Abstract
In optimizing anticoagulation therapy, it is essential to balance treatment efficacy with the major adverse effect of anticoagulant treatment, bleeding risk. This narrative review examines the efficacy and safety of the non-vitamin K antagonist oral anticoagulants (NOACs) dabigatran, rivaroxaban, apixaban, and edoxaban compared with standard anticoagulation or placebo. NOAC therapies provide equivalent to superior protection versus standard therapy, with similar or superior safety, and potential benefits in convenience. We will review the phase III evidence for each of the available NOACs in different antithrombotic indications, including atrial fibrillation (in the absence of significant mitral stenosis or mechanical heart valves); prophylaxis of venous thromboembolism (VTE) in patients undergoing orthopedic surgery; and acute and long-term treatment of VTE. Further, we will illustrate scenarios in which the evidence is stronger for a particular agent in the context of the overall positive safety and efficacy profile of NOACs in general. Limitations of the factor Xa inhibitors include the lack of a specific antidote in case of a bleeding emergency (an approved agent is available for reversing the effect of the direct thrombin inhibitor). We discuss the options for mitigating bleeding and describe the ongoing developments towards specific reversal agents. In conclusion, the available data for efficacy and safety, together with reliable pharmacokinetics obviating the need for regular monitoring, indicate that NOACs may offer substantial benefits for patients with nonvalvular atrial fibrillation or VTE.
Keywords
Introduction
Classical anticoagulation approaches, while effective in reducing thrombus propagation, are hampered by several drawbacks. Heparins, in the form of unfractionated heparin or low-molecular-weight heparin (LMWH), must be administered parenterally, are associated with variable anticoagulant response, and carry a risk for heparin-induced thrombocytopenia, albeit small. 1 Vitamin K antagonists (VKAs), most notably warfarin, have been the standard of care for oral anticoagulation for over 50 years, but anticoagulation with VKA therapy has several specific limitations that can contribute to its underuse. 2 Multiple food and drug interactions exist, complicating VKA management. VKAs have a narrow therapeutic window, which contributes to bleeding risk and suboptimal anticoagulation in a significant number of patients. Frequent monitoring and dose adjustment to maintain treatment within the therapeutic range [which in the case of venous thromboembolism (VTE) and nonvalvular atrial fibrillation (NVAF) is an international normalized ratio (INR) 2.0–3.0] are imperative. 3 Although advanced systems have been developed to address these management concerns, even well-controlled trials report that the time in the therapeutic range is only ~60%.4–14 Factors such as these contribute to almost half of patients eligible for VKA use not receiving it. 15 Attempts were made to address these clinical shortcomings with ximelagatran, darexaban, and otamixaban, but these agents were unsuccessful.16–18 Subsequently, four non-vitamin K oral anticoagulants (NOACs; dabigatran, rivaroxaban, apixaban, and edoxaban) were developed and are now in use in the United States (US), European Union, and numerous other countries.19–27 A fifth agent, betrixaban, has been approved for the indication of VTE prophylaxis in medically ill patients. 28
The availability of the NOACs in recent years has broadened the choice of anticoagulants used to manage thrombotic disease and thromboprophylaxis. This advance mandates reassessment of the risk–benefit ratios used in decision making regarding treatment and prophylaxis of VTE disorders and in reducing the risk of stroke in patients with NVAF. Mandate is a strong word, not often used in the field of evidence-based medicine. However, the changes in pharmacokinetics, pharmacodynamics, and monitoring frequency represented by the new drugs have truly altered our calculus of risk–benefit ratios. The NOACs offer the potential for more convenient management without requiring routine laboratory monitoring.19–26 The need to optimally manage bleeding associated with these new agents prompts thorough examination of clinical trial evidence of the likelihood of each NOAC to cause bleeding events and the severity of such events when they occur. There are no trials directly comparing the NOACs, and therefore care must be taken when making comparisons between agents.
The aim of this review is to characterize the risk of bleeding associated with each of these agents (Figure 1 shows major bleeding rates across phase III trials5,6,8–14,29–43), and to provide a narrative of the published phase III efficacy and safety evidence for each of the NOACs available in the US across the different antithrombotic indications (Table 1). Additionally, we describe the results of subgroup analyses from these trials, with particular attention to results among elderly patients and those with decreased renal function, along with the agents available for the reversal of NOAC activity.

BID, twice daily; CrCl, creatinine clearance; DVT, deep vein thrombosis; NOAC, non-vitamin K antagonist oral anticoagulants; NVAF, nonvalvular atrial fibrillation; PE, pulmonary embolism; P-gp, P-glycoprotein; QD, once daily; SE, systemic embolism; VTE, venous thromboembolism.
Trials of NOACs for reducing the risk of stroke in patients with NVAF
Primary efficacy and safety outcomes
All of the evaluated NOACs (dabigatran, rivaroxaban, apixaban, and edoxaban) were either noninferior or superior to VKA therapy in reducing the risk of stroke or systemic embolism (SE) in patients with NVAF.4–7,30 The risk of major bleeding (generally defined in accordance with International Society of Thrombosis and Haemostasis standards (see captions to Table 2 and subsequent tables for details) in these patients was significantly reduced compared with VKAs by dabigatran 110 mg twice daily, 31 apixaban 5 mg twice daily, 5 and edoxaban 60 or 30 mg once daily, 7 with an equivalent risk of major bleeding for dabigatran 150 mg twice daily 31 and rivaroxaban 20 mg once daily. 6 In addition, apixaban was found to be superior to aspirin for reduction in the risk of stroke or SE in patients with NVAF with an equivalent risk of major bleeding. 30 Efficacy and bleeding results from the phase III trials of the NOACs for reducing the risk of stroke or SE in patients with NVAF are summarized in Table 2.4–7,30,31,45–47 Compared with standard warfarin therapy, each NOAC was associated with reduced rates of intracranial hemorrhage in patients with NVAF. However, dabigatran 150 mg twice daily, rivaroxaban 20 mg once daily, and edoxaban 60 mg once daily were associated with higher rates of gastrointestinal bleeding than warfarin, while edoxaban 30 mg once daily was associated with lower rates of gastrointestinal bleeding than warfarin.
Results of phase III trials of NOACs for reducing the risk of stroke in NVAF. In general, in patients with NVAF, clinical trial evidence indicates that the NOACs are as effective as standard therapy without the requirement for routine monitoring or dose adjustment; dabigatran 110 mg twice daily, apixaban, and edoxaban have shown safety advantages over standard therapy. Generally, NOACs are associated with reduced risks of intracranial hemorrhage compared with warfarin in patients with NVAF.

