Abstract
Objective
The pathogenesis of polycystic ovarian syndrome (PCOS) remains unclear, but is known to be related to increased insulin resistance, diabetes, obesity, oxidative stress, inflammation, and cardiovascular disease. No meta-analyses have examined whether PCOS is associated with nitric oxide (NO). Therefore, we aimed to evaluate the association between serum/plasma nitrite levels and PCOS.
Methods
We performed a meta-analysis. To identify eligible original articles, we searched online computerized databases, including EMBASE, Medline, and Web of Science. The characteristics of each single study, as well as the standard mean difference (SMD) with corresponding confidence intervals (CIs), were calculated and subgroup analysis was performed to examine heterogeneity.
Results
We included 12 articles with 16 studies and a total of 895 patients were included in the meta-analysis. We found a significant association between PCOS and serum or plasma nitrite levels, with a pooled SMD of −0.61 (95% CI: −1.05 to −0.16). The final pooled data were determined by the random effects model because significant high heterogeneity (I2 = 89%) was found.
Conclusions
Our meta-analysis shows that decreased serum or plasma nitrite levels are associated with PCOS. The pathogenesis of PCOS may be due to endothelial dysfunction; however, further research to confirm this possibility is required.
Keywords
Introduction
Polycystic ovarian syndrome (PCOS) is a common reproductive endocrine and metabolic disorder that affects 6% to 10% of women. PCOS is characterized by ovulatory dysfunction, menstrual irregularities hyperandrogenism, and polycystic ovaries.1,2 The pathogenesis of PCOS remains unclear, but is known to be related to increased insulin resistance, diabetes, obesity, oxidative stress, inflammation, and cardiovascular diseases. PCOS remains a syndrome and no single diagnostic criterion is sufficient for clinical diagnosis.
Insulin resistance plays an important role in the pathogenesis of PCOS. Hyperinsulinemia may contribute to the development of endothelial dysfunction, hypertension, diabetes, and dyslipidemia, and leads to an increased risk for metabolic disorders.3,4 Recent studies showed that high levels of advanced glycation end products and androgen induced inflammation and oxidative stress.5,6 These studies also showed a strong association between endothelial dysfunction and oxidative stress in the pathogenesis of PCOS, and these are prognostic indicators of cardiovascular disease, as well as recurrent pregnancy loss. Nitric oxide (NO) is one of the most important regulators of vascular physiology, such as neuronal transmission. NO is a relatively stable gas free radical that diffuses through the cell membrane. 7 Nitric oxide is synthesized by nitric oxide synthase (NOS) during conversion of L-arginine using oxygen and NADPH as the cofactors. NO can adversely affect carbohydrates, proteins and lipids and, together with other inflammatory mediators, results in cell damage. 8 NO plays an inevitable role under physiological and pathological conditions involved in regulation of vascular tone, blood pressure, vascular remodeling, and inflammation. 9 Because of the short half-life of NO in vivo, measurement of NO levels in human is extremely difficult. However, NO produces the stable NO products of nitrate and nitrite. Previous studies have shown a causal relationship between NO and nitrite. 10
A recent study concluded that there was endothelial dysfunction in women with PCOS, even if they were young and non-obese. 11 Studies have indicated that patients with PCOS are under high oxidative stress. 6 In the past decade, various studies have examined nitrite in patients with PCOS and have investigated the associations between them. There is contradictory evidence on the level of nitrite in PCOS. Some studies have reported decreased nitrite levels in patients with PCOS, while some studies have found the opposite result.12,13 However, no meta-analyses have examined whether PCOS is associated with NO. Therefore, we conducted a meta-analysis to further examine the association between serum/plasma nitrite levels and PCOS (Supporting information: PRISMA Checklist). 14
Material and methods
Search strategy
Studies on PCOS and serum or plasma nitrite levels were identified. To identify all of the relevant original articles, we comprehensively searched a series of online computerized databases, including EMBASE, Medline, and Web of Science, using the key words “nitric oxide”, “nitrate”, “nitrite”, “nitrosamine”, “polycystic ovary syndrome”, and “PCOS” using the operator OR or AND. Papers were searched up to December 2018 and without limits of language. Reference sections from the identified literature were also reviewed in case of missing extra qualified papers. We first screened the titles and abstracts of the searched studies, and then evaluated the full article in case it contained data regarding PCOS and serum or plasma nitrite levels. Two authors (MC and XXY) reviewed all abstracts and screened the reference lists. Disagreements were resolved by consensus.
