21st century. Over the past two decades, there has been a reported increase in the incidence of IE in North America and a twofold increase across Europe.3 –5 This is partly owing to a growing, at-risk, and aging population and an increase in prosthetic valve and cardiac device implantation. 6 The improved detection of IE is also potentially playing a role in this rising incidence due to an increase in the use of echocardiography across hospital centers and the introduction of more advanced imaging techniques such as dedicated cardiac computed tomography (CCT), nuclear imaging, and cardiac magnetic resonance imaging (MRI). 4 The utility of multimodality imaging has in fact been highlighted in the recently updated American College of Cardiology (ACC)/American Heart Association (AHA) and European Society of Cardiology (ESC) guidelines6,7 and continues to garner greater recognition in clinical practice. In this review, we focus on the strengths and limitations of these imaging modalities as they pertain to the evaluation of IE in both native and prosthetic valves. We also present a focused section on cardiac implantable device related IE and infection of surgical grafts with relevant illustrative cases. All the cases presented in this review are original and unique.
Epidemiology
In a nationwide study using the National Inpatient Sample, it was determined that hospitalizations for IE reached approximately 600,000 between 2003 and 2016 in the United States. 8 The same study noted that the incidence of IE hospitalizations had increased significantly over the years from 34,488 in 2003 to 54,405 in 2016. 8 A similar trend was observed in a systemic review by Talha et al. of IE cases in Europe, noting an increase in yearly incidence of IE of 0.27 cases per 100,000 people, effectively resulting in a doubling of IE hospitalizations in Europe over the past two decades. 4 Mortality associated with IE remains high, with an estimated in-hospital mortality of 14%–22% and 1-year mortality up to 40%.9 –13 A large analysis using the Centers for Disease Control and Prevention’s Wide-Ranging Online Data for Epidemiologic Research database from 1999 to 2020 found that while mortality rates have seen a significant decline for those over the age of 55, there was a notable acceleration in IE-associated morality in the younger 25- to 45-year age group. This increase coincided with specific regions of the United States including Kentucky, Tennessee, and West Virginia, which have seen a notable rise in intravenous drug use and the opioid epidemic.5,10
The incidence of native valve infective endocarditis (NVIE) is approximately 2–10 cases per 100,000 person-years.1,14 Prosthetic valve procedures including transcatheter aortic valve replacement (TAVR) have significantly increased over the last decade,15,16 which has led to an increase in the incidence of prosthetic valve infective endocarditis (PVIE), now accounting for approximately 20%–30% of all IE cases. 15 The incidence of PVIE is approximately 1%–4% in the first year following surgery with an estimated incidence of 1% per year thereafter. 17 The crude rate of IE is 0.43 per 100 patients for mechanical valves and 0.60 per 100 patients for bioprosthetic valves within 1–5 years following aortic valve replacement. 18
While cardiovascular implantable electronic devices (CIEDs) such as pacemakers and defibrillators are widely used, the exact epidemiology and natural history of CIED-related infective endocarditis (CIED-IE) is still not well-defined, and evidence is mainly driven by sporadic case reports and case series.1,2 A study from the EURO-ENDO registry demonstrated that diagnosis of CIED-IE was dependent on imaging findings in at least 85% of cases and that almost 20% could not reach definite diagnostic criteria, with underutilization of multimodality imaging in these cases. 19 Prognosis for CIED-IE also remains poor, 19 similar to other cardiac prosthetic infections including aortic graft and ventricular assist device infections.20 –22
Clinical manifestations
IE has a wide spectrum of clinical manifestations.23,24 Fever is the most common presenting symptom reported in approximately 70%–80% of cases.23–25 A new murmur and worsening of a known murmur are also common and were reported in 48%–70% of cases.23,24 “Classic” examination manifestations such as splinter hemorrhages, Janeway lesions, and Roth’s spots are much less frequent in routine clinical practice, in the order of 0.5%–5%.6,24 Systemic complications including sepsis, meningitis, heart failure, pulmonary emboli, stroke, acute peripheral arterial occlusion, and renal failure are all well described in IE.23,24
Diagnosis
The diagnosis of IE has classically relied on the modified Duke criteria, first established in the year 2000. These criteria are based on microbiology findings, clinical features with an emphasis on evidence of embolic events by physical examination, and imaging findings via echocardiography.6,26,27 Despite the widespread use of the modified Duke criteria, major limitations have surfaced, particularly in the presence of culture-negative organisms28,29 and cardiac prosthetic material that renders evaluation by echocardiography more challenging, with up to 30% of IE cases reported to be normal or inconclusive by transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE).6,30 For this reason, recent ESC guidelines in 2023 considered findings suggestive of IE in other imaging modalities (CCT, 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18FDG-PET)/CT, white blood cell single-photon emission CT/CT (WBC-SPECT/CT), and cerebral MRI) as major and minor criteria for the diagnosis of IE and its extracardiac manifestations, resulting in superior diagnostic performance compared to the traditional Duke criteria. 6 The 2023 Duke International Society of Cardiovascular Disease (ISCVD) criteria have proposed updates to the diagnostic criteria of IE. These updates included more imaging modalities (CCT and (18FDG-PET)/CT) as a new major clinical criterion. 30
