Abstract
For decades, plasma arginine vasopressin (AVP) levels have been known to be elevated in patients with congestive heart failure (HF). Excessive AVP signaling at either or both the V1a and V2 receptors could contribute to the pathophysiology of HF by several mechanisms. V1a activation could cause vasoconstriction and/or direct myocardial hypertrophy as intracellular signaling pathways are closely related to those for angiotensin II. V2 activation could cause fluid retention and hyponatremia. A hemodynamic study with the pure V2 antagonist tolvaptan (TV) showed minimal hemodynamic effects. Compared with furosemide in another study, the renal and neurohormonal effects of TV were favorable. Several clinical trials with TV as adjunctive therapy in acute HF have shown beneficial effects on fluid balance and dyspnea, with no worsening of renal function or neurohormonal stimulation. Two smaller studies, one in acute and one in chronic HF, have shown comparable clinical and more favorable renal and neurohormonal effects of TV compared with loop diuretics. However, long-term treatment with TV did not alter outcomes in acute HF. No data are available other than single-dose studies of an intravenous pure V1a antagonist, which showed a vasodilating effect if plasma AVP levels were elevated. One hemodynamic study and one short-duration clinical trial with the balanced intravenous V1a/V2 antagonist conivaptan (CV) showed hemodynamic and clinical effects largely similar to those with TV in similar studies. A new orally effective balanced V1/V2 antagonist (pecavaptan) is currently undergoing phase II study as both adjunctive and alternative therapy during and after hospitalization for acute HF. The purpose of this review is to summarize what we have learned from the clinical experience with TV and CV, and to suggest implications of these findings for future work with newer agents.
Introduction
It has been nearly 40 years since the first reports appeared using accurate radioimmunoassay of increased arginine vasopressin (AVP) levels in patients with heart failure (HF).1,2 From the standpoint of cardiovascular pathophysiology, excessive AVP signaling via the V1a and the V2 receptors could have multiple adverse effects including vasoconstriction, direct myocardial stimulation leading to inappropriate ventricular remodeling, fluid retention, increased diastolic wall stress and hyponatremia. The case for blocking these receptors in HF has been made repeatedly over the years.3,4 There was an early report of the effects of a single dose of a V1a antagonist in stable HF patients. 5 However most of what we have learned about AVP antagonism in HF has come from clinical trials with the selective V2-antagonist tolvaptan.6–16 It has been difficult to develop orally effective and safe antagonists to the V1a receptor, but we do have one hemodynamic study in patients with chronic HF and one trial in those with acute HF using the balanced V1a/V2 antagonist conivaptan.17,18
There is a new compound now in phase II clinical trials, which is an orally effective and apparently well-tolerated balanced V1a and V2 antagonist.19,20 In this trial the new drug is being studied both as adjunctive and alternative therapy to loop diuretics in acute and subacute HF. Interpreting the results of this trial will depend on both the results per se and how they compare with similar experience with tolvaptan and to a lesser extent with conivaptan, as it is not likely there would be a comparison of the new agent with tolvaptan or conivaptan any time soon.
The purpose of this brief review is therefore to summarize concisely what we have learned from the present body of information gained from the major hemodynamic and clinical trials in patients with HF using the selective V2 antagonist tolvaptan and the combined V1a/V2 antagonist conivaptan and to place this information in the context of directions for future work with AVP antagonism in HF.
Hemodynamic and renal studies with tolvaptan and conivaptan
As discussed above, it is known that plasma AVP concentrations are elevated in patients with HF with reduced ejection fraction (HFrEF), and the rationale for studying antagonists to AVP was based on the possibility that increased AVP levels could play a role in the fluid retention and hemodynamic abnormalities seen in patients with HF.3–5 The hemodynamic and renal effects of vasopressin antagonists in stable chronic HF patients have been investigated in studies with the selective V2 antagonist tolvaptan as well as with the dual antagonist conivaptan (Table 1).
Review of Studies.
