Abstract
Introduction:
Data about plasma levels of neutrophil gelatinase-associated lipocalin (NGAL) in children with heart failure (HF) are very limited. NGAL is used widely as a biomarker for the diagnosis of renal injury in numerous clinical studies. The aim of this study is to investigate the plasma NGAL in children with HF caused by idiopathic dilated cardiomyopathy (IDCM) and its relation to the severity of HF.
Material and methods:
In a case-control study, 30 nondiabetic children, aged –16 years (all have IDCM) recruited from the pediatric department of our institute together with 30 healthy children were prospectively enrolled in this study. Patients underwent a detailed history taking, clinical examination, New York Heart Association (NYHA) class assessment and echocardiographic evaluation. Plasma levels of NGAL were measured by enzyme-linked immunosorbent assay.
Results:
Plasma levels of NGAL were significantly higher in children with HF compared with healthy controls (mean: 290.97 versus 144.33, p < 0.0001). The relationship between NGAL and the severity of HF was investigated. However, we did not find any statistically significant relationship between plasma NGAL levels and indices of myocardial function.
Conclusions:
NGAL levels were significantly increased in children with HF caused by IDCM. However, there was no significant relationship between plasma NGAL levels and indices of myocardial function. Future multicenter clinical studies in a large population addressing the natural course of NGAL in HF and its potential as a treatment target are needed in the near future.
Introduction
Heart failure (HF) is a severe disorder associated with high morbidity and mortality. It has a significant impact on the healthcare system and its occurrence is increasing in adults as well as in the pediatric population. Early diagnosis of HF is very important to improve outcomes and for therapy optimization [Yndestad et al. 2007; Palazzuoli et al. 2012].
The ability to recognize cardiac insufficiency at an early stage before signs and symptoms manifest depends on the specificity, sensitivity and accuracy of the tools used. For this reason, there is an increasing interest in the development of new biomarkers and numerous laboratory tests has recently been put forward for early diagnosis of HF [Palazzuoli et al. 2012]. During the past two decades, persistent inflammation has been proposed to play a pathogenic role in HF by influencing heart contractility, inducing matrix degradation and fibrosis, and promoting apoptosis, thereby contributing to myocardial remodeling. But despite numerous lines of evidence supporting the involvement of inflammation in the pathogenesis of HF, there are still issues to be clarified, including the identification and characterization of the different biomarkers involved [Yndestad et al. 2007; Palazzuoli et al. 2012].
Neutrophil gelatinase–associated lipocalin (NGAL) is a 25 kDa protein covalently bound to matrix metalloproteinase-9 which was first isolated from neutrophils. It belongs to the lipocalin family, and is best known clinically as a novel and early marker of acute kidney injury (AKI) [Devarajan, 2008; Aghel et al. 2010; Cruz et al. 2011; Damman et al. 2011]. NGAL has been found to be elevated in both plasma and urine in patients with HF. It is produced by a wide variety of cells, including respiratory and intestinal epithelial cells, endothelial cells, renal tubular cells and cardiomyocytes. Due to its association with kidney injury, inflammation and matrix remodeling, NGAL has been proposed as a marker of prognosis in patients with HF. NGAL plays a role in tumor development and may have a protective role against bacterial infection, apoptosis and oxidative stress. Under physiological conditions, NGAL is detected at very low levels in several human tissues including kidney, heart, stomach, lungs and colon [Devarajan, 2008; Poniatowski et al. 2009; Yndestad et al. 2009; Aghel et al. 2010; Cruz et al. 2011; Palazzuoli et al. 2012].