2023 Duke-ISCVD criteria could be superior in detecting IE. Goehringer et al. performed a retrospective analysis to evaluate the sensitivity and specificity of the 2023 Duke-ISCVD criteria in detecting IE compared to the traditional modified Duke criteria and the 2015 ESC criteria. 2023 Duke-ISCVD criteria had higher sensitivity (97.6% vs 93.2% and 95%, p < 0.001), respectively, but had lower specificity on the other hand (46% vs 61.3% and 60.5%), respectively. 31 Table 1 summarizes the benefits and limitations of these different imaging modalities, with a review of the current literature below.
Comparison between different cardiac imaging modalities in their applications for infective endocarditis evaluation.

CCT, cardiac computed tomography; CIED-IE, cardiovascular implantable electronic device-related infective endocarditis; 18F-FDG-PET/CT, 18 fluorodeoxyglucose positron emission tomography/computed tomography; MRI, magnetic resonance imaging, NVIE, native valve infective endocarditis; PVIE, prosthetic valve infective endocarditis; TEE, transesophageal echocardiogram; TTE, transthoracic echocardiogram; WBC-SPECT/CT, white blood cell single-photon emission computed tomography/computed tomography.
Echocardiographic imaging in IE
Technical aspects, indications, and main findings
TTE is a widely available, affordable, noninvasive tool that can be performed rapidly and with no radiation exposure.31,32 Due to these features, TTE remains the cornerstone modality in the evaluation of endocarditis. Echocardiography is the first-line imaging modality for suspected IE per both the 2020 AHA/ACC and 2023 ESC guidelines.3,4 TTE should be also considered in all cases for further evaluation and prognostication, particularly, in patients with prosthetic valves or implanted devices.3,4
In addition to vegetation visualization, echocardiography can be used to identify infectious complications such as perivalvular extension of the infection, abscess or fistula formation, or prosthesis malfunction and dehiscence. It also plays an important role in quantification of valvular regurgitation and stenosis. 33 Echocardiography also serves a key role in the clinical evaluation of the patient and can provide important hemodynamic information, including stroke volume, biventricular function, filling pressures, and more. TEE can provide peri-operative assessment and assist in determining the optimal surgical approach and the feasibility of valve repair. 33
Sensitivity, specificity, and limitations
In a large meta-analysis of 16 studies, the sensitivity and specificity of TTE for IE detection of all types of IE were reported to be 61% and 94%, respectively. 34 Despite its many advantages, echocardiography, and TTE specifically, does have several important limitations. It is highly operator dependent and image quality can be significantly affected by patient factors that result in suboptimal imaging windows. Vegetations usually <5 mm can be missed on TTE such that early cases of IE may require repeat imaging 5–7 days after initial TTE or further evaluation with TEE. 6 The evaluation of PVIE presents a particular challenge in TTE, for which the sensitivity is as low as 30% compared to approximately 70% in NVIE.35,36,37 Figure 1 clearly highlights the challenge of imaging prosthetic material by TTE, even in the presence of a large vegetation. This is in part due to acoustic shadowing from the prosthesis thereby limiting visualization but also the manifestation of PVIE itself, which frequently involves the paravalvular space or part of the prosthesis without overt formation of vegetations, but rather thickening or irregularity around the prosthetic ring. TEE can be used in these scenarios and yields a very high sensitivity for diagnosing PVIE in a range of 86%–94% and specificity of 88%, as noted in a large series of patients with PVIE by Daniel et al. 38 Cardiac complications like abscess formation can likewise be difficult to detect on TTE but has reasonable detection in TEE; in a French study of 233 patients with IE by Choussat et al., sensitivity for abscess detection with TTE and TEE were found to be 36% and 80%, respectively. 39 3D TEE has also emerged as a helpful complementary tool. Notably 3D assessment tends to yield larger measurements of vegetation given its ability for volumetric reconstruction, which can have important implications for determining embolization risk and surgical timing. 40 A retrospective study by Montané et al. noted a significant sensitivity increase in IE detection with TEE from 85.7% in 2011 to 95.3% in 2019, thought to be partly due to the improved frame rate and resolution of modern TEE probes, as well as a significant increase of almost 27% in the use of 3D technology during that time. 41 In summary, TEE should be considered in most cases of IE when appropriate since it improves the diagnostic performance and detection of the lesions and associated complications.