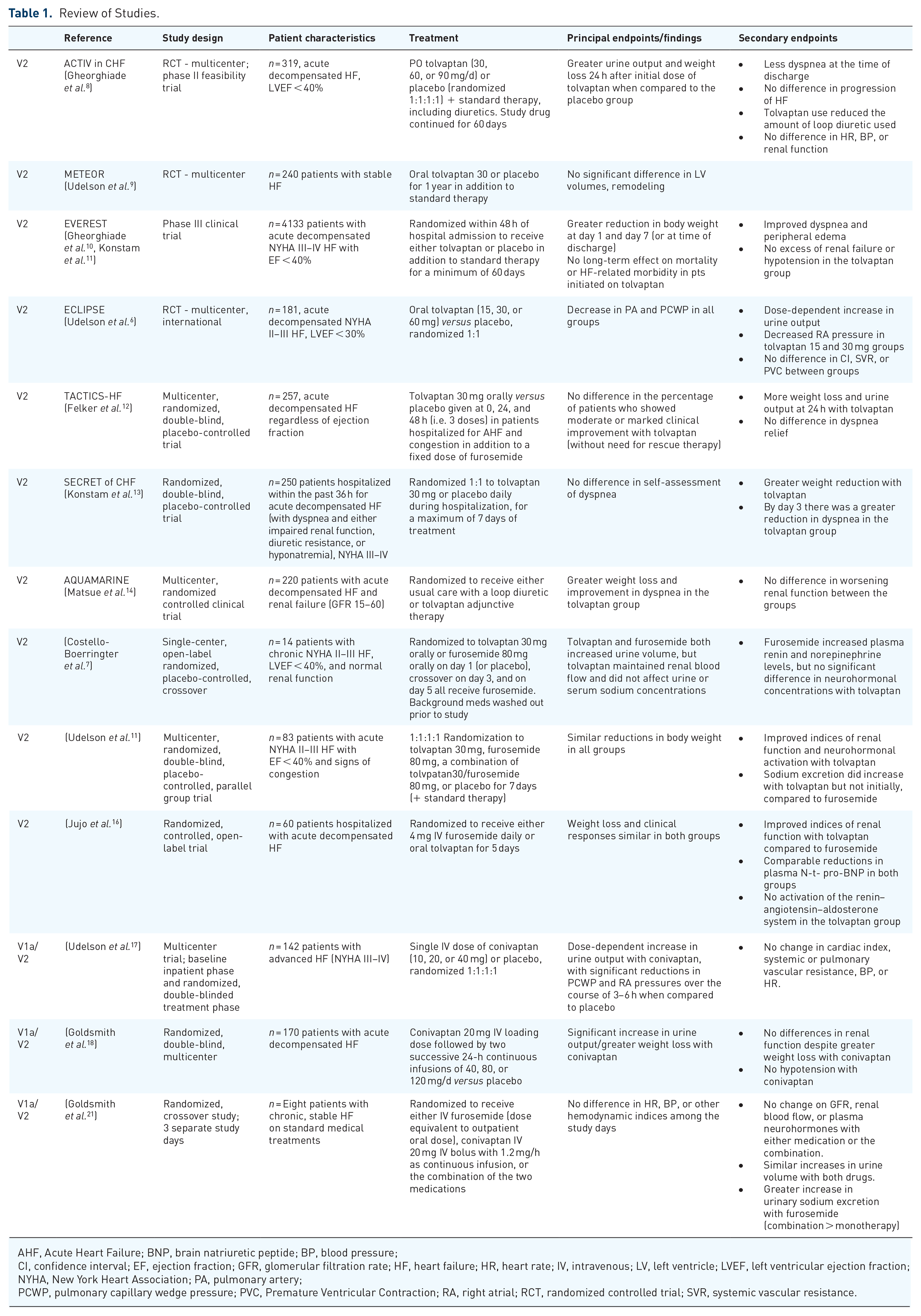
AHF, Acute Heart Failure; BNP, brain natriuretic peptide; BP, blood pressure;
CI, confidence interval; EF, ejection fraction; GFR, glomerular filtration rate; HF, heart failure; HR, heart rate; IV, intravenous; LV, left ventricle; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; PA, pulmonary artery;
PCWP, pulmonary capillary wedge pressure; PVC, Premature Ventricular Contraction; RA, right atrial; RCT, randomized controlled trial; SVR, systemic vascular resistance.
