Abstract
Objective:
To investigate, in patients with known or suspected coronary artery disease (CAD) undergoing myocardial perfusion single-photon emission computed tomography (MPS),the interaction between patient symptoms and single-photon emission computed tomography (SPECT) results, and their effects on patient outcomes. Previous data have shown that medical treatment may be as beneficial as invasive treatment for most patients with stable CAD. Nonetheless, patient presentation with typical angina (TA) seems to still lead to more aggressive management, regardless of the amount of myocardial ischemia detected by noninvasive methods.
Methods:
Over 33 ± 20 months, 2958 patients undergoing stress/rest cardiac SPECT were followed. Summed stress, rest and difference scores (SSS, SRS and SDS, respectively) were analyzed. Abnormal SPECT was defined as a SSS>3, and ischemic SPECT as a SDS>1. During follow up, cardiac catheterization (CATH), myocardial revascularization (either percutaneous or surgical), myocardial infarction (MI) and all-cause death were evaluated.
Results:
TA was found in 228 patients (7.7%). Comparing patients with TA with those without it, the former more often had had abnormal (43.0% versus 34.3%, p < 0.001) and ischemic SPECT (25.9% versus 13.6%, p < 0.001). Also, higher rates of MI (2.0% versus 0.6%, p < 0.001), CATH (10.1% versus 4.7%, p < 0.001) and revascularization (7.8% versus 3.0%, p < 0.001) were observed, while death was not significantly different (1.5% versus 1.0%, p = 0.2). Even in the absence of ischemia in SPECT, patients with TA had higher CATH and revascularization rates; death, again, was not significantly increased.
Conclusion:
Although patients with TA more often had ischemic SPECT, all-cause death was not significantly increased. Nonetheless, TA was associated with more frequent referrals for CATH and revascularization, even with nonischemic SPECT. This may suggest that despite current evidence, the management of stable patients with known or suspected CAD is influenced by symptom type.
Keywords
Introduction
The importance of coronary heart disease remains unequivocal, as current data estimate that it affects over 15 million people in the USA alone [Go et al. 2013]. Among the several presentations of coronary heart disease, chronic, stable angina pectoris is found in up to 15% of adult populations worldwide [Hemingway et al. 2008] and therefore takes part of the daily practice of most cardiologists as well as of many clinicians and family practitioners. As a result, stable coronary artery disease (CAD) has been the subject of several large trials assessing the impact of different therapies, all of them ultimately aimed at finding the best way to relieve symptoms and prolong survival [Boden et al. 2007; Hueb et al. 2004].
Stable CAD may be manifested as typical angina (TA), atypical symptoms or may even be asymptomatic. Even though prognosis may not be directly related to symptoms [Zellweger et al. 2004], TA may lead doctors to use more aggressive diagnostic as well as therapeutic interventions, including for example cardiac catheterization (CATH), followed or not by coronary angioplasty. Notwithstanding, recent evidence shows no significant reduction of the risk of cardiovascular events offered by percutaneous coronary intervention with intensive pharmacologic therapy as an initial management strategy in patients with stable CAD, when compared with intensive medical therapy alone [Boden et al. 2007]. Therefore, decisions based only on the type of symptom may be misleading.
Myocardial perfusion single-photon emission computed tomography (MPS) is widely employed for the diagnostic and prognostic evaluation of CAD as well as for the evaluation of the response to different therapies for CAD. Indeed, in the COURAGE Nuclear Substudy, MPS demonstrated different degrees of ischemia reduction in response to medical therapy alone or in conjunction with coronary angioplasty [Shaw et al. 2008]. Also, a recent large study emphasized the importance of noninvasive risk stratification to increase the diagnostic yield of cardiac catheterization in routine clinical practice [Patel et al. 2010]. This study sought to evaluate the roles of symptoms (more specifically, of TA in comparison with other symptoms) and of scintigraphically demonstrated myocardial ischemia on the management of stable patients with known or suspected CAD in the current, ‘post-COURAGE’ era of therapy for CAD.
