Abstract
Objectives:
Microalbuminuria (MA) is associated independently with cardiovascular risk and progression of renal disease in patients with diabetes and the normal population. MA is an accepted factor for microvascular defects, in particular in patients with diabetes, and is associated with inflammation.
Activated transmigrating macrophages are key cells in these inflammatory processes. Based on the theory that myeloid-related protein 8/14 complex (MRP8/14) is secreted by transmigrating macrophages, we hypothesized that MA was accompanied by elevated MRP8/14 and investigated whether MA predicts MRP8/14 in patients with type 2 diabetes.
Methods:
A total of 86 men with type 2 diabetes were grouped according to urinary albumin excretion in normoalbuminuria and MA. Serum MRP8/14 levels were measured by enzyme-linked immunosorbent assay. Established cardiovascular risk factors were quantified in both groups and compared with urinary albumin excretion.
Results:
Albuminuria (mg/day) was positively associated with MRP8/14 (r = 0.34) and systemic blood pressure (r = 0.33). Patients with type 2 diabetes and MA demonstrated significantly higher MRP8/14 levels than patients with normoalbuminuria [median (interquartile range), 1.24 (0.97–2.28) µg/ml versus 0.97 (0.67–1.35) µg/ml, p < 0.05]. Serum creatinine levels, systolic blood pressure (SBP), very low density lipoprotein levels and the incidence of hypertension and coronary artery disease were significantly higher in the group with MA. Both groups did not differ significantly in other cardiovascular risk factors. MA was an independent predictor of serum MRP8/14 levels (β = 0.454) as well as SBP (β = 0.625) and haemoglobin A1c (β = 0.322).
Conclusion:
Our data demonstrate that albumin excretion is associated with the process of macrophage activation determined by MRP8/14 levels. These data not only suggest tissue inflammation as a factor for elevated cardiovascular risk in patients with type 2 diabetes, they further point to a role of macrophage activation in this process.
Keywords
Introduction
Diabetes mellitus is a global health problem with as many as 246 million people being affected worldwide [Fioretto et al. 2010]. Diabetes is one of the leading causes of blindness and end-stage renal failure which is due to changes in the microvasculature [Burrows et al. 2010]. But even early in the development of end-organ damages we see changes in the microvasculature, for example by measurement of the urinary excretion of albumin which is a well established marker of glomerular damage and an accepted risk factor for cardiovascular disease and progression of renal disease [Gansevoort et al. 2010]. Recent findings indicate that the development of organ damage in patients with type 2 diabetes is dependent in part on the accumulation of tissue macrophages [Gustafson, 2010]. Especially in chronic kidney disease caused by diabetes but also other kinds of kidney diseases, the accumulation of macrophages is one of the main features and is significantly associated with renal fibrosis [Lim et al. 2010; Tesch, 2010].
Myeloid-related protein 8/14 complex (MRP8/14, in the literature also termed calprotectin, leukocyte protein L1 complex or cystic fibrosis antigen), a heterodimer of two intracellular, calcium-binding proteins (S100A8 and S100A9, also referred to as MRP8 and MRP14), is critically involved in proinflammatory signalling through toll-like receptor 4 and the receptor for advanced glycation end products, cell differentiation, cell cycle progression and cytoskeletal reorganization [Hessian et al. 1993; Guignard et al. 1995; Rammes et al. 1997; Vogl et al. 2007; Pouliot et al. 2008]. MRP8 and MRP14 are mainly expressed in cells of myeloid origin, particularly in monocytes/macrophages and neutrophils [Altwegg et al. 2007] but also platelets [Healy et al. 2006]. Upon activation, the MRP8/MRP14 complex is secreted and leads to an inflammatory response in endothelial cells [Teigelkamp et al. 1991; Lemarchand et al. 1992; Viemann et al. 2005]. Furthermore it has been shown that MRP8/MRP14 complex is essential for proper leukocyte migration capacity [Manitz et al. 2003; Nacken et al. 2003]. MRP8/14 and apolipoprotein E double-deficient animals show an attenuation of atherosclerotic lesions in response to high-fat diet providing direct evidence for an implication of MRP8/14 in atherogenesis [Croce et al. 2009]. As a clinical biomarker levels of MRP8/MRP14 complex could predict microvascular alterations in patients with type 2 diabetes [Burkhardt et al. 2009], cardiovascular death or myocardial infarction 30 days after an acute coronary syndrome (ACS) [Morrow et al. 2008]. MRP8/14 assists in the detection of an ACS [Altwegg et al. 2007] and can predict a first cardiovascular event [Healy et al. 2006].
