Abstract
Background:
Secundum atrial septal defect (ASD) is treated following trans-catheter closure in alternative to surgical treatment. Per-intervention selection of device size with balloon occlusive diameter (BOD) often cause tearing or enlarging, causing arrhythmias and hypotension. We assessed the suitability of percutaneous device closure for ASD using 3-dimensional transthoracic echocardiography (3DTTE).
Objectives:
This study was conducted to investigate if 3DTTE could be an alternative of balloon sizing for selection of device size in atrial septal defect device closure.
Design:
It was a cross-sectional comparative study.
Methods:
This study was conducted at the department of Pediatric Cardiology, Bangabandhu Sheikh Mujib Medical University for a period of 2 years. Thirty-three purposively selected secundum ASD patients suitable for device closure were included in the study. Ethical permission was taken from the Institutional Review Board and written consent was taken from each patient’s guardian. In this study, 3DTTE derived ASD diameter and BOD were compared with that of deployed device size using correlation analysis.
Results:
Out of 33 patients, 63.6% were female and 36.4% were males had a mean age of 18.07 ± 14.58 years (range 2–55 years). Mean diameter of ASD measured by 2-dimensional (2D) and 3-dimensional (3D) echocardiography were 17.09 ± 6.08 mm and 21.30 ± 6.56 mm, respectively, yielding a significant difference (p < 0.001). 3D echocardiography derived ASDs diameter were highly correlated with device size than BOD and 2D echocardiography derived diameter (2D echocardiography: r = 0.796, p = <0.001, 3D echocardiography: r = 0.960, p = <0.001, BOD: r = 0.840, p = <0.001).
Conclusion:
3DTTE can accurately measure ASD diameter and can be used as an alternate, effective, and safe method to select device size.
Keywords
Introduction
While the atrial septal defect (ASD) is common in congenital heart diseases,1,2 ostium secundum also remain very frequent type (approximately 7–10%)3,4 of common lesions that suits much for percutaneous device closure.3,4 This procedure demands accurate morphological characteristics of ASD including its size measurement. Nonetheless spatial relationship remains crucial to assess, between ASD and neighboring structures for successful deployment of trans-catheter septal occluder. 5
Evidence-based observations suggest that pediatric interventional cardiologists must know the exact morphology of ASD in a beating heart to facilitate deploying a therapeutic device more safely. Similar to earlier experiences, Cooke et al.
6
commented that factors that decide suitability for percutaneous closure of ASD include the defect size allowing adequate tissue rims around it. Conventionally, ASD up to 35 mm diameter with a margin
Several authors reported that most traditional non-invasive method of ASD size evaluation and positioning remains 2-dimensional transthoracic echocardiography (2DTTE) that usually underestimates maximal size and provides limited at a time.9–12 We, thus, used balloon occlusive diameter (BOD) to select appropriate device size for implantation.13–15 Reportedly, balloon inflation causes tear or enlarges defect, cause arrhythmias or hypotension owing to decreased diastolic filling16,17 staying apart from ASD size overestimation by overstretching balloon sizing. 18 This cumbersome procedure, therefore, be regarded as unnecessary rather, echocardiographic measuring methods remain preferable in selecting size of ASD occluder device, being more rational.
The 2-dimensional transesophageal echocardiography (2DTEE) provides much clearer imaging of ASDs than 2DTTE. 12 However, that has certain obvious clinical limitation requiring deep sedation or general anesthesia in children. On the contrary, the newly introduced real-time 3-dimensional (3D) echocardiography can visualize the overall structure of atrial septum, yielding pertinent information on position and size during cardiac cycle with exact morphology of surrounding rims. 5
Based on this aforementioned background, we conducted this cross-sectional study to assess the suitability of percutaneous device closure for ASD cases using 3-dimensional transthoracic echocardiography (3DTTE). We compared its echocardiographic diameter and BOD with deployed device size to ascertain the accuracy of real-time 3DTTE.
