Abstract
Hyperglycemia is a frequent condition in patients with acute coronary syndromes (ACS). Hyperglycemia during ACS is caused by an inflammatory and adrenergic response to ischemic stress, when catecholamines are released and glycogenolysis induced. Although the involved pathophysiological mechanisms have not yet been fully elucidated, it is believed that hyperglycemia is associated with an increase in free fat acids (which induce cardiac arrhythmias), insulin resistance, chemical inactivation of nitric oxide and the production of oxygen reactive species (with consequent microvascular and endothelial dysfunction), a prothrombotic state, and vascular inflammation. It is also related to myocardial metabolic disorders, leading to thrombosis, extension of the damaged area, reduced collateral circulation, and ischemic preconditioning. In the last few years, several observational studies demonstrated that hyperglycemia in ACS is a powerful predictor of survival, increasing the risk of immediate and long-term complications in patients both with and without previously known diabetes mellitus. Glucose management strategies in ACS may improve outcomes in patients with hyperglycemia, perhaps by reducing inflammatory and clotting mediators, by improving endothelial function and fibrinolysis and by reducing infarct size. Recent clinical trials of insulin in ACS have resulted in varying levels of benefit, but the clinical benefit of an aggressive treatment with insulin is yet unproved.
Introduction
Numerous studies have shown that hyperglycemia is a commonly encountered issue in critically ill patients in the intensive care setting, even in patients without diabetes mellitus [Umpierrez et al. 2002; Finney et al. 2003; Bartnik et al. 2004; Jacobi et al. 2012; Viana et al. 2014].
A recent analysis of medical records showed that hyperglycemia was present in 38% of adult patients admitted to hospital, of whom 26% had a known history of diabetes, and 12% had no history of diabetes before the admission [Umpierrez et al. 2002].
In addition, patients with hyperglycemia had a longer length of hospital stay, a higher admission rate to an intensive care unit, and were less likely to be discharged home, frequently requiring transfer to a transitional care unit or nursing home facility [Umpierrez et al. 2002].
Similarly, the Euro Heart Survey on diabetes and the heart analyzed the prevalence of abnormal glucose regulation in adult patients with coronary artery disease (CAD) [Bartnik et al. 2004]. The survey engaged 110 centers in 25 countries recruiting 4196 patients referred to a cardiologist due to CAD, of which 2107 were admitted on an acute basis and 2854 had an elective consultation [Bartnik et al. 2004]. In patients with acute CAD, 58% had impaired glucose regulation. In the stable group this proportion was 51% [Bartnik et al. 2004].
The presence of hyperglycemia is also associated with increased morbidity and mortality, regardless of the reason for admission (e.g. acute myocardial infarction, surgery, stroke, sepsis) [Umpierrez et al. 2002; Finney et al. 2003; Bartnik et al. 2004; Jacobi et al. 2012; Viana et al. 2014].
In this context, the adverse prognostic impact of hyperglycemia at the time of acute coronary syndromes (ACS) has focused considerable attention from expert groups and professional societies [Capes et al. 2000; Deedwania et al. 2008; Inzucchi, 2008; Angeli et al. 2010]. It is now well established that glycometabolic status upon hospital admission is an important prognostic marker for all-cause death in patients with ACS, whether or not they had previously known diabetes mellitus [Deedwania et al. 2008; Angeli et al. 2010].
Although the association between hyperglycemia and increased mortality and in-hospital complications in patients with ACS has been confirmed in different studies [Capes et al. 2000; Angeli et al. 2010], the pathophysiology, the definition, and the treatment of hyperglycemia remain controversial [Deedwania et al. 2008; Inzucchi, 2008].
Epidemiology
A certain amount of controversy still surrounds the cutoff value that should be used as a diagnostic marker of hyperglycemia in ACS. Further, it is not clear whether the glucose index value should be that taken on admission, after overnight fasting, or during an oral glucose tolerance test [Deedwania et al. 2008; Inzucchi, 2008].