Methods
Study Population
Consecutive patients ≥18 years with known or suspected CAD undergoing MPS between March 2008 and March 2010 were prospectively enrolled (n = 3184) and followed for 3.0 ± 0.3 years (all for >1 year). Patients with Canadian Cardiovascular Society (CCS) class IV angina or known significant left main coronary artery disease were considered ineligible for the study. A total of 86 patients (2.7%) were censored from the analysis due to early (<60 days after single-photon emission computed tomography [SPECT]) revascularization, and from the remaining, 4.4% (n = 140) were lost to follow up, leaving a final population of 2958 patients. There were no significant differences between followed patients and those lost to follow up (Table 1). The research was approved by the institutional ethics committee and was performed with ethical standards, in compliance with the Declaration of Helsinki 1975, revised Hong Kong 1989.
Comparison between patients followed and those lost to follow up.

Values are expressed as n (%) or mean ± standard deviation.
LVEF, left ventricular ejection fraction.
All patients answered a questionnaire with clinical information. The current use of aspirin, beta-blockers, calcium channel blockers with negative chronotropic properties or statins was registered. Hypertension was defined by a history of BP ≥ 140/90 mmHg or antihypertensive drug use; diabetes mellitus was defined by history and use of insulin or oral hypoglycemic medications. Hypercholesterolemia, prior myocardial infarction (MI), or myocardial revascularization (either percutaneous or with coronary artery bypass surgery) were defined by history. TA was considered present when a patient reported current exercise-induced chest discomfort, relieved by rest or sublingual nitrates [Diamond, 1983], which might be reproduced or not during stress testing. For this study, asymptomatic patients and those with any other symptoms were studied together.
MPS
A 2-day protocol was used. Rest images were obtained after the injection of 99mTc-sestamibi (10–12 mCi). Stress images were obtained after a treadmill test or pharmacologic stress with dypiridamole or dobutamine.
Exercise protocol
A symptom-limited exercise treadmill test was performed with the standard Bruce protocol. At near-maximal exercise, 99mTc-sestamibi (10–12 mCi) was injected intravenously, and exercise was continued at maximal workload for at least 1 minute. Exercise was terminated if there was increasing angina, a fall in blood pressure >20mmHg, limiting dyspnea or fatigue or sustained arrhythmias. Significant ST-segment depression was defined as that ≥1.5mm, 80 ms after the J-point. Patients achieving peak heart rate <85% of maximal predicted heart rate (defined as 220-age in years) in the absence of limiting, typical exercise-induced angina (with or without concomitant significant ST-segment depression) were switched to pharmacologic stress.
Dipyridamole protocol
Dipyridamole (0.56 mg/kg) was infused over 4 minutes with the patient in the supine position and with continuous electrocardiographic monitoring. 99mTc-sestamibi (10–12 mCi) was injected at 8 minutes. Aminophylline (120–240 mg) was intravenously given 2 minutes after radiotracer injection.
Dobutamine protocol
Dobutamine was infused incrementally starting at 10 μg/kg/min, and increasing at 3-minute intervals up to 40 μg/kg/min. Intravenous atropine was added if 85% of the age-corrected maximal predicted heart rate was not achieved at the peak dose or interruption criteria occurred (systolic blood pressure >220 mmHg or diastolic blood pressure > 110 mmHg, relevant cardiac arrhythmias, limiting angina or hypotension). When the target heart rate was achieved, 99mTc-sestamibi (10–12 mCi) was injected intravenously and dobutamine infusion was stopped.