Material and methods
Settings
This cross-sectional comparative study was conducted between the periods of September 2015 and August 2017 in the Department of Pediatric Cardiology, Bangabandhu Sheikh Mujib Medical University, Dhaka, Bangladesh a center of excellence with regard to skilled manpower, modern equipment, and volume of service. Objective of this study was to compare and correlate the size of ASDs measured by 3DTTE and balloon sizing maneuver with deployed device size.
Sample
For estimating sample size following formula was used N = [(Z α + Z β )/C]2 + 3. Here, α (two-tailed) is threshold probability for rejecting the null hypothesis, type 1 error rate = 0.05, β is probability of failing to reject the null hypothesis under the alternative hypothesis, type 2 error rate = 0.20, and r is expected correlation coefficient. In a previous study, it was 0.930. But in this study, presumed r was 50% of the previous study (r = 0.47). The standard normal deviate for α = Z α = 1.96 and β = Z β = 0.842. C = 0.5 *In [(1 + r)/(1 − r)] = 0.510. Thus, the total sample size was =[(Z α + Z β )/C]2 + 3 = [(1.96 + 0.842)/0.510]2 = 30 + 3 = 33.
Patient selection and data collection
Total 33 consecutive patients attending Pediatric Cardiology Department of Bangabandhu Sheikh Mujib Medical University aged above 2 years and weighing 10 kg or more who were diagnosed as secundum ASDs suitable for device closure having size up to 35 mm, left to right shunt with features of right ventricular volume overload and acceptable pulmonary vascular resistance were included in the study. Critically ill patients, presence of multiple defects, unwilling to enroll in the study, or having financial constrain to bear the cost of device were excluded from the study.
Based on a literature review,5,8,9,14,19 a semi-structured questionnaire was developed and finalized after a consultative meeting with public health professionals, pediatric cardiologists. Prior to data collection, this questionnaire was pre-tested with four children (12% of final sample population) to determine its suitability for data collection. This pre-tested questionnaire in English are translated into easy local Bengali were used for interviewing the patients. Sociodemographical data, clinical history, and physical examination findings were recoded. After that, all eligible patients underwent echocardiography and essential laboratory testing as well as per-interventional balloon sizing. 3DTTE and BOD measurement during percutaneous device closure was performed by different investigators, blindly, on 3D echocardiography findings.
Echocardiography
Initially patients having secundum ASD were assessed both by 2DTTE and 3DTTE regarding suitability for device closure. The maximum diameter and rims of the secundum ASDs were measured by both 2-dimensional (2D) and 3D echocardiography. Real-time 3D echocardiography studies were performed using Vivid E9 ultrasound system (GE Healthcare, Horton, Norway) equipped with X matrix probe and software package including 3D data acquisition and analysis. Data acquisition were based on recommendations of Adhoc 3D Echo Protocol Working Group endorsed by International Society of Cardiovascular Ultrasound. 20 Once a 3D data set is acquired, 2 click crop method was applied to visualize atrial septum from both atrial sides. 3D analysis involved isolating the atrial septum including all the relevant margins to produce an en-face moving image of the whole atrial septum from left and right atrial aspects, throughout the cardiac cycle. This allowed the area, maximum diameters, margins, shape, and anatomical details including fenestrations to be assessed. Planimetry was performed including measurement of diameters. 19 The maximal diameter of the defect was measured in each ASD patient by picking up the two farthest points from the 3D curved plane during cardiac cycle. 21 Analysis of the various margins were performed by measuring the shortest distance connecting the edge of the septal defect to the structure defining the rim, from both right and left atrial en-face views of the septum. 19 Echocardiography was also done during device closure procedure, especially before device release, immediately after device release, 2 h after procedure, and next day before discharging the patient.