Nevertheless, current classification of hyperglycemia in hospital includes diabetes diagnosed and treated before admission (known history of diabetes); existing, but unrecognized, diabetes (fasting glucose more than 6.9 mmol/liter or random glucose higher than 11.1 mmol/liter during hospital stay and confirmed after discharge; new-onset stress hyperglycemia or hospital-related hyperglycemia (fasting glucose more than 6.9 mmol/liter or random glucose higher than 11.1 mmol/liter during hospital stay that reverts to normal range after discharge) [Deedwania et al. 2008; Inzucchi, 2008].
Regardless of its classification, prevalence of hyperglycemia in different epidemiological studies (Table 1) ranges from 3% to 71% of patients admitted with ACS [Angeli et al. 2010, 2013].
Main clinical studies evaluating the prognostic significance of hyperglycemia in patients with acute coronary syndromes.

ACS, acute coronary syndrome; adm, admission; CCP, Cooperative Cardiovascular Project; GRACE, Global Registry of Acute Coronary Events; HAS, Heart Attack Study; HR, high risk; ICONS, Improving Cardiovascular Outcomes in Nova Scotia; JACCS, Japanese Acute Coronary Syndrome Study; LVSD, left ventricular systolic dysfunction; PCI, percutaneous coronary intervention; Rand, randomization; VALIANT, VALsartan In Acute myocardial iNfarcTion.
Data from the following references [Ravid et al. 1975; Lewandowicz et al. 1979; Soler and Frank, 1981; Oswald et al. 1986; Bellodi et al. 1989; Sewdarsen et al. 1989; O’Sullivan et al. 1991; Wahab et al. 2002; Aguilar et al. 2004; Stranders et al. 2004; Timmer et al. 2004; Ainla et al. 2005; Kosiborod et al. 2005; Kosuge et al. 2005; Nordin et al. 2005; Suleiman et al. 2005; Meisinger et al. 2006; Schiele et al. 2006; Shen et al. 2006; Goldberg et al. 2007; Ishihara et al. 2007; Petursson et al. 2007; Scott et al. 2007; Gasior et al. 2008].
As depicted in Figure 1, a number of contemporary investigators have shown a significant inverse relationship [r = −0.75; 95% confidence interval (CI) –0.88 to −0.49, p < 0.0001] between the threshold of blood glucose for defining hyperglycemia and its prevalence in patients with ACS [Angeli et al. 2010, 2013]. As expected, the lowest prevalence is observed with serum glucose levels of 9.4–17 mmol/liter (169–306 mg/dl) [Angeli et al. 2010], while lower thresholds for diagnosing hyperglycemia (6.1 mmol/liter; 108 mg/dl) are associated with the highest prevalence [Angeli et al. 2010, 2013].

Relationship with 95% confidence ellipse between threshold of blood glucose for defining hyperglycemia and its prevalence in contemporary studies recruiting patients with myocardial infarction. CCP, Cooperative Cardiovascular Project; GRACE, Global Registry of Acute Coronary Events; HAS, Heart Attack Study; ICONS, Improving Cardiovascular Outcomes in Nova Scotia; JACCS, Japanese Acute Coronary Syndrome Study; VALIANT, VALsartan In Acute myocardial iNfarcTion. See Table 1 for other details. Data from ref. [Ravid et al. 1975, Lewandowicz et al. 1979, Soler et al. 1981, Oswald et al. 1986, Bellodi et al. 1989, Sewdarsen et al. 1989, O’sullivan et al. 1991, Wahab et al. 2002, Aguilar et al. 2004, Stranders et al. 2004, Timmer et al. 2004, Ainla et al. 2005, Kosiborod et al. 2005, Kosuge et al. 2005, Nordin et al. 2005, Suleiman et al. 2005, Meisinger et al. 2006, Schiele et al. 2006, Shen et al. 2006, Goldberg et al. 2007, Ishihara et al. 2007, Petursson et al. 2007, Scott et al. 2007, Gasior et al. 2008].
Mechanisms
Stress hyperglycemia is the result of sympathetic nervous system activation and the hypothalamic–pituitary axis with consequent raised production of catecholamines and cortisol that stimulate processes of glyconeogenesis, glycogenolysis, and lipolysis [McCowen et al. 2001; Huberlant and Preiser, 2010] (Figure 2).

Cardiovascular effects of hyperglycemia during the acute phase of myocardial infarction.