Growing evidence suggests that NGAL may be upregulated in ‘stressed’ cells, especially in cardiomyocytes, in which case the correlation with HF is important [Yndestad et al. 2009; Palazzuoli et al. 2012]. Due to the high incidence and poor prognosis associated with worsening renal function (WRF) in HF, previous reports have highlighted a role for NGAL in predicting WRF in decompensated HF [Aghel et al. 2010]. Both systemic and urine NGAL also constitute sensitive markers of renal impairment in patients with chronic HF [Poniatowski et al. 2009; Damman et al. 2011]. Yndestad and colleagues have suggested that NGAL may be upregulated in cardiomyocytes, within the failing myocardium itself, in response to pro-inflammatory cytokines [Yndestad et al. 2009]. Therefore, it is postulated that NGAL may constitute an important biomarker of acute and chronic inflammatory and oxidative stress within the failing myocardium in patients with HF [Tong et al. 2005; Roudkenar et al. 2008; Bahmani et al. 2010; Ding et al. 2010].
This study was designed to investigate the plasma NGAL levels in children with HF caused by idiopathic dilated cardiomyopathy (IDCM) and its relation to the severity of HF.
Patients and methods
Study design and population
This single-center prospective case-control study was carried out at Assiut University Children’s Hospital. The study was approved by the Assiut University ethics committee and was conducted in accordance with the Declaration of Helsinki. An informed written consent in accordance with Assiut University Ethical Committee guidelines was taken from all guardians of patients and controls.
The study included 30 pediatric patients (14 males and 16 females), with clinical evidence of HF [New York Heart Association (NYHA) functional class II–IV], recruited from the Children’s Hospital of our institute. All patients have IDCM as an underlying cause of HF. In addition, 30 age- and gender-matched healthy controls recruited from patients’ relatives were included in the study. Subject demographics, full history taking, clinical examination, relevant comorbidities and documented left ventricular ejection fraction (LVEF) percentage were obtained and recorded upon enrollment.
Baseline measurements included standard weight, height, body mass index (BMI: kg/m²), blood pressure, electrocardiogram (ECG), blood urea nitrogen (BUN), serum creatinine and hemoglobin (Hb) levels. The assessment of the stages of HF was made according to the NYHA functional classification system and two-dimensional echocardiography for assessment of left ventricle (LV) dimensions and LVEF% by the modified Simpson’s method.
Exclusion criteria
Patients with pre-existing renal insufficiency, or suffering from peripheral vascular diseases, or on nephrotoxic drugs before the study, as well as known diabetics were excluded from the study. In addition, patients with acute intercurrent noncardiac inflammatory illness (e.g. postoperative wound infection), malignancy or autoimmune diseases were excluded.
Blood sampling
Fasting blood samples (10 cm3 venous blood) were obtained by venipuncture from all participants (patients and controls). Blood samples were collected into test tubes containing disodium ethylenediaminetetraacetate (Na2 EDTA) or no anticoagulant. Care was taken to prevent any physical damage which might cause hemolysis. The tubes were allowed to stand at room temperature for 10 min and then centrifuged at 3000g for 15 min. After separation, the clear, nonhemolyzed supernatant serum or plasma was quickly removed and aliquoted into equal volumes in 2 ml tubes and stored frozen at –70°C until sample analysis for biomarkers. CBC, BUN and serum creatinine were measured in fresh blood samples.
Assays for plasma NGAL
Plasma NGAL was measured by a research enzyme-linked immunosorbent assay (ELISA) using commercial reagents (Hycult Biotech, NGAL, Human ELISA kit, catalog: HK330-01, HNL, lipocalin 2, oncogene 24p3, LCN2) and following the instructions supplied with the kit. A 2000-fold dilution was used to bring concentrations of each sample within the 100-fold sensitivity range of the assay (0.4–100 ng/ml). All diluted samples fell within the range of the calibration curve. The minimum detection limit of the assay was 0.4 ng/ ml. The intra-assay and inter-assay coefficient of variation was 1.1% and 3.2%, respectively, at 65 ng/ml. The results are expressed in ng/ml.
Statistical analysis
Clinical and laboratory data were collected and analyzed using Minitab Version 14 for Windows (Minitab Inc., State College, Philadelphia, PA, USA). Assumptions of normality and homogeneity of variance were initially checked using the Shapiro–Wilk W-test. Noncategorical (numerical) data are expressed as mean and standard deviation (SD). The differences between two groups were assessed by using the t-test or Mann–Whitney U test, as appropriate. A value of p < 0.05 was considered statistically significant for all tests.