Thirty-eight-year-old woman with a history of intravenous drug use and prior endocarditis requiring tricuspid valve replacement presents with 2 weeks of fevers and shortness of breath. Parasternal short-axis and four-chamber views on TTE (a, c) and TEE (b, d). A bioprosthetic tricuspid valve is seen by TTE but no vegetation can be clearly appreciated. In comparison, large mobile vegetation on the prosthetic tricuspid valve is clearly appreciated by TEE (arrow), with 3D reconstruction (e) yielding a vegetation size of 2.3 cm.
Even with the availability of modern TTE and TEE technology, certain populations remain difficult to image with echocardiography. Patients with TAVR-related PVIE, for instance, can present with atypical imaging findings that make applying the modified Duke Criteria for diagnosis more challenging.41,42 Valve dehiscence typically does not occur in this population as the valve is maintained in place via radial force rather than by sutures, and valve regurgitation from PVIE can be mistaken for postoperative paravalvular leak. 43 In a 55-patient study with TAVR PVIE, among whom 85% underwent TEE, vegetation was only noted in a quarter of patients, and almost a third had no overt imaging findings at all. Signs of TAVR PVIE are often more subtle and, if present, may show only leaflet thickening or increased valvular gradients. 44 Similarly, CIED-related IE represents a challenging population to evaluate; again, sensitivity with TEE is significantly higher than with TTE (90% vs 22%–43%), 45 but vegetations can be missed if they lie in extracardiac segments of the lead that cannot be visualized, and TEE cannot delineate infected vegetation from sterile mass or thrombus.46,47 In addition, challenging clinical conditions including IE affecting mitral annular calcification can be difficult to diagnose using echocardiography alone48,49 (Table 2).
Sensitivities and specificities of different imaging modalities in diagnosing different types of infective endocarditis.

CIED-IE, cardiac implanted electronic devices infective endocarditis; 18F-FDG PET/CT, 18F-Fluorodeoxyglucose positron emission tomography/computed tomography; LVAD, left ventricular assisted device; NVIE, native valve infective endocarditis; PVIE, prosthetic valve infective endocarditis; WBC-SPECT/CT, white blood cell single-photon emission computed tomography/computed tomography.
Summary
TTE is the first-line modality in suspected IE, and TEE should be considered in most cases. Considering TEE is particularly important in patients with PVIE, suspected abscess, and CIED-IE. 3D images should be acquired when performing the TEE due to its advantage in allowing multiplanar reconstruction leading to higher diagnostic sensitivity.
CCT in IE
Technical aspects
Cardiac CT has gained increasing attention over recent years in the work-up of IE due to its ability to provide both a detailed anatomical evaluation of the infection and important preoperative planning information, including defining coronary anatomy and the relationship between cardiac structures and the sternum. Specialized protocols are required for optimal diagnostic yield, including the use of multiphase electrocardiogram (ECG)-synchronized imaging, thin sections (ideally 0.75 mm or less), and appropriately timed contrast agents to highlight the anticipated infected sites.47,50
Indications according to different guidelines
Incremental developments in CCT have made it an important complementary tool to echocardiography, and this has been reflected in updated IE guidelines including the 2023 ESC and 2020 ACC/AHA guidelines.6,7 The former has in fact incorporated the presence of embolic events by imaging as a minor criterion, 6 while the latter has made a class 2A recommendation for the use of CCT as adjunctive imaging in IE. 7 The 2023 ESC and the 2023 Duke-ISCVID diagnostic criteria, on the other hand, consider as a major imaging criterion any findings typical of IE (i.e., vegetation, perforation, abscess, pseudoaneurysm).5,30
Sensitivity, specificity, and limitations
Given the rising importance of CCT in the evaluation of IE, a number of studies have sought to define the sensitivity of CCT, particularly as it compares to TEE. The evidence discussed below reveals that TEE is generally superior for the detection of vegetation, owing to its greater temporal resolution, while CCT can provide comparable or better diagnostic accuracy in the evaluation of paravalvular complications. Figure 2 highlights how these modalities can complement one another and provide different clinical information.