Although the mechanisms underlying the association between new hyperglycemia and mortality in patients with ACS are not fully understood, evidence that use of insulin to lower glucose concentrations at the time of ACS decreases mortality suggests that hyperglycemia is not simply an epiphenomenon of the stress response mediated by cortisol and noradrenaline [Huberlant and Presier, 2010; Senthinathan et al. 2011].
In particular, the higher risk of poor outcome in patients with hyperglycemia during ACS may reflect several potential underlying mechanisms (Figure 2).
Acute or stress hyperglycemia has a facilitating role in the development of ACS and accentuates the consequences of cellular damage caused by acute myocardial ischemia [Ceriello, 2005].
It has been shown that hyperglycemia increases the release of inflammatory and vasoconstrictive factors which impair coronary endothelial function, contributes to the production of reactive oxygen species with consequent oxidative stress, and increases platelet aggregation [Ray et al. 2007; Worthley et al. 2007].
In addition, acute hyperglycemia produces a significant lengthening of the QT interval, reduces ischemic preconditioning and increases no-reflow and insulin resistance (impaired glucose utilization which can potentially worsen ischemia through elevated oxygen consumption) [Oliver and Opie, 1994; Zarich, 2006; Bauters et al. 2007] (Figure 3).

Cardiac effects of hyperglycemia during the acute phase of myocardial infarction.
Moreover, the abnormal metabolic state that accompanies hyperglycemia alters the functional properties of multiple cell types, including platelets [Ferroni et al. 2004].
An altered platelet metabolism and changes in intraplatelet signaling pathways may contribute to the development of atherothrombotic complications. Hyperglycemia represents a causal factor for in vivo platelet activation, and is responsible for nonenzymatic glycation of platelet glycoproteins, causing changes in their structure and conformation, as well as alterations of membrane lipid dynamics [Ferroni et al. 2004] (Figure 4). Furthermore, hyperglycemia-induced oxidative stress is responsible for enhanced peroxidation of arachidonic acid to form biologically active isoprostanes, which represents an important biochemical link between impaired glycemic control and persistent platelet activation [Ferroni et al. 2004]. Finally, increased oxidative stress is responsible for activation of transcription factors and expression of redox-sensitive genes leading to a phenotypic switch of endothelium toward an adhesive, prothrombotic condition, initial platelet activation, adhesion, and subsequent platelet aggregate formation [Ferroni et al. 2004].
