Abstract
Objectives:
Randomized acute coronary syndrome (ACS) trials testing various antithrombotic (AT) regimens have largely excluded patients with coexisting conditions and indications for anticoagulation (AC). The purpose of this study is to examine the 2-year clinical outcomes of patients with ACS with indication for AC due to venous thromboembolism (VTE) during hospitalization for the ACS event or a prior or new diagnosis of atrial fibrillation (AF) with a CHADS2 (Congestive heart failure; Hypertension; Age; Diabetes; previous ischemic Stroke) score ⩾2.
Methods:
ACS patients with AC indication from 2004 to 2009 were identified (
Results:
A total of 261 (42.2%) patients had a MACE event. AT strategy was not a significant factor for MACE (all
Conclusions:
In this real world observational study, the at-discharge AT strategy was not significantly associated with the 2-year risk of MACE. These findings do not negate the need for randomized trials to generate evidence-based approaches to management of this important population.
Introduction
Current guidelines recommend the use of aspirin and a thienopyridene, or ‘dual antiplatelet therapy’ (DAP) for the majority of patients following an acute coronary syndrome (ACS) [Vandvik et al. 2012]. For the most part, physicians follow these recommendations [Zeymer et al. 2013; Maier et al. 2014; Potpara et al. 2014; Knight et al. 2015]. However, many ACS patients have comorbidities [e.g. atrial fibrillation (AF) and venous thromboembolism (VTE)] for which guidelines also recommend chronic oral anticoagulation (AC) [Kearon et al. 2012; You et al. 2012; Lip et al. 2014]. The well-documented increase in the risk of major bleeding with combined DAP and AC [Orford et al. 2004; Mattichak et al. 2005; Khurram et al. 2006; Lamberts et al. 2012; Dewilde et al. 2013; Rubboli et al. 2014] often accounts for physicians’ reluctance to prescribe three antithrombotic (AT) agents and instead use more conservative strategies. A recent observational study confirmed that ACS patients prescribed AC medication were three times more likely to be prescribed a single antiplatelet (SAP) than DAP therapy [Knight et al. 2015]. However, there is evidence that use of combined DAP and AC is on the rise [Maier et al. 2014] and currently between 40% and 50% of patients with both ACS and AF are prescribed DAP and AC [Maier et al. 2014; Potpara et al. 2014]. The long-term outcomes of various AT strategies in these complex patients with ACS and comorbid AF or VTE are lacking. Indeed, randomized ACS trials testing various AT regimens have largely excluded these patients.
In order to better understand the use patterns and consequences of AT agents used in ACS patients, we studied the 2-year risk of a composite endpoint of major adverse cardiovascular events (MACE) including death, myocardial infarction (MI) and cerebrovascular accident (CVA) in ACS patients with documented coexisting indications for AC. We hypothesized that the choice of AT strategy at discharge would impact the likelihood of subsequent MACE. To test this hypothesis, a cohort of ACS patients with indications for AC within an integrated healthcare system was defined by the onset of ACS and subsequent endpoints were identified by review of comprehensive electronic medical records. Further, we examined the risk of major bleeding events based on the AT strategy at discharge from the index ACS event.
Methods
This study was approved by the Intermountain Healthcare institutional review board. Investigations were in accordance with the Declaration of Helsinki.
Study population:
The study employed an observational cohort design using electronic data from the Intermountain Heart Collaborative Study (IHCS) registry [Taylor et al. 1998] and the Intermountain Enterprise Wide Data Warehouse (EDW). The subjects were ACS patients in the IHCS registry with an indication for AC therapy for the years 2004 to 2009 (
Primary endpoints
The primary efficacy outcome was a composite of MACE within 2 years of the index hospitalization. Major events included death (all-cause), MI or CVA. Death was determined by linkage of Intermountain Healthcare patient records to both the Utah Department of Health Death Certificate database and the Social Security Death Master File. Cardiovascular (CV) death was defined using ICD10 coded cause of death codes from the Utah Department of Health Death Certificate data (Supplementary Table 1). Nonfatal MI and CVA were determined using subsequent visits in the Intermountain Healthcare System.
The safety endpoint was major bleeding during the 2-year follow-up period. Major bleeding was defined by the following criteria: symptomatic bleeding, defined using ICD9 codes (Supplementary Table 1) in a critical area or organ (such as intracranial bleeding, intraspinal intraocular, retroperitoneal, intra-articular or pericardial, or intramuscular with compartment syndrome) and/or transfusion of two or more units of packed red blood cells.