Fifty-four-year-old man with a history of bacterial endocarditis and subsequent valve homograft implantation presenting with 3 weeks of fatigue. Transesophageal echocardiography (mid-esophageal short-axis view) shows a homograft prosthetic aortic valve with a large pseudoaneurysm/abscess cavity from the left coronary sinus (a, *) with abnormal flow on color Doppler (b). A large mobile vegetation with multiple fronds (arrow) is also seen inside the abscess cavity. A gated cardiac CT further defines the consequences of the infection. Sagittal images during diastole (c) and systole (d) show dynamic compression (circle) of the left main coronary artery between the pulmonary artery and the pseudoaneurysm cavity. Axial view (e) more clearly shows the extension of the large pseudoaneurysm from the left coronary sinus communicating with the left ventricle along the lateral wall (*). The large vegetation is again seen by CT (arrow).
In a 75-patient 2018 study by Kim et al., the detection rate for vegetations was found to be higher in TEE compared to CCT (97.3% and 72.0%, respectively), noting that small vegetations under 10 mm were more often underdiagnosed with CCT compared to TEE (52.2% vs 94.4%). 51 Sims et al. published a retrospective review of 137 patients with IE and reported a sensitivity of 70% for vegetation detection using CCT when compared to surgical findings. 52 In a large 872-patient contemporary meta-analysis, a similar pattern was found, with a vegetation detection sensitivity of 96% and 85% for TEE and CCT, respectively, noting that in the PVIE subgroup, CCT in fact had higher specificity compared to TEE (94% vs 74%). 53
With regard to paravalvular complications, the same study above by Jain et al. found comparable diagnostic performance between CCT and TEE for detecting fistulae, paravalvular leaks, and prosthetic valve dehiscence, with a trend favoring CCT for detection of paravalvular complications and TEE for leaflet perforation. 53 Other studies found that CCT could be superior to TEE for the evaluation of paravalvular complications in certain clinical cases. Sifaoui et al. published a 68-patient study of left-sided PVIE and NVIE, and found CCT to be more sensitive than TEE in pseudoaneurysm detection (100% vs 67%). 54 In a 122-patient study with surgically proven IE, sensitivity and specificity for pseudoaneurysm/abscess detection were greater with CCT compared to TEE (81% vs 64% and 75% vs 33%, respectively). 55 Another retrospective study published in 2018 by Ouchi et al. found that CCT again offered superior pseudoaneurysm detection in a cohort of surgically proven IE. 56 Importantly, and especially relevant for clinical practice, several of the studies listed above have also found that the combination of CCT with TEE can significantly improve diagnostic yield, with an increased sensitivity of approximately 15% when combining imaging modalities.53,55,57 In addition to relatively lower temporal resolution resulting in poorer detection of small vegetations and leaflet perforation, CCT has several important limitations that clinicians must remember. Radiation exposure can be high, particularly in the retrospective, four-dimensional ECG-gated protocols generally used for IE. Several techniques can be used to minimize radiation like tube current reduction and iterative reconstruction; however, these measures can affect the assessment of mobile vegetation and cardiac valves. 47 Nephrotoxicity secondary to dye use is a concern, although typically more so for patients with estimated glomerular filtration rates under 30 mL/min/1.73 m. 47 Image acquisition can also be challenging in critically ill patients who may be tachycardic and unable to perform a breath-hold, making patient selection and heart rate control to ensure good diagnostic accuracy very important. 58 The technology of photon counting has led to improvement in spatial resolution in the diagnosis of IE. This is an advanced feature that is evolving and has an advantage over the traditional energy-integrating dual-source scanners. Scanners with the ability to perform photon counting have better spatial resolution which could expand the role of CCT in IE 58 (Table1).
