Abstract
Background:
The blood pressure lowering effect of sesamin has been demonstrated to be associated with the increase in vascular nitric oxide (NO) biological activity by our previous studies and others. The present study was designed to explore the underlying mechanisms involved in the effect of sesamin on aortic NO bioactivity in spontaneously hypertensive rats (SHRs).
Methods:
Sesamin was orally administered for 8 consecutive weeks in SHRs. Systolic blood pressure (SBP) was measured using the tail-cuff method. The aortas were isolated and in vitro vascular reactivity studies were performed. Superoxide anion production in carotid arteries was assessed by dihydroethidium fluorescence staining. The protein expression of endothelial nitric oxide synthase (eNOS), phosphorylated eNOS (P-eNOS), dihydrofolate reductase (DHFR), nicotinamide adenine dinucleotide phosphate oxidase subunit p47phox, and copper, zinc superoxide dismutase (Cu/Zn-SOD) in aortas was detected by Western blotting. The dimeric form of eNOS in aortas was determined by low-temperature sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Aortic level of nitrotyrosine and activities of antioxidant enzymes, namely, total SOD (T-SOD), glutathione peroxidase (GPx) and catalase were also detected.
Results:
In SHRs, sesamin treatment reduced SBP, improved vascular relaxation induced by acetylcholine and enhanced aortic NO bioactivity. Sesamin treatment enhanced NO biosynthesis in SHR aortas was due to upregulated P-eNOS and suppressed eNOS uncoupling, and the latter effect might be attributed to decreased nitrotyrosine and upregulated DHFR. Sesamin also reduced the NO oxidative inactivation and decreased the superoxide anion production through downregulation of p47phox and amelioration of eNOS uncoupling. In addition, sesamin treatment did not alter the levels of GPx and catalase activity but obviously reduced the compensatory elevated T-SOD activity and Cu/Zn-SOD protein expression.
Conclusion:
Chronic treatment with sesamin could reduce hypertension and improve endothelial dysfunction through enhancement of NO bioactivity in SHR aortas.
Introduction
Sesamin, one of the major lignans, is a natural substance found in sesame oil and seeds, and exerts a variety of pharmacological properties, including lipid-lowering, antioxidative and antihypertensive effects [Gu et al. 1998; Kita et al. 1995; Kong et al. 2009; Nakano et al. 2002, 2003; Zhang et al. 2013]. Our group and others have demonstrated that chronic treatment with sesamin lowers blood pressure in different models of hypertensive rats such as the two-kidney, one-clip (2K1C) renal hypertensive rat [Kita et al. 1995], the deoxycorticosterone acetate (DOCA) salt hypertensive rat [Nakano et al. 2003], the 2K1C rat fed with a high-fat, high-sucrose diet [Kong et al. 2009], and the spontaneously hypertensive rat (SHR) [Zhang et al. 2013]. Interestingly, Miyawaki and colleagues reported that administration of sesamin 60 mg per day for 4 weeks reduces both systolic and diastolic blood pressure in patients with mild hypertension [Miyawaki et al. 2009].
Nitric oxide (NO), the pivotal endothelium-derived vasodilator, is generated within vascular endothelium by endothelial NO synthase (eNOS). Current evidence suggests that NO plays an important role in the antihypertensive effects of sesamin. First, sesamin has been demonstrated to increase NO concentration and enhance eNOS expression in the medium of human umbilical vein endothelial cells [Lee et al. 2004]. Second, the metabolites of sesamin induce relaxation in both endothelium-intact and denuded rat aortic rings in vitro. Pretreatment with these isolated aortic rings with a NOS inhibitor, NG-nitro-L-argininethe (L-NOARG), could attenuate vasorelaxation [Nakano et al. 2006]. Moreover, the blood pressure-lowering effect of sesamin is not observed in L-NOARG-treated hypertensive rats or in DOCA-salt-treated eNOS-deficient mice [Nakano et al. 2006]. Third, our previous study showed that treatment with sesamin directly ameliorates the aortic NO bioactivity reduction in the 2K1C rat fed with a high-fat, high-sucrose diet [Kong et al. 2009]. However, the molecular mechanisms by in vivo sesamin treatment, leading to the enhancement of NO biological activity, are not completely clarified.
