Abstract
Background:
Heart failure (HF) is a highly prevalent disease, among the primary factors contributing to morbidity and death. One of its types is heart failure with preserved ejection fraction (HFpEF) comprising 40%–50% of newly diagnosed HF cases. Despite the high prevalence of HFpEF, there is still a lack of knowledge regarding the best drugs and treatment approaches to be used. However, the sodium-glucose co-transporter 2 (SGLT2) inhibitors could be a promising treatment.
Objectives:
To examine SGLT2 inhibitors’ effect on hospitalization, cardiovascular death, and estimated glomerular filtration rate (eGFR) in HFpEF patients.
Search methods:
We conducted searches for randomized controlled trials (RCTs) in PubMed, Embase, Scopus, and Web of Science up to July 2024.
Selection criteria:
We chose RCTs that examined the effects of SGLT2 inhibitors and placebo in individuals with higher than 40% ejection fraction (HFpEF).
Data collection and analysis:
The methodology for the systematic review and meta-analysis was in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis.
Main results:
We included 8 studies with 16,509 participants. Drugs examined in our paper included empagliflozin, dapagliflozin, sotogliflozin, and ertugliflozin. Various outcomes were analyzed in different papers. However, different SGLT2 inhibitors lead to a decreased risk of cardiovascular hospitalization and kidney injury. Our meta-analysis showed a decreased risk of cardiovascular hospitalization but not death due to cardiovascular causes or other causes. These results were regardless of baseline status of eGFR, systolic blood pressure, atrial fibrillation or flutter, diabetes mellitus, sex, body mass index, and nt-proBNP. The included studies were of moderate to high quality.
Conclusion:
For individuals with HFpEF, SGLT2 inhibitors have been proven to be a safe and effective medication. However, more studies are needed for longer durations, reporting adverse events, effects on exercise tolerance, and other secondary outcomes.
Keywords
Introduction
Heart failure (HF) persists as a prevalent, expensive, incapacitating, and life-threatening medical ailment frequently encountered by various healthcare providers in both primary and specialized settings. 1
The main source of morbidity and death among non-communicable diseases is cardiovascular disease, which is the main cause of it. 2 In developed nations, the prevalence of HF varies between 1.2% and 3.3% of adults, with HF prevalence reaching 10.0% in those over 70.3,4 The chance of having HF during life recently increased from 20% to 24% and its prevalence was also estimated to increase by almost 50% in the United States from 2012 to 2030.5,6
There are several ways that HF can be categorized; however, classification based on the left ventricle ejection fraction (EF) measurement is considered the basis for diagnosis and treatment strategy.7,8
It belongs to one of the following main phenotypes: mid-range EF HF, heart failure with preserved ejection fraction (HFpEF), heart failure with decreased ejection fraction (HFrEF), or HF with improved EF.9,10
Research conducted on newly occurring cases of HF within community-based populations indicates that HFpEF makes up around 40%–50% of all newly diagnosed cases of HF. 11 Likewise, when studying individuals without a history of HF over an extended period in cohorts such as PREVEND, the Framingham Heart Study, and the Cardiovascular Health Study, it has been observed that HFpEF comprises approximately 37%–53% of newly developed HF cases.12,13
While cardiovascular death rates in HFpEF are lower in comparison to HFrEF, it has been shown that the Lifetime Risk of HFpEF is almost double that of HFrEF reaching 19.3% and 11.4% respectively, 14 there is also a high occurrence of hospital readmissions, and the quality of life for individuals with HFpEF remains subpar and can be worse than HFrEF in some cases. 15 Furthermore, clinical trials have yet to identify a definitively effective treatment for HFpEF, as medications approved for HFrEF are considered mostly futile for treating HFpEF.16,17
However, in recent years, a new class of drugs has shown promising results in the treatment of HF, and sodium-glucose co-transporter 2 (SGLT2) inhibitor is a novel class of drugs commonly prescribed to treat diabetes that functions by preventing the proximal tubules from reabsorbing glucose. 18
Unlike other hypo-glycemic agents which offered very limited cardiorenal protection, SGLT2 inhibitors decreased the likelihood of hospitalization, HF, and cardiovascular death, 9 it also showed a relevant decrease in visceral obesity, albuminuria, and progression of nephropathy across the full spectrum of EF regardless of the status of diabetes mellitus.19–21
Using a systematic review and meta-analysis of randomized controlled trials, we evaluated the safety and effectiveness of using different SGLT2 inhibitors in HFpEF patients.
