Abstract
The prevalence of juvenile-onset gout has been increasing. Hereditary factors and secondary diseases should be considered in these patients. Adipsic diabetes insipidus (ADI) is characterized by arginine vasopressin (AVP) deficiency, which results in hypotonic polyuria, and dysfunction of thirst osmoreceptors, which results in failure to generate a thirst sensation in response to hypernatremia. We herein report a case of a boy with gouty arthritis, refractory hyperuricemia, prominent hypernatremia, a high creatinine concentration, and a history of surgery for a hypothalamic hamartoma. The patient was diagnosed with central diabetes insipidus after endocrine evaluation. Because he never had symptoms of thirst, the final diagnosis was corrected to ADI. This is the first report of gout due to chronic ADI in an adolescent. Volume contraction due to ADI might be one cause of hyperuricemia and renal impairment in such patients. Moreover, AVP deficiency might directly lead to low urate clearance due to the lack of vasopressin receptor 1 stimulation. Lack of polydipsia and polyuria may delay the diagnosis of ADI and lead to severe complications of a chronic hyperosmolar status. Sufficient and effective establishment of normovolemia is critical for these patients.
Keywords
Introduction
The prevalence of gout has been steadily increasing, and the age at onset has decreased.1,2 However, juveniles are not at high risk, and further research is therefore needed in such patients. Hereditary factors and secondary diseases should be considered in these patients. Adipsic diabetes insipidus (ADI) is a very rare disorder characterized by arginine vasopressin (AVP) deficiency, which results in hypotonic polyuria, and dysfunction of thirst osmoreceptors, which results in failure to generate a thirst sensation in response to hypernatremia. We herein report a case of gout due to chronic ADI, present the results of a literature review, and discuss the possible mechanisms of ADI-induced gout. This case is being reported to alert physicians to the possibility of encountering patients with this condition in clinical practice.
Case report
A 16-year-old male patient was admitted to the Peking Union Medical College Hospital in April 2017 with a 6-year history of hyperuricemia and a 3-year history of joint pain. In October 2010, the patient had undergone laboratory tests that showed an elevated serum uric acid (UA) level (533–647 µmol/L) and elevated creatinine (Cr) level (131 µmol/L); his previous test results in May 2009 showed that both the UA and Cr levels were normal. In October 2013, the patient suddenly developed pain in his right hip and could not walk. He had no fever or other joint pain. His symptom was relieved after 1 day without any treatment. No abnormalities were found on a radiographic examination, but the pain in his right hip intermittently recurred. Joint swelling and pain successively developed in the fourth right distal interphalangeal (DIP) joint, bilateral ankles, and bilateral first metatarsophalangeal (MTP) joints beginning in February 2015, with skin redness, increased skin temperature, and limited joint movement. Ibuprofen was effective, and the symptoms lasted for 1 to 3 days during each episode. The patient’s right hand then exhibited gradual deformation of the fourth proximal interphalangeal (PIP) joint. The symptoms became aggravated at the beginning of 2017, and he was transferred to our hospital in April of that year.
Routine laboratory tests revealed significantly elevated serum UA (925 µmol/L), sodium (Na) (178 mmol/L), and Cr (319 µmol/L) levels. Other laboratory findings revealed an elevated erythrocyte sedimentation rate (29 mm/h; reference range, 0–20 mm/h) and C-reactive protein level (42.58 mg/L; reference range, 0–3 mg/L); however, no abnormalities were found in the levels of antinuclear antibody, human leukocyte antigen B27, rheumatoid factor, anti-cyclic citrullinated polypeptide antibody, anti-keratin antibody, and anti-perinuclear factor antibody. Ultrasound examination showed bilateral renal diffuse lesions without urolithiasis. Radiographic examination showed visible bone erosion in the left first MTP and right second metacarpophalangeal (MCP) joint, soft tissue swelling, and joint deformation of the fourth finger of the right hand (Figure 1(b), (c)). Computed tomography showed mild bone destruction of the bilateral sacroiliac joints. The patient was given an intramuscular injection of betamethasone (5 mg) and was encouraged to drink at least 2 L of water every day. His symptoms were relieved in 2 days. Another test 1 week later revealed improved serum UA (631 µmol/L), Na (151 mmol/L), and Cr (173 µmol/L) levels.

Multi-joint malformation and bone destruction due to frequent acute flares of gouty arthritis. (a) Physical examination and (b) a radiograph of both hands and (c) a radiograph of both feet showed that the right third, fourth, and fifth fingers had flexion deformity (metacarpophalangeal, proximal interphalangeal, and distal interphalangeal joint malformation) and that the left first metatarsophalangeal joint was deformed. (d) Dual-energy computed tomography of both hands and feet showed a spot green marker only on the second metacarpophalangeal joint of the right hand and bone destruction in the joints of both (e) hands and (f) feet.