SPECT images were processed with Evolution for Cardiac software (GE Healthcare, Milwaukee, WI, USA). Image interpretation was visual and semiquantitative, using a 17-segment model of the left ventricle, scored by consensus of 2 expert observers using a 5-point scale (0 = normal; 1 = equivocal; 2 = moderate; 3 = severe reduction of tracer uptake; 4 = absence of detectable radiotracer activity). The summed stress score (SSS) and summed rest score (SRS) were obtained by means of adding the scores for the 17 segments of the stress and rest images, respectively. The sum of the differences between the SSS and SRS was defined as the summed difference score (SDS). These variables incorporate the extent and severity of perfusion defects [Hachamovitch et al. 2011], and were converted to the percentage of the total myocardium involved with stress (% total myocardium), fixed (% fixed myocardium) or ischemic (% ischemic myocardium) defects, respectively, by dividing the summed scores by 64, the maximal potential score (4 × 17), and multiplying by 100. In addition, two categorical variables were defined: abnormal MPS (SSS >1), and ischemic MPS (SDS >1).
After automatic reorientation, post-stress gated short-axis images were processed using quantitative gated SPECT software (Cedars-Sinai Medical Center, Los Angeles, CA, USA), and left ventricular ejection fraction (LVEF) was automatically calculated.
Follow up
Follow up was performed by telephone interview, and all-cause death, MI, CATH or myocardial revascularization (by angioplasty or coronary artery bypass surgery) were registered. Events were confirmed through review of hospital charts or physician’s records.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation and compared by Student’s t-test or Mann–Whitney test, when appropriate. Categorical variables were expressed as number and percentage and compared by Chi-square or Fisher’s exact test. Kaplan–Meier estimates of event-free survival were plotted and compared with the log-rank test. Analyses were performed with SPSS software, version 17.0. A p-value <0.05 was considered significant.
Results
A total of 228 patients (7.7%) had TA (88.2% CCS II and 11.8% CCS III). Comparing patients with TA with those without it (Table 1), clinical and demographic characteristics were not significantly different. However, the use of aspirin, beta-blockers or calcium channel blockers as well as statins was more frequent in the former. Exercise stress was overall more often employed than pharmacologic stress, and exercise-induced angina was more prevalent in patients with TA. MPS was abnormal in 43.0% of the patients with TA and in 34.3% of the other patients (p < 0.001), and myocardial ischemia was found in 25.9% versus 13.6% (p < 0.001). The percentages of total myocardium with perfusion defects and with ischemia were higher in patients with TA, but not the percentage of fixed defect or mean LVEF.
Figure 1 shows annualized event rates according to the presence or absence of TA. MI, CATH and revascularization were more frequent in patients with TA (2.0% versus 0.6%, 10.0% versus 4.7% and 7.8% versus 3.0%, respectively, p < 0.001), while death was not significantly increased (1.5% versus 1.0%, p = 0.2). Conversely, when evaluating event rates according to the presence of, or absence of, ischemia in SPECT (Figure 2), myocardial ischemia was associated with death, in addition to MI, CATH and revascularization.