Summary
CCT plays a major role in evaluating paravalvular complications, while TEE is superior in detecting vegetation. It also has higher sensitivity in detecting pseudoaneurysms and abscesses compared to TEE.
Nuclear imaging in IE
Technical aspects
The addition of hybrid nuclear medicine imaging modalities such as 18F-FDG PET/CT has been very valuable as a complementary tool in the evaluation of endocarditis. The basis of FDG-PET lies in the use of 18F-FDG, a radioactive analog of glucose, which preferentially enters cells with high glucose transporter membrane expression and therefore high metabolic activity. 59 The formation of biofilm is also common on prosthetic material and renders it more metabolically active. Understanding this fundamental principle is key, as it may explain why 18F-FDG PET/CT has been particularly effective in evaluating PVIE and infection of other synthetic materials, including CIED and infected aortic grafts, but less so for NVIE.
Indications according to different guidelines
Recently updated ESC guidelines now recommend the use of 18F-FDG PET/CT in the diagnostic algorithms of NVIE, PVIE, and CIED-IE. 6 Abnormal cardiac findings typical of IE are considered a major imaging criterion in both the 2023 ESC and 2023 Duke-ISCVI diagnostic criteria.5,30 It should be noted that in patients with a valvular prosthesis implanted less than 3 months, positive 18F-FDG PET/CT findings are only considered a minor criterion in the 2023 Duke classification. 5
The ACC/AHA guidelines, in contrast, have yet to make a formal recommendation on its routine use despite acknowledging its diagnostic utility. 7
Sensitivity, specificity, and limitations
In a retrospective study of 303 patients, the sensitivity of 18F-FDG PET/CT was noted to be much higher in PVIE (93%) compared to NVIE (22%); on review of histopathologic findings, the NVIE group was found to have a significantly lower number of polymorphonuclear cells and higher tissue fibrosis extent, conferring a lesser localized inflammatory process consistent with the reduced 18F-FDG uptake observed in these patients. 60 In a meta-analysis of seven largely prospective studies, Kamani et al. similarly showed quite a low sensitivity (36.9%) albeit excellent specificity (99.1%) for the diagnosis of NVIE using 18F-FDG PET/CT. 59 Even in those studies where patient preparation for the scan and myocardial glucose uptake suppression were optimized such that sensitivity reached a high of 80%, 61 18 F-FDG PET/CT continued to prove less sensitive for NVIE than traditional imaging modalities such as TEE. The role of 18F-FDG PET/CT therefore remains limited in evaluating for NVIE but should be considered if there remains equipoise for the diagnosis or if TEE cannot be performed.