Patient and clinical characteristics
Patient and clinical data including age, sex, cardiac risk factors, clinical presentation diagnosis, medications and procedures were electronically derived from the Intermountain Healthcare Enterprise Data Warehouse (EDW). Comorbidities were defined based on ICD9 codes at prior visits in the Intermountain Healthcare System or at the time of the index hospitalization, except for hypertension and hyperlipidemia which were derived from physician notes. The burden of comorbidities was calculated using the Charlson comorbidity index [Charlson et al. 1987]. The risk of mortality was calculated for each study subjects using the Intermountain Risk Score (IMRS) [Horne et al. 2009]. The IMRS is calculated by weighting various levels of complete blood count (CBC) and basic metabolic profile (BMP) laboratory values from the index hospitalization. The score was stratified by low/moderate
Statistical analysis
The primary efficacy outcome was MACE (all-cause death, MI or CVA) within 2 years of index hospitalization and the primary explanatory variable of interest was at-discharge AT therapy following the index hospitalization. Kaplan–Meier curves were generated for the primary efficacy outcome of MACE by at-discharge AT therapy. A landmark analysis of 0–6 months and 6–24 months was also performed. A landmark analysis sets a fixed time point (for this study 6 months) and looks at survival responses before and after this time point [Dafni, 2011]. Kaplan–Meier curves were generated for these landmark analyses. Univariate Cox proportional hazards models with primary efficacy outcome of MACE and demographic and clinical characteristics were performed. In addition to at-discharge AT therapy, significant factors from the univariate analyses (
SAS 9.2 (SAS Institute, Cary, North Carolina) and R 2.15.3 (Comprehensive R Archive Network, CRAN) were used for the analyses. All analyses results were considered statistically significant at
Results
There were 619 subjects; 547 (88.3%) had AF with CHADS2 score ⩾2 and 103 (16.6%) had a VTE during the ACS hospitalization. Table 1 shows the demographic and clinical characteristics of the subjects stratified by MACE. There were a total of 261 (42.2%) patients with a MACE event in the 2 year period. Of these 155 (59.3%) were deaths (all-cause), 36 (13.7%) were nonfatal CVA and 136 (52.1%) were nonfatal MI. The breakdown of the MACE events by at discharge AT therapy is shown in Table 2.
Subject and clinical characteristics by major adverse cardiovascular events (MACE).

AC, anticoagulation; ACE, angiotensin converting enzyme; AF, atrial fibrillation; CABG, coronary artery bypass graft; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; CVD, cardiovascular disease; DAP, dual antiplatelet therapy; ICH, intracranial hemorrhage; IMRS, Intermountain Risk Score; MACE, major adverse cardiovascular events; MI, myocardial infarction; PCI, percutaneous coronary intervention; SAP, single antiplatelet therapy; STD, standard deviation; STEMI, ST segment elevation myocardial infarction; VTE, venous thromboembolism.
Outcomes by at-discharge AT strategy.
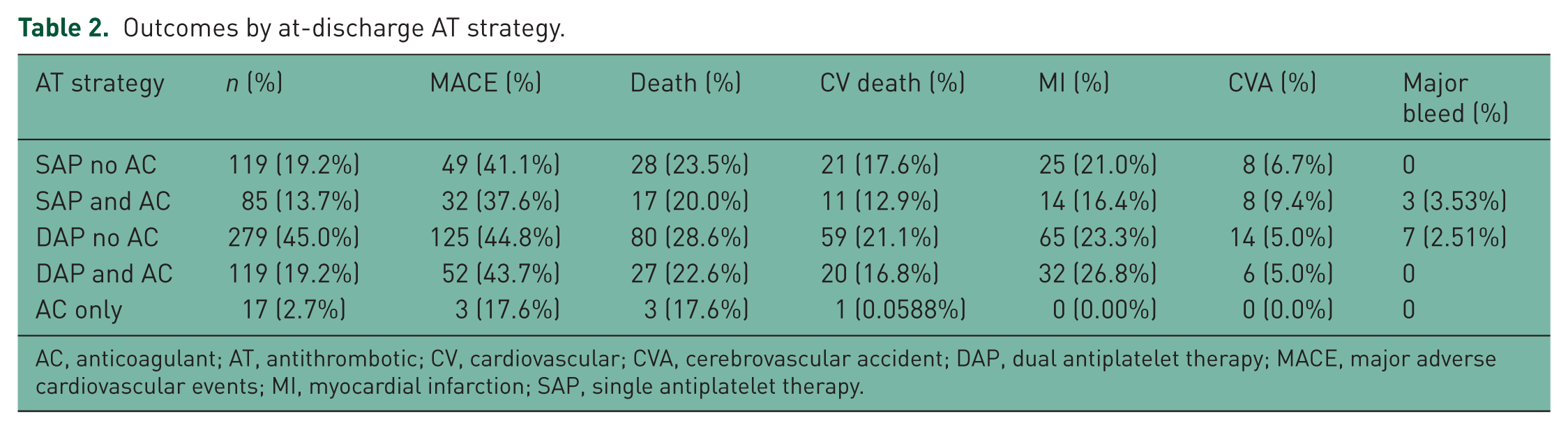
AC, anticoagulant; AT, antithrombotic; CV, cardiovascular; CVA, cerebrovascular accident; DAP, dual antiplatelet therapy; MACE, major adverse cardiovascular events; MI, myocardial infarction; SAP, single antiplatelet therapy.