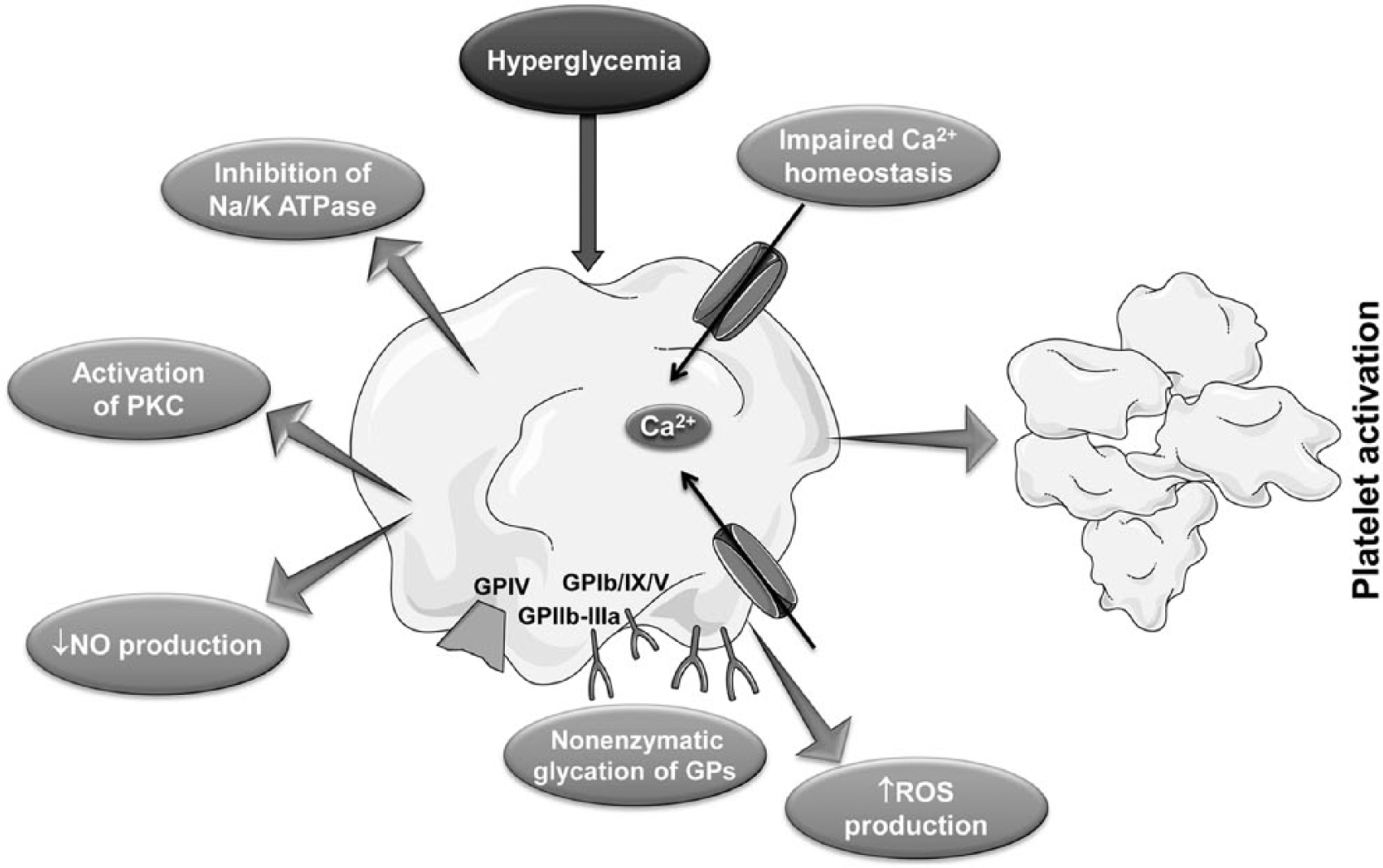
Impact of hyperglycemia on platelet function.
Prognosis
In recent years many studies have focused on the prognostic value of hyperglycemia in ACS, independent of whether the patients are known to have diabetes [Capes et al. 2000; Foo et al. 2003; Goyal et al. 2006]. In particular, hyperglycemia predicts mortality in patients with ACS, including ST-segment elevation myocardial infarction (STEMI), thrombolysis, and percutaneous coronary revascularization [Capes et al. 2000; Zeller et al. 2005; Pandey et al. 2009; Timmer et al. 2011; Eitel et al. 2012; Planer et al. 2013; Chen et al. 2014; Ekmekci et al. 2014; Lazzeri et al. 2014].
A recent analysis of the Cooperative Cardiovascular Project which included a large cohort of patients (n = 141,680) with ACS showed that depending on different levels of glucose on admission (⩽110, 110–140, 140–170, 170–240, and ⩾240 mg/dl), new hyperglycemia was associated with a significant 13–77% relative increase in short-term mortality and a 7–46% relative increase in 1-year mortality [Kosiborod et al. 2005]. Notably, after multivariable adjustment for confounders (including age, sex, prior vascular events, hypertension, smoking status, Killip class, and atrial fibrillation) [Angeli et al. 2012a], patients with higher glucose levels at admission remained at increased risk of both 30-day and 1-year mortality compared with patients whose admission glucose was up to 110 mg/dl [Kosiborod et al. 2005].
In a study involving 2127 patients with ACS, Foo and coworkers stratified admission plasma glucose concentrations above 5.8 mmol/liter into quartiles, and reported a relationship between increasing glycemia and ventricular dysfunction, and in-hospital death [Foo et al. 2003]. The prognostic value of this variable was not affected by the known presence of diabetes at admission, and was similar in patients with ACS with and without ST segment elevation.
Furthermore, Goyal and colleagues demonstrated that admission glucose and changes in glucose level within the first 24 h predict the risk of mortality at 30 days in patients with ACS without a history of diabetes and that the risk of 30-day mortality decreased by 9% per 11 mg/dl reduction in glucose within the first 24 h after ACS [Goyal et al. 2006].