Inclusion and exclusion criteria and data extraction
Original examinations were cautiously checked and there were no country limitations in our study. The inclusion criteria were as follows: (1) all examination and control subjects were limited to adults with PCOS and analyzed reliably by either the consensus statement declared at Rotterdam 15 or National Institute of Health (NIH) 16 criteria; (2) there was no significant difference in terms of body mass index (BMI) and age in the study and control groups; (3) studies were restricted to humans, contained original information, and were present in either full-text or abstract form; (4) studies reported serum/plasma nitrite levels; and (5) studies in which all PCOS group subjects did not have any conceivable inclining factors (such as Cushing syndrome, thyroid dysfunction, ovarian tumors) that may be related to their PCOS or those who were taking drugs (e.g., oral contraceptives, other hormonal treatments) at the time of the study. The exclusion criteria were as follows: (1) duplicate publications, case reports, abstracts, non-English, review articles, and editorials; and (2) the data were not sufficient for investigation or extraction of information.
Data extraction
Data were gathered for each study concerning the name of the first author, publication year, study design, sample size, age, nitrite levels, and BMI (mean ± standard deviation) in patients with PCOS and in control subjects.
Risk of bias and statistical analysis
We evaluated the quality of each individual included study using the Newcastle–Ottawa Scale. 17 Meta-analysis was performed using Cochrane statistical software Review Manager 5.3 (Copenhagen, Denmark). When P was < 0.05, the random effects model was applied according to the P value of chi-squared statistics. Heterogeneity was assessed by calculating the I2 metric. An I2 value > 75% was considered as high heterogeneity, an I2 value between 50% and 75% as moderate heterogeneity, and an I2 value between 25% and 50% as low heterogeneity. An I2 value < 25% was considered homogeneous. If the I2 value was > 50% the random effects model was used and if the I2 value was < 50% the fixed effects model was used to combine effect size. We also performed subgroup analysis and meta-regression to determine the sources of heterogeneity. Repeated sensitivity analysis was performed to estimate the effect of each individual study in the meta-analysis by removing different individual studies each time. Nitrite levels in each study were calculated using the standard mean difference (SMD) with the 95% confidence interval (CI).
Results
Study selection
Using our search strategy, we identified 98 potentially relevant articles from online electronic databases and one study from reference records. Subsequent to excluding copies, 88 records remained. After reading the titles or abstracts of these 88 studies, we excluded 54 articles according to inclusion criteria. When we screened the full text of the remaining 33 articles, 19 were excluded because they did not report adequate data, one was not in English, and one study was a duplicate. Eventually, 12 articles including 16 studies satisfied the eligibility criteria.12,18–27 The selection process is shown in Figure 1 and the information of each study is shown in Table 1.

Search strategy to identify articles on the relationship between polycystic ovary syndrome and prostatic-specific antigen levels.
Description of included studies in the meta-analysis.