ECLIPSE: The Effect of Tolvaptan on Hemodynamic Parameters in Subjects with Heart Failure (ECLIPSE) study was designed to evaluate the hemodynamic effects of tolvaptan, a vasopressin V2 receptor antagonist in 181 patients with advanced New York Heart Association (NYHA) II–IV HF with left ventricular ejection fraction (LVEF) <40%. Using pulmonary artery (PA) catheters to assess baseline hemodynamics, patients were randomized to receive tolvaptan (15, 30, or 60 mg) or placebo (randomized in a 1:1:1:1 fashion) if they were found to be congested [pulmonary capillary wedge pressure (PCWP) was >18 on two consecutive readings]. There was a significant increase in urine output along with reductions in PCWP and right atrial (RA) pressures with all tolvaptan doses when compared with placebo, without changes in heart rate (HR), arterial pressure, cardiac output (CO) or systemic vascular resistance (SVR). 6
Costello-Boerringter 2006: Studies have shown that V2 receptor antagonism causes aquaresis without alteration in renal hemodynamics. 7 In this single-center, open-label, randomized, placebo-controlled trial, 14 patients with NYHA II–III HF with LVEF < 40% and normal renal function were randomized to receive either tolvaptan 30 mg orally or furosemide 80 mg orally on day 1 (or placebo) and were crossed over to the other study medication on day 3. On day 5, all patients received furosemide 80 mg orally. Days 2 and 4 were washout periods and all background medications were washed out for 3 days prior to the start of the trial period. Both tolvaptan and furosemide increased urine volume, but tolvaptan was shown to maintain renal blood flow (whereas it was decreased with furosemide) without effect on urine or serum sodium concentrations. There was no change in neurohormone concentrations in the tolvaptan group while furosemide increased renin and norepinephrine levels.
Udelson 2001: The short-term hemodynamic effects of conivaptan (V1a/V2 receptor antagonist) were studied versus placebo in 142 patients with symptomatic HF (NYHA III–IV). As in the ECLIPSE study, all patients were studied using PA catheter-derived data. At baseline, patients had elevated PCWP but normal CO and SVR. Baseline AVP levels were also essentially normal. Patients received a single intravenous (IV) dose of conivaptan (10, 20, or 40 mg) or placebo, and there was a dose-dependent increase in urine output with conivaptan, with significant reductions in PCWP and RA pressures over the course of 3–6 h when compared to placebo. There was no change in cardiac index, systemic or pulmonary vascular resistance, blood pressure (BP), or HR. 17
Goldsmith 2011: The renal effects of conivaptan, furosemide and the combination of conivaptan with furosemide was studied in eight patients with chronic, stable HF on standard medical treatments. 21 Patients were randomized to receive either IV furosemide (dose equivalent to outpatient oral dose), conivaptan 20 mg IV bolus with 1.2 mg/h as continuous infusion, or the combination of the two medications. The main goal of this study was to look for differences in HR, BP, noninvasively measured hemodynamics, glomerular filtration rate (GFR), and renal blood flow between conivaptan and furosemide and the combination. Sodium excretion was also assessed. There were no differences among the groups in HR, BP, or other noninvasively measured hemodynamic indices, nor were there changes in GFR, renal blood flow, or plasma neurohormones with either medication or the combination. However, sodium excretion in the group receiving both furosemide and conivaptan was greater than in the group receiving furosemide alone, although as expected conivaptan had no independent effect on urine sodium. This result suggests a possible effect of intrarenal V1a antagonism, a topic which has received considerable experimental study, with the suggestion that V1a activation may influence renal medullary blood flow.22–24
Clinical trials with tolvaptan
The following clinical trials all examined the effects of tolvaptan as adjunctive therapy to loop diuretics in patients with acute HF.
ACTIV in CHF: The Acute and Chronic Therapeutic Impact of a Vasopressin Antagonist in Congestive Heart Failure was a placebo-controlled trial that included 319 patients with NYHA III–IV acute decompensated HF with an LVEF <40%. 8 These patients were randomized to oral tolvaptan in addition to standard therapy, including loop diuretics. Patients who received tolvaptan had a significantly greater weight reduction 24 h after initial dose when compared to the placebo group, along with a greater urine volume. The enhanced diuretic effect was maintained throughout the hospitalization, and patients in the tolvaptan group reported less dyspnea at the time of discharge when compared to the placebo group. At the end of the 60-day follow-up period, there was no difference in worsening HF between the two groups, though the tolvaptan group used less furosemide and had no difference in HR, BP, or renal function. While the study was not powered to evaluate for mortality, a post hoc analysis of the study data suggested that patients with hyponatremia, “severe” congestion and/or a blood urea nitrogen (BUN) of >29 mg/dl that were treated with tolvaptan may have a reduction in mortality. 25
METEOR: The effect of vasopressin receptor antagonism with tolvaptan on left ventricular remodeling was studied in the Multicenter Evaluation of Tolvaptan Effect on Remodeling (METEOR) trial. 9 In this trial, 240 patients were randomized to tolvaptan or placebo as adjunctive therapy to standard HF background medications and followed for 1 year of therapy. The primary endpoint was change in left ventricular (LV) volumes. At the end of the study period, there was no significant difference in LV volumes between groups. The study was not powered for mortality but no differences in survival were seen between the two groups.