In the specific setting of patients with STEMI, elevated plasma glucose levels on admission confirmed to be independent prognosticators of both in-hospital and long-term outcome regardless of diabetic status [Malmberg et al. 1999; Chen et al. 2014].
For every 18 mg/dl increase in glucose level, there is a 4% increase in mortality in subjects without diabetes, and when admission glucose level exceeds 200 mg/dl, the risk of all-cause mortality is similar in patients with and without a history of diabetes [Stranders et al. 2004].
Nevertheless, in some reports fasting glucose the day after admission appeared to be a better predictor of early mortality than glucose level on admission in patients with STEMI [Suleiman et al. 2005]. Patients with both an elevated admission glucose and an elevated fasting glucose the next day have a threefold increased risk of mortality. Similarly, failure of an elevated glucose level to fall within 24 h of admission is associated with excess mortality in patients with STEMI without diabetes mellitus [Goyal et al. 2006].
To further elucidate the significance of new hyperglycemia in ACS, we recently reviewed the literature on this topic and we performed a meta-analysis of observational studies [Angeli et al. 2010]. Overall, 24 clinical studies were included in the analysis (Figure 5). Patients with new hyperglycemia showed a 3.6-fold [odds ratio (OR) 3.62, 95% CI 3.09–4.24; p < 0.0001] increased risk of death during hospitalization compared with those who were normoglycemic [Angeli et al. 2010]. Similarly, new-onset hyperglycemia was associated with an increased risk of occurrence of death during a follow up of 30 days (OR 4.81, 95% CI 2.18–10.61; p < 0.0001) [Angeli et al. 2010]. Addressing long-term prognosis, new hyperglycemia was also associated with an excess risk of mortality (OR 2.02, 95% CI: 1.62 − 2.51, p < 0.0001) [Angeli et al. 2010].

Prognostic impact of new hyperglycemia on in-hospital (upper panel), 30-day (middle panel) and long-term mortality (lower panel) in patients with acute coronary syndromes. Odds ratios for individual studies are reported. Bars and diamonds denote the 95% confidence intervals (CI) for individual studies and pooled estimates, respectively. See Table 1 for other details. CCP, Cooperative Cardiovascular Project; GRACE, Global Registry of Acute Coronary Events; HAS, Heart Attack Study; ICONS, Improving Cardiovascular Outcomes in Nova Scotia; JACCS, Japanese Acute Coronary Syndrome Study; VALIANT, VALsartan In Acute myocardial iNfarcTion. Data from ref. [Angeli et al., 2010].
Of note, trial level covariates (including timing of blood glucose determination, threshold glucose concentration used to define hyperglycemia, starting year of enrolment, and publication year) did not exert a statistically significant influence on estimates (all p > 0.05) of in-hospital, 30-day and long-term mortality [Angeli et al. 2010].
In addition, admission hyperglycemia seems to predict mortality in patients with ACS, offering an incremental predictive value when added to risk prediction scores (including the Thrombolysis in Myocardial Infarction and the Global Registry of Acute Coronary Events (GRACE) scores) [Greig et al. 2010; Timoteo et al. 2014].
In this context, a recent analysis of a single center registry of ACS (2099 patients, 69% men), evaluated the ability of two models (GRACE risk score alone and in combination with blood glucose) to predict death. The inclusion of admission blood glucose in the model with GRACE score increased the area under the receiver operating characteristic curve (p = 0.018) and was associated with an increase in both net reclassification improvement (37%) and integrated discrimination improvement (0.021) [Timoteo et al. 2014].
Therapeutic implications
The substantial harm associated with hyperglycemia in patients with ACS suggests that glucose management strategies might improve outcome in patients with hyperglycemia [Angeli et al. 2010, 2013; Senthinathan et al. 2011].
Experimental studies suggested that treatment with insulin reduces inflammatory and clotting mediators in the plasma, improves endothelial function and fibrinolysis, and reduces infarct size in patients with ACS [Chaudhuri et al. 2004; Senthinathan et al. 2011; Wong et al. 2011].