So far, several mechanisms, including decreased biosynthesis of NO, exaggerated superoxide anion mediated inactivation of NO and reduced responsiveness of vascular smooth muscle cells (VSMCs) to NO, have been implicated in the etiology of NO bioactivity reduction. The SHR is an animal model of genetic hypertension which has an obvious diminished bioactivity of endothelium-derived NO [Hong et al. 2000; Sanchez et al. 2006; Ulker et al. 2003]. Therefore, the purpose of this study was to explore whether chronic administration of sesamin increased the aortic NO bioactivity in SHRs through these aforementioned mechanisms.
Methods
Drugs
Sesamin (>94% purity) was provided by Tianyi Lvbao Technology Co. (Wuhu, China). Its structure and invention patent number were described in our previous studies [Kong et al. 2009; Zhang et al. 2013]. Norepinephrine, phenylephrine (Phe), acetylcholine (ACh), sodium nitroprusside (SNP) and mercaptopropionylglycine (MPG) were purchased from Sigma (St Louis, MO, USA).
Animals and experimental design
Male Wistar-Kyoto (WKY) rats and SHRs aged 12 weeks were purchased from SLAC Laboratory Animal Co. (Shanghai, China). All rats received humane care in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication no. 85-23, revised 1996). The study protocol was approved by the Animal Care and Use Committee of Wannan Medical College.
After 1 week of accommodation in the new environment, 30 SHRs were randomly separated into three groups: SHR model group (n = 10) and two sesamin treatment groups (given sesamin by gavage at the daily dose of 160 or 80 mg/kg, n = 10 each). WKY rats (n = 10) were used as normotensive controls. Sesamin was suspended in 0.5% carboxymethylcellulose sodium and orally administrated for 8 weeks in two treatment groups. Untreated groups received an equal volume of carboxymethylcellulose sodium as a control.
The systolic blood pressure (SBP) was measured in conscious rats using the tail-cuff method (ALC-NIBP, Shanghai, China) as described in our previous studies [Kong et al. 2009, 2011]. At the end of the study, all rats were fasted overnight and anaesthetized by an intraperitoneal injection of sodium pentobarbital. Aortas from the rats were carefully removed, cleaned of adhering tissue and divided into two parts. Two transverse rings (3 mm in length) were cut from part of the descending thoracicaortas and used for vascular reactivity experiments. The remaining aortas were quickly frozen in liquid nitrogen and stored at −80°C for future processing. In addition, carotid arteries were removed from rats and immediately frozen in Tissue-Tek OCT embedding medium (Sakura Finetek, Torrance, CA, USA) for further superoxide anion detection.
Vascular reactivity studies
As mentioned in our previous study [Kong et al. 2009, 2014], the aortic ring was suspended by a pair of stainless steel hooks in a water-jacketed organ bath containing 10 ml Krebs solution (composition in mM: NaCl 118.3, KCl 4.7, CaCl2 2.5, KH2PO4 1.2, MgSO4 1.2, NaHCO3 25 and glucose 11.1) at 37°C, and continuously bubbled with 95% O2 and 5% CO2. The rings were stimulated with 3 × 10−7 M norepinephrine to evaluate their viability and were serially washed back to baseline levels and equilibrated once again.
The concentration–relaxation response curves to ACh (10−8 to 10−4 M) and sodium SNP (10−9 to 10−6 M) were performed in aortic rings, which were precontracted by 10−6 M Phe. The relaxant responses to ACh and SNP were calculated as a percentage of the response to Phe.
In order to evaluate the superoxide anion-mediated NO inactivation, the response to ACh was tested in Phe-precontracted aortic rings previously incubated for 20 min with 10 μM MPG. Incubation with MPG, a potent superoxide anion scavenger, has been reported to improve endothelial function in SHR aortic rings [Ulker et al. 2003]. In the present experiment, the disparity between ACh-induced maximum relaxation values obtained in the absence and presence of MPG was used to reflect NO oxidative inactivation.