Results
This study included 30 pediatric patients with IDCM (14 males and 16 females), with clinical evidence of HF (NYHA class II–IV). Their ages ranged from 2 to 16 years. In addition, 30 age- and gender-matched healthy controls were included in the study. Sociodemographic, clinical and laboratory data of the patients and control groups are shown in Table 1. There were no significant differences between the two groups with respect to age, gender, creatinine and Hb levels. Significant differences were noticed between the patient group and healthy controls regarding weight, height and BMI (p < 0.01 in all) (Table 1).
Clinicodemographic and laboratory data of IDCM and control groups.

Data are presented as number and percentage, mean and standard deviation (SD).
p < 0.05 is significant.
BMI, body mass index; Hb, hemoglobin; IDCM, idiopathic dilated cardiomyopathy; LVEF, left ventricular ejection fraction; NGAL, neutrophil gelatinase-associated lipocalin; NYHA, New York Heart Association.
As regards the clinical severity of HF in IDCM group; 13 patients (43.3%) had NYHA class II and 17 patients (56.7%) had NYHA class III–IV (Table 2).
Comparison between different groups of children with heart failure.
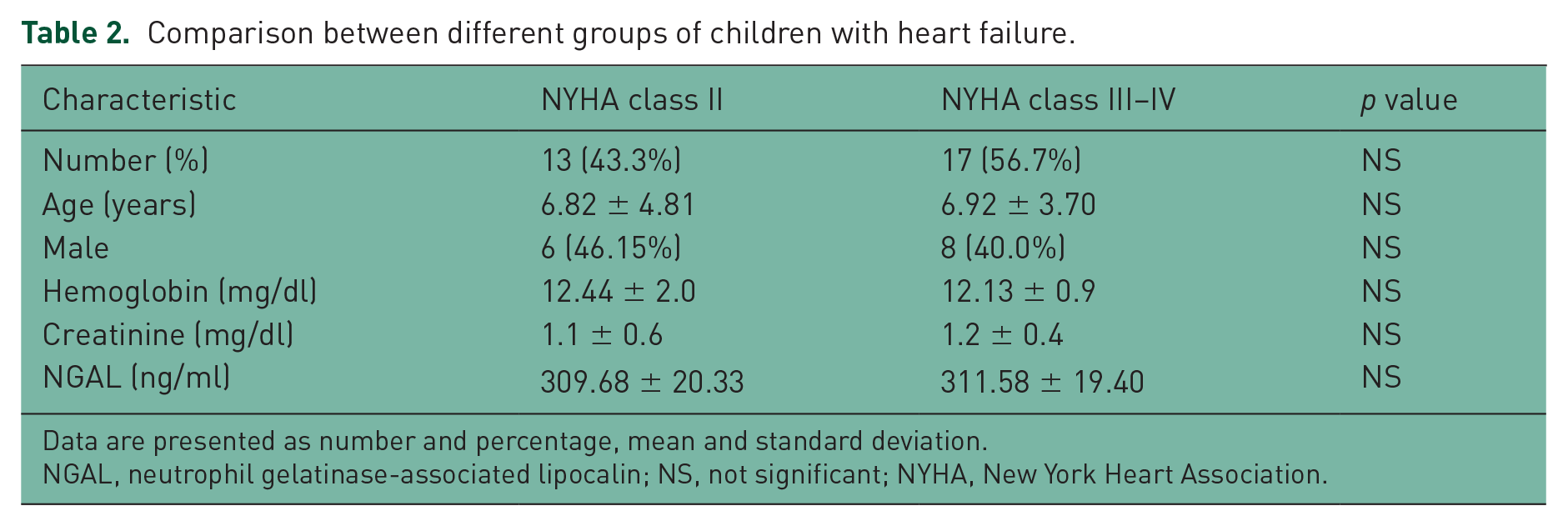
Data are presented as number and percentage, mean and standard deviation.