Clinical trials
In the last few years, controlling hyperglycemia during ACS has been the target of a great deal of clinical research, with results primarily trending toward a benefit [Angeli et al. 2010, 2013].
In the Clinical Trial of Reviparin and Metabolic Modulation in Acute Myocardial Infarction Treatment Evaluation-Estudios Clinicos Latino America (CREATE-ECLA) study, which enrolled 20,000 patients, a glucose-insulin-potassium (GIK) regimen did not demonstrate improved outcomes in ACS [Mehta et al. 2005]. In this trial, high-dose GIK infusion was initiated immediately following randomization and continued for 24 h, and 40% of the total subjects became hyperglycemic (>144 mg/dl) in the postrandomization period; of these, the majority (62%) were in the GIK group [Mehta et al. 2005]. Subjects with a postrandomization glucose of more than 144 mg/dl had a 2.5-fold higher risk of mortality compared with subjects with a glucose level less than 126 mg/dl [Mehta et al. 2005]. While GIK was associated with a significant reduction in mortality and heart failure at 30 days after ACS, the overall effect was neutralized by an increase in mortality and heart failure with this regimen in the immediate 3-day post-ACS period. This initial increase in poor outcomes was likely mediated by the fact that GIK infusion frequently induced hyperglycemia and volume overload, which may have mitigated the beneficial effects of insulin in the regimen [Mehta et al. 2005].
Diabetes Mellitus Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI) [Malmberg, 1997, Malmberg et al. 1999] and DIGAMI-2 [Malmberg et al. 2005] examined the effect of glycemic control in patients with ACS with either a history of diabetes or admission glucose levels greater than 11 mmol/liter (198 mg/dl).
The DIGAMI trial [Malmberg, 1997; Malmberg et al. 1999] evaluated the effect of intensive insulin treatment on mortality and morbidity in patients with diabetes and ACS. Insulin infusion was initiated as soon as possible after the onset of symptoms and continued for at least 24 h [Malmberg, 1997; Malmberg et al. 1999]. Patients were randomized to an intensive insulin treatment (insulin-glucose infusion followed by multidose subcutaneous insulin for ⩾3 months) or to a control group receiving conventional treatment at the discretion of the physician in charge [Malmberg, 1997; Malmberg et al. 1999]. Subjects assigned to more aggressive insulin therapy showed a significant reduction in mortality compared with the control group (estimate relative mortality reduction of 28%) [Malmberg, 1997; Malmberg et al. 1999].
On the contrary, in the DIGAMI-2 trial [Malmberg et al. 2005] there were no differences in mortality rates at 2 years between patients who received insulin and those who received routine care. Specifically, this trial compared three different management strategies in patients with type 2 diabetes and acute myocardial infarction: a 24 h insulin–glucose infusion followed by a subcutaneous insulin-based long-term glucose control (group 1); a 24 h insulin–glucose infusion followed by standard glucose control (group 2); and routine metabolic management according to local practice (group 3) [Malmberg et al. 2005]. Overall, mortality was not significantly different among the three groups: the estimated mortality was 23.4% in group 1 and 21.2% in group 2 [hazard ratio (HR) 1.03; 95% CI 0.79–1.34; p = 0.832). In group 3, the observed mortality (17.9%) was lower compared with group 1 but this difference did not achieved significance (HR 1.26; 95% CI 0.92–1.72; p = 0.157). Comparing groups 2 and 3, the HR was 1.23 (95% CI 0.89–1.69; p = 0.203) [Malmberg et al. 2005].
More recently, in the Hyperglycemia Intensive Insulin Infusion in Infarction Study (HI-5) [Cheung et al. 2006], 240 subjects with ACS and either known diabetes or without diabetes but blood glucose level at least 7.8 mmol/liter were randomized to receive insulin/dextrose infusion therapy for at least 24 h to maintain a glucose level less than 10 mmol/liter or conventional therapy.
Insulin/dextrose infusion did not reduce mortality at the inpatient stage (4.8 versus 3.5%, p = 0.75), 3 months (7.1 versus 4.4%, p = 0.42), or 6 months (7.9 versus 6.1%, p = 0.62) [Cheung et al. 2006].