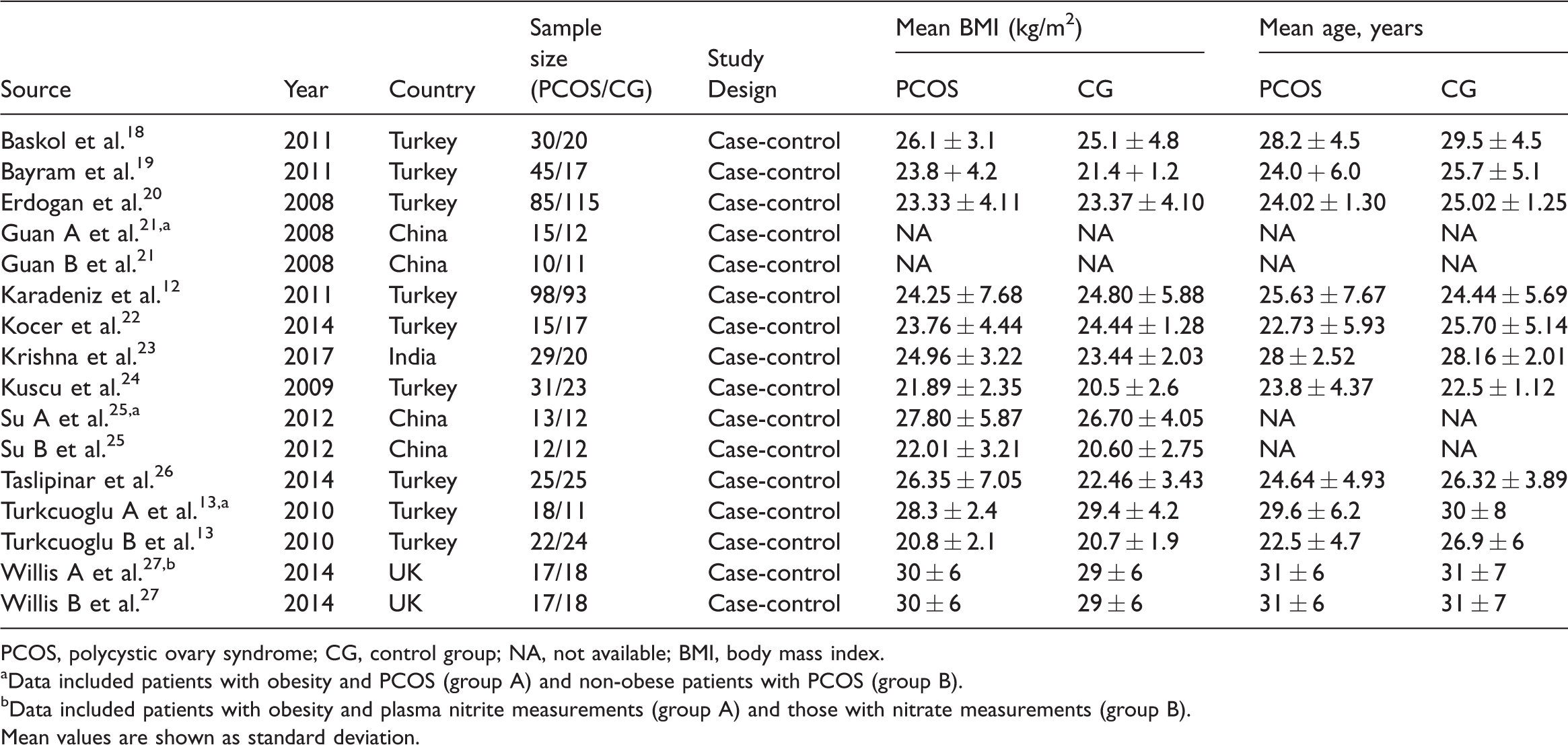
PCOS, polycystic ovary syndrome; CG, control group; NA, not available; BMI, body mass index.
aData included patients with obesity and PCOS (group A) and non-obese patients with PCOS (group B).
bData included patients with obesity and plasma nitrite measurements (group A) and those with nitrate measurements (group B).
Mean values are shown as standard deviation.
The meta-analysis included a total sample size of 895 (465 cases and 430 controls) and all of them were case–control studies. Three studies13,21,25 reported results separately for patients with obesity and PCOS and non-obese patients with PCOS, and one study 27 reported plasma nitrite and nitrate study groups. Nine studies12,13,18–20,23,24,26,27 investigated European populations and three studies21,23,25 analyzed Asian populations.
Meta-analysis results
A Forest plot of the relationship of PCOS with serum or plasma nitrite levels is shown in Figure 2. We found a significant association between PCOS and serum or plasma nitrite levels, with a pooled SMD of −0.61 (95% CI: −1.05 to −0.16; P < 0.01). High significant heterogeneity (I2 = 89%) was found among the studies. Therefore, the pooled data were determined by the random effects model.

Relationship between polycystic ovary syndrome with serum or plasma nitrite levels. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval.