Numbers analyzed for efficacy (see original publications for numbers analyzed for safety or other populations).5–7 Shading indicates significant (p < 0.05) difference between treatments. All rows show NOAC versus warfarin, except AVERROES, which compared apixaban versus aspirin. All data are presented as annual rates per 100 patients; numbers within brackets indicate 95% confidence intervals (97.5% for ENGAGE AF-TIMI efficacy). Major bleeding was generally defined in accordance with International Society of Thrombosis and Haemostasis standards (fatal bleeding; bleeding causing a decrease in the hemoglobin level of ⩾2 g/dl within a 24-h period, or necessitating a transfusion of ⩾2 units of packed red cells; and/or symptomatic bleeding in a critical area or organ). 47 In ROCKET AF, bleeding that caused permanent disability was also included. 6 Additionally, in RE-LY, bleeding events requiring inotropic agents or surgery were included in life-threatening bleeding (a subcategory of major bleeding). 4 CRNM was defined in ROCKET AF as bleeding not meeting criteria for major bleeding but requiring medical intervention, unscheduled contact (visit or telephone) with a physician, temporary interruption of study drug (i.e. delayed dosing), pain, or impairment of daily activities; 6 in ENGAGE AF as a nonmajor bleed that required/prolonged hospitalization or required laboratory evaluation, imaging studies, nasal packing or compression, therapeutic procedure, interruption of study medication, or change in concomitant therapy; 46 and in ARISTOTLE and AVERROES as clinically overt bleeding that did not satisfy the criteria for major bleeding and that led to hospital admission, physician-guided medical or surgical treatment, or a change in antithrombotic therapy.5,30
BID, twice daily; CRNM, clinically relevant nonmajor; GI, gastrointestinal; HR, hazard ratio; INR, international normalized ratio; ITT, intent-to-treat; mITT, modified intent-to-treat; NOAC, non-vitamin K antagonist oral anticoagulant; NVAF, nonvalvular atrial fibrillation; OT, on treatment; PP, per protocol; QD, once daily; RR, relative risk; SE, systemic embolism.
Subgroup analysis of bleeding events
Dabigatran
In further analyses of bleeding outcomes in RE-LY, among 7258 patients aged ⩾75 years, those receiving dabigatran 150 mg were at increased risk of extracranial bleeding versus those receiving warfarin [relative risk (RR) 1.39; 95% confidence interval (CI) 1.13–1.70], while those receiving dabigatran 110 mg showed extracranial bleeding rates similar to those receiving warfarin. 48 No significant interaction was seen between treatment and creatinine clearance (CrCl) for major bleeding, suggesting that age-related decline in kidney function was not responsible for the different results in older patients. Further, no significant interactions were seen between the treatment effect of dabigatran versus warfarin and CHADS2 [Cardiac failure, Hypertension, Age >75 years, Diabetes, Stroke/transient ischemic attack (TIA) history (2 points)] category (0–1, 2, or 3–6) for the outcomes of major or intracranial bleeding; rates of both bleeding endpoints increased with CHADS2 score for patients receiving either dose of dabigatran or warfarin. 49
Among 4591 patients in RE-LY undergoing surgery, major bleeding rates were similar in warfarin-treated patients (4.6%) and in dabigatran-treated patients (3.8% and 5.1% for the 110-mg and 150-mg doses, respectively; p > 0.05 for both versus warfarin). 50 Dabigatran was last given an average of 49 (interquartile range, 35–85) hours before the procedure, versus an average of 114 (87–144) hours in patients receiving warfarin (p < 0.001).
Rivaroxaban
Among ROCKET AF patients in the ⩾75-years category (n = 6229), the hazard ratio (HR) for major bleeding (rivaroxaban versus warfarin) was 1.11 (95% CI 0.92–1.34); among the younger subgroup (n = 8035), the HR was 0.96 (95% CI 0.78–1.19); p = 0.34 for interaction. Hemorrhagic stroke rates were similar in both age groups; there was no interaction between age and rivaroxaban response. 51 No significant interaction was seen between CHADS2 category (2, 3, 4, 5, or 6) and treatment difference in major/clinically relevant nonmajor (CRNM) bleeding. 6 Patients with moderate renal insufficiency at enrollment (CrCl 30–49 ml/min; n = 2950) assigned to rivaroxaban received a reduced dose (15 mg). Rates of major bleeding were similar with rivaroxaban 15 mg and warfarin in patients with moderate renal insufficiency (4.49% versus 4.70%, respectively; HR 0.95; 95% CI 0.72–1.26), and rivaroxaban 20 mg and warfarin in patients with normal renal function (3.39% versus 3.17%; HR 1.07; 95% CI 0.91–1.26; p = 0.48 for interaction). 52
In the ROCKET AF study, 968 patients assigned to rivaroxaban and 1162 assigned to warfarin had a temporary interruption of study drug for purposes of a surgical/invasive procedure without adverse event bleeding; study drug was stopped ⩾3 days before the procedure in 90% of these cases. 53 Major bleeding occurred at similar rates in both groups (0.99/30 days and 0.97/30 days in rivaroxaban and warfarin groups, respectively; HR 1.02; 95% CI 0.50–2.06; p = 0.96).
Apixaban
In a subgroup analysis of ARISTOTLE by age (<65 years, 65–<75 years, and ⩾75 years), the rates of stroke, all-cause death, and major bleeding were shown to increase with increasing age; however, the benefit of apixaban versus warfarin was consistent, regardless of age. 54 This suggests that the absolute benefits of apixaban were greater in elderly patients with higher risks.