During the most recent 4 years before the current presentation, the patient had experienced nocturia. No significant change in his body weight had occurred. His medical history showed he had been born at full term and was postnatally normal until repeated seizures appeared in 2004. A tumor was found in the hypothalamus in 2008; that same year, he underwent resection of the tumor, which was confirmed to be a hamartoma. No further seizures appeared, but his intelligence development was slow. His parents were nonconsanguineous, his two younger brothers were healthy, and no other family member had gout or hyperuricemia.
Physical examination revealed that the patient had almost normal comprehension but poor computational and language abilities. Other nervous system examinations showed no obvious abnormality. A suspected tophus was found on the left auricle (Figure 2(a)). The right third, fourth, and fifth fingers showed flexion deformity (MCP, PIP, and DIP joint malformation) (Figure 1(a)). The left first MTP joint showed tenderness and deformity. The movements of the bilateral hips, knees, and ankles were limited, and the patient could not walk by himself.
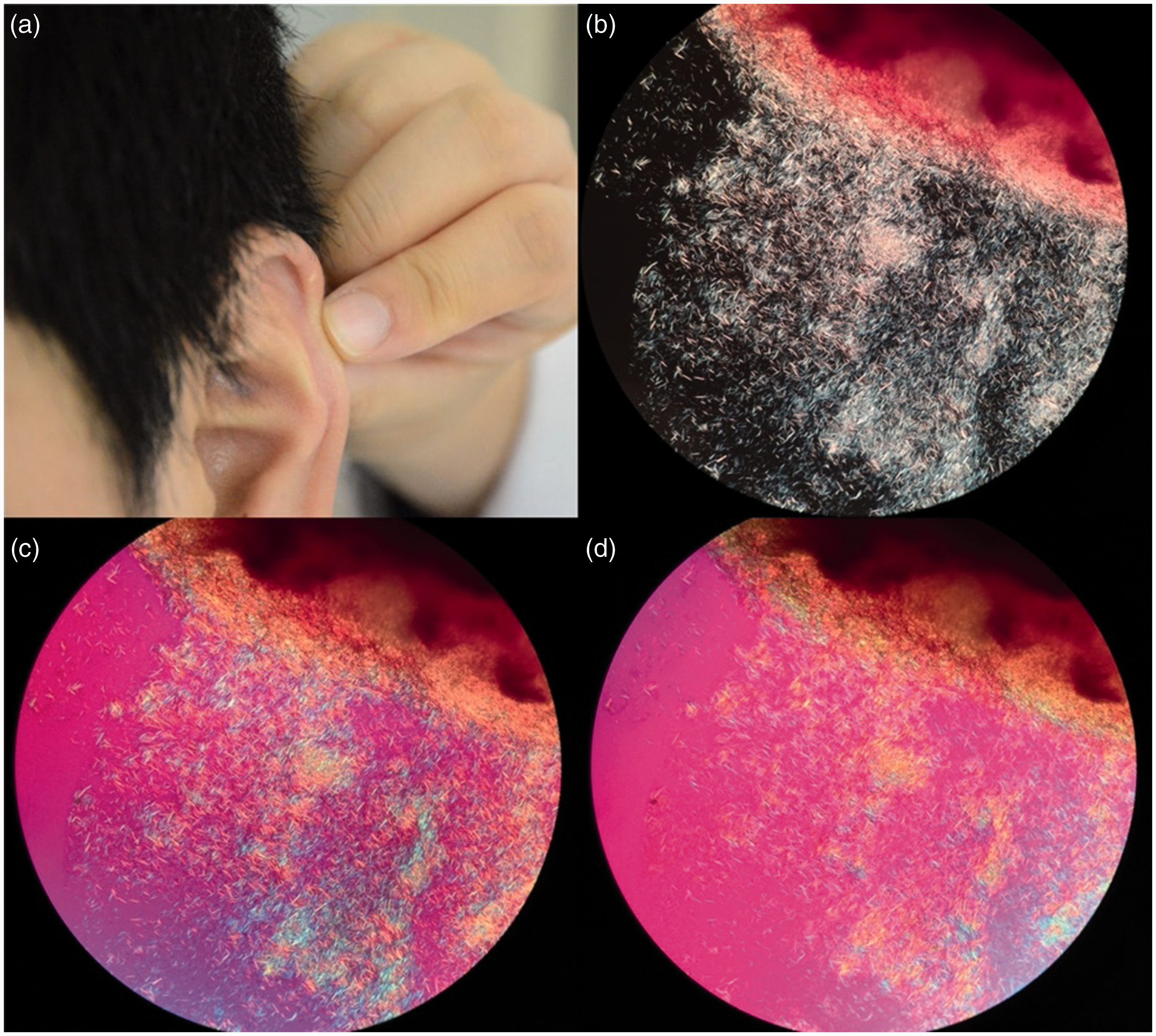
(a) Tophus on the auricle. (b–d) Polarized light microscopy showed numerous needle-like crystals with negative birefringence.