In situ detection of vascular superoxide anion
Carotid arteries were included in OCT embedding medium. Ten-micrometer cross sections were obtained in a freezing microtome (Leica, Germany, Wetzlar, Germany). Sections were incubated for 30 min with 10 μM dihydroethidium (DHE, Beyotime Institute of Biotechnology, Shanghai, China) to evaluate carotid arterial superoxide anion levels in situ. DHE is oxidized by superoxide anion to yield ethidium which stains DNA. In preliminary experiments, DHE fluorescence was almost abolished by the superoxide anion scavenger MPG, indicating the specificity of this reaction. Sections were photographed and DHE fluorescence was quantified using image J software. The mean fluorescence was quantified and normalized to values obtained from WKY rats.
Biochemical analysis
Total superoxide dismutase (T-SOD), glutathione peroxidase (GPx) and catalase activity assay kits were purchased from Beyotime Institute of Biotechnology. T-SOD activity in the aortas was measured using the xanthine oxidase method as described previously [Kong et al. 2012]. The activity of catalase was estimated by following the breakdown of hydrogen peroxide according to the method described previously [Quan et al. 2009]. The activity of GPx was determined in aortic homogenates according to the manufacturer’s protocol.
Determination of aortic nitrotyrosine level
Peroxynitrite is a strong oxidant formed in the reaction between NO and superoxide anion. Then, the subsequent reaction of peroxynitrite with protein results in the formation of nitrotyrosine, which is frequently used as a stable footprint of in vivo peroxynitrite mediated oxidative/nitrative stress and superoxide anion mediated inactivation of NO [Ma et al. 2001], and a surrogate index of in vivo uncoupled eNOS-dependent damage [ter Steege et al. 1998].
Aortic nitrotyrosine level was measured using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Cell Biolabs, San Diego, CA, USA) according to the manufacturer’s instructions, as described in detail previously [Kong et al. 2014; Li et al. 2013].
Western blot analysis
As described in our previous study [Kong et al. 2009, 2012, 2013], equal amounts (30 μg) of proteins were applied to an 8–10% sodium dodecyl sulfate polyacrylamide separating gel and transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore, Bedford, MA, USA). After blocking with 5% skimmed milk or 1% bovine serum albumin (BSA) in Tris buffered saline with Tween 20 (TBST) at room temperature for 1 h, the membrane was incubated with the primary antibodies against eNOS (BD Transduction Laboratories, San Diego, CA, USA), phosphorylated eNOS (Ser1177) (Cell Signaling Technology, Danvers, MA, USA), nicotinamide adenine dinucleotide phosphate (NADPH) oxidase subunit p47phox (Santa Cruz Biotechnology, Santa Cruz, CA, USA), dihydrofolate reductase (DHFR) (Epitomics, Hangzhou, China) or copper, zinc superoxide dismutase (Cu/Zn-SOD; Epitomics) overnight. After washing the membranes three times, the immunoblots were incubated with the appropriated secondary antibodies for 1 h. Antibody-bound proteins were detected by a Millipore enhanced chemiluminescence kit. The blots were evaluated by densitometry using image J software. The intensity of the bands was normalized to that of tubulin.
For immunoblot analysis of the dimeric and monomeric form of eNOS protein, arterial samples were not heated and the temperature of the gel was maintained below 15°C during low-temperature sodium dodecyl sulfate-polyacrylamide gel electrophoresis [Satoh et al. 2005; Yamamoto et al. 2007]. The remaining steps were the same as those described above.
Statistical analysis
Data were expressed as mean ± SD. Comparisons among the four groups were determined by the use of analysis of variance (ANOVA) followed by a Newman–Keuls test. Changes in the SBP over time and dose–response curves were analyzed by two-way repeated-measures ANOVA followed by a Newman–Keuls test. p less than 0.05 was considered statistically significant.