Balloon sizing and selection of device size
The stretched BOD was measured by the balloon sizing maneuver using Amplatzer sizing balloons (AGA Medical Corporation, Golden Valley, MN, USA). In left anterior oblique view, the balloon was inflated within the ASD with diluted contrast until a waist appeared. 5 The BOD was determined outside the body by passing the same amount of contrast medium into the balloon and measuring the size by passing it through a calibrated sizing plate. Angiographic measurement of balloon also was taken. The size of the device was chosen same as BOD or 1–2 mm larger.
Statistical analysis
All the data were entered into Statistical Package for the Social Sciences (SPSS) version 21, rechecked and cleaned and were subjected to statistical analysis. All quantitative variables were expressed as mean and standard deviation (SD), and categorical variables were subjected to frequency and percentage; mean diameter of ASDs measured by 2D, 3D echocardiography, and balloon occlusion were compared using t tests. Measurements of maximal ASD size obtained by 2DTTE, 3DTTE, and balloon sizing method were compared with the deployed device size using correlation analysis. p Value < 0.05 was considered to be statistically significant.
Results
A total of 33 patients were included in the study. Male and female were 36.4% and 63.6%, respectively. The mean ± SD of age of the respondents was 18.07 ± 14.58 years with a range of 2–55 years. Among the respondents, two-thirds (66.7%) were in pediatric age group (<18 years) and one-third (33.3%) were adult (>18 years). The mean ± SD of weight and height were 34.33 ± 18.13 kg and 138.70 ± 21.34 cm, respectively. Socioeconomic status of the respondents according to wealth index were very low 24.2%, low 21.2%, medium 15.2%, high 21.2%, and very high 18.2%.
All patients underwent successful transcatheter closure with no residual shunt.
The mean diameter of ASDs measured by 2D and 3D echocardiography were 17.09 ± 6.08 mm and 21.30 ± 6.56 mm, respectively, yielding 3D echocardiography being significantly higher (p value < 0.001). The individual mean length of all rims were sufficient (>5 mm) both in 2D and 3D echocardiography except aortic rims (<5 mm), but mean length of mitral, aortic, posterosuperior, and inferior vena cava (IVC) rims were measured significantly higher in 3D than 2D echocardiography and no significance difference in superior vena cava (SVC) and posterior rims (Table 1).
Comparison of 2D and 3D echocardiographic findings.

Paired t-test was done to measure the level of significance.
ASD, atrial septal defect; 2D, 2-dimensional; 3D, 3-dimensional; IVC, inferior vena cava; SVC, superior vena cava.
Mean diameter of ASDs measured by balloon sizing of 21.30 (±6.56) mm was significantly higher than that of 2D echocardiography 17.09 (±6.08) mm (p < 0.001), whereas, it did not vary when measured using 3D echocardiography (21.30 ± 6.56 mm versus 20.36 ± 7.78 mm, p = 0.24), (Table 2).
Comparison of ASD size measured by 2DTTE and 3DTTE with BOD.

Paired t test was done to measure the level of significance.
ASD, atrial septal defect; BOD, balloon occlusive diameter; 2DTTE, 2-dimensional transthoracic echocardiography; 3DTTE, 3-dimensional transthoracic echocardiography.
Pearson correlation test among different methods of ASD diameter measurements and device sizes that yielded the following:
The 2DTTE and 3DTTE derived diameter of ASDs were positively correlated with invasively determined BOD, but stronger correlation revealed among 3D echocardiography than 2D echocardiography (2DTTE; r = 0.662, p < 0.001, 3DTTE; r = 0.816, p < 0.001) (Figure 1).
2DTTE, 3DTTE derived diameter and BOD of ASDs showed excellent correlation with device size (2DTTE; r = 0.796, p < 0.001, 3DTTE; r = 0.960, p < 0.001, BOD; r = 0.840, p < 0.001), but the correlation was better in 3D echocardiography derived diameter with device size than 2D echocardiography and BOD (Figures 2 and 3).

Correlation of size of ASD measured by 3DTTE with BOD.

Correlation of size of ASD measured by 3DTTE with device size.