The time to each event based on at-discharge AT strategy is shown in Figure 1. The AT strategy at discharge did not appear to be associated with the time to a MACE event (all

Kaplan–Meier curves for MACE.

Kaplan–Meier curves for MACE landmark study of (a) 0–6 months and (b) 6–24 months.
From the univariate Cox proportional hazards analyses, prior MI, HF, renal failure, presentation [i.e. ST segment elevation myocardial infarction (STEMI), non-STEMI, unstable angina], number of vessels with CAD, index hospital length of stay, discharged to home, discharged on anti-arrhythmic medication, and the IMRS were all associated with the outcome of MACE (Figure 3a). In a multivariable analysis adjusting for these factors simultaneously in a Cox proportional hazards regression model, the at-discharge DAP and AC was not a significant risk factor for MACE compared with all the other at-discharge AT therapies (Figure 3b). Compared with unstable angina, STEMI [hazard ratio (HR) = 2.70 95% CI: 1.61–4.51;

Cox regression hazard ratio (HR) for MACE (a) univariate and (b) multivariate.
For the subgroup of VTE patients, at-discharge AT strategy was not associated with the outcome of MACE (all
AT strategy at discharge was not a significant risk factor for the individual outcomes of all-cause death, CV death, MI (fatal and nonfatal) and CVA (fatal and nonfatal) (Figure 4). For all-cause death, high IMRS score (HR = 2.42, 95% CI: 1.62–3.62;

Multivariable Cox regression hazards ratio (HR) for (a) individual outcomes of death, (b) CV death, (c) MI (fatal and nonfatal) and (d) CVA (fatal and nonfatal).
Discussion
In this observational series of ‘real world’ ACS patients with indications for long-term anticoagulant prophylaxis, we found that the AT strategy prescribed by clinicians at discharge did not appear to have a significant impact on the risk of MACE. This observation must be carefully interpreted. Our findings could lead to a face-value interpretation that the chosen discharge AT strategy in this complex patient population is not a matter of importance; however, we caution against such a conclusion. A second possibility is that the risk of subsequent MACE events in this complex population is too high to be incrementally associated with the use of multiple AT agents. While there has not been a randomized trial looking at AT strategies for ACS patients with indications for long-term anticoagulant use, there are several studies that have examined AT strategies in separate ACS, AF or VTE patient populations. These studies have shown a protective effect of appropriate AT strategies. For ACS patients, studies have shown that the use of DAP decreased the risk of MACE events from a high of 18% in the CURE trial [Yusuf et al. 2001] to a 7% for the CHARISMA trial [Bhatt et al. 2006], with a recent meta-analysis of randomized trials of DAP
Therefore, a more nuanced interpretation of our results suggests that, largely in the absence of applicable evidence and guidelines, clinicians are generally successful at determining a reasonable AT strategy for individual patients. These clinical decisions are based on evaluation of the patient’s comorbidities that contribute to the risk of both ischemic and bleeding complications. The overall equalization of outcomes among AT strategies, in this interpretation, indicates the aggregate success of these clinical decisions. The clinical decision making process involves a myriad of individual patient clinical factors, but also includes current guidelines and recommendations [Zeymer et al. 2013; Knight et al. 2015]. In our study of ACS patients, for those who received a stent, a majority (62%) were given DAP and, for those who had a CABG, 53% were given SAP. These findings suggest that, in general, clinicians were following guidelines for ACS patients. In fact, we found that for patients who had a CAGB, SAP and no AC was associated with decreased MACE events compared with DAP and no AC. While this finding would not be significant after correction for multiple testing, it is suggestive that the guidelines are appropriate. In contrast, the rate of AC prophylaxis was substantially lower (35.7%) for our study population of patients with VTE or AF with a CHADS2 score ⩾2, suggesting that these patients were managed differently from guidelines with regard to AC use. However, this low rate of AC use is similar to the report of triple therapy in other studies of ACS patients with AF, with these studies reporting a range from 33% to 49% [Maier et al. 2014; Potpara et al. 2014].