Results
Systolic blood pressure
As shown in Figure 1, throughout the experimental period, the SBP level was significantly higher in vehicle-treated SHRs compared with control WKY rats. Daily oral administration of both 160 and 80 mg/kg sesamin induced a progressive reduction in SBP (reduced by 16% and 12% at the end of 8 weeks respectively, p < 0.05 versus vehicle-treated SHRs). These results confirmed previous evidence that sesamin has a blood pressure lowering effect.
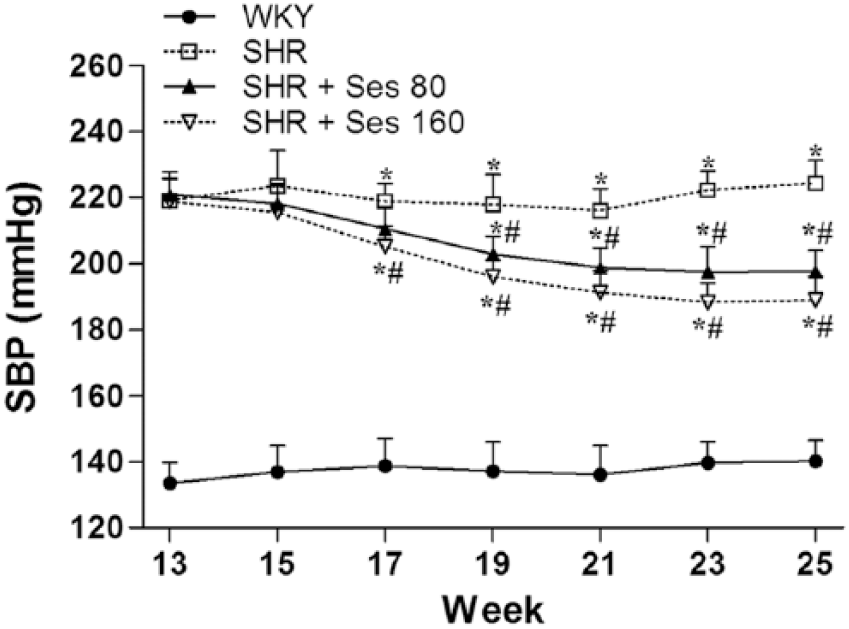
Effects of sesamin on SBP in rats from each group. Values are expressed as mean ± SD, n = 10 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. SD, standard deviation; Ses, sesamin; SBP, systolic blood pressure; SHR, spontaneously hypertensive rat; WKY, Wistar-Kyoto.
In vitro vascular activity
As shown in Figure 2(a), the aortic endothelial-dependent relaxation induced by ACh was obviously reduced in vehicle-treated SHRs. Treatments with sesamin obviously ameliorated the impairment of endothelial-dependent vasodilator response. Moreover, no differences were observed in the endothelium-independent vasorelaxation induced by NO donor SNP among all experimental groups. Sesamin did not modify the relaxant response to SNP [Figure 2(b)].

In vitro vascular activity. Endothelial-dependent relaxation induced by ACh in the absence (a) and presence (c) of 10 μM MPG (a potent superoxide anion scavenger), and endothelium-independent relaxation induced by SNP (b) in aortic rings from each group. The disparity between ACh-induced maximum relaxation values obtained in the absence and presence of MPG was used to reflect NO oxidative inactivation (d). Values are expressed as mean ± SD, n = 5–6 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. ACh, acetylcholine; MPG, mercaptopropionylglycine; NO, nitric oxide; SD, standard deviation; Ses, sesamin; SHR, spontaneously hypertensive rat; SNP, sodium nitroprusside; WKY, Wistar-Kyoto.
As shown in Figure 2(c), the superoxide anion scavenger MPG (10 μM) added to the bath significantly enhanced ACh-induced relaxation in aortic rings from vehicle-treated SHRs. Therefore, NO oxidative inactivation was reflected by the disparity between ACh-induced maximum relaxation values obtained in the absence and presence of MPG. Figure 2(d) showed that vehicle-treated SHRs were associated with obvious NO oxidative inactivation. A significant improvement in NO inactivation was found in the sesamin 160 mg/kg treated group.