In the evaluation of PVIE, several studies have demonstrated that the addition of 18F-FDG PET/CT can help improve diagnostic assessment. In a pooled meta-analysis of 28 studies totaling 1358 patients, the sensitivity for diagnosing PVIE using 18F-FDG PET/CT was 86% with a concomitant specificity of 84%. 62 Similar findings have been reproduced across several studies. In a 72-patient prospective study by Saby et al., the scan sensitivity for PVIE was 73%, and it was found that adding abnormal FDG uptake around the prosthetic valve as a diagnostic criterion increased the sensitivity of the classically used modified Duke Criteria from 70% to 97%. 63 The 188-patient study by de Camargo et al. likewise included 18F-FDG cardiac uptake as a major criterion at admission and recategorized 76% of its patients from “possible” to “definite” PVIE. 60
The use of 18F-FDG PET/CT has also been of interest in cases of CIED, albeit with a wide range of reported sensitivity from 16.3% in the EURO-ENDO registry 16 to 76.2% in the large meta-analysis by Wang et al. 62 This may be explained by the distinction in test sensitivity between the intracardiac and extracardiac components of the device, with much lower sensitivity in detecting lead infection compared to pocket infection as noted in both single-center studies and meta-analyses.64,65 18F-FDG PET/CT has also proved useful in the evaluation of vascular graft infections, which can be difficult to diagnose clinically given the nonspecific symptoms and often negative serum microbiology. Mikail et al. showed the benefit of combining FDG-PET and CTA in a four-patient case series. 66 Interestingly, a meta-analysis combining 13 studies found that the pattern of FDG uptake rather than its presence and intensity was an important diagnostic tool for aortic graft infection evaluation. 67 Lastly, in addition to extracardiac CIED-IE, the presence of extracardiac foci of infection can be determined with 18F-FDG PET/CT 68 and it could be useful in excluding IE, which can have important clinical implications; in a 40-patient study by Orvin et al., detection of extracardiac complications was shown to affect treatment planning in 35% of cases, including a 15% increase in surgical referrals. 69 Duval et al. investigated the impact of systematic whole body 18F-FDG PET/CT on the management of patients suspected of IE including 70 suspected PVIE patients and 70 suspected NVIE patients. Management was modified in 21.4% of patients with PVIE and 31% of patients with NVIE (p = 0.005) mainly due to paravalvular uptake in patients with PVIE and extracardiac uptake in patients with NVIE. 70
The value of 18F-FDG/CT was also tested in the era of machine learning. Godefroy et al. evaluated 108 patients retrospectively from a single center between 2015 and 2021 with suspected aortic PVIE. 18F-FDG/CT machine learning-based analysis had 59% sensitivity and 83% specificity in detecting aortic PVIE. 71