We hypothesized that circulating MRP8/14 secreted by transmigrating macrophages is augmented in patients with microalbuminuria (MA) and type 2 diabetes. Therefore MRP8/14 could be used as a marker for tissue inflammation in patients with type 2 diabetes and hence as a marker for microvascular disease. Thus we investigated whether MA in patients with type 2 diabetes predicted elevated MRP8/14.
Methods
Patients
A total of 86 patients with type 2 diabetes were included in this study. Due to gender differences in cardiovascular diseases and diabetic nephropathy we decided to create a homogenous group by including only male patients with type 2 diabetes, which is in line with previous cardiovascular investigations [Von Eynatten et al. 2008; Schneider et al. 2009]. Two different cohorts of male patients were studied: 46 individuals with type 2 diabetes and normoalbuminuria; and 40 individuals with type 2 diabetes and MA. MA was defined as levels of urinary albumin between 30 and 300 mg/day. All patients included in this study were recruited from clinical routine procedures at the Department of Medicine, University Hospital Heidelberg, according to the guidelines of the local ethics committee and after giving written informed consent.
Type 2 diabetes was defined according to the criteria of the American Diabetes Association [American Diabetes Association 2011]. A fasting plasma glucose level of at least 7.0mmol/liter and macroalbuminuria were exclusion criteria for the study. Severe kidney or liver diseases were exclusion criteria in both groups.
Coronary artery disease (CAD) was diagnosed angiographically and defined as a stenosis of greater than 50% of the luminal diameter in at least one of the coronary arteries or their major branches. Hypertension was diagnosed according to the European guidelines of hypertension. The body mass index (BMI) was calculated using the formula: weight (kg)/[height (m)]2.
Analysis of lipids/lipoproteins and routine blood parameters were part of the normal clinical setting in these patients.
Total cholesterol, high-density lipoprotein (HDL) cholesterol and triacylglycerol levels were determined enzymatically in a Synchron LX-20 (Beckman Coulter, Krefeld, Germany). Low-density lipoprotein (LDL) cholesterol was separated by ultracentrifugation in a centrifuge (LM-8 ultra; Beckman Coulter) in 100 μl volumes by use of a VT-51.2 rotor (Beckman Coulter). Venous blood was drawn after an overnight fasting period for the measurement of glucose and insulin.
Serum insulin immunoreactivity was determined in frozen serum by enzyme-linked immunosorbent assay (ELISA) (CIS bio international, Gif-Sur-Yvette, France). The degree of insulin resistance was estimated by homeostasis model assessment of insulin resistance index (HOMA-IR) according to the method recommended by Levy and colleagues [Levy et al. 1998].
Detection of serum levels of MRP8/14 complex and interleukin 6
MRP8/14 was detected in serum using a sandwich ELISA (Bühlmann Laboratories, Schönenbuch, Switzerland) with an interassay variation of 7.2% and a detection limit of 3.0 mg/liter. The specific monoclonal antibody for the MRP8/14 heterodimer (mAb 27E10) was used as primary antibody and a polyclonal antibody coupled with horseradish peroxidase was used as secondary antibody [Burkhardt et al. 2009]. The antibody is specific for the MRP8/14 heterodimer or higher order complexes and does not bind MRP8 or MRP9 monomers [Altwegg et al. 2007].
Interleukin-6 (IL-6) serum levels were determined using commercially available ELISA kits (R&D Systems, Minneapolis, MN, USA).
Statistical analyses
Statistical analyses were performed using SPSS software version 11.0 (SPSS, Chicago, IL, USA). Comparison between two cohorts of patients was performed by unpaired t test for values with standard distribution or the Mann–Whitney U test for values with skewed distribution. The results are expressed as mean ± SD or median with interquartile range. A p value of less than 0.05 was considered to be statistically significant.