Methods
Protocol and registration
This review complies with the Preferred Reporting Items for Systematic Review and Meta-Analyses protocol and was registered on PROSPERO with registration number CRD42023489104. 22
Inclusion criteria
The following studies fulfilled the inclusion criteria: (1) We included only randomized clinical trials; (2) we included only studies that were published in the English language; (3) studies throughout the previous 5 years; (4) only patients with preserved EF selected; and (5) we included only adult patients (>18 years).
Exclusion criteria
Excluded studies had the following criteria: (1) research involving HF patients with a reduced EF (<40%), (2) any study including other than empagliflozin drug, (3) trials that were non-randomized observational trials, case reports, case series, cohort, and systematic reviews, and (4) any animal studies.
Electronic search
We looked through the following electronic medical databases: PubMed, Embase, Scopus, and Web of Science from November 15 to July 15, 2024. We used the following strategy: ((“Therapeutics”[MeSH] OR “Management” OR “Treatment”) AND (“Sodium-Glucose Transporter 2 Inhibitors”[MeSH] OR “SGLT2 Inhibitors” OR “SGLT2i”) AND “Heart Failure”[Mesh] AND (“preservation, biological”[MeSH Terms] OR preserved[Text Word]) AND ejection[All Fields] AND fraction[All Fields].
Selection criteria
After importing studies from PubMed, Scopus, Embase, and Web of Science and eliminating duplicates, the remaining articles were assessed based on their titles and abstracts in the EndNote Reference Library software. Subsequently, the studies fulfilling the criteria underwent a full-text review to evaluate their relevance. Two reviewers independently carried out the screening (A.K.H. and A.A.T.) Any disagreement was settled through conversation.
According to the flow diagram in Figure 1, the Preferred Reporting Items for Systematic Reviews and Meta-Analyses standards were followed during the literature screening procedure to identify relevant studies. 22
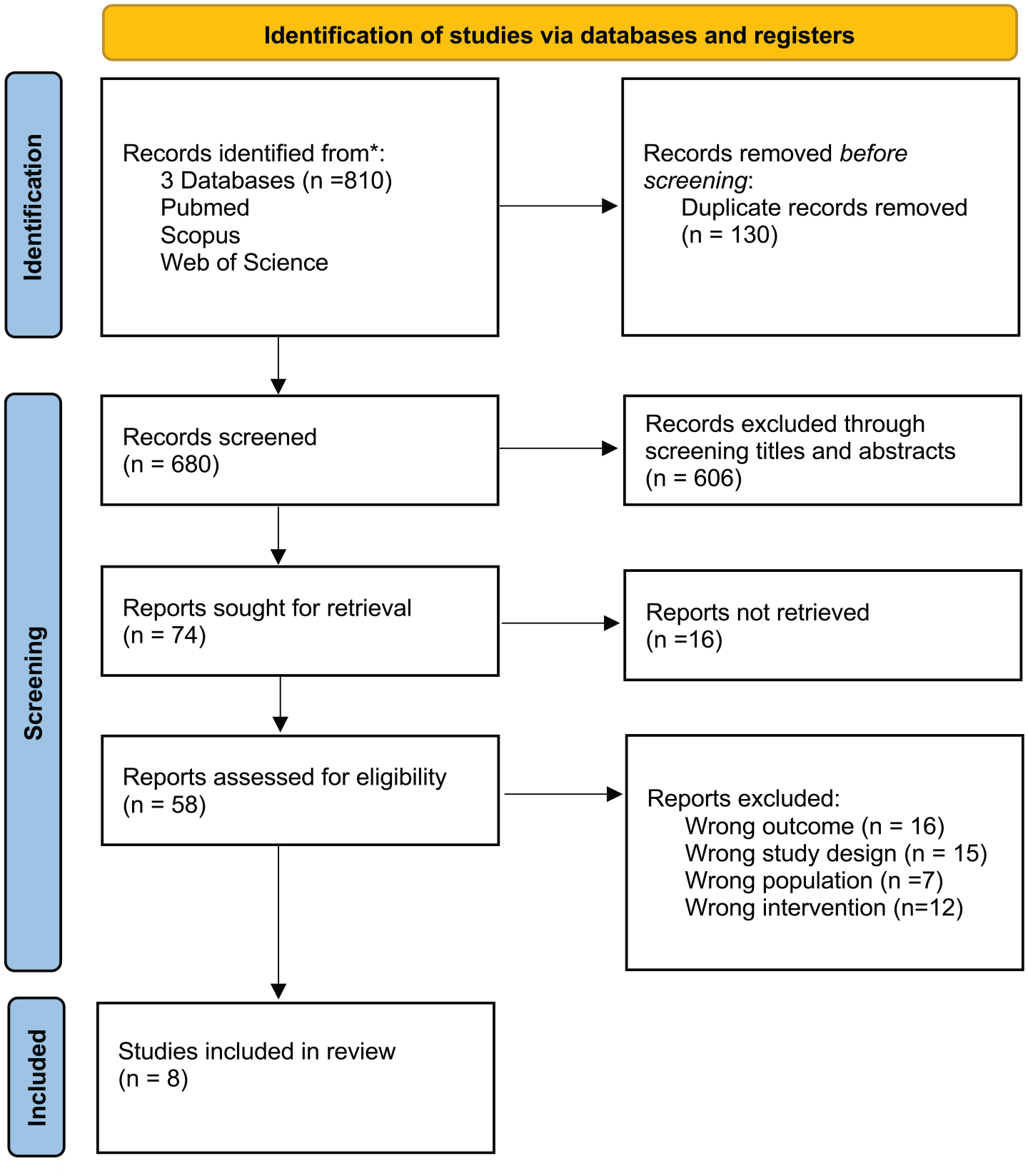
Diagram showing the flow of the PRISMA.
Risk of bias of individual studies