Key limitations to 18F-FDG PET/CT should be considered. Patients in the early postoperative period, particularly the first several months of a prosthetic valve or graft implantation, may have an FDG-avid result at the surgical site, which may not correlate with infection but rather with expected ongoing inflammation. Figure 3 highlights the difference between low-level FDG uptake in a normal surgical graft and the high-intensity, heterogeneous uptake in an infected graft. In a 39-patient study by Rouzet et al., the majority of patients who had false-positive findings by FDG-PET were in fact within 2 months of their cardiac surgery; intensity of FDG-avidity was noted to wane with time but could still persist years after the surgery. 72 For this reason, 2015 ESC and 2023 Duke-ISCVID guidelines do not recommend PET within 3 months of valve implantation. 6 Interestingly, Swart et al. published a multi-center trial in which recent valve implantation was not found to be a predictor of false-positive results, but the use of surgical adhesives was. 73 Biologic adhesives such as Bioglu® were also reported as a cause of false positives in integrated PET/CT. 74 Other inflammatory processes like vasculitis, myocarditis, malignancy, and even atherosclerotic or thrombotic plaque may also cause a false-positive result. 71 Conversely, patients who have been on prolonged antibiotics may have a decreased FDG signal owing to the lower inflammatory activity resulting in a false negative study, making the timing of 18F-FDG PET/CT implementation critical. 70 Lastly, PET imaging requires dedicated expertise for interpretation, specialized equipment, and strict oral intake preparation, which has limited its widespread use and availability.
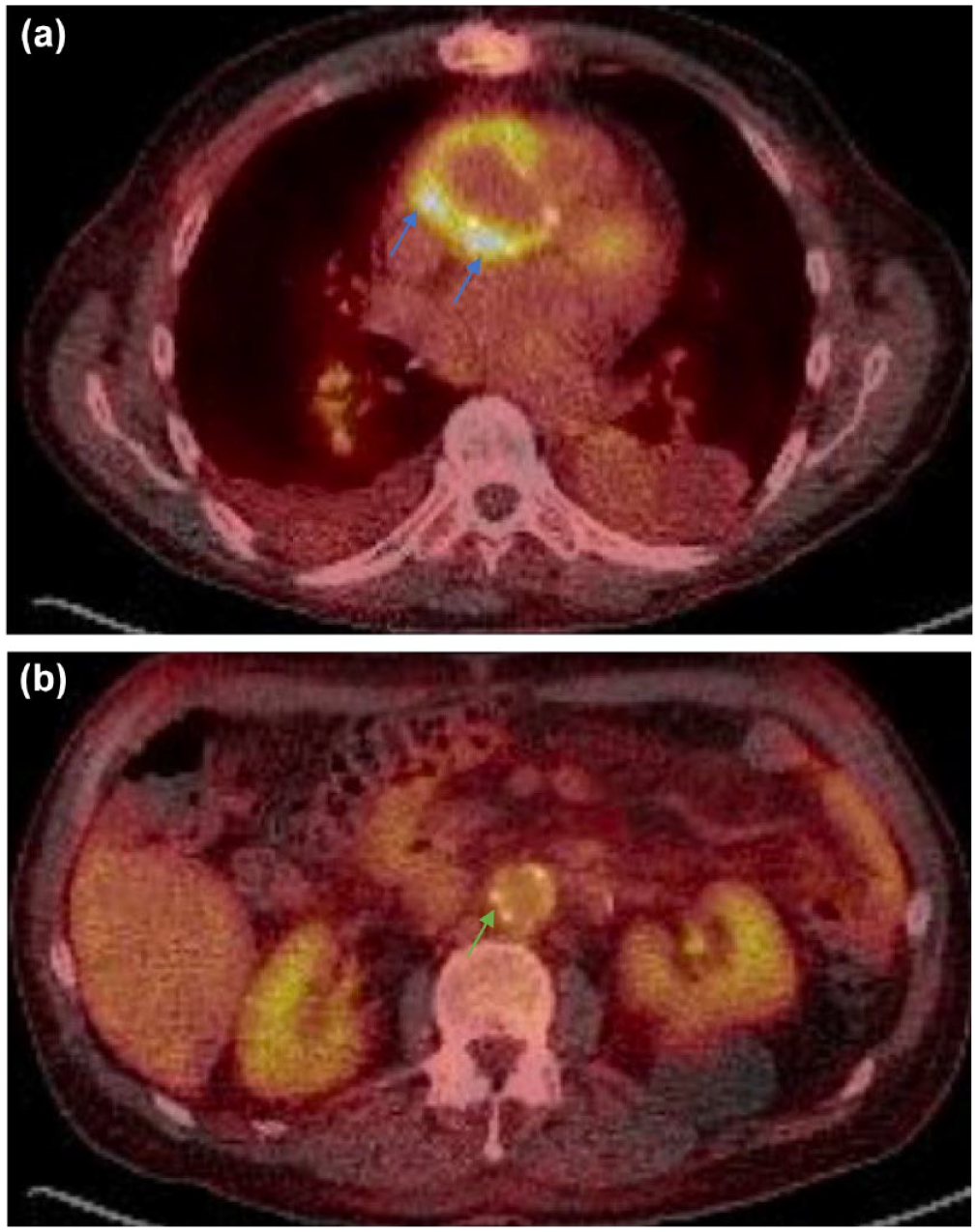
Whole body 18F-FDG PET/CT of a patient with prior aortic root, ascending aorta, and arch replacement and aorto-bifemoral bypass graft, presenting with fevers and methicillin-sensitive Staphylococcus aureus (MSSA) bacteremia. Note the intense heterogeneous uptake in the ascending aorta graft consistent with infection (a, blue arrows), in comparison to low-level more homogeneous uptake in the abdominal graft (b, green arrow) consistent with expected post-surgical changes.
Summary
18 F-FDG PET/CT has higher sensitivity in patients with PVIE. It should not be routinely performed in patients with NVIE and only be considered in patients who are unable to have a TEE. It has great value in detecting vascular graft infections. Its role in CIED-ID is limited due to low sensitivity. Finally, it plays an important role in detecting extracardiac foci of infection which could be useful in excluding IE.
Radiolabeled-WBC SPECT/CT in IE
Technical aspects
Similar to 18F-FDG PET/CT, WBC scintigraphy with hybrid SPECT/CT imaging utilizes radiolabeled white blood cells with the understanding that increased uptake of the cells will preferentially be directed toward infected tissues, there is actually more limited data on WBC scintigraphy for IE, largely restricted to single-center trials and case series.