In analyses of data from ARISTOTLE, the level of renal function showed a significant interaction with treatment effect for the outcome of major bleeding (p = 0.03 for interaction); patients with severe or moderate renal impairment showed reduced major bleeding with apixaban versus warfarin (3.2% versus 6.4%, respectively) and those with no impairment showed similar rates of major bleeding between treatment groups (1.5% versus 1.8%, respectively). To note, patients with two out of three criteria, including age ⩾80 years, body weight ⩽60 kg, or serum creatinine level ⩾ 1.5 mg/dl (133 μmol/l) assigned to apixaban (n = 428) received a reduced (2.5 mg twice daily) dose. 5 Other subgroup analyses by CHADS2 score showed no significant interaction with treatment. Additional analyses found that apixaban consistently reduced major bleeding and intra-cranial hemorrhage versus warfarin across categories of risk level as indicated by CHADS2, CHA2DS2VASc (Congestive heart failure, Hypertension, Age ⩾75, Diabetes, Stroke/TIA, Vascular disease, Age 65–74, Sex), and HAS-BLED (Hypertension, Abnormal renal and liver function, Stroke, Bleeding, Labile INRs, Elderly, Drugs or alcohol) scores. 55
In ARISTOTLE, 5439 patients underwent 9260 invasive procedures within 7 days of having taken a dose of study drug; the study drug was interrupted before 5792 of these procedures. Major bleeding rates were 1.62% for apixaban and 1.93% for warfarin [odds ratio (OR) 0.846; 95% CI 0.614–1.166]. 56
In analyses of bleeding rates between aspirin and apixaban groups in AVERROES, RRs of major or CRNM bleeding were similar for apixaban and aspirin in key subgroups based on baseline characteristics. There were no significant interactions among age, sex, body mass index, study dose of aspirin or aspirin-placebo, or estimated glomerular filtration rate and randomized treatment for the outcome of major and CRNM bleeding. 57 Sites of bleeding events were similar with apixaban and aspirin.
Edoxaban
No significant interaction was seen between treatment (edoxaban 30 mg versus warfarin or edoxaban 60 mg versus warfarin) and subgroups defined according to age <75 or ⩾75 years in a subgroup analysis of ENGAGE AF for the primary safety endpoint of major bleeding. In another analysis, there was no significant effect modification by age group (<65, 65–74, and ⩾75 years) on the relative treatment effect of edoxaban 60 mg as compared with warfarin for this endpoint.7,58 Comparisons of the efficacy and safety of edoxaban and warfarin among patients <80 or ⩾80 years were similar to analyses considering the three age groups. The HRs for high-dose edoxaban versus warfarin for major bleeding were lower in patients with CrCl ⩾ 80 ml/min (HR 0.70; 95% CI 0.55–0.89), compared with patients with mild renal impairment (CrCl >50 and <80 ml/min; HR 0.90; 95% CI 0.74–1.08). 59 Among those with moderate renal dysfunction (CrCl 30–50 ml/min), the HR for patients randomized to the high-dose edoxaban regimen (the dose was reduced for 84% of these patients) was 0.76 (95% CI 0.58–0.98; p = 0.036), while among those with CrCl >50–95 ml/min the HR was 0.89 (95% CI 0.75–1.04; p = 0.15). 60
Summary
In general, in patients with NVAF, clinical trial evidence indicates that the NOACs are as effective as standard therapy without the requirement for routine monitoring or dose adjustment; dabigatran 110 mg twice daily, 31 apixaban, 5 and edoxaban 7 have shown safety advantages over standard therapy. There may be certain subpopulations or clinical situations in which assessment of exposure may prove useful (for instance, to test for compliance, or in the case of a bleeding event or VTE, overdose, or emergent surgical intervention). Generally, NOACs are associated with reduced risks of intracranial hemorrhage compared with warfarin in patients with NVAF.
Trials of NOACs for VTE prevention in orthopedic surgery
Primary efficacy and safety outcomes
The NOACs have been studied in several phase III trials for VTE prevention after orthopedic surgery; either total knee replacement (TKR) or total hip replacement (THR) as shown in Table 3.32–35,37–43,44,61 The majority of trials had the primary efficacy endpoint of VTE or all-cause death, with the exception of the edoxaban trials in which VTE alone was the primary efficacy endpoint. Dabigatran 220 mg once daily was found to be noninferior to enoxaparin 40 mg once daily for the primary efficacy outcome in the RE-NOVATE I, RE-NOVATE II, and RE-MODEL trials, but did not meet noninferiority criteria versus enoxaparin 30 mg twice daily in the RE-MOBILIZE trial (dabigatran 150 mg once daily was found to be noninferior to enoxaparin in RE-NOVATE I and RE-MODEL, but did not meet noninferiority in RE-MOBILIZE).32,33,35,38 Rivaroxaban 10 mg once daily was found to be superior to enoxaparin for the primary efficacy endpoint in RECORD-1, RECORD-2, RECORD-3, and RECORD-4 (enoxaparin was given 40 mg once daily in RECORD-1, -2, and -3, and 30 mg every 12 h in RECORD-4).34,39,40,44 In the ADVANCE-1 trial, apixaban 2.5 mg twice daily did not meet the criteria for noninferiority to enoxaparin 30 mg every 12 h for the primary efficacy endpoint, 41 while in the ADVANCE-2 and ADVANCE-3 trials, apixaban 2.5 mg twice daily was found to be superior to enoxaparin 40 mg once daily.42,43 In ADVANCE-1, apixaban 2.5 mg twice daily was associated with significantly lower rates of the combined endpoint of major and CRNM bleeding (2.9% versus 4.3%; p = 0.03). 41 Edoxaban 30 mg once daily was found to be superior to enoxaparin 2000 IU (equivalent to 20 mg) twice daily for the primary endpoint of symptomatic and asymptomatic deep vein thrombosis (DVT) and pulmonary embolism (PE) in the STARS-J-V and STARS E-3 trials.37,61 Rates of bleeding were generally similar in the NOAC and LMWH arms, expect for the reduction in bleeding seen with apixaban in ADVANCE-1. 41
Results from phase III clinical trials of VTE prevention in patients undergoing THR or TKR. Rates of bleeding were generally similar in the NOAC and LMWH arms, save for the reduction in bleeding seen with apixaban in ADVANCE-1.