Subgroup analysis
The SMD (−0.09) for a BMI ≥ 25 kg/m2 in studies with a mean BMI ≥ 25 kg/m2 was not significant (95% CI: −0.49 to 0.30, P > 0.01) (Figure 3). The SMD (−1.01) for a BMI < 25 kg/m2 in studies with a mean BMI < 25 kg/m2 was not significant (95% CI: −1.78 to −0.24, P > 0.01) (Figure 4). The SMD (−0.32) for age ≥ 25 years in studies with a mean age ≥ 25 years was not significant (95% CI: −1.11 to 0.48, P > 0.01) (Figure 5). The SMD (−0.80) for an age < 25 years in studies with a mean age < 25 years was not significant (95% CI: −1.62 to 0.01, P > 0.01) (Figure 6). The SMD (−1.27) for Asian ethnicity in studies with an Asian population was not significant (95% CI: −2.24 to −1.30, P > 0.01) (Figure 7). The SMD (−0.32) of European ethnicity in studies with a European population was not significant (95% CI: −0.77 to 0.13, P > 0.01) (Figure 8).

Forest plot of body mass index ≥25 kg/m2 in subgroup analysis. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval.

Forest plot of body mass index <25 kg/m2 in subgroup analysis. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval.

Forest plot of age ≥25 years in subgroup analysis. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval
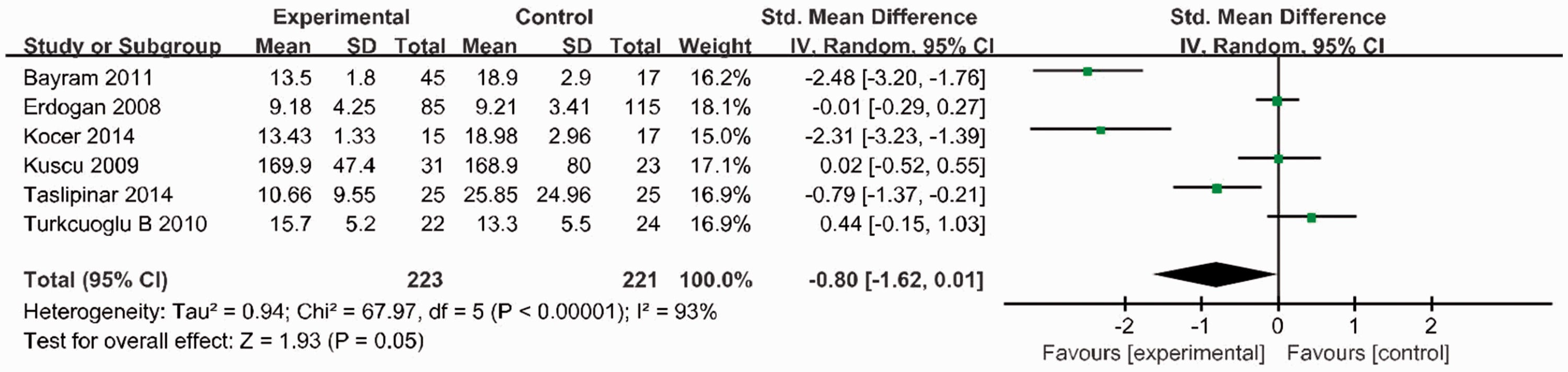
Forest plot of age <25 years in subgroup analysis. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval.

Forest plot of the Asian population in subgroup analysis. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval.

Forest plot of the European population in subgroup analysis. SD, standard deviation; std, standard; IV, inverse variance; CI, confidence interval.
Sensitivity analysis
Forest plots showed that our results had high heterogeneity (I2 = 89%). Sensitivity analysis results showed that the pooled value ranged from −0.43 (95% CI: −0.82 to −0.05) to −0.68 (95% CI: −1.14 to −0.22). Additionally, subgroup heterogeneity was also analyzed (I2 = 53% for BMI ≥ 25 kg/m2; I2 = 94% for BMI < 25 kg/m2; I2 = 91% for age ≥ 25 years; I2 = 93% for age < 25 years; I2 = 85% for an Asian population; I2 = 88% for a European population). However, sensitivity analysis results were different in the subgroups. In the BMI ≥ 25 kg/m2 subgroup, the results dramatically changed when Taslipinar et al.’s study 26 was removed (I2 decreased from 53% to 0%). In the age ≥ 25 years subgroup, when we removed Krishna et al.’s study, 23 the I2 decreased from 91% to 0%. In the Asian population subgroup, when we removed Krishna et al.’s study, 23 the I2 decreased from 85% to 0%. Meta-regression analysis showed that the publication date may have been the source of heterogeneity (publication date: ≤2010, P = 0.03; country: Europe, P = 0.10; sample size: PCOS ≤30, P = 0.78).