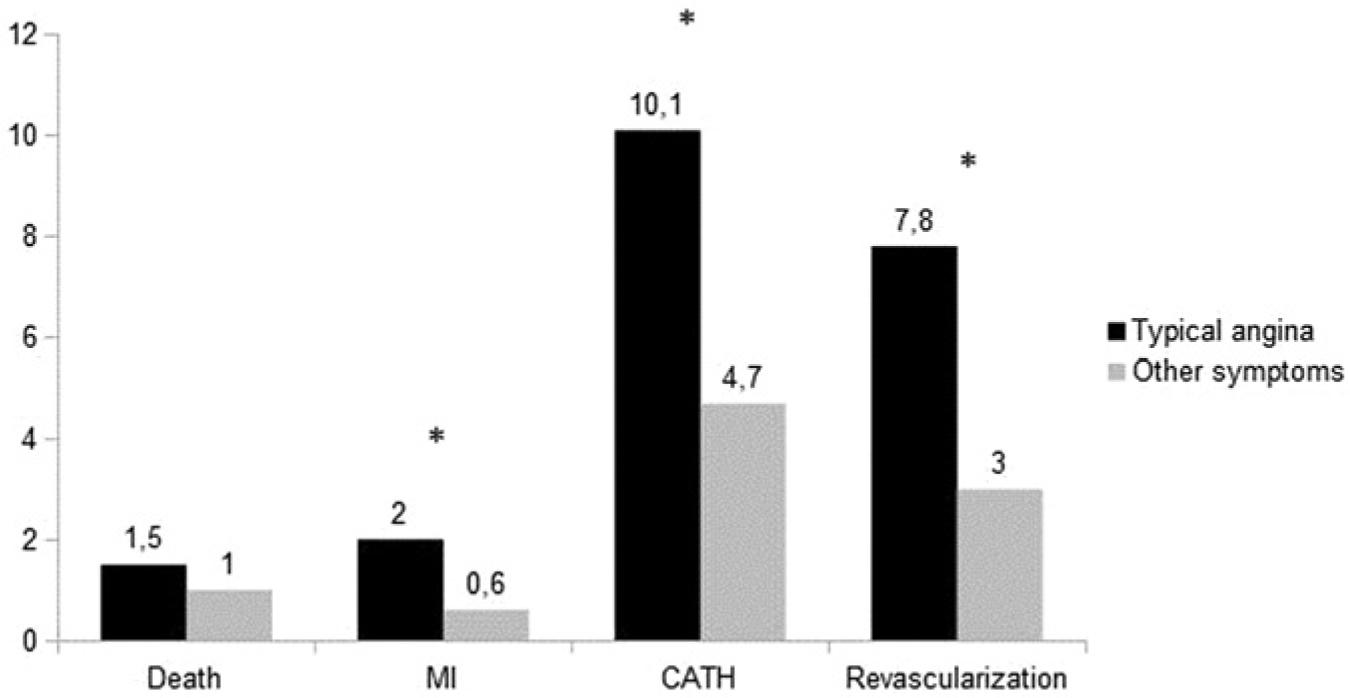
Annualized event rates according to the presence or absence of typical angina.
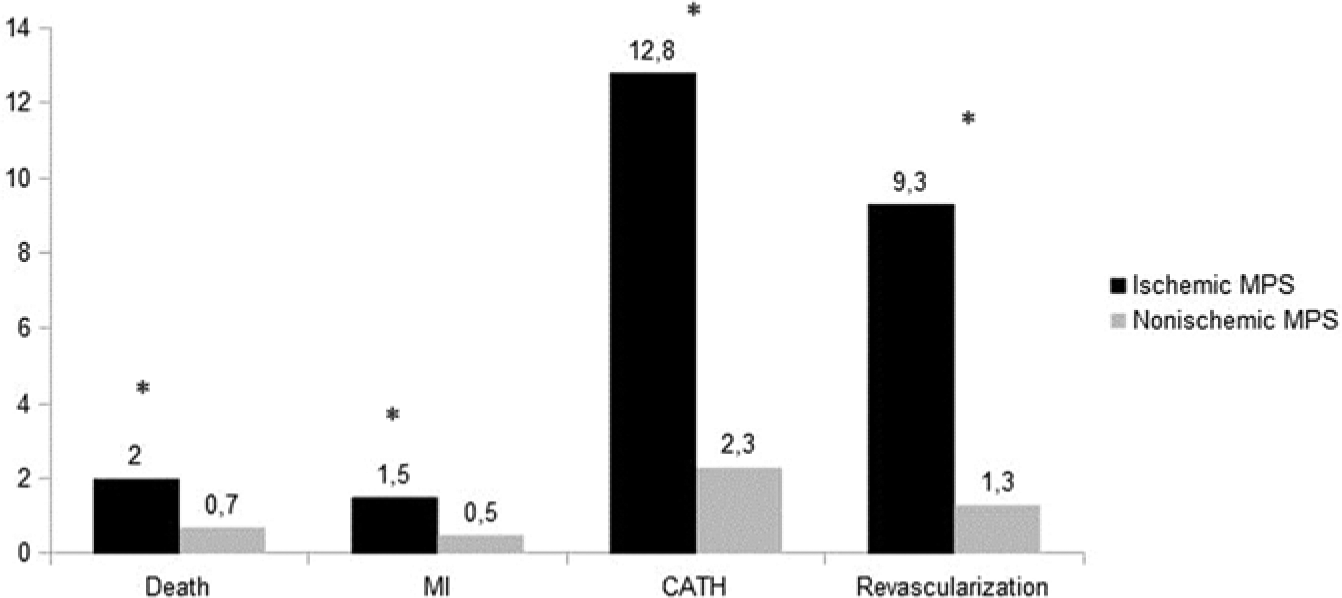
Annualized event rates according to the presence or absence of ischemia in myocardial perfusion single-photon emission computed tomography.