Although the varying glucose target levels in these trials make it difficult to compare their results and infer a conclusion, it is worth mentioning that in the DIGAMI-2 [Malmberg et al. 2005] and HI-5 trials [Cheung et al. 2006] blood glucose levels were not adequately lowered (the aggressive treatment groups achieved similar reductions in blood glucose compared with the control treatment groups). In addition, these trials were underpowered to show an effect on mortality and the infusions of insulin were started some hours after symptom onset, when it may have been too late to salvage the myocardium.
In-hospital therapy
Although recent large prospective studies of insulin in ACS have resulted in varying levels of benefit, the only acceptable treatment modality of hyperglycemia in the ACS setting is continuous intravenous regular insulin infusion, which should be initiated when blood glucose levels are greater than 10/11 mmol/liter (180/200 mg/dl) [Wilson et al. 2007; Houlden et al. 2013; Cheng et al. 2014].
Subcutaneous regimens with insulin and oral antihyperglycemic agents are not recommended because of the increased risk of hypoglycemia.
While evidence favors a tighter control in the range of 6.1–7.8 mmol/liter (110–140 mg/dl) in surgical patients, a less aggressive target may suit patients with ACS (7.8–10.0 mmol/liter; 140–180 mg/dl) [Wilson et al. 2007; Houlden et al. 2013; Cheng et al. 2014].
There are many insulin infusion protocols available, but those protocols which contain orders that take into account both current blood glucose values and rate of change of blood glucose are preferred [Goyal et al. 2008; Lipton et al. 2011]. Initial monitoring of blood glucose should be done on an hourly basis (interval of testing can be increased when two to three consecutive readings are consistently around the target) and insulin dose should be titrated according to blood glucose levels [Wilson et al. 2007; Houlden et al. 2013; Cheng et al. 2014].
Conclusion
Hyperglycemia during ACS is a common finding and also an adverse prognostic marker that increases the risk of immediate and long-term complications in patients both with and without diabetes mellitus [Capes et al. 2000; Deedwania et al. 2008; Angeli et al. 2010, 2013]. In particular, new hyperglycemia during ACS portends an excess risk of mortality not only in the hospital setting, but also in the short term (30 days) and long term (up to 108 months) [Angeli et al. 2010].
Despite hyperglycemia as a factor of worse outcome is documented in several prospective studies, many gaps in knowledge currently exist in our understanding of the association between elevated glucose levels and adverse outcomes in patients with ACS [Deedwania et al. 2008; Jacobi et al. 2012; Angeli et al. 2013; Viana et al. 2014].
First, there is currently no consensus about the precise glucose value that should be considered abnormal (i.e. no minimum blood glucose concentration that could carry an increased risk of worse outcome has been clearly established) [Finney et al. 2003; Bartnik et al. 2004; Worthley et al. 2007; Jacobi et al. 2012].
Of note, the meta-analysis by Capes and coworkers has reported that patients without diabetes with ACS are at risk for in-hospital complications for blood glucose levels over 110 mg/dl, while patients with diabetes and ACS are at risk for in-hospital complications for admission blood glucose concentrations equal to or over 180 mg/dl [Capes et al. 2000].
Second, there is no consensus about the most suitable method to initially measure and subsequently monitor blood glucose levels in the acute setting of ACS.
Third, the benefits of treating hyperglycemia have not been established definitively, and the target value of blood glucose to be achieved with treatment remains undefined. The clinical benefits of an aggressive treatment with insulin are yet unproven [Senthinathan et al. 2011] and the potential role of concomitant drug therapies, including renin–angiotensin system blockers [Verdecchia and Angeli, 2003; Reboldi et al. 2009] and statins [Angeli et al. 2012b, 2012c], to ameliorate the detrimental effect of hyperglycemia need to be further evaluated.
Thus, intervention trials are needed to optimize the definition of new hyperglycemia and to establish appropriate modalities and goals of glucose-lowering treatment.
Footnotes
Acknowledgements
The authors meet the criteria for authorship as recommended by the International Committee of Medical Journal Editors and were fully responsible for all content and editorial decisions, and were involved in all stages of manuscript development. The authors received no compensation related to the development of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