Indications according to different guidelines
The use of WBC-SPECT/CT should be considered in patients with high clinical suspicion of PVIE or cardiac implanted devices when echocardiography is negative or inconclusive, and when 18F-FDG PET/CT is not available.3,4
Sensitivity, specificity, and limitations
One of the first studies to demonstrate the utility of WBC scintigraphy in IE was a 131-patient study by Erba et al., which showed a 90% sensitivity, 94% negative predictive value, and 100% specificity and positive predictive value in using scintigraphy for IE; both NVIE and PVIE were included. 76 Interestingly, there were no false-positive results in the study, even in patients who were in the early 2-month postoperative period. 72 False-negative findings were hypothesized to be secondary to the organism type such as Candida or Enterococcus species that may form biofilm or produce proteases to minimize leukocyte infiltration. 72 In a later study by Rouzet et al., 39 patients were evaluated with both 18F-FDG PET/CT and WBC scintigraphy; the former modality was found to have sensitivity and specificity of 93% and 71%, while the latter had a lower sensitivity of 64% but a specificity of 100%, once again demonstrating the discriminatory power of WBC scintigraphy in the early postoperative period (in the first 2 months after surgery), but the higher false-negative rate with non-pyogenic microorganisms (such as Coxiella or Candida). 72 A retrospective French study comprising 36 patients who underwent cardiac surgery 30 days after WBC-SPECT imaging noted similar sensitivity and specificity of 87% and 100%, and found that patients with negative imaging were more likely to have a longer antibiotic duration and absence of perivalvular abscess. 73
Data on CIED-IE specifically are limited with this modality, but one study by Holcman et al. found that tagged WBC-SPECT/CT could help reclassify patients with possible CIED-IE and increase original modified Duke criteria accuracy from 83% to 88%. 78 In another study by Erba et al., 63 patients with suspected CIED-IE underwent WBC scintigraphy, which had a sensitivity of 94% and specificity of 100%. 75 Several small studies of patients with suspected LVAD infections also reported 100% specificity in using WBC scintigraphy,76,77 with one study noting a sensitivity of 71.4%. In a small 23-patient study of endovascular grafts where patients routinely underwent WBC scintigraphy, there were no false positives in the first month after prosthesis implantation. 78 Taken together, these data highlight the potential utility of radiolabeled WBC SPECT/CT, particularly in cases of clinical equipoise where echocardiography is limited and 18F-FDG PET/CT may yield false-positive results in the early postoperative setting.
Summary
18 F-FDG PET/CT has higher sensitivity in patients with PVIE. It should not be routinely performed in patients with NVIE and only be considered in patients who are unable to have a TEE. It has great value in detecting vascular graft infections. Its role in CIED-ID is limited due to low sensitivity. Finally, it plays an important role in detecting extracardiac foci of infection which could be useful in excluding IE.
MRI in IE
While the use of brain MRI to evaluate for subclinical embolization events can allow for early diagnosis of IE and affect treatment strategy, 79 the use of cardiac MRI has a less well-defined role in the evaluation of IE. 6 Only small single-center studies have evaluated the diagnostic potential of cardiac magnetic resonance (CMR), and with limited direct comparison to other imaging modalities. Dursun et al. published a 16-patient study that showed CMR could detect 14 vegetations in 11 patients, in addition to detecting endothelial inflammation of cardiac structures via late gadolinium enhancement (LGE) imaging. 84 A study by Zatorska et al. noted that LGE was present in up to 40% of their 20 patients, and often extended into the myocardium itself. 85 The study also highlighted the limited spatial resolution of CMR that made vegetation detection inferior to echocardiography. 81 Bhuta et al. built on these earlier findings and reported on a 14-patient study, where CMR failed to identify vegetations readily seen by echocardiography in two patients. 86 They also found the diagnostic accuracy of CMR was significantly affected by a patient with a prosthetic aortic valve. 82 They too found evidence of LGE in several of their patients but noted that these findings did not ultimately help guide clinical decision-making. 82
The present data suggest that CMR has a limited role as a primary diagnostic modality in the evaluation of IE. It should be highlighted that CMR can provide valuable information on cardiac anatomy, valvular function, hemodynamic assessment, myo-pericardial inflammatory status, and mass characterization. 82 However, CMR is a timely and costly tool that is generally limited to specialized centers with larger volumes of patient cases and expertise on appropriate imaging protocols. 82 As was noted in some of the above studies, even in specialized centers, CMR evaluation can be significantly affected by magnetic artifacts from prosthetic valves or CIEDs, as well as patient factors including poorly controlled heart rates and inability to perform breath-holds.71,82 As this imaging modality continues to evolve, spatial and temporal resolution are likely to improve and CMR may find a greater role in the evaluation of IE in the future, particularly when used in conjunction with other imaging modalities.83–87
Conclusion
IE is a serious infection with elevated morbidity and mortality. The role of multimodality imaging in the evaluation of IE continues to evolve, and clinicians should familiarize themselves with the advantages and limitations of these modalities as they pertain to NVIE, PVIE, and other cardiac implantable device- and graft-associated infections to improve the diagnosis and management of patients with IE.