NGAL, neutrophil gelatinase-associated lipocalin; NS, not significant; NYHA, New York Heart Association.
The LVEF% was significantly lower in IDCM patients compared with healthy controls (mean: 39.7 versus 61.7; p < 0.001) (Table 1).
Children with HF showed a significantly higher plasma NGAL levels compared with the control group (mean: 290.97 versus 144.33; p < 0.0001) (Table 1).
Table 2 shows the comparison between patients with different NYHA classes. There were no significant differences between patients with NYHA class II and NYHA class III–IV with respect to age, gender, creatinine and Hb levels. The relationship between NGAL and the severity of HF was investigated. However, we did not find any statistically significant relationship between NYHA class II and NYHA class III–IV patients (mean: 309.68 versus 311.58; p < 0.52 (Table 2).
Discussion
Early diagnosis of HF is very important for therapy optimization and to improve outcomes. The ability to recognize cardiac insufficiency at an early stage before signs and symptoms manifest depends on the specificity, sensitivity and accuracy of the tools used [Palazzuoli et al. 2012]. This study was designed to investigate the plasma level of NGAL in pediatric HF caused by IDCM and to explore any relationship between NGAL and the severity of HF.
NGAL is synthesized in the bone marrow during granulocyte maturation. Granulocytes, epithelial cells, renal tubular cells and hepatocytes release NGAL during injury, and its levels are significantly elevated in epithelial damage [Devarajan, 2008; Yndestad et al. 2009]. Many studies have suggested that NGAL may be involved in cell survival, inflammation and matrix degradation. Through direct binding to matrix metalloproteinases (MMP) 9, NGAL inhibits its inactivation, leading to enhanced proteolytic activity with prolonged effects on collagen degradation [Devarajan, 2008; Yndestad et al. 2009].
In this study, we found that plasma NGAL levels in children with HF were significantly higher than in the control group. Our results are consistent with the findings of Yndestad and colleagues, who investigated the role of NGAL in HF, where they observed increased expression of NGAL levels in experimental and clinical HF both systemically and within the failing myocardium [Yndestad et al. 2009].
During recent years, persistent inflammation has been suggested to play a pathogenic role in HF by influencing heart contractility, inducing matrix degradation and fibrosis [Rutschow et al. 2006]. But although several lines of evidence support the involvement of inflammation in the pathogenesis of HF, there are still issues to be clarified, including the identification and characterization of the different actors involved [Yndestad et al. 2009].
Damman and colleagues reported that serum NGAL levels were associated with increased risk of subsequent worsening of renal function in admitted patients with HF [Damman et al. 2011]. Moreover, in patients with acutely decompensated HF, Maisel and colleagues found that serum NGAL measured at discharge was a better risk predictor of 30 day events than B-type Natriuretic peptide, creatinine or estimated glomerular filtration rate (eGFR) [Maisel et al. 2011]. Interestingly, Daniels and colleagues studied plasma NGAL levels in 1393 participants without cardiovascular disease (CVD), who were followed for a mean of 11 years. NGAL was a significant predictor of mortality and CVD in this adult cohort, independently of traditional risk factors and kidney functions [Daniels et al. 2012].
Van Deursen and colleagues assessed the prognostic value of serum NGAL in HF patients with or without renal dysfunction [van Deursen et al. 2014]. They observed that plasma NGAL predicted mortality in HF patients, either with or without chronic kidney disease (CKD) and it was a stronger predictor for mortality than frequently used biomarkers of CKD. Similarly, Nymo and colleagues investigated whether serum NGAL could provide prognostic information in chronic HF [Nymo et al. 2012]. They found that circulating NGAL was predictive of total mortality from cardiovascular causes, both before and after adjustment for several clinical variables. However, these findings were not significant after adjustment for additional variables, indicating that biomarkers other than systemic NGAL levels may provide more prognostic information in this population. Consistent with the earlier studies, Shrestha and colleagues observed that elevated systemic NGAL levels were associated with renal insufficiency in both acute and chronic systolic HF. They also observed associations between elevated systemic NGAL levels and poorer prognosis in patients with chronic HF [Shrestha et al. 2011].