Aortic eNOS expression
As shown in Figure 3, the protein expression of total eNOS was not different among all experimental groups. Nevertheless, the phosphorylated level of eNOS was obviously reduced in vehicle-treated SHRs, and these abnormal changes were essentially reversed by treatment with 160 mg/kg sesamin for 8 weeks.

Effects of sesamin on the protein expression of total eNOS (T-eNOS) and phosphorylated eNOS (P-eNOS) in aortic tissues by Western blotting. The panel shows representative bands and the histogram represents optical density values normalized to the corresponding T-eNOS. Values are expressed as mean ± SD, n = 4 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. eNOS, endothelial nitric oxide synthase; SD, standard deviation; Ses, sesamin; SHR, spontaneously hypertensive rat; WKY, Wistar-Kyoto.
Aortic eNOS dimers and DHFR protein expression
As shown in Figure 4(a), vascular DHFR protein levels were significantly downregulated in vehicle-treated SHRs compared with control WKY rats. Administration of sesamin effectively increased the expression of DHFR in SHR aortas.

Effects of sesamin on the DHFR protein expression and eNOS uncoupling in aortic tissues by Western blotting. (a) The panel shows representative bands of DHFR and the histogram represents optical density values normalized to the corresponding tubulin. Values are expressed as mean ± SD, n = 4 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. (b) Panel shows representative blots of eNOS after low-temperature SDS-PAGE. The disruption of eNOS dimer indicates the uncoupling of eNOS occurred in SHR aortas. DHFR, dihydrofolate reductase; eNOS, endothelial nitric oxide synthase; SD, standard deviation; Ses, sesamin; SHR, spontaneously hypertensive rat; WKY, Wistar-Kyoto.
The uncoupled eNOS in vehicle-treated SHR aortas was confirmed by the decrease in eNOS dimer form [Figure 4(b)]. The disruption of aortic eNOS protein dimers was significantly prevented by treatment with sesamin for 8 weeks [Figure 4(b)].
Vascular superoxide anion product and p47phox protein expression
Ethidium red fluorescence in sections of carotid arteries obtained from incubation with DHE was used to characterize and localize superoxide anion production within the vascular wall. As shown in Figure 5(a), positive red nuclei could be observed in adventitial, medial and endothelial cells. As shown in Figure 5(b), the content of superoxide anion in vehicle-treated SHR carotid arteries was markedly higher than those in control WKY rats. Chronic treatment with sesamin induced a significant decrease in superoxide anion production.

Effects of sesamin on the superoxide anion production in carotid arteries. (a) The panel shows representative fluorescence photomicrographs. (b) The histogram represents the average values in each group. Values are expressed as mean ± SD, n = 7 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. SD, standard deviation; Ses, sesamin; SHR, spontaneously hypertensive rat; WKY, Wistar-Kyoto.
Significant protein overexpression of NADPH oxidase subunit p47phox was displayed in aortic tissue from vehicle-treated SHRs compared with control WKY rats. Treatment with sesamin obviously ameliorated the increase in p47phox protein expression (Figure 6).