After admission, the suspected tophus on the auricle was punctured, and numerous needle-like crystals with negative birefringence were found under polarized light microscopy (Figure 2(b)–(d)). Dual-energy computed tomography showed a spot green marker only on the right second MCP joint (Figure 1(d)). Thus, the diagnosis of gout was established.
Other laboratory findings revealed impaired renal function (Cr, 160 µmol/L), high serum Na (149 mmol/L), and high UA (564 µmol/L). Other routine test results were normal, including the serum potassium level, glucose level, lipid level, liver function, erythrocyte sedimentation rate, and C-reactive protein level. The 24-hour urinary urate and Cr excretions were 2.364 and 15.24 mmol, respectively, and the 24-hour urinary urate clearance rate, Cr clearance rate, and fractional excretion of UA were 2.91 mL/minute, 66.15 mL/minute, and 4.4%, respectively. Endocrine evaluation was performed for the previous operation to remove the hypothalamic hamartoma. The patient’s anterior pituitary function was normal, including the levels of all thyroid hormones, growth hormones, insulin-like growth factor-1, adrenocorticotropic hormone, plasma and urine cortisol, and gonadotropins. However, his posterior pituitary function was abnormal: laboratory tests revealed low urine osmolality (274 mOsm/kg·H2O), slightly high plasma osmolality (315 mOsm/kg·H2O), and low urine specific gravity (1.005), all of which were consistent with central diabetes insipidus (CDI). Renal diabetes insipidus was ruled out by a fluid deprivation–vasopressin test. However, the patient had no symptoms of thirst, and his daily urine volume was only about 1000 mL. Thus, the diagnosis was corrected to ADI. The patient was given desmopressin (Minirin; Ferring Pharmaceuticals, Saint-Prex, Switzerland) at 0.05 mg once every 12 hours, and he increased his water intake to ensure a urine volume of >2 L/day. Ten days after admission, his laboratory test results significantly improved (serum UA, 289 µmol/L; Na, 140 mmol/L; Cr, 121 µmol/L). The 24-hour urinary excretion of urate and Cr were 1.978 and 16.71 mmol, respectively, and the 24-hour urinary UA clearance rate was 4.75 mL/minute, 24-hour urinary Cr clearance rate was 95.90 mL/minute, and fractional excretion of UA was 4.9%.
During hospitalization, the patient developed sudden-onset redness, swelling, heat, and pain in the left fourth DIP and second MCP. Acute flare of gout was diagnosed. The patient was treated with anti-inflammatory therapy (intramuscular injection of betamethasone, 5 mg), and his symptoms were relieved within the same day. Colchicine (0.5 mg twice a day) was given to prevent acute flares during urate-lowering therapy. Table 1 shows the changes the patient’s laboratory findings during the different stages of the disease.
Laboratory findings of the patient with juvenile-onset gout and adipsic diabetes insipidus.

UA: uric acid, Cr: creatinine, Na: sodium, ESR: erythrocyte sedimentation rate, CRP: C-reactive protein, ANA: antinuclear antibody, HLA: human leukocyte antigen, RF: rheumatoid factor, CCP: cyclic citrullinated polypeptide, AKA: anti-keratin antibody, APF: anti-perinuclear factor, FE.UA: fractional excretion of uric acid
After discharge, the patient was followed up every 2 months. His water intake was inadequate, and his serum levels of UA, Cr, and Na fluctuated. Additionally, his parents reported an acute gout flare. We educated the parents to ensure that the patient’s water intake was adequate. All symptoms and laboratory test results were stable at the last follow-up in October 2017.
This report was approved by the institutional review board of Peking Union Medical College Hospital, and informed consent for participation was obtained from the patient.
Discussion
Gout is a common disease that typically manifests as acute, self-limiting episodes of monoarthritis in the initial stage. After the development of chronic arthritis, tophi deposition leads to bone destruction, joint malformation, and joint dysfunction. The prevalence of gout has steadily increased, while the age at onset has decreased.1,2 Risk factors for gout include male sex, an age of >40 years, a high-purine diet, manifestations of metabolic syndrome, and cardiovascular and cerebrovascular diseases. No definite risk factors were present in our case, so hereditary factors and secondary diseases were considered.