Quality assessment
The trim and fill technique showed that no investigation should have been statistically corrected. Table 2 summarizes the consequences of the quality evaluation. The methodological quality was evaluated as high in three studies, moderate in six studies, and low in three studies.
Methodological assessment according to the Newcastle–Ottawa scale.

FU, follow-up.
− indicates no stars (*).
aWe considered a study to be of high quality when the total score was 8 or 9 stars, moderate quality when the total score was 6 or 7 stars, and low quality when the total score was 5 stars or fewer.
Discussion
This meta-analysis showed that PCOS was associated with serum or plasma nitrite levels. Furthermore, in patients with PCOS, serum or plasma nitrite levels were reduced compared with controls. The value of I2 = 89 showed that there was high heterogeneity. We also performed subgroup analysis and meta-regression to determine the sources of heterogeneity. Additionally, sensitivity analysis showed that when any individual study was overlooked, the general outcomes remained the same. Therefore, our meta-analysis results appear to be reliable.
The mechanisms of PCOS are not completely understood, but recent studies suggest that insulin resistance plays an important role in the pathogenesis of PCOS. 3 Endothelial dysfunction, increased oxidative stress, decreased antioxidant defense, excess androgen levels, and dyslipidemia have important effects on cardiovascular disease in patients with PCOS. 28 Orio et al. 29 assessed youthful patients with PCOS and no metabolic and cardiovascular disease and reported disturbance in endothelial functions in the early period of this disease. However, Mather et al. 30 showed normal endothelial function in patients with PCOS. Kandarakis et al. 31 indicated that high serum androgen levels in women with PCOS may add to impairment of endothelial structure and increased risk of cardiovascular disease. Our meta-analysis results support the outcomes from most previous studies 18 because serum or plasma nitrite levels were lower in patients with PCOS than in controls.
Sorensen et al. 32 reported that endothelial dysfunction in patients with PCOS is independent from age and BMI. Obesity is an additional burden and a risk factor in PCOS, and is involved in the pathogenesis of hyperandrogenism and insulin resistance. Cupisti et al. 33 found that a high BMI (≥25 kg/m2) was the main factor for endocrinological and metabolic disturbances in women with PCOS. Willis et al. 27 found that plasma nitrite and nitrate levels were unaltered in women with PCOS compared with age- and BMI-matched controls. Consistent with previous studies, 32 our BMI and age subgroup analysis results were not significant. However, further research is required to determine the relationship between serum or plasma nitrite levels and obesity or BMI. There have been no studies on racial differences in NO levels across different ethnicities in women with PCOS. In our meta-analysis, there was no obvious difference in nitrite levels between Asian and European populations, but more studies in other ethnicities are required to evaluate if serum or plasma nitrite levels are influenced by ethnicity.
The relationship of PCOS with serum or plasma nitrite levels is controversial. Our meta-analysis is the first to support a relation of PCOS with serum or plasma nitrite levels. However, there are some potential limitations of our meta-analysis that should be considered when interpreting the results. First, despite that we investigated the source of heterogeneity, we only examined heterogeneity from the publication date because there was inadequate information and a limited amount of studies. Second, the number of studies was generally insufficient and may have affected the precision of our outcomes. Further larger scale studies need to be performed to confirm our results. Third, the outcomes may have been one-sided by various estimation methods and instruments used to identify serum or plasma nitrite levels. Furthermore, plasma nitrite and nitrate levels reflect not only endogenous NO production, but also dietary nitrate ingestion. Magnesium and vitamin E can increase plasma NO levels. 34 Therefore, dietary variables may have affected our findings. Additionally, plasma NO status may sometimes not reflect tissue NO status. Finally, we could only include studies in the English language because of difficulty in obtaining abstracts or full texts of articles published in other languages. Therefore, our results need confirmation by further research and should be interpreted with caution.
Conclusions
Our meta-analysis shows that serum or plasma nitrite levels are reduced in patients with PCOS. The pathogenesis of PCOS may be due to endothelial dysfunction, but further research on this issue is required in the future.
Footnotes
Author contributions
All authors contributed significantly to this work. M.C. designed and performed the research; M.C. and X.X.Y. wrote and revised the manuscript. All authors approved the final draft of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