Considering the influence of symptoms (TA versus other symptoms) on patient management after cardiac SPECT, in the absence of demonstrable ischemia, TA was associated with higher MI, CATH and revascularization rates, but not increased death. In patients with ischemic SPECT, TA had borderline association with increased CATH, and was significantly associated with increased revascularization rates (Table 2). These relationships are demonstrated in Figures 3, 4 and 5. Survival curves show that in patients with TA, ischemia in MPS significantly decreases survival (Figure 3), but the converse is not true: in patients with ischemic MPS, TA does not influence survival significantly (Figure 4). Finally, in patients with nonischemic MPS, TA has a modest, nonsignificant effect on survival (Figure 5).
Baseline data of patients with typical angina or other symptoms.

Values are expressed as n (%) or mean ± standard deviation.
LVEF, left ventricular ejection fraction; MPS, myocardial perfusion single-photon emission computed tomography.
p < 0.05.
Annualized event rates according to symptoms and MPS results.

Values are expressed as %.
CATH, cardiac catheterization; MPS, myocardial perfusion single-photon emission computed tomography.
p < 0.05.
p = 0.06.

Survival curves for patients with typical angina with (gray line) or without (black line) ischemic myocardial perfusion single-photon emission computed tomography.

Survival curves for patients with ischemic myocardial perfusion single-photon emission computed tomography with (gray line) or without (black line) typical angina.

Survival curves for patients with nonischemic myocardial perfusion single-photon emission computed tomography with (gray line) or without (black line) typical angina.
Discussion
As CAD is one of the most frequent conditions found in medical practice worldwide [Go et al. 2013; WHO, 2013], it is not surprising that, for decades, numerous studies have been assessing the best way to manage patients with stable CAD, ultimately aiming at improving symptoms and prolonging survival. The first goal has been progressively refined both with medications and invasive procedures such as coronary artery bypass surgery and coronary angioplasty; however, regarding the second, recent evidence has shown no significant difference in death or nonfatal MI in patients receiving revascularization in addition to optimal medical therapy alone [Boden et al. 2007; Hueb et al. 2004]. Notwithstanding, in many cases the type of symptom (or, more specifically, the presence of TA) is the sole reason for recommending invasive diagnostic and therapeutic procedures to patients, regardless of its severity and without correlation with the objective evaluation and quantification of myocardial ischemia. Considering TA as a marker of myocardial ischemia, it is easy to understand that it may cause more concern for doctors than atypical symptoms, even when chest pain is nonlimiting and symptoms are stable. However, it has been shown that optimal medical therapy may reduce myocardial ischemia even in high risk patients [Mahmarian et al. 2006], and the diagnostic yield of elective coronary angiography performed without prior functional evaluation is low [Patel et al. 2010]. This study aimed to assess the influence of TA and of objectively demonstrated myocardial ischemia on the management and clinical evolution of patients referred for MPS.