EVEREST: The Efficacy of Vasopressin Antagonism in Heart Failure: Outcome Study with Tolvaptan (EVEREST) was a phase III clinical trial designed to study the short-term and long-term effects of tolvaptan (published separately as they were separate trials from a regulatory standpoint) in patients hospitalized with acute decompensated HF when added to standard therapy.10,11 A total of 4133 patients with NYHA III–IV HF with EF < 40% were randomized within 48 h of hospital admission to receive either tolvaptan or placebo in addition to standard therapy for a minimum of 60 days. For the short-term or hospitalization phase, the primary endpoint was a composite of global clinical status and body weight at 7 days. Secondary endpoints included the individual components of the primary endpoint, as well as measures of dyspnea and peripheral edema. Tolvaptan showed greater reduction in body weight at day 1 and day 7 (or at time of discharge). The primary endpoint was met, due to the effects on weight loss, but there was no difference in the clinical wellbeing scores. Tolvaptan also improved dyspnea and peripheral edema. Adverse event frequencies did not differ between groups, and there was no excess of renal failure or hypotension in the tolvaptan group. The long-term outcomes phase of the study featured dual primary endpoints: (1) all-cause mortality and (2) cardiovascular death or HF hospitalization. Secondary endpoints included signs and symptoms of vascular congestion, including dyspnea, body weight, and edema. Despite the apparent short-term clinical benefits, the long-term clinical outcomes did not show an effect on mortality or HF-related morbidity in patients whom were initiated on tolvaptan. A post hoc analysis of the hyponatremic patients in EVEREST; however, did show improved greater improvements in dyspnea early on with greater weight loss and improved survival in the patients with serum sodium <130 meq/l with tolvaptan. 26
TACTICS-HF: The Targeting Acute Congestion with Tolvaptan in Congestive Heart Failure Trial (TACTICS-HF) was designed to evaluate the use of tolvaptan versus placebo when added to standard diuretic therapy in patients admitted to the hospital with acute decompensated HF. 12 A total of 257 patients with HF regardless of EF were randomized without distinction. There was no difference in the primary outcome, which was a measure of the percent of patients who showed moderate or marked clinical improvement with tolvaptan as predefined by the investigators (without need for rescue therapy). There was more fluid loss at 24 h with tolvaptan, but no difference in dyspnea relief at the time of the primary endpoint, though differences emerged later. Renal function was assessed as a binary secondary endpoint, defined as a change in serum creatinine of >0.3 mg/dl within 72 h. While the tolvaptan group had significantly more patients that met this definition of worsening renal function at 72 h, this effect seems to be driven by a change in creatinine within the first 24 h (there was no difference in serum creatinine at 48 or 72 h).
SECRET of CHF: Study of the Short-Term Clinical Effects of Tolvaptan in Patients Hospitalized for Worsening Heart Failure with Challenging Volume Management (Secret of CHF) randomized 250 patients regardless of EF who were hospitalized within the past 36 h for acute decompensated HF with dyspnea and either impaired renal function, diuretic resistance, or hyponatremia. As in the other studies patients received either tolvaptan or placebo in addition to standard diuretic therapy. 13 The primary endpoint was a change in self-assessment of dyspnea measured at 8 h and 16 h after the initial dose of tolvaptan. While the primary endpoint was not met, there was ultimately a greater reduction in dyspnea in the tolvaptan group by day 3 of the study period. The tolvaptan group did show a greater weight reduction in this study, as seen in previous clinical trials.
AQUAMARINE: The clinical effectiveness of tolvaptan in patients with acute decompensated HF and renal failure was studied in the AQUAMARINE trial, which was a multicenter, randomized but not placebo-controlled clinical trial that enrolled 220 patients with acute decompensated HF regardless of EF with GFR between 15 and 60. 14 Patients were randomized to receive either usual care with a loop diuretic or tolvaptan adjunctive therapy. This study showed significant incremental weight loss and improvement in dyspnea without worsening renal function in the tolvaptan group.