Our patient had undergone surgical treatment of a hypothalamic hamartoma. Besides hyperuricemia, his severe laboratory abnormalities were prominent hypernatremia and a high Cr concentration. After further confirmation, the patient was diagnosed with CDI, which is a common disorder after brain surgery or trauma and in the presence of brain inflammatory diseases that influence the hypothalamus, where AVP (also known as antidiuretic hormone) is synthesized and secreted. CDI may be transient or permanent and can occur only a few days to years after surgery. 3 The fluid deprivation–vasopressin test is a useful method to establish a clear diagnosis in 83% of affected patients. The classic presenting symptoms are polyuria and polydipsia. However, some patients present with no thirst. 4 The patient in the present report had significant hypernatremia but never had symptoms of thirst; therefore, we changed the diagnosis to ADI. Arima et al. 5 reviewed 149 patients with CDI and identified 23 patients with adipsia. Six patients died during follow-up, among whom four were adipsic. ADI is a very rare disorder characterized by hypotonic polyuria due to AVP deficiency and failure to generate the sensation of thirst in response to hypernatremia. Adipsia implies damage to the circumventricular organs of the anterior hypothalamus, where the osmoreceptors for thirst are located. 6 Causes of ADI include surgery for treatment of an anterior communicating artery aneurysm, tumors and related surgery, neurosarcoidosis, head injury, intracranial inflammatory disease, hydrocephalus, and chemical exposure. ADI is associated with a high mortality rate because it can lead to several complications including rhabdomyolysis, seizures, central nervous system damage, thromboembolism, sleep apnea, and severe infections, some of which may be life-threatening.7–12
To the best of our knowledge, this is the first report of gout due to chronic ADI in an adolescent. After our literature review, we hypothesized two potential explanations for the relationship between gout and chronic ADI. In patients with CDI, the serum Na, Cr, and UA levels are often normal or slightly high. However, our patient had volume contraction due to adipsia and impaired renal function due to hypoperfusion. The serum UA level is partially dependent on renal clearance, which is influenced by different factors, among which establishment of effective normovolemia has a major influence.13–15 Our patient’s laboratory parameters were obviously improved after treatment with desmopressin and increased daily water intake. However, the serum UA level increased again during follow-up, and his urate clearance was very low after treatment. Therefore, another hypothesis is that AVP deficiency may directly lead to low urate clearance. Modulation of renal urate clearance occurs mainly in the proximal tubule, where it is coupled to Na reabsorption, while urea majorly relies on distal reabsorption. When patients with CDI are treated with desmopressin, about half of them remain hyperuricemic and present low urate clearance despite normonatremia and normal Cr clearance. 16 Thus, hyperuricemia in a patient with normonatremia, polydipsia, and polyuria is thought to be highly suggestive of CDI. 14 There are three types of vasopressin receptors: V1, V2, and V3. The V2 receptor is mainly distributed in the basement membrane of the collecting duct where it increases the reabsorption of water and concentrates the urine, playing the role of antidiuresis. Therefore, it is used for the treatment of CDI. Desmopressin specifically acts as a V2 receptor agonist. The V1 receptor mainly acts as a vasoconstrictor. It is also expressed in renal medullary interstitial cells and collecting duct epithelial cells, antagonizing the antidiuretic effect of the V2 receptor. The role of the V3 receptor is mainly to promote the release of adrenocorticotropic hormone and prolactin in the pituitary. In patients with CDI, lack of V1 and V2 receptor stimulation may contribute to low urate clearance. Studies have shown that urate clearance remained low in patients with CDI after treatment with Minirin. However, the urate clearance rate significantly increased when coupled with terlipressin (triglycyl-lysine vasopressin), a selective V1 receptor agonist.16,17 Therefore, the lack of V1 receptor stimulation could explain why patients with CDI present with hyperuricemia. Although the mechanism remains unclear, some hypotheses have been proposed. Stimulation of the V1a receptor in patients with syndrome of inappropriate antidiuretic hormone secretion (SIADH) induces downregulation of the reabsorption urate transporter together with upregulation of secretion urate transporters. 18 V1 receptor stimulation may affect systemic or intrarenal hemodynamics and thus indirectly influence proximal Na and urate reabsorption.16,17,19
Before admission, the serum UA level of our patient continued to increase with frequent fluctuations, which led to frequent acute gout flares and a continuous, severe whole-body inflammatory state. Therefore, bone destruction and joint deformity rapidly appeared. In future follow-ups, we will continue to monitor both the serum and urine UA levels and administer terlipressin if necessary.
In summary, ADI is a very rare disorder with a high mortality rate. Lack of polydipsia and polyuria may delay the diagnosis of ADI and lead to severe complications of a chronic hyperosmolar status. Establishment of sufficient and effective normovolemia is critical for these patients. Their fluid intake needs to be supervised daily.
Footnotes
Acknowledgements
We thank all of the physicians from the Division of General Internal Medicine of PUMCH who participated in the care of the patient.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Chinese Research Special Fund for Public Welfare Industry of Health (grant number 201502024).