Our population was predominantly male and, interestingly, no clustering of risk factors for CAD was found in patients with TA. Among them, the use of ‘guideline-recommended’ medications [Qaseem et al. 2012] was higher than that of patients with atypical or no symptoms, but still relatively infrequent, with one third of patients using aspirin and slightly over 50% using beta-blockers or calcium channel blockers. In the current ‘era of optimal medical therapy’ for CAD, this might sound like patient undertreatment, although it may be argued that many physicians would be waiting to initiate cardioprotective medications after SPECT results. While somewhat low, the frequency of medication use was similar to that described in the recent SPARC study for patients undergoing SPECT, in whom baseline use of aspirin, beta-blockers or lipid-lowering drugs was 41%, 30% and 44%, respectively [Hachamovitch et al. 2009].
Interestingly, only a quarter of patients with TA had exercise-induced angina. This finding may be explained by the subjective nature of chest pain symptoms, which may not be reproduced in the stress test. Zehender and colleagues prospectively studied patients with daily angina, scintigraphic evidence of exercise-induced myocardial ischemia, and coronary artery stenosis >75%; angina during the exercise test was absent in 47% of the patients [Zehender et al.1995]. Absence of chest pain during the exercise test may be associated with less extensive coronary artery disease [Visser et al. 1990]. On the other hand, in the study by Mahmarian and colleagues [Mahmarian et al. 1990], the extent of SPECT perfusion defects was nearly identical in patients with and without exertional chest pain undergoing SPECT and concurrent coronary angiography, and no differences in the extent, severity, or distribution of coronary stenosis were found in patients with silent or symptomatic ischemia.
Abnormal MPS was significantly more frequent in patients with TA, and myocardial ischemia was almost twice as frequent when compared with patients with atypical or no symptoms; also, the percentage of ischemic myocardium was higher in the former. In that sense, a link between TA and myocardial ischemia was objectively demonstrated. In line with that, CATH and revascularization were more frequent in patients with TA, in the common cascade of events initiated by the detection of myocardial ischemia, and as described before [Bateman et al. 1995]. Nonetheless, it is worth noting that mean ischemic myocardium was only 4% of the left ventricle, which might be considered a small amount of ischemia and would not a priori indicate any advantage of revascularization over medical therapy [Hachamovitch et al. 2003, 2011].
Also of note is the fact that only a quarter of the patients with TA had ischemic SPECT. A nonischemic MPS in the setting of typical chest pain might be either a consequence of the subjective nature of chest pain symptoms (i.e. if even with ‘TA’ the patient has normal coronary arteries or nonobstructive disease; in this case, the nonischemic MPS would be a ‘true-negative’ scan), or a ‘false-negative’ scan caused by ‘balanced’ ischemia/multivessel coronary artery disease (which is a limitation of MPS). Regarding the first hypothesis, in the study by Mahmarian and colleagues, the majority of patients who developed ischemia during exercise testing were asymptomatic, although they exhibited an angiographic profile and an extent of abnormally perfused myocardium similar to those of patients with symptomatic ischemia [Mahmarian et al. 1990]. Regarding the second hypothesis, in the study by Lima and colleagues, among patients with known 3-vessel coronary artery disease, 18% had a normal SPECT scan by perfusion analysis alone [Lima et al. 2003]. This phenomenon might explain our findings.
It may be surprising that patients with nonischemic MPS and TA had more MI than those with other symptoms. As we discussed before, a nonischemic MPS in the setting of typical chest pain might be a ‘false-negative’ study caused by ‘balanced’ ischemia/multivessel coronary artery disease. In our study, as the event numbers are small, even a few false-negatives would make the MI rate similar to the MI rate of ischemic MPS. On the contrary, in patients with ischemic MPS, MI rates were not significantly different whether the patients had TA or not, indicating that the type of symptom did not have a strong influence on this outcome. It may be argued that small event numbers may make this difference nonsignificant, but there are other examples in the literature underscoring that symptoms are poorer predictors of death or MI than the objective demonstration of ischemia [Zellweger et al. 2004]. For all-cause death, ischemia in MPS makes death rates notably different and higher than in patients without ischemia in MPS. In fact, patients with nonischemic MPS and TA have the same low death rate (0.7%) as patients with nonischemic MPS and other symptoms, which reinforces the fact that TA symptoms are not predictors of all-cause death.