There may be several possible reasons for our observed AT patterns of fairly high guideline-based use of DAP or SAP but much lower guideline-based use of AC. First, the index hospitalization was for ACS; this immediate event may have had more impact on the choice of AT strategy. Second, the addition of an AC to DAP increases the risk of subsequent bleeding. However, we observed few major bleeding events (1.6%) in our study population. This rate of major bleeding is similar to rates reported by other studies in AF, VTE or ACS populations using similar measurements for major bleeding events with the rates ranging from 0.4% to 5% [Patel et al. 2011; Castellucci et al. 2014; Kawaji et al. 2014; Navarese et al. 2015]. However, it does not represent all possible bleeding events and other studies have reported bleeding events to be as high as 10–12% on DAP and AC [Fosbol et al. 2012; Rubboli et al. 2014]. Finally, AT strategies are largely untested and unknown in this patient population as ACS and AC trials have typically excluded these patients. As such physicians are reliant on their own or institutional experience and observation data.
Given our observation and the results reported in many of the other studies, the major bleeding rate is relatively low with AC therapy, especially when compared with the risk of MACE, and clinicians considering risk
There are several limitations with this study. First the duration of, and compliance with, AT strategies was not captured. The AT strategy at discharge could have changed in the 2-year follow-up period. To assess persistence with a strategy including warfarin, we searched for International Normalized Ratio (INR) testing. For those patients discharged on warfarin, the median time to last INR test was 201 days, suggesting that over half of all patients discharged on AC continued AC use for over 6 months. In addition, only 14% of those who were discharged without AC had an INR test during the observation period. Therefore, we believe failure to persist with the prescribed strategy had minimal impact on our findings. We were also not able to determine the exact medication dosage for the AT medications. However, Intermountain has implemented an at-discharge AT dosage guideline and has an AT clinic that helps track patients with indications for AC therapy. These systems help minimize variation in AT dosage strategies. Thus, we believe the impact of variation in dosage is minimal in this study.
A relatively small sample size is a second limitation; this may have limited the power to detect small differences in the outcomes associated with an at-discharge AT strategy. We had 80% power to detect an absolute increase of 13%, assuming a baseline of 42% MACE events. Except for the few subjects who received AC only, the percentage of subjects that had a MACE event varied from 37.7% to 43.7% between AT groups. Thus, we did not have the power to detect the differences of the magnitude we observed. However, the differences reported are still relatively small and may not be as clinically important as some of the other factors such the severity of underlying disease states. This is an observational study and as such we are not able to capture and adjust for all possible confounders. We were also unable to determine minor bleeding events as these would most likely not result in medical treatment, and thus our bleeding events are restricted to only major bleeding events that would have resulted in medical treatment.
There are several strengths in our study. First, patients received care in an integrated healthcare system and were less likely to have changes in medication as care was coordinated between cardiologist and primary care physician. Second, the Intermountain Healthcare EDW has data from nearly all the facilities and clinical offices in the Intermountain Healthcare system. This allowed for comprehensive tracking of patients and provided us with real world estimates of efficacy and safety of at-discharge AT strategy, without much lost-to-follow-up or self-reports.
Conclusion
In this observational study of real world patients presenting with ACS and a coexisting indication for chronic oral AC, the at-discharge AT strategy was not significantly associated with a 2-year risk of MACE. In contrast, individuals’ comorbidity and severity of illness at presentation played a major role in determining the risk of major adverse cardiovascular events in the observation period. Major bleeding rates were generally low in all groups studied. Although reassuring, these findings are observational in nature and do not negate the continued need for randomized trials to generate evidence-based approaches for physicians to management of this important population.
Footnotes
Funding
This work was supported by Janssen Scientific Affairs, LLC.
Conflict of interest statement
Z.Y. and R.M. were salaried employees of Janssen Research and Development, LLC at time of this research. T.B. is a consultant for Boston Scientific Corporation, St Jude Medical, Sanofi Aventis and Biosense Webster. J.M. is a member of the speaker bureau for Daiichi-Sankyo, Merck, Lilly and GSK. The other authors declare no conflicts of interest in preparing this article.