The risk of bias in papers that qualified was evaluated by two authors separately. They assessed the quality of 11 studies according to Cochrane Risk of bias (RoB) assessment tool. 23 then each one considered RoB “high” or “low” as shown in Figure 2.

Quality assessment of RCT using the Cochrane assessment tool.
Results
Our study included eight papers, which reported the results of eight trials, EMPERIAL, EMPA-REG OUTCOME, EMPEROR-Preserved, Declare TIMI 58, SCORED, SOLOIST-WHF, VERTIS CV trial, and DELIVER trial (Table 1). These trials were done on a pooled population of 16,509 patients with HFpEF.
The study’s design and results are part of the meta-analysis.

CKD, chronic kidney disease; HF, heart failure.
Two trials, DELIVER 24 and DECLARE, 25 examined the drug dapagliflozin and showed a significant reduction in the composite outcome of cardiovascular death and hospitalization. EMPERIAL, 26 EMPEROR, 27 and EMPAREG, 28 examined the drug empagliflozin. However, each of these studies examined different outcomes with different follow-up durations. In EMPEROR 27 and EMPAREG 28 trials, patients treated with empagliflozin had a decreased risk of the composite outcome of cardiovascular death and hospitalization. In the EMPERIAL trial, 26 patients had no significant improvement in functional symptoms after treatment with Empagliflozin. SCORED trial 29 examined the drug sotagliflozin and demonstrated a decreased rate of renal failure in patients treated with the drug. SOLOIST trial 30 demonstrated a lower chance of a composite result of cardiovascular death and hospitalization using Sotagliflozin. VERTIS trial 31 showed a decreased risk of renal failure with the use of ertugliflozin.
Studies examined the influence of different variables on SGLT2 inhibitor effectiveness in HFpEF patients. HF hospitalization and cardiovascular death were both reduced by SGLT2i; however, all-cause and cardiovascular death were not. It did not decrease the risk of one secondary outcome: any cause of death.
Main outcomes
Cardiovascular death or hospitalization for HF
Cardiovascular death or hospitalization for HF analysis was conducted in seven studies (Figure 3). Pooled studies analysis showed statistically significant results between SGLT2i group and placebo group favoring SGLT2i group (Mean Difference (MD) = 0.78; 95% CI = (0.72–0.85)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.63).