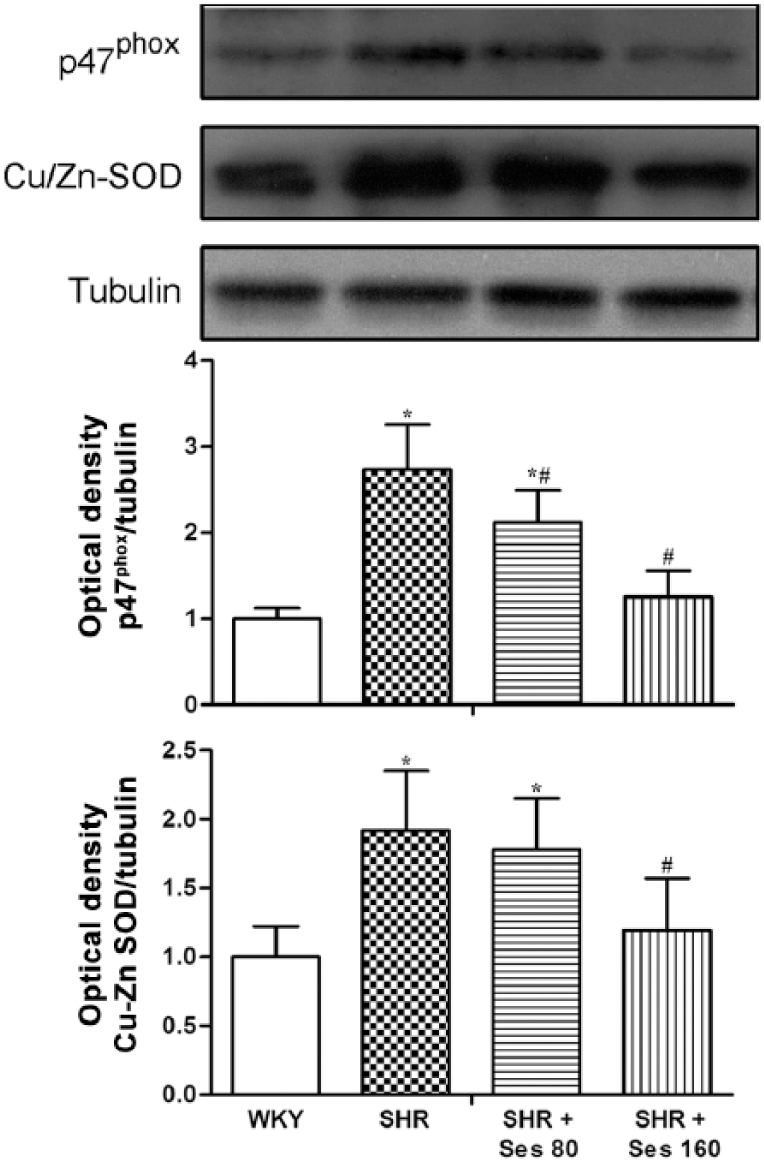
Effects of sesamin on the protein expression of p47phox and Cu/Zn-SOD in aortic tissues by Western blotting. The panel shows representative bands and the histogram represents optical density values normalized to the corresponding tubulin. Values are expressed as mean ± SD, n = 4 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. Cu/Zn-SOD, copper, zinc superoxide dismutase; SD, standard deviation; Ses, sesamin; SHR, spontaneously hypertensive rat; WKY, Wistar-Kyoto.
Aortic nitrotyrosine level, antioxidant enzymes activities and Cu/Zn-SOD protein expression
As shown in Figure 7(a), aortic nitrotyrosine levels were markedly increased in vehicle-treated SHRs compared with control WKY rats. Treatment with sesamin reduced the vascular nitrotyrosine levels.

Effects of sesamin on the aortic nitrotyrosine level and activities of T-SOD, GPx and catalase. Values are expressed as mean ± SD, n = 5 per group. *p < 0.05 versus WKY control rats. #p < 0.05 versus vehicle-treated SHRs. GPx, glutathione peroxidase; SD, standard deviation; Ses, sesamin; SHR, spontaneously hypertensive rat; T-SOD, total superoxide dismutase; WKY, Wistar-Kyoto.
As shown in Figure 7(b)–(d), T-SOD activity was found to be significantly higher in vehicle-treated SHR aortic homogenates. However, both GPx and catalase activities were similar in vehicle-treated SHRs and control WKY rats. Sesamin treatment did not modify the levels of GPx and catalase activity but obviously reduced the elevated T-SOD activity.
In accordance with the observed increase in SOD activity, protein expression of aortic Cu/Zn-SOD was upregulated in vehicle-treated SHRs. Administration of sesamin decreased the expression of Cu/Zn-SOD in SHR aortas (Figure 6).