Numbers analyzed for efficacy (see original publication for numbers analyzed for safety or other populations).32–35,37–42,44,61 The primary endpoint was VTE or all-cause deaths, with the exception of the STARS-J-V and STARS E-3 trials in which the primary endpoint was VTE. No deaths occurred in either of the STARS trials. Shading indicates significant (p < 0.05) difference between treatments. Major bleeding was generally defined in accordance with International Society of Thrombosis and Haemostasis standards (fatal bleeding; bleeding causing a decrease in the hemoglobin level of ⩾2 g/dl within a 24-h period or necessitating a transfusion of ⩾2 units of packed red cells; and/or symptomatic bleeding in a critical area or organ); bleeding necessitating reoperation was also included. In RE-NOVATE I and II, RE-MODEL, and RE-MOBILIZE, bleeding requiring treatment cessation was also included.32–35,37–43,44 In the ADVANCE trials, CRNM bleeding included acute, clinically overt episodes such as wound hematoma, bruising or ecchymosis, gastrointestinal bleeding, hemoptysis, hematuria, or epistaxis that did not meet the criteria for major bleeding.41–43 In the RECORD-4 publication, CRNM was defined as multiple-source bleeding, unexpected hematoma (>25 cm2), excessive wound hematoma, nose bleeding (>5 min), gingival bleeding (>5 min), macroscopic hematuria, rectal bleeding, coughing or vomiting blood, vaginal bleeding, blood in semen, intraarticular bleeding with trauma, or surgical-site bleeding. 44 In RE-NOVATE I and II, RE-MODEL, and RE-MOBILIZE, clinically significant (or CRNM) bleeding events were defined as spontaneous skin hematoma ⩾25 cm2, wound hematoma ⩾100 cm2, epistaxis >5 min, spontaneous macroscopic hematuria or that lasting >24 h if associated with an intervention, spontaneous rectal bleeding, gingival bleeding >5 min, and any other bleeding event judged as clinically significant by the investigator not qualifying as a major bleed.32,33,35,38 In STARS E-3, CRNM bleeding was defined as bleeding not meeting the criteria for major bleeding and included hematoma of ⩾5 cm in diameter, epistaxis or gingival bleeding lasting ⩾5 min, gastrointestinal bleeding, or gross hematuria persistent after 24 h of onset. 37 Definitions used in other trials were not published.
BID, twice daily; CRNM, clinically relevant nonmajor; GI, gastrointestinal; LMWH, low-molecular-weight heparin; mITT, modified intent-to-treat; NOAC, non-vitamin K antagonist oral anticoagulant; NR, not reported; QD, once daily; THR, total hip replacement; TKR, total knee replacement; VTE, venous thromboembolism.
The efficacy and safety for each agent are given in detail in Table 3.32–35,37–43,44,61 When interpreting the rates of major bleeding in the NOAC VTE prevention trials, it is important to note that the apixaban and dabigatran trials included surgical-site bleeding in their definition of major bleeding, whereas the rivaroxaban trials did not.
Subgroup analysis of bleedings
Dabigatran
Pooled results from the RE-NOVATE, RE-MOBILIZE, and RE-MODEL studies found a significant reduction in major bleeding with dabigatran 150 mg versus enoxaparin in patients >75 years of age (OR 0.38; 95% CI 0.15–0.99) despite higher rates of major bleeding in this subgroup across all treatment groups. 62 No significant differences were seen between dabigatran and enoxaparin in major bleeding rates in subgroups divided by renal function.
Rivaroxaban
In pooled results of the four RECORD studies comparing rivaroxaban and enoxaparin, no significant interaction between treatment and subgroup was seen for the endpoint of major and CRNM bleeding for subgroups stratified by age, sex, body weight, and CrCl. 63
Apixaban
In a pooled analysis of the ADVANCE-2 and ADVANCE-3 studies, univariate analyses identified statistically significant (at the prespecified p < 0.10 threshold) interactions for both age (p = 0.07) and body weight (p = 0.07) with treatment effect for the outcome of major bleeding, and for CrCl (p = 0.03) with treatment effect for the composite outcome of major and CRNM bleeding. 64 Apixaban was associated with absolute risk reductions versus enoxaparin in major bleeding of −0.41% (95% CI −0.90–0.08) in patients aged <65 years and −0.14% (95% CI −0.53–0.25) in patients >60 kg. However, apixaban was associated with absolute risk increases compared with enoxaparin of 0.13% (95% CI −0.49–0.75) in those aged 65–74 years, 1.02% (95% CI −0.18–2.22) in those aged ⩾75 years, and 1.04% (95% CI −0.02–2.10) in patients <60 kg. For major or CRNM bleeding, apixaban was associated with an absolute risk reduction of −2.46% (95% CI −4.21 to −0.70) in those with CrCl 51–80 ml/min, but an absolute risk increase of 1.17% (95% CI −3.21–5.55) in those with CrCl ⩽50 ml/min and 0.44% (95% CI −0.63–1.51) in those with CrCl >80 ml/min. It should be noted that patients with severe renal impairment made up a small (<5%) portion of the population.
Edoxaban
No data have been published discussing edoxaban use in renally impaired or elderly patients undergoing prophylaxis after orthopedic surgery.
Summary
Based on the available evidence, NOACs offer generally similar safety to enoxaparin in patients undergoing orthopedic surgery, and dabigatran may have a safety advantage over enoxaparin in patients over 75 years of age undergoing orthopedic surgery. Subgroup analyses of the ADVANCE trials suggest there may be a link between age, body weight, and renal function and the relative benefit of apixaban versus enoxaparin.
Trials of NOACs for VTE treatment
Primary efficacy and safety outcomes
The efficacy and safety results from phase III trials of NOACs for acute treatment of VTE are shown in Table 4 and summarized briefly below.8–10,12–14 With regard to efficacy in the phase III trials of NOACs for treatment of VTE (RE-COVER I and II, EINSTEIN-DVT and EINSTEIN-PE, AMPLIFY and Hokusai-VTE), dabigatran 150 mg twice daily, apixaban 10 mg twice daily for 7 days, followed by 5 mg twice daily, and edoxaban 60 mg once daily were found to be noninferior to VKA for the primary efficacy endpoint of recurrent VTE or VTE-related death and rivaroxaban 15 mg twice daily for 3 weeks, followed by 20 mg once daily was found to be noninferior to warfarin for the outcome of recurrent VTE alone.8–10,12–14 Differences in treatment regimens should be noted, in that the dabigatran and edoxaban protocols included a lead-in of at least 5 days of parenteral anticoagulation prior to NOAC treatment whereas the apixaban and rivaroxaban protocols did not require prior heparin treatment.
Results from phase III clinical trials of NOACs for acute VTE treatment. Compared with LMWH/VKA, in the general population of patients being treated for VTE, apixaban was associated with reduced major bleeding, and edoxaban was associated with reductions in the composite of major and CRNM bleeding.