A forest plot comparing the risk of outcomes for patients with HFpEF in response to SGLT2 inhibitors against a placebo. The forest plot displays the hazard ratios of SGLT2 inhibitors in patients with HFpEF in comparison to a placebo. Cardiovascular death or hospitalization for heart failure; cardiovascular death; heart failure hospitalization; all-cause death. (a) Main outcomes and (b) secondary outcomes.
Cardiovascular death
Cardiovascular death analysis was conducted in five studies. Pooled studies analysis showed statistically insignificant results between the SGLT2i group and the placebo group (MD = 0.92; 95% CI = (0.81–1.03)). Pooled results were non-heterogeneous (I2 = 14%, p = 0.32).
HF hospitalization
HF hospitalization analysis was conducted in four studies. Pooled studies analysis showed statistically significant results between the SGLT2i group and the placebo group favoring the SGLT2i group (MD = 0.74; 95% CI = (0.67–0.83)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.87).
All-cause death
All-cause death analysis was conducted in five studies. Pooled studies analysis showed statistically insignificant results between the SGLT2i group and the placebo group (MD = 0.97; 95% CI = (0.89–1.06)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.86).
Secondary outcome
Death from any cause
Death from any cause analysis was conducted in two studies. Pooled studies analysis showed statistically insignificant results between the SGLT2i group and the placebo group (MD = 0.97; 95% CI = (0.88–1.06)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.54).
Prespecified analysis of main outcomes
The effect of SGLT2i on the incidence of primary outcome events was generally consistent across prespecified subgroups, including estimated glomerular filtration rate (eGFR; CKD-EPI) at baseline, systolic blood pressure (SBP), atrial fibrillation or flutter, diabetes mellitus, sex, body mass index (BMI), NYHA, NT-proBNP, and overall race. However, the result was heterogeneous in patients with NYHA class III and IV and non-white races, with increased risk in black patients (Figure 4 and Table 2).

Forest plot for prespecified analysis of main outcomes.
Baseline characteristic of prespecified analysis.

GFR, glomerular filtration rate.
eGFR (CKD-EPI) at baseline
eGFR (CKD-EPI) at baseline analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between patients with eGFR ⩾60 ml/min/1.73 m2 and those with eGFR < 60 ml/min/1.73 m2 favoring the SGLT2i group (MD = 0.80; 95% CI = (0.74–0.88)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.94).
Subgroup analysis showed statistically significant results for patients with eGFR ⩾60 ml/min/1.73 m2 favoring the SGLT2i group (MD = 0.82; 95% CI = (0.72–0.95)) and pooled results were non-heterogeneous (I2 = 0%, p = 0.79) and the same was for patients with eGFR < 60 ml/min/1.73 m2 (MD = 0.79; 95% CI = (0.71–0.88)) and pooled study results were also non-heterogeneous (I2 = 0%, p = 0.74).
SBP at baseline
SBP at baseline analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between patients with SBP < median and patients with SBP > median favoring the SGLT2i group (MD = 0.81; 95% CI = (0.74–0.88)). Pooled results were non-heterogeneous (I2 = 43%, p = 0.15).
Subgroup analysis showed statistically significant results for patients with SBP < median (MD = 0.85; 95% CI = (0.75–0.96)) and for patients with SBP > median (MD = 0.76; 95% CI = (0.67–0.86)) both favoring the SGLT2i group.
Atrial fibrillation or flutter at baseline
Atrial fibrillation or flutter at baseline analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between patients with atrial fibrillation or flutter and patients without atrial fibrillation or flutter favoring the SGLT2i group (MD = 0.80; 95% CI = (0.73–0.87)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.97).
Subgroup analysis showed statistically significant results for patients with atrial fibrillation or flutter (MD = 0.80; 95% CI = (0.70–0.90)) and for patients without atrial fibrillation or flutter (MD = 0.80; 95% CI = (0.71–0.91)) both favoring the SGLT2i group.
Diabetes mellitus at baseline
Diabetes mellitus at baseline analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between patients with diabetes mellitus and patients without diabetes mellitus favoring the SGLT2i group (MD = 0.80; 95% CI = (0.74–0.88)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.98).