Discussion
NO, generated by eNOS from L-arginine, within vascular endothelium plays pivotal roles in the maintenance of blood pressure and vascular tone [Loscalzo and Welch, 1995]. Among the various endothelium-derived molecules, endothelium-dependent vasodilatation relies almost entirely on the release of NO in rat aortas [Nagao et al. 1992]. Therefore, the most characteristic feature of endothelial dysfunction is a diminished bioactivity of endothelium-derived NO. In the present study, vehicle-treated SHRs were accompanied by a decreased aortic endothelium-dependent relaxant response to Ach, an endothelium- and NO-dependent vasodilator. Sesamin was able to ameliorate the endothelial dysfunction in SHRs. These results suggest that sesamin improves endothelial function in SHRs by increasing NO bioactivity.
A decline in NO bioavailability is mainly caused by the reduction of NO synthesis by eNOS, the increased oxidative inactivation of NO by superoxide anion, and the reduced responsiveness of VSMCs to NO [Griendling et al. 2000; Nagao et al. 1992; Rodriguez-Rodriguez et al. 2007]. The magnitude of endothelium-independent vasodilation induced by SNP, a soluble guanylate cyclase activator, was similar in SHRs and WKY rats, thereby suggesting the presence of an intact and functional VSMC layer in SHRs. In addition, the SNP-induced relaxation was unaffected by sesamin, indicating that the improvement in endothelium-derived NO bioactivity after treatment with sesamin is not attributed to the downstream effects on VSMCs. Thus, the aforementioned third potential mechanism could be ruled out.
It is well known that eNOS within the normal endothelium is the major contributor to NO production. Several potential mechanisms would be involved in reduced NO biosynthesis, such as the impaired expression of eNOS, its phosphorylation and the uncoupled state of eNOS. In this study, the aortic expression of total eNOS was not altered in SHRs, but the phosphorylation level of eNOS was significantly decreased in the aortas. Treatment with sesamin obviously increased phosphorylated eNOS in SHR aortas. Furthermore, the formation of eNOS protein homodimers is necessary for enzymatic activity and NO biosynthesis. The uncoupling of eNOS refers to a process in which the enzyme generates superoxide anion rather than NO [Landmesser et al. 2003]. In our study, the aortic uncoupled state of eNOS occurring in SHRs was confirmed and characterized by the disruption of eNOS dimers. Peroxynitrite can disrupt eNOS protein dimers through oxidation and displacement of the zinc metal ion, resulting in the reduction of NO production [Zou et al. 2002]. DHFR is a key enzyme in the synthesis of tetrahydrobiopterin (eNOS cofactor). Downregulated DHFR causes the uncoupling of eNOS, leading to a decrease in NO biosynthesis [Chalupsky and Cai, 2005]. We showed that the uncoupled state of eNOS in SHR aortas was associated with the enhancement of nitrotyrosine (an indicator of peroxynitrite) level and the downregulation of DHFR protein. Sesamin treatment essentially reversed these abnormalities. Taken together, these data demonstrate for the first time that the enhancement of aortic NO biosynthesis in SHRs after sesamin treatment is due to the upregulation of phosphorylated eNOS expression and improvement of uncoupled eNOS, and the latter might be attributed, at least in part, to the decrease in nitrotyrosine and upregulation of DHFR.
Enhanced production of reactive oxygen species, in particular superoxide anion, inactivates NO thereby diminishing its half life, and leads to the generation of peroxynitrite, which reacts with tyrosine residues to produce nitrotyrosine. Superoxide mediated NO inactivation could contribute to hypertension and endothelial dysfunction by limiting the availability of biologically active NO. The elevation of aortic superoxide anion production and nitrotyrosine abundance was observed in SHRs. In accordance with our previous studies [Kong et al. 2009; Zhang et al. 2013] and others [Nakano et al. 2003], sesamin was effective in decreasing superoxide anion production and reducing nitrotyrosine accumulation in aortas. However, this evidence for aortic NO inactivation is indirect. In the current study, we obtained direct and tangible evidence for the existence of superoxide anion mediated NO inactivation within the vascular wall. Preincubation of aortic rings with the superoxide scavenger MPG enhanced the endothelial function in SHRs. The difference between ACh-induced maximum relaxation values in the absence and presence of MPG was used to reflect superoxide anion mediated NO inactivation. Our analysis revealed that exaggerated NO inactivation occurred in the aortas of SHRs and was prevented by sesamin treatment. These results indicate that another cause of the increase in aortic NO bioactivity provided by sesamin may possibly be the improvement in NO oxidative inactivation.