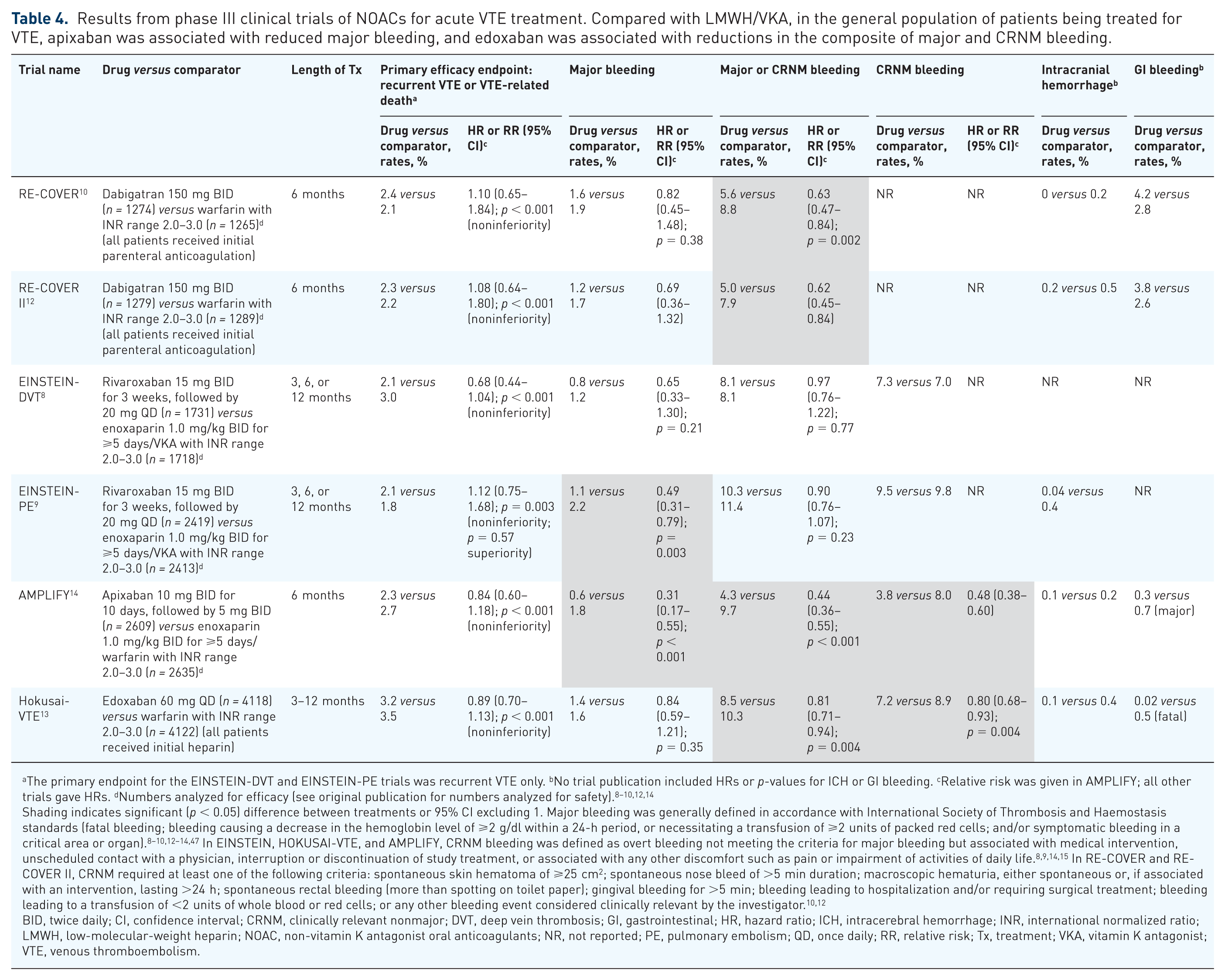
The primary endpoint for the EINSTEIN-DVT and EINSTEIN-PE trials was recurrent VTE only. bNo trial publication included HRs or p-values for ICH or GI bleeding. cRelative risk was given in AMPLIFY; all other trials gave HRs. dNumbers analyzed for efficacy (see original publication for numbers analyzed for safety).8–10,12,14
Shading indicates significant (p < 0.05) difference between treatments or 95% CI excluding 1. Major bleeding was generally defined in accordance with International Society of Thrombosis and Haemostasis standards (fatal bleeding; bleeding causing a decrease in the hemoglobin level of ⩾2 g/dl within a 24-h period, or necessitating a transfusion of ⩾2 units of packed red cells; and/or symptomatic bleeding in a critical area or organ).8–10,12–14,47 In EINSTEIN, HOKUSAI-VTE, and AMPLIFY, CRNM bleeding was defined as overt bleeding not meeting the criteria for major bleeding but associated with medical intervention, unscheduled contact with a physician, interruption or discontinuation of study treatment, or associated with any other discomfort such as pain or impairment of activities of daily life.8,9,14,15 In RE-COVER and RE-COVER II, CRNM required at least one of the following criteria: spontaneous skin hematoma of ⩾25 cm2; spontaneous nose bleed of >5 min duration; macroscopic hematuria, either spontaneous or, if associated with an intervention, lasting >24 h; spontaneous rectal bleeding (more than spotting on toilet paper); gingival bleeding for >5 min; bleeding leading to hospitalization and/or requiring surgical treatment; bleeding leading to a transfusion of <2 units of whole blood or red cells; or any other bleeding event considered clinically relevant by the investigator.10,12
BID, twice daily; CI, confidence interval; CRNM, clinically relevant nonmajor; DVT, deep vein thrombosis; GI, gastrointestinal; HR, hazard ratio; ICH, intracerebral hemorrhage; INR, international normalized ratio; LMWH, low-molecular-weight heparin; NOAC, non-vitamin K antagonist oral anticoagulants; NR, not reported; PE, pulmonary embolism; QD, once daily; RR, relative risk; Tx, treatment; VKA, vitamin K antagonist; VTE, venous thromboembolism.