Of note, in the setting of nonischemic SPECT, patients with TA more frequently underwent CATH and revascularization than patients with other symptoms. The higher CATH rate might be an effect of persisting diagnostic uncertainty; however, increased revascularization might be due to true obstructive CAD missed at SPECT due to multivessel CAD [Lima et al. 2003]. However, a nonischemic scintigraphic study in patients with TA might also be the result of effective medical treatment, with suppression of demonstrable ischemia; in that case, revascularization of lesions found at coronary angiography would be a resource for symptom alleviation, if these are judged to be limiting or unbearable by the patient, even though no evidence to date has pointed towards any survival benefit in this setting [Boden et al. 2007; Shaw et al. 2008]. Next, the increased MI rate of patients with nonischemic SPECT might reflect on the underlying pathophysiology of acute CAD; the destabilization of a small, usually non-flow-limiting coronary plaque [Ambrose et al. 1988]. At last, and in line with other studies [Boden et al. 2007; Shaw et al. 2008], death was not increased anyway.
One might argue that coronary interventions might alter patient outcomes and therefore influence the rates of MI and death. For that reason, we excluded patients with early revascularization, and consequently all events would be linked to the baseline disease, with or without progression during follow up. It is worth noting that this study did not aim to evaluate the effect of either medical treatment or interventions driven by the index SPECT study on patient outcomes; patients’ prognoses were interpreted from the perspective of baseline symptom status.
Although underpowered to define prognosis according to SPECT results, the COURAGE Nuclear Substudy suggested that patients fared better with coronary interventions in the setting of larger amounts of ischemia [Shaw et al. 2008]. As a result, it left the message that treatment should evolve from anatomic-driven interventions to ischemia-guided therapies. As a parallel to this, there is no clinical trial evidence that intervening in patients with stable, controlled angina results in improved ‘hard’ (death or MI) outcomes or in a decrease in long-term medical costs [Shaw et al. 1999]. There is also concern about the potential risks associated with invasive procedures.
It is worth emphasizing that scientific papers and the overall medical literature should always be put into context, and the extrapolation of trial data to clinical practice is sometimes impossible. Of course, trials or guidelines are not intended to be substitutes for clinical judgment, but, on the other hand, some of them are the best evidence to be considered. In that sense, the presence of TA in itself, without accounting for the intensity of symptoms or the degree of medication used, for instance, should not be viewed as an indication for invasive evaluation, followed or not by revascularization. The results of stress tests, and in this particular case, of MPS, may help map the best way to proceed. The management of stable CAD based on clinical information and functional evaluation, while taking into account a physician’s judgment and a patient’s preference, is probably the best way to better healthcare delivery.
Limitations
This was an observational study aiming at documenting patterns of referral for CATH and revascularization and the occurrence of ‘hard’ events after MPS, and according to the study design the changes in prescribed medications after the SPECT study, which might affect patient prognosis, were not registered. Our data were obtained at single, outpatient, nonacademic practice settings, which may limit the generalizability of the results, but may otherwise reflect most ‘real-world’ settings.
Conclusions
In this observational study, TA was associated with a greater amount of myocardial ischemia in MPS, but not with death during follow up. Nonetheless, TA was associated with more frequent referrals for CATH and revascularization, even with nonischemic SPECT. This may suggest that despite current evidence regarding therapy for stable CAD, the management of stable patients with known or suspected CAD differs according to symptoms.
Footnotes
Acknowledgements
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