NADPH oxidase has been found in all layers of the vessel wall and shown to be one of the main sources of vascular superoxide, along with the uncoupled state of eNOS [Bayraktutan et al. 2000; Hong et al. 2000]. Uncoupling of eNOS reduces NO production and also promotes superoxide generation by NO syntheses. Therefore, the disruption of eNOS dimers and upregulation of p47phox (a cytoplasmic subunit of NADPH oxidase) expression observed in this study could raise superoxide production and nitrotyrosine level, and thereby contribute to NO oxidative inactivation in the aortas of SHRs. Treatment with sesamin decreased the protein level of p47phox and restored the uncoupled eNOS, suggesting that sesamin rescues the superoxide anion mediated NO inactivation by these mechanisms.
SOD, GPx and catalase represent important components of the antioxidant defense system. Excessive release of oxygen radicals, if not controlled by one or more of these enzymes, can lead to oxidant stress. To the best of our knowledge, this is the first time the in vivo effect of sesamin on the vascular antioxidant enzymes has been evaluated. Consistent with a previous report [Ulker et al. 2003], T-SOD activity and Cu/Zn-SOD expression were significantly increased in SHR aortas while GPx and catalase activities were not altered. SOD plays an important role in converting superoxide to hydrogen peroxide. The upregulation of SOD may be a compensatory response in the face of elevated superoxide anion derived from both NADPH oxidase and eNOS uncoupling. The increase in superoxide anion production may greatly exceed the enhancement of antioxidant enzyme SOD. Thus, the increased SOD activity and expression in SHR aortas does not prevent but may slow down the progression of vascular oxidative injury. Sesamin treatment did not modify the activities of GPx and catalase but obviously decreased the compensatory elevated T-SOD activity and Cu/Zn-SOD expression. Although SOD was decreased in SHRs treated with sesamin, the generation of superoxide anion was greatly improved by sesamin treatment, resulting in the correction of oxidant and antioxidant imbalance and the improvement of superoxide mediated NO inactivation.
Conclusion
In summary, the present study confirms and extends previous evidence about the antihypertensive effect and the improvement in endothelial dysfunction of sesamin in hypertensive rats. These effects seem to be related to restore aortic NO bioactivity in SHRs, resulting from elevated NO biosynthesis by upregulation of phosphorylated eNOS and suppression of eNOS dimer disruption; and reduced NO oxidative inactivation through downregulation of p47phox and amelioration of eNOS uncoupling (Figure 8). These findings suggest that sesamin may be considered as a potential strategy for the prevention or therapy of cardiovascular diseases, including hypertension.

Summary of the mechanisms of sesamin that enhance aortic NO bioactivity in SHRs. DHFR, dihydrofolate reductase; eNOS, endothelial nitric oxide synthase; NADPH, nicotinamide adenine dinucleotide phosphate; NO, nitric oxide; ONOOˉ, peroxynitrite; O2ˉ, superoxide anion; P-eNOS, phosphorylated eNOS; SHR, spontaneously hypertensive rat; SOD, superoxide dismutase.
Footnotes
Acknowledgements
Xiang Kong and Wei Li contributed equally to this study as cofirst authors. XK and JRY conceived and designed the experiments. XK, WL, LQG and JXZ performed the experiments. XK and WL analyzed the data. XK, XPC, WYL and JRY wrote the paper. All authors read and approved the final manuscript.
Funding
This research was supported by the Anhui Provincial Natural Science Foundation (grant number 1308085QH145).
Conflict of interest statement
The authors declare that they have no competing interest.