The primary safety outcomes differed between trials, with the RE-COVER I, RE-COVER II, and AMPLIFY trials having major bleeding as the primary safety outcome, and the EINSTEIN-DVT/PE and Hokusai-VTE trials having major or CRNM bleeding. Dabigatran showed no significant difference versus warfarin in major bleeding in the RECOVER I and II trials (pooled analysis showed a reduction in major bleeding after the parenteral lead-in period).10,12 Significantly fewer instances of major bleeding were reported with apixaban versus LMWH/VKA in AMPLIFY. 14 Rivaroxaban showed no significant difference in rates of the composite of major and CRNM bleeding in patients with acute DVT or PE compared with standard therapy. This was true irrespective of whether patients experienced an acute DVT or an acute PE event.8,9 Edoxaban was found to be superior to warfarin for reduction in the risk of major or CRNM bleeding in Hokusai-VTE. 13
Patient subgroups
Dabigatran
Pooled data from the RE-COVER I and RE-COVER II trials showed that the risk reduction of major or any bleeding with dabigatran versus warfarin was similar across subgroups including CrCl, sex, ethnicity, body mass index, and previous VTE. 12 Analysis by age group (<75 years or ⩾75 years) found major bleeding, major or CRNM bleeding, and any bleeding were less frequent with dabigatran than warfarin in both age groups during the double-dummy, oral-only treatment period. Additionally, regression analysis showed no significant interaction between treatment and baseline CrCl for such bleeding events. 65
Rivaroxaban
Analysis of subgroups in the EINSTEIN-DVT and EINSTEIN-PE trials, including age and renal function found generally consistent results in both trials.8,9 A pooled analysis of data from both trials examined effects in subgroups including ‘fragile’ patients (fragility was defined as ⩾1 of the following criteria: age >75 years, CrCl <50 ml/min, or body weight ⩽50 kg). 66 In fragile patients (n = 1567), a statistically significant difference for major bleeding in favor of rivaroxaban (1.3%) versus standard therapy (4.5%) was observed (HR 0.27; 95% CI 0.13–0.54), while no such difference was seen in nonfragile patients (n = 6679; 0.9% versus 1.1%; p = 0.01 for interaction).
Apixaban
A subgroup analysis of the AMPLIFY trial showed that the RR of major bleeding was lower with apixaban compared with warfarin across age subgroups (<65, 65–<75, and ⩾75 years) with no significant treatment–subgroup interaction. 14
Edoxaban
A subgroup analysis of the Hokusai-VTE trial showed that age and renal function subgroups showed consistent benefit in major or CRNM bleeding risk for edoxaban over warfarin; however, a significant interaction was seen between treatment and sex, race, and center-level time in therapeutic range, with results suggesting greater benefit for edoxaban in male patients, Asian patients, and patients at centers at which the time in therapeutic range for warfarin was <60%. 13
Summary
Compared with standard therapy, in the general population of patients being treated for VTE, apixaban was associated with reduced major bleeding, and edoxaban was associated with reductions in the composite of major and CRNM bleeding.
Trials of NOACs for VTE extended therapy
Overall, five trials have reported the efficacy and safety results of the NOACs for the extended treatment of VTE (Table 5); however, it should be noted that the study designs of these trials differ between agents.8,11,29,67 Rates of bleeding were low in all trials investigating the use of NOACs for the extended treatment of VTE.8,11,29,67 Dabigatran 150 mg twice daily was noninferior to warfarin for the outcome of VTE or VTE-related death in patients at increased risk of recurrent VTE who had previously received 3–12 months of anticoagulation (RE-MEDY), and significantly reduced the risk of recurrent or fatal VTE or unexplained death compared with placebo in patients who had completed 6–18 months of treatment (RE-SONATE); both studies enrolled patients who had been treated with an approved anticoagulant or had received dabigatran in RE-COVER or RE-COVER II. 11 In RE-MEDY, major bleeding was rare in both the dabigatran and warfarin groups; dabigatran was associated with less major or CRNM bleeding compared with warfarin. 11 Major bleeding was rare in the dabigatran and placebo groups in RE-SONATE, and dabigatran was associated with significantly more major or CRNM bleeding compared with placebo. 11 After an initial 6–12 months of anticoagulant treatment, rivaroxaban 20 mg once daily significantly reduced the risk of recurrent VTE compared with placebo in the EINSTEIN extension, and rivaroxaban 20 mg and 10 mg both significantly reduced the risk of recurrent VTE compared with aspirin 100 mg in EINSTEIN CHOICE.8,67 Major bleeds were rare across groups in both trials; rivaroxaban was associated with an increased rate of first major or CRNM bleeds compared with placebo (6.0% versus 1.2%; p < 0.001), with no significant difference in major or CRNM bleeding rate between aspirin and rivaroxaban 20 mg or 10 mg.8,67 Both doses of apixaban (2.5 mg and 5 mg twice daily) reduced the risk of recurrent VTE or all-cause death after an initial 6–12 months of treatment compared with placebo in the AMPLIFY-EXT trial. 29 Major bleeds were again rare; both the 2.5-mg and the 5-mg dose of apixaban achieved efficacy without increasing major or CRNM bleeding (3.2%, 4.3%, and 2.7% for apixaban 2.5 mg, 5 mg, and placebo, respectively). 29
Results from phase III clinical trials of long-term treatment with NOACs for prevention of VTE recurrence. Rates of major bleeding were low across NOAC trials for the long-term treatment of VTE, with no subgroups identified that had an effect on the rate of major bleeding in any of the NOAC trials. Overall, dabigatran and rivaroxaban, but not apixaban, were associated with an increased rate of major or CRNM bleeding versus placebo.