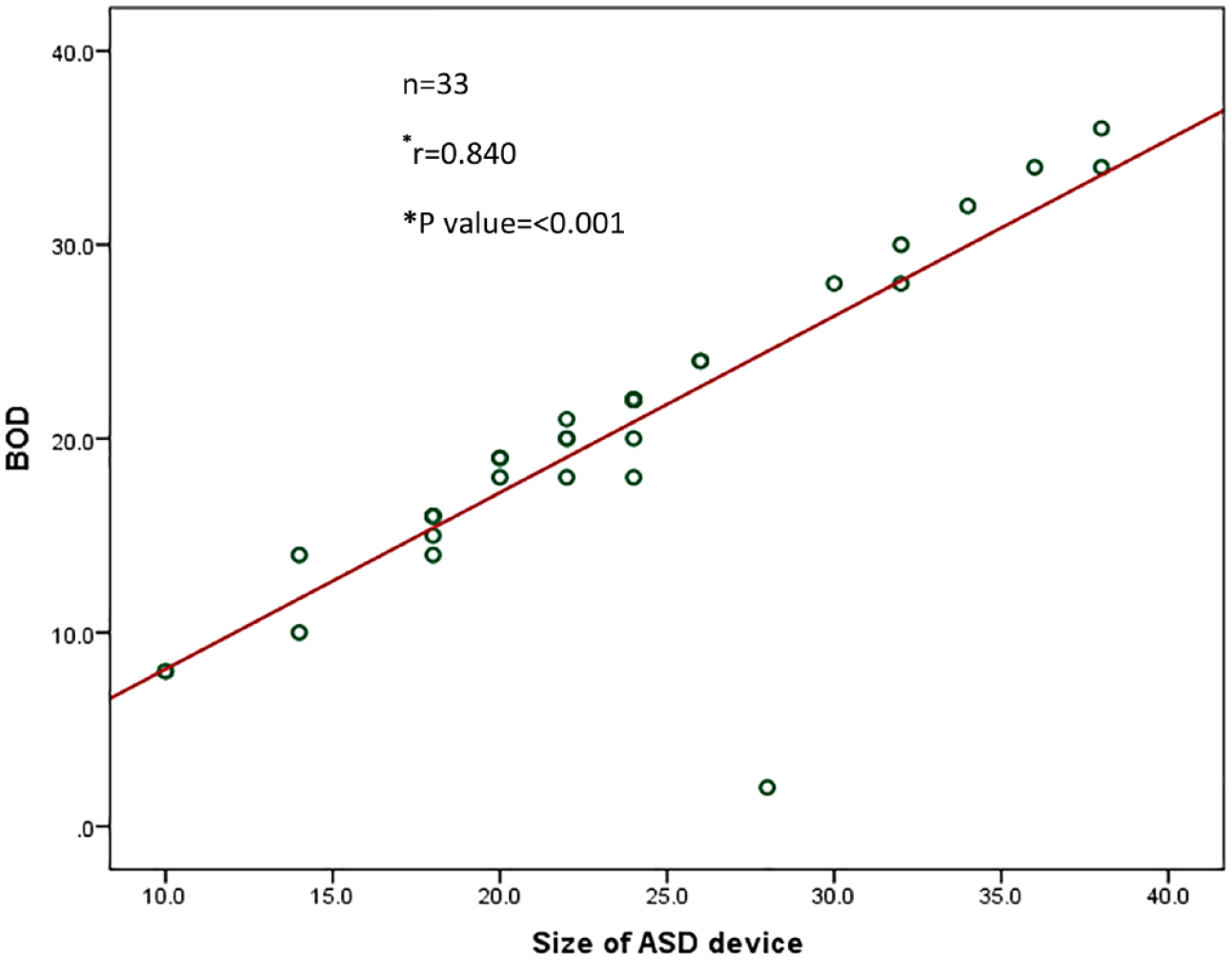
Correlation of BOD with device size.