The following two studies looked at tolvaptan as alternative therapy to loop diuretics in chronic and acute HF.
Udelson 2011: Tolvaptan as monotherapy was compared to furosemide and the combination of tolvaptan and furosemide for up to 7 days in 83 patients with NYHA II–III HF with EF < 40%. 15 In this study, 83 patients were randomized to either tolvaptan 30 mg, furosemide 80 mg, a combination of tolvpatan/furosemide, or placebo for 7 days in addition to standard therapy without fluid restriction. Tolvaptan monotherapy led to a reduction in body weight when compared to placebo which was not different than that produced by furosemide or the combination. While there was no change in urinary sodium levels between tolvaptan and furosemide, serum sodium levels were higher in the tolvaptan group for the first week after randomization, though levels returned to baseline after this. Plasma brain natriuretic peptide (BNP) levels were similar with furosemide and tolvaptan. After 1 week of therapy, aldosterone levels were suppressed in the tolvaptan group compared to the furosemide group, consistent with the expected renin–angiotensin system activation with loop diuretics, an effect not seen with V2 antagonism.
Jujo 2016: This was a pilot trial that compared tolvaptan with furosemide in acute HF. 16 A total of 60 patients who were hospitalized with acute decompensated HF were randomized to receive either 40 mg IV furosemide daily or oral tolvaptan for 5 days. At the end of the study period, weight loss and symptom relief were similar between the two groups. Plasma BNP levels and plasma catecholamines declined similarly in both groups. Plasma renin activity and aldosterone levels, serum creatinine, BUN, and BUN/creatinine ratios all increased in the furosemide group and did not change in the tolvaptan group.
Clinical trials with conivaptan
Only one clinical trial has been reported with conivaptan.
Goldsmith 2008: The efficacy and safety of conivaptan in the treatment of acute decompensated HF were studied in a double-blind, multicenter trial with 170 hospitalized patients who were randomized to receive either conivaptan or placebo. 18 Conivaptan was administered as a 20-mg IV loading dose followed by two successive 24-h continuous infusions of 40, 80, or 120 mg/d. There was significantly more urine output and greater weight loss in the conivaptan groups though there was no difference in patient or clinician assessments of symptoms. Serum creatinine and BUN were similar in the two groups despite the greater weight loss in the conivaptan group. There was no clinically significant hypotension in the conivaptan group.
Conclusions and implications for future study
Modulating neurohormonal imbalance in patients with HFrEF has been up to now the basis for all of the successful therapies that have improved patient survival. Prior to the introduction of the combination of angiotensin II antagonism and neprilysin inhibition (ARNI) therapy had been directed at antagonizing or inhibiting maladaptive neurohormonal systems. The use of ARNI also promotes the activity of beneficial neurohormonal activity. 27 While it is known that plasma AVP levels are elevated in HFrEF, it remains unclear at this point if this hormone plays a role in disease progression. As summarized in this review, we now have a clear understanding of the effects of pure V2 antagonism as adjunctive therapy in acute and chronic HFrEF with most of the evidence coming from short-term studies, and one large long-term outcomes study. Over the course of an admission for acute HF, V2 antagonism consistently improves decongestion as reflected in weight loss, with improvements in dyspnea at variable time points, and neutral or beneficial effects on renal function relative to loop diuretics alone. In patients with normal EF in the trials that included these patients the responses were not different from those with reduced EF. In HFrEF, however, adjunctive therapy with pure V2 antagonism did not reduce LV volumes or improve mortality with long-term use as shown in METEOR and the chronic phase of EVEREST.