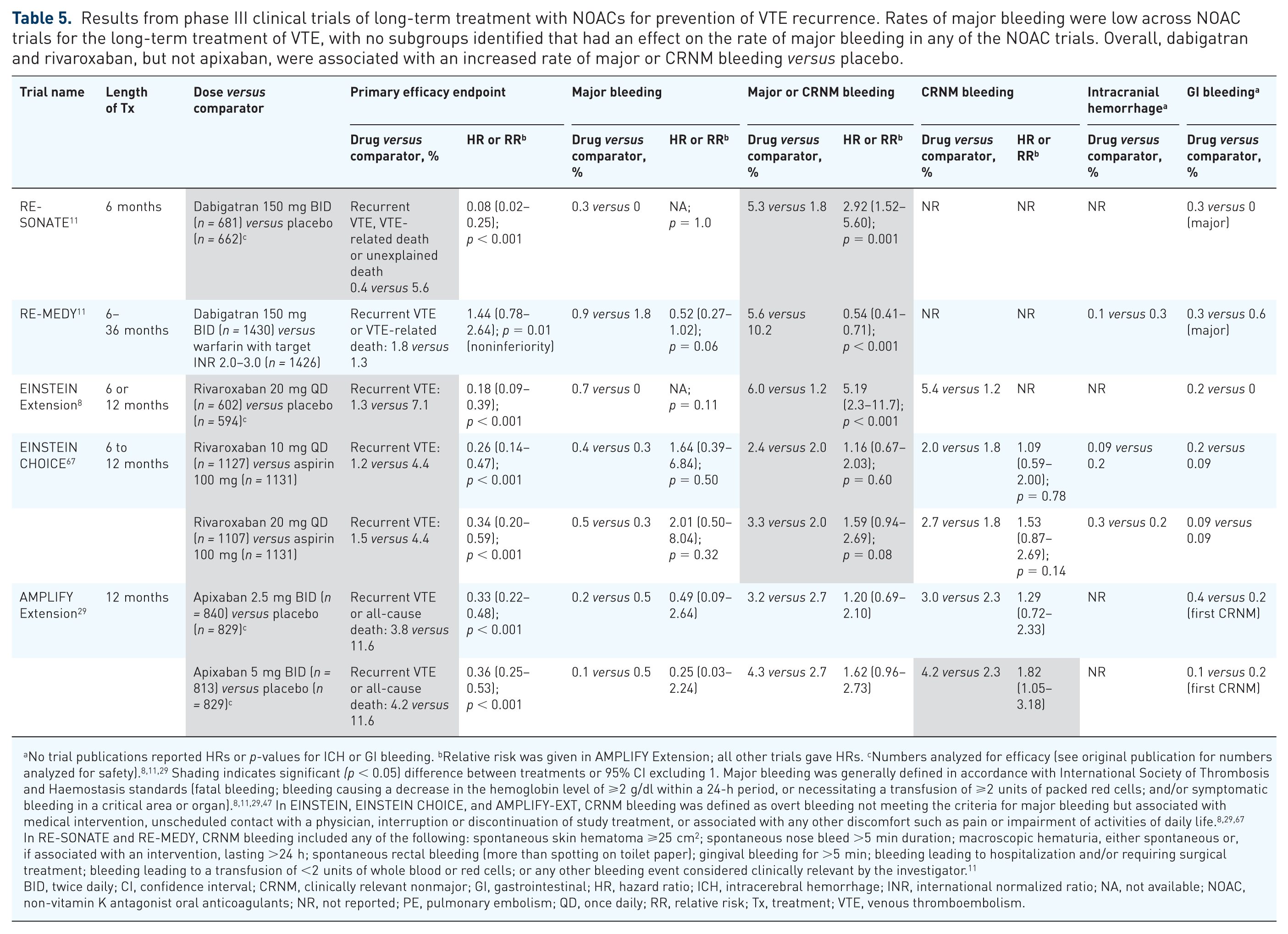
No trial publications reported HRs or p-values for ICH or GI bleeding. bRelative risk was given in AMPLIFY Extension; all other trials gave HRs. cNumbers analyzed for efficacy (see original publication for numbers analyzed for safety).8,11,29 Shading indicates significant (p < 0.05) difference between treatments or 95% CI excluding 1. Major bleeding was generally defined in accordance with International Society of Thrombosis and Haemostasis standards (fatal bleeding; bleeding causing a decrease in the hemoglobin level of ⩾2 g/dl within a 24-h period, or necessitating a transfusion of ⩾2 units of packed red cells; and/or symptomatic bleeding in a critical area or organ).8,11,29,47 In EINSTEIN, EINSTEIN CHOICE, and AMPLIFY-EXT, CRNM bleeding was defined as overt bleeding not meeting the criteria for major bleeding but associated with medical intervention, unscheduled contact with a physician, interruption or discontinuation of study treatment, or associated with any other discomfort such as pain or impairment of activities of daily life. 8,29,67 In RE-SONATE and RE-MEDY, CRNM bleeding included any of the following: spontaneous skin hematoma ⩾25 cm2; spontaneous nose bleed >5 min duration; macroscopic hematuria, either spontaneous or, if associated with an intervention, lasting >24 h; spontaneous rectal bleeding (more than spotting on toilet paper); gingival bleeding for >5 min; bleeding leading to hospitalization and/or requiring surgical treatment; bleeding leading to a transfusion of <2 units of whole blood or red cells; or any other bleeding event considered clinically relevant by the investigator. 11
BID, twice daily; CI, confidence interval; CRNM, clinically relevant nonmajor; GI, gastrointestinal; HR, hazard ratio; ICH, intracerebral hemorrhage; INR, international normalized ratio; NA, not available; NOAC, non-vitamin K antagonist oral anticoagulants; NR, not reported; PE, pulmonary embolism; QD, once daily; RR, relative risk; Tx, treatment; VTE, venous thromboembolism.
Subgroup analysis of bleedings
Dabigatran
No significant differences in the risk of bleeding according to study treatment in predefined subgroups were seen in RE-MEDY; subgroup bleeding results were not presented in RE-SONATE. 11
Rivaroxaban
The relative safety in the EINSTEIN extension study was generally consistent across subgroups, although 25/371 (6.7%) of patients on rivaroxaban with normal renal function (CrCl ⩾80 ml/min) had major or CRNM bleeding versus 3/373 (0.8%) of patients on placebo; corresponding rates among patients with CrCl <50 ml/min were 1/37 (2.7%) for rivaroxaban and 2/49 (4.1%) for placebo. 8 In EINSTEIN CHOICE, subgroup analyses of the composite outcome of major or CRNM bleeding provided results that were consistent with the overall treatment effects. 67 However, among patients with CrCl ⩾80 ml/min rates of major or CRNM bleeding were 21/774 (2.7%) versus 13/790 (1.6%) for rivaroxaban 10 mg and aspirin 100 mg respectively, while corresponding rates among patients with CrCl <50 ml/min were 0/51 (0%) for rivaroxaban 10 mg versus 4/64 (6.3%) for aspirin 100 mg. 67
Apixaban
RRs indicated no significant difference in major and CRNM bleeding between either apixaban dose and placebo for all predefined subgroups, including those defined by age or renal function. 29
Summary
Rates of major bleeding were low across NOAC trials for the long-term treatment of VTE, with no subgroups identified that had an effect on the rate of major bleeding in any of the NOAC trials. Overall, dabigatran and rivaroxaban, but not apixaban, were associated with an increased rate of major or CRNM bleeding versus placebo. Subgroup analyses suggested an effect of renal function on major or CRNM bleeding for rivaroxaban.