Subgroup analysis showed statistically significant results for patients with diabetes mellitus (MD = 0.81; 95% CI = (0.72–0.91)) and for patients without diabetes mellitus (MD = 0.80; 95% CI = (0.70–0.91)) both favoring the SGLT2i group.
Sex
Sex analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between females and males favoring the SGLT2i group (MD = 0.80; 95% CI = (0.74–0.88)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.90).
Subgroup analysis showed statistically significant results for males (MD = 0.82; 95% CI = (0.73–0.92)) and for females (MD = 0.78; 95% CI = (0.68–0.90)) both favoring the SGLT2i group.
Body mass index
BMI analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between patients with BMI ⩾ 30 and those below 30 favoring the SGLT2i group (MD = 0.80; 95% CI = (0.73–0.87)). Pooled results were non-heterogeneous (I2 = 8%, p = 0.35).
Subgroup analysis showed statistically significant results for patients with BMI ⩾ 30 (MD = 0.79; 95% CI = (0.69–0.89)) and for those with BMI below 30 (MD = 0.81; 95% CI = (0.72–0.92)) both favoring the SGLT2i group.
NYHA class
NYHA class analysis was conducted in three studies. Overall pooled studies analysis showed statistically significant results between class II and class III or IV favoring the SGLT2i group (MD = 0.88; 95% CI = (0.81–0.95)). Pooled results were heterogeneous (I2 = 84%, p < 0.0001).
Subgroup analysis showed statistically significant results for class II (MD = 0.86; 95% CI = (0.78–0.94)) favoring the SGLT2i group and pooled study results were heterogeneous (I2 = 88%, p = 0.0002). Results were insignificant for class III or IV (MD = 0.92; 95% CI = (0.80–1.06)) and pooled study results were heterogeneous (I2 = 85%, p = 0.002 but they are non-conclusive.
NT-proBNP at baseline
NT-proBNP at baseline analysis was conducted in two studies. Overall pooled studies analysis showed statistically significant results between patients with NT-proBNP ⩾ median value and patients with NT-proBNP < median value favoring the SGLT2i group (MD = 0.79; 95% CI = (0.73–0.86)). Pooled results were non-heterogeneous (I2 = 0%, p = 0.95).
Subgroup analysis showed statistically significant results for patients with NT-proBNP ⩾ median value (MD = 0.79; 95% CI = (0.71–0.88)) and for patients with NT-proBNP < median value (MD = 0.80; 95% CI = (0.69–0.93)) both favoring the SGLT2i group.
Race
Race analysis was conducted in three studies. Overall pooled studies analysis showed statistically significant results between White, Black, Asian, and other races favoring the SGLT2i group (MD = 0.90; 95% CI = (0.84–0.98)). Pooled results were heterogeneous (I2 = 72%, p < 0.0001) but they are non-conclusive.
Subgroup analysis showed statistically significant results for White race favoring the SGLT2i group (MD = 0.86; 95% CI = (0.78–0.94)), Black race favoring the placebo group (MD = 1.30; 95% CI = (1.06–1.60)), Asian favoring the SGLT2i group (MD = 0.84; 95% CI = (0.68–1.02)), and pooled results were heterogeneous for White and Black races and non-heterogeneous in Asian race. Results were insignificant for other races (MD = 0.88; 95% CI = (0.64–1.23)).
Discussion
Previous studies have demonstrated that SGLT2 inhibitors reduce the risk of significant renal problems and hospitalizations for HF in diabetic patients when compared to other antihyperglycemic medications. The SGLT2 inhibitor class in particular offers advantages for the cardiovascular system and renal function.32–34
Multiple studies and meta-analyses35,36 were done regarding the efficacy of SGLT2 inhibition in the management and control of HF symptoms. Empagliflozin, an SGLT2 inhibitor class medication known for its use in diabetes mellitus management plans, is now showing promising consistent results that could potentially be the cornerstone of a newer, better approach to managing HF symptoms regardless of the other different parameters and their variations between HF patients.