In patients with HF, diminished perfusion may be a vital harmful factor for the kidneys. Because the renal tubules are more vulnerable to hypoxic damage, it is possible that minor declines in renal functions result in tubular damage where the renal cortex stays intact, maintaining GFR. Therefore, tubular damage might better reflect a decreased perfusion than eGFR. Also, in the acute conditions, tubular markers are increased, whereas creatinine is still normal. Renal blood flow can fall by 30–40% without apparently disturbing the functional GFR. Therefore, plasma NGAL levels are likely to be more sensitive than creatinine levels [Schmidt-Ott et al. 2007; van Deursen et al. 2014].
In children undergoing cardiopulmonary bypass, NGAL was increased significantly in plasma and urine 2–6 hours after surgery in those patients who subsequently developed AKI; the rise in creatinine was not evident until 48–72 hours later. Both urine and plasma NGAL were good predictors of AKI. Similar findings have been reported in adults who developed AKI after cardiac surgery, with NGAL elevation occurring 1–3 hours after surgery [Schmidt-Ott et al. 2007; Maisel et al. 2011].
The relationship between NGAL and the severity of HF was investigated in our study. However, we did not find any statistically significant relationship between systemic NGAL levels and indices of myocardial function. Contrary to our results, previous researchers have observed increased serum NGAL levels in both acute and chronic HF, which correlated significantly with the disease severity as assessed by clinical and neurohormonal parameters [Yndestad et al. 2009]. Similarly, Shrestha and colleagues found that elevated plasma NGAL levels were associated with worsening LV diastolic dysfunction in patients with systolic HF and independently predictive of adverse long-term outcomes [Shrestha et al. 2010]. Moreover, Bolignano and colleagues observed a clear correlation between systemic NGAL levels and the clinical severity of HF expressed according to the NYHA classification and ejection fraction [Bolignano et al. 2009].
Both serum and urine NGAL were measured in many disorders other than HF. Breidthardt and colleagues reported that adults with lower respiratory tract infections are followed by an increase in the serum NGAL [Breidthardt et al. 2012]. They concluded that NGAL is a powerful predictor of early AKI in patients with lower respiratory tract infection. Decavele and colleagues investigated the effects of urinary tract infection and urinary neutrophil counts on urinary NGAL values [Decavele et al. 2011]. They reported that urinary tract infections were the cause of high urinary NGAL. Other researchers found elevated levels of urinary NGAL in children with congenital obstructive urinary tract anomalies [Wasilewska et al. 2011]. In a recent study, Wang and colleagues examined the clinical utility of NGAL as an indicator of mortality and multiple organ dysfunction syndrome (MODS) in severe sepsis and septic shock [Wang et al. 2015]. They reported that high plasma NGAL independently predicts mortality and MODS in patients with severe sepsis and septic shock. Smertka and colleagues [2014] determined serum and urine NGAL concentrations in 73 infants (51 with sepsis, 22 with severe sepsis) admitted to the intensive care unit in the first month of life [Smertka et al. 2014]. They found powerful correlations between the concentration of serum and urinary NGAL and inflammatory markers, such as C-reactive protein (CRP) and procalcitonin. They concluded that serum and urinary NGAL values are markers of AKI and more accurately reflect the severity of inflammatory status.
The limitations of our study were the small number of HF patients and that all patients were from a single center. As a consequence, the statistical power of this comparison between patients and controls is limited.
Conclusion
The present study showed that children with HF due to IDCM had significantly higher plasma NGAL levels than healthy controls. Multicenter clinical studies in a large population addressing the natural course of NGAL in HF and its potential as a treatment target are needed in the near future.
Footnotes
Acknowledgements
We thank the two anonymous reviewers for providing helpful comments on the earlier version of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