Betrixaban in medically ill patients
An additional recent trial (APEX) evaluated another NOAC, betrixaban 80 mg once daily, compared with enoxaparin 40 mg once daily for the prevention of VTE among acutely ill medical patients. 68 Among the initial cohort of patients with an elevated baseline D-dimer level, the composite endpoint of asymptomatic proximal DVT (days 32–47), symptomatic proximal or distal DVT, symptomatic nonfatal PE, or death from VTE (days 1–42) occurred in 6.9% and 8.5% of the betrixaban and enoxaparin groups, respectively (RR 0.81; 95% CI 0.65–1.00; p = 0.054). Major bleeding in the safety population occurred at rates of 0.7% and 0.6% in the betrixaban and enoxaparin groups, respectively (RR 1.19; 95% CI 0.67–2.12; p = 0.55). It should be noted that this included major bleeding until 7 days after discontinuation of study medication, and that betrixaban was given for 35–42 days compared with 10 ± 4 days for enoxaparin.
NOAC reversal strategies
NOACs have relatively short half-lives and their anticoagulant effects are significantly reduced within 24 h of the last dose.19–26 However, some situations can require emergency reversal; the International Society for Thrombosis and Haemostasis identifies ‘life-threatening bleeding, bleeding into a critical organ or closed space, prolonged bleeding despite local hemostatic measures, high risk of recurrent bleeding because of overdose or delayed clearance of [NOACs], and need for an urgent intervention associated with a high risk of bleeding.’ 69 In such emergency cases, validated, specific antidote therapy is available for the direct thrombin inhibitor dabigatran. 70 Idarucizumab, a humanized monoclonal antibody fragment, was approved in 2015 for the use in patients treated with dabigatran when reversal of the anticoagulant effect is needed for emergency surgery or urgent procedures, and in life-threatening or uncontrolled bleeding.70,71 A recombinant protein (andexanet alfa) for the reversal of factor Xa inhibitors, including the LMWH enoxaparin, an indirect factor Xa inhibitor, has recently been approved,72–74 and a small synthetic molecule (PER977, also known as ciraparantag or aripazine) is in development for the reversal of all the NOACs as well as unfractionated heparin and LMWH.75–77
Other potential approaches for reversing the anticoagulant effects of NOACs have been proposed. Fresh frozen plasma is not likely to be of benefit. 78 Strategies such as prothrombin complex concentrate (PCC), activated PCC (aPCC), or recombinant factor VIIa (rFVIIa) have not been proven effective in clinical trials to reverse emergency bleeding or overdose associated with NOAC use. However, cohort studies have found PCC to be effective in most cases for reversing major bleeds in patients who had received dabigatran or apixaban, and that aPCC might provide benefit over supportive care in some patients with major bleeding who had received dabigatran.79,80 There has also been positive human in vitro evaluation of aPCC for reversal of dabigatran, evidence for reversal of rivaroxaban by PCC in healthy volunteers and by aPCC in vitro, and in vitro evidence for PCC to reverse the effects of apixaban; additionally, in vitro evidence for rFVIIa exists for all three agents.19–22,25,26,81–84 It has been shown that PCC, aPCC, and rFVIIa reverse the effect of edoxaban on prothrombin time in a rat model. 85 rFVIIa and factor VIII inhibitor bypass activity rapidly reverse edoxaban-mediated anticoagulation effects as per prothrombin time and activated partial thromboplastin time in ex vivo human blood, but have minimal effect on intrinsic factor X activity. 86
While idarucizumab (if readily available) is the preferred approach for life-threatening bleeding in a patient who has taken dabigatran, expert recommendations and European Heart Rhythm Association guidelines suggest that PCC or aPCC, starting at a dose of 50 U/kg, can be considered in a patient with life-threatening bleeding who has taken NOAC therapy if immediate hemostatic support is required.27,78 Oral activated charcoal may be considered for reducing the absorption of apixaban or rivaroxaban.19,20,25,26,87 In healthy volunteers, activated charcoal reduced the apixaban area under the concentration–time curve (AUCINF) by 50% and 27% when administered 2 h and 6 h after a 20-mg dose, respectively;20,87 activated charcoal has also been shown to reduce dabigatran concentrations in vitro.88,89 Hemodialysis can remove dabigatran; based on limited clinical evidence including case studies, this may be associated with a reduction in duration and/or severity of bleeding.21,90 Apixaban, rivaroxaban, and edoxaban are not dialyzable.19,20,23–26
Conclusions
While the majority of patients may be anticoagulated with VKAs with reasonable efficacy, management of these agents is complicated by the need for frequent phlebotomy, dose adjustment, and drug and dietary restrictions. The narrow therapeutic window of VKA therapy also makes anticoagulation with these agents unsuitable for a substantial number of patients. NOAC therapies are established to provide equivalent to superior protection in multiple indications, with similar or superior safety without these inconvenient drawbacks and limitations. In NVAF trials, dabigatran 110 mg, apixaban, and edoxaban reduced the risk of major bleeding compared with VKA therapy, while the risk of major bleeding seen with dabigatran 150 mg and rivaroxaban was similar to that with VKA, and apixaban had a risk of major bleeding similar to aspirin. Compared with warfarin, all NOAC therapies have a marked impact in reducing the risk of the most dreaded bleeding complication, intracranial hemorrhage, in patients with NVAF. When compared with standard therapy for prophylaxis of VTE in patients undergoing orthopedic surgery, NOACs have generally been associated with comparable bleeding rates. In VTE treatment trials, dabigatran, edoxaban, and apixaban each reduced the risk of major and CRNM bleeding compared with standard therapy; major bleeding was reduced versus standard therapy by rivaroxaban (in patients with PE) and by apixaban. The safety and convenience benefits associated with NOAC therapy, as well as the emergence of specific antidotes, may prove useful in ensuring that patients who are appropriate candidates for oral anticoagulation receive adequate treatment.
Footnotes
Acknowledgements
Professional medical writing and editorial assistance was provided by Robert Coover, MPH, CMPP, and Nicole Draghi, PhD, CMPP, at Caudex, and was funded by Bristol-Myers Squibb and Pfizer. The authors also acknowledge David Kuten, MD, of Texas Heart Institute and Baylor St. Luke’s Medical Center, TX, USA for his contributions to the conception and design of previous drafts of the manuscript.
A. Bracey, W. Shatila, and J. Wilson contributed to the conception and design of this study; contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
Professional medical writing and editorial assistance was funded by Bristol-Myers Squibb, Princeton, NJ, and Pfizer, New York, NY.
Conflict of interest statement
Arthur Bracey is on the speaker’s bureau for Bristol-Myers Squibb and Pfizer. Wassim Shatila and James Wilson have nothing to disclose.