Different trials have reported an improvement in patients’ HF symptoms frequency and burden assessment based regarding the clinical summary score (CSS) of the Kansas City Cardiomyopathy Questionnaire (KCCQ), especially those with a reduced EF HFrEF. However, the assessment revealed a slight to no improvement in HFpEF.26,37 Nonetheless, another trial demonstrated an enhancement of the score assessment and standard of living of both HFrEF (40%–49%) and HFpEF (⩾50%) participants on empagliflozin compared to their counterparts on placebo control.19,38 Furthermore, the use of empagliflozin has caused the risk to be significantly lower of heart disease-related hospitalization, death, and eGFR decline which were consistent in both HFrEF and HFpEF unrelated to SBP baseline values. There was no correlation shown between baseline SBP and the effects of empagliflozin as it has only demonstrated a slight insignificant increase in hypotension and volume depletion hence deeming physicians’ reluctance to prescribe empagliflozin to patients with SBP < 110 unnecessary. 19
Multiple studies have also demonstrated that older patients (>75) tend to be female with HFpEF, higher blood pressure, and reduced eGFR. However, empagliflozin has shown similar positive results, improved quality of life, and an increase in KCCQ-CSS up to 5 points across all ages. It has also been speculated to be involved in decreasing endothelial, cardiac stromal cells, and renal cellular senescence. Therefore, empagliflozin has been proven effective regardless of age, SBP, and renal function. 39
Empagliflozin’s effects on renal outcomes in persons with and without chronic kidney disease (CKD) have also been explored, which has shown a slowed eGFR decline, and a decreased risk of progression to macroalbuminuria and hyperkalemia in patients with and without CKD. These benefits were consistent across a broad spectrum of kidney function.40,41 The effect of empagliflozin in mineralocorticoid receptor antagonists users and nonusers has also been investigated with no significant differences observed between them. 41
Another study has explored the effect of Empagliflozin and diabetes mellitus on one another. It has been demonstrated to lower the likelihood that patients with preserved EF may get cardiac failure, regardless of diabetes status. 20 It has also shown a consistent reduction in risk of HF hospitalization in T2DM patients with predicted HFpEF, predicted HF with mid-range EF/HFrEF, and those without HF history. These findings support evaluating empagliflozin’s benefits across EF range in HF patients. 28
Due to the close association and frequent coexistence of heart disease with liver disease, the effects of liver parameters and empagliflozin on each other have been explored, revealing that due to the diuretic effect of empagliflozin, albumin values increased over time, but it had no other measurable effect on the other liver parameters. It has also been suggested that this diuretic effect is only useful in acute HF as it is transient, lasting about 3 months thus is of little use in chronic cases. 42 Empagliflozin did not improve liver tests in HFpEF patients compared to placebo and liver parameters did not alter the action of empagliflozin, hence allowing its use irrespective of their values even if they were pathological. 42
Despite all empagliflozin numerous benefits, interestingly, it also induces various adverse effects, including genital fungal infections and urinary tract infections, symptomatic hypotension, the potential for hypoglycemia when combined with insulin or insulin-secreting medications, and rarely seen occurrences of ketoacidosis.35,43,44 Cardiovascular, renal, and other outcomes are summarized in Figure 5.

Summary outcomes of empagliflozin in HFpEF.
To investigate the possible effects of SGLT2 inhibitors, we carried out a systematic review and meta-analysis on various parameters, such as HF hospitalization, cardiovascular death, cardiovascular death or hospitalization for HF, all cause death, eGFR (CKD-EPI) at baseline ⩾60 ml/min/1.73 m2, and eGFR (CKD-EPI) at baseline < 60 ml/min/1.73 m2. These findings may support the consideration of SGLT2 inhibitors as potential therapeutic options for heart failure patients with preserved ejection fraction (HFpEF). Pooled studies showed that SGLT2 inhibitors have a promising effect regarding improving the number of hospitalizations needed for HF patients. Not only is it found to improve that, but also it showed that it contributes significantly to reducing the rates at which predicted GFRs are declining regardless of baselines. But cases of cardiovascular deaths and all-cause death were not influenced by the SGLT2 inhibitors. In our study, it was shown that all elements had low heterogeneity. The results of our study were consistent with the previously conducted studies which solidifies the need to consider this class of drugs to be used in HFpEF patients.