Discussion
Recent literature evidence percutaneous closure of secundum ASD as an attractive yet effective alternative to surgical repair in selected cases. Our earlier observation yielded that a precise assessment of ASD remains crucial for device selection and procedural success. Traditionally 2DTTE or 2DTEE was used to assess the secundum ASDs for device closure using a most frequently methods for device selection and deployment include balloon sizing maneuver with 2D transthoracic or transesophageal monitoring.13–15 Currently, the 3D echocardiography has been shifted from research laboratory to daily clinical tool since it is non-invasive yet inexpensive method to demonstrate dynamic morphology. 19 However, currently it remains a considerable debate regarding the most appropriate imaging technique to guide device closure of ASD both in terms of pre-procedure selection and device placement in the catheter laboratories. 19 Most frequently, 2D echocardiographic diameter of ASD does not correlate with BOD9–12,22 a dangerous per-operative method that requires selection of device size.
The current study described our initial experience of real-time 3DTTE application in 33 secundum ASD patients. In addition, our aim extended to standardize the technique in order to facilitate routine use of 3DTTE for assessment of ASDs prior to device closure without per-operative use of BOD.
Though our findings revealed a female preponderance among the patients we dealt with ASDs, that remains similar to other study reports, 21 but the exact reason could not be identified, but this preponderance must have certain genetic basis, as we assume.
Furthermore, our findings revealed a significant difference between the mean diameter of ASDs measured by 2D echocardiography and balloon sizing, which showed a close relationship between ASD sizes assessed and quantified by 3DTTE and balloon sizing, finally. Contrary to some previously published reports, revealing echocardiographic measurements being smaller than BOD,23,24 our findings on 3DTTE measurement were slightly larger than that of BOD. The 2D and 3D echocardiography derived diameter of ASDs correlated with the invasively determined BOD (2DTTE; r = 0.662, p < 0.001 and 3DTTE; r = 0.816, p < 0.001); however, degree of correlation was better with 3D than 2D echocardiography. This finding of ours remain similar to that of other’s study where 3DTTE derived maximum diameter of ASDs best correlated with invasively determined BOD. 25
Again, we observed, 2D, 3D echocardiography derived ASDs diameter and BOD yielding a strong correlation with the device sizes (2DTTE; r = 0.796, p = <0.001, 3DTTE; r = 0.960, p = <0.001 and BOD; r = 0.840, p = <0.001) though strength of correlation was better in 3D echocardiography derived diameter with device size than 2D echocardiography and BOD with device size. These detailed findings also remain similar to that of study reports from other countries, too.5,26
We found an excellent correlation (r = 0.960) between 3D echocardiography derived diameter of ASDs and device size, which was even better than the correlation (r = 0.840) between BOD and device size. These facts represent that real-time 3D echocardiography showed not only the immediate anatomical characteristics, but also the accurate measurement of ASD size to select the device size. Previous reports have confirmed that 3D echocardiography is a useful method to evaluate ASD size.19,23,24 Our study adds a new information on quantitative use for real-time 3D echocardiography that, it proposes a standardized plane for accurate measurement of ASDs and also it defines a method for the selection of a device, thus eliminating the cumbersome balloon sizing maneuver.
Our research work has several limitations. Patients with multiple ASD were excluded. There are some limitations of 3D echocardiography such as proper gain acquisition to avoid under or overestimation of measurements of the defects and especially in children limitation related to high heart rate, breathing, and movement artifact. Sometimes, atomical detailing of the interatrial septum is insufficient by 3DTTE and transesophageal echocardiography (TEE) resolution is superior considering its posterior location, but it requires deep sedation or general anesthesia in children. Another limitation is unavailable acoustic window; therefore, some patients may need TEE/Balloon sizing for device size selection though we did not find much difficulties to acquire images of interatrial septum. Small sample size in our study may also be a limitation which can be waived through further similar studies.
Conclusion
3D echocardiography is a useful tool in the assessment of ASD with regard to suitability for device closure, and it can measure the diameter of ASD accurately to select device size and hence can replace the balloon sizing.