A beneficial effect of blocking AVP signaling on LV remodeling and mortality would more likely be seen, however, with a V1a antagonist since reverse remodeling and reduced mortality are linked in prior studies of neurohormonal modulation, and the signaling pathway for the V1a receptor is analogous to that for angiotensin II. In the METEOR trial using a pure V2 antagonist, no effects were seen on remodeling (and while the trial was not powered for events, survival was not different between the groups). The failure to block the V1a receptor might also have undercut any potential benefit of V2 antagonism in the EVEREST trial since AVP levels rose in the tolvaptan group. 28 We have not had a V1a antagonist available for chronic use to test this hypothesis, either alone or in combination with a V2 receptor. Ideally, one would study a dual antagonist, as if AVP levels were displaced from receptor sites with a selective V1a antagonist, unwanted water retention could occur from enhanced V2 signaling in the kidney. Our only experiences with V1a antagonism suggest that the hemodynamic effects may be related to plasma levels as the SVR fell with a single injection of an early IV antagonist only in patients with increased AVP levels, while the only hemodynamic experience with a dual antagonist in patients with chronic HF but normal AVP levels showed no effects on CO, BP or SVR.5,17 The only clinical trial of a combined antagonist was of short duration, hemodynamics and AVP levels were not assessed, and the results resemble those seen with pure V2 antagonism. 18
The studies which have explored V2 antagonism as an alternative to loop diuretics are intriguing since they both showed that comparable decongestive effects were seen, suggesting that one does not require a natriuretic agent to produce clinically important decongestion. As well, in the Jujo report, plasma BNP levels declined comparably in both groups suggesting that despite radically different mechanisms of diuresis, ventricular wall stress is similarly affected. In both studies, tolvaptan was associated with a superior renal profile accompanied by major differences in activation of the renin–angiotensin–aldosterone axis. This finding was buttressed by the mechanistic study showing superior renal and neurohormonal effects of tolvaptan versus furosemide. 7 Given the consistently poor outcomes in acute HF using loop diuretics as the predominant decongestive therapy, further exploration of V2 antagonism as alternative therapy would certainly be justified. To avoid any unwanted effects of additional V1a stimulation; however, it might be best if a dual antagonist was used in such studies, though it could be that simply removing (or at least limiting) the adverse effects of loop diuretics could allow for benefit to be seen with a pure V2 antagonist alone.
This review has focused on the impact of AVP antagonism in HF. V2 antagonism has also been studied in euvolemic and hypervolemic hyponatremia, including that seen in HF. The pivotal study establishing the efficacy of tolvaptan for hyponatremia showed equivalent responses regardless of etiology of the hyponatremia. 29 Hyponatremia in chronic HF is common, though levels <130 meq/l are not common in acute HF. 11 Hyponatremia is associated with poor outcome in HF but whether it is just a marker for poor outcome or a contributor to the pathophysiology of HF is not clear. 30 Further study of this important issue is needed especially in view of the results of the post hoc analyses from ACTIV and EVEREST showing improved outcomes in the group with hyponatremia, in particular with serum sodium <130 meq/l.25,26 Whether hyponatremia is symptomatic in HF and whether correction of hyponatremia improves any symptoms or survival has not yet been addressed but would certainly seem to be justified based on the available data. 31
Worldwide, experience with AVP antagonism is conditioned by location and regulatory approval. Most of the HF studies have been in the United States or Japan. In the United States, regulatory approval for tolvaptan and conivaptan is confined to hypervolemic and euvolemic hyponatremia. Neither is approved for HF per se, despite tolvaptan in the inpatient phase of EVEREST having met its primary endpoint. In Japan, regulatory approval for tolvaptan includes edema in HF patients not responding to loop diuretics. The situation is evolving in other Asian countries regarding HF and hyponatremia. In Europe, tolvaptan is indicated only for euvolemic hyponatremia.
In conclusion, based on what we have to date it may be unlikely that further study of a pure V2 antagonist would show improved outcomes in the broad run of patients with HFrEF as adjunctive therapy though as noted, we do not have data on this approach in the absence of loop diuretics. In theory, blocking both receptors would perhaps make the most sense for the reasons discussed above. As noted earlier there is a new dual antagonist undergoing phase II studies during and after hospitalization for acute HF.19,20 A recent study with this agent in experimental HF suggests hemodynamic advantages compared with the pure V2 antagonist tolvaptan. 32 The main potential limitation to V1a antagonism or dual blockade would be hypotension but it is reassuring that in the experience with conivaptan in acute HF this did not occur, neither was it seen in the experimental study cited.18,32 Finally, in chronic HF outcomes are now quite good with the addition of ARNI therapy and it might be difficult to show benefit of a V2 antagonist as alternative therapy, or a dual antagonist as either alternative or adjunctive therapy without studying many thousands of patients. However, since outcomes after acute HF remain poor it may be possible to demonstrate potential benefits of either approach in this setting with many fewer patients.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Dr. Urbach declares no conflict of interest.Dr. Goldsmith has in the past received research support and speaking fees from Otsuka and Astellas Pharmaceuticals and currently receives consulting fees from Bayer Pharmaceuticals.