Regarding the prespecified analysis of the above main outcomes of our study, the results were in favor of the SGLT2 inhibitors group in terms of eGFR at baseline, SBP at baseline, atrial fibrillation or flutter at baseline, DM at baseline, sex, BMI, NYHA class, NT-proBNP at baseline, and race except black race which showed prespecified analysis favoring the placebo group.
When testing secondary outcomes, there was nothing significant found in patients with death from any cause to support the effect of the SGLT2 inhibitors.
More studies have discussed the effects of SGLT2 inhibitors on patients with diabetic47,48 and non-diabetic 49 kidney diseases. Insulin secretion was found to be enhanced by the inhibition of the SGLT2 inhibitors due to their main action on glycemic control, which improved its tissue sensitivity too. The loop-like diuretic action of these agents through acting on Na–H exchange was found to improve kidney functions through lowering intraglomerular pressure, which is one of the mechanisms that make SGLT2 inhibitors a good option in non-diabetic kidney diseases that could help hyperkalemia or Acute kidney injury (AKI) by compensating the action of the drugs that cause these complications. 49 Not only do they have a diuretic action but also their effects on beta-OHB and hematocrit levels improved the consumption and production of oxygen and energy, respectively. 47 These effects make SGLT2 inhibitors a good complementary future option for RAS inhibitors in diabetic kidney disease. 48 The EMPA-KIDNEY study is a significant ongoing trial, the awaited results of which are expected to explain if empagliflozin reduces the risk of cardiac and renal problems, such as overt albuminuria and low eGFR, in individuals with CKD. 49
The cardioprotection associated with the use of SGLT2 inhibitors is explained partially by its protective effects on the kidneys. The relative volume depletion attributed to the diuretic and natriuretic effect of SGLT2 inhibitors along with the increase in the levels of erythropoietin and increased RBCs levels are associated with a hemoconcentration effect along with a higher rate of oxygen delivery to the heart and kidney and are accountable for approximately 50% of the observed cardiac benefits of SGLT2 inhibitors.49,50
However, other effects were also hypothesized to be involved in the decrease in HFpEF complications, like reducing arterial wall stiffness, limiting oxidative stress and tissue inflammation, decreasing sympathetic nervous activity in both the Cardiovascular System (CVS) and kidneys, and improving cardiac metabolism, by increasing fatty acid breakdown and ketone utilization associated with the hypoglycemic effect of empagliflozin. Other renoprotective mechanisms may also be indicated. However, its effect on the glycemic index itself does not appear to provide significant CVS protection. Likewise, it is insufficient to prevent major kidney injury.47,48
Limitation
Due to the small number of studies and short duration of our systematic review and meta-analysis, more research is required to assess the renoprotective and cardioprotective benefits of SGLT2 inhibitors in patients with HFpEF.
Recommendation
Additional research with extended follow-up durations and longer enrolment window is necessary to establish a conclusive determination about the effectiveness and safety of SGLT2i in patients with HFpEF, particularly its impact on exercise tolerance, the incidence of adverse events, and secondary outcomes rather than cardiac benefits within a larger population of individuals.
Conclusion
In conclusion, the present systematic review and meta-analysis highlights that SGLT2 inhibitors are safe and efficacious treatment for individuals with HFpEF. Meta-analysis showed decreased hospitalizations in patients treated with SGLT2 inhibitors regardless of baseline status of eGFR, SBP, atrial fibrillation or flutter, diabetes mellitus, sex, BMI, and nt-proBNP.
However, SGLT2 inhibitors did not decrease cardiovascular death or all-cause death in our meta-analysis. SGLT2i showed improvement in kidney function and eGFR and decreased cardiac remodeling in patients taking SGLT2 inhibitors.
Supplemental Material
sj-docx-1-tak-10.1177_17539447241289067 – Supplemental material for Empagliflozin and other SGLT2 inhibitors in patients with heart failure and preserved ejection fraction: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tak-10.1177_17539447241289067 for Empagliflozin and other SGLT2 inhibitors in patients with heart failure and preserved ejection fraction: a systematic review and meta-analysis by Abdulrahman Khaldoon Hamid, AbdulJaber A’Ed Tayem, Sandra Thair Al-Aish, Ahmed Sermed Al Sakini, Dalia Dhia Hadi and Rami Thair Al-Aish in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