Multiple linear regression analysis was performed with the degree of albuminuria and different variables, including MRP8/14, systolic blood pressure (SBP), haemoglobin A1c (HbA1c), C-reactive protein (CRP), age, BMI, HDL, HOMA-IR and serum creatinine. R denoted the strength of the correlation. The β coefficient showed the relationship between albuminuria and the variable. The t test was used to show the significance of the correlation.
Results
Biological parameters and cardiometabolic risk factors in patients with normo- versus microalbuminuria
Table 1 shows the distribution of basic clinical characteristics, as well as cardiometabolic risk factors and biological parameters, in the two cohorts. Parameters with a significant difference in the two groups were hypertension (as %) and SBP, treatment with an angiotensin-converting enzyme (ACE) inhibitor, CAD, very low density lipoprotein (VLDL) (but not other lipoproteins or lipids), serum creatinine, albuminuria and MRP8/14. Of those, there was a highly significant difference in albuminuria, serum creatinine and CAD in the two cohorts (p < 0.001). MRP8/14 plasma levels were significantly higher in the microalbuminuric group of patients with diabetes with 1.24 (0.97–2.28) µg/ml versus 0.97 (0.67–1.35) µg/ml in the normoalbuminuric group (p < 0.05). Serum creatinine levels were also higher in patients with MA (1.08 ± 0.4 mg/dl) compared with the normoalbuminuric group (0.85 ± 0.2 mg/dl; p < 0.001). Mean values of CAD were also significantly different in the two cohorts (MA: 85% versus normoalbuminuria: 47%; p < 0.001). Of the patients with MA, 73% against 43% of the patients with normoalbuminuria were treated with ACE inhibitors.
Clinical characteristics of patients with type 2 diabetes overall and separated in normo- and microalbuminuria.

Values are presented as mean ± SD or $median (interquartile range).
p < 0.05, **p < 0.001 versus normoalbuminuria (unpaired t test).
p < 0.01, ****p < 0.001 versus normoalbuminuria (Mann–Whitney U test).
ACE, angiotensin-converting enzyme; BMI, body mass index; CAD, coronary artery disease; HbA1c, haemoglobin A1c; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment of insulin resistance; hsCRP, high-sensitivity C-reactive protein; IL-6, interleukin 6; LDL, low-density lipoprotein; MRP8/14, myeloid-related protein 8/14 complex; VLDL, very low density lipoprotein.
In the observation of all patients in our cohort separated by treatment with an ACE inhibitor or not (49 against 37 patients), there was no difference in the MRP8/14 levels between the two groups (1.05 ± 0.86 with ACE inhibition against 1.10 ± 1.18 μg/ml without ACE inhibition; p > 0.05).
Multiple linear regression analysis with albuminuria and different variables, including MRP8/14, systolic blood pressure, haemoglobin A1c and C-reactive protein
Regression analysis showed the predictive value of albuminuria and different cardiovascular and inflammatory parameters (Table 2). In our cohort MA is significantly and independently associated with MRP8/14, SBP and HbA1c (data shown in Table 2).
Independent associations between pathophysiological cardiovascular risk factors and microalbuminuria.

Bold p values indicate statistical significance (p < 0.05).
BMI, body mass index; HbA1c, haemoglobin A1c; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment of insulin resistance; MRP8/14, myeloid-related protein 8/14 complex; SBP, systemic blood pressure.
Figure 1 shows the relationship between normoalbuminuria (open circles) or MA (black dots) and MRP8/14 [Figure 1(a)], SBP [Figure 1(b)], HbA1c [Figure 1(c)] and CRP [Figure 1(d)]. R values show the corresponding strength of the correlation. In Figure 1(a) MRP8/14 was positively correlated with the degree of albuminuria (r = 0.34, p = 0.002). Patients with an increased albuminuria tended to display higher levels of MRP8/14 in the blood as a marker for an active monocyte/ macrophage transmigration process. Additionally, MA was strongly associated with SBP [r = 0.33, p = 0.002; Figure 1(b)]. HbA1c, as a marker of long-term elevation of blood sugar levels, was only weakly associated with the amount of albuminuria [r = 0.15, p = 0.16; Figure 1(c)]. In contrast to MRP8/14, no correlation between CRP and MA was observed [r = 0.03, p = 0.8; Figure 1(d)).
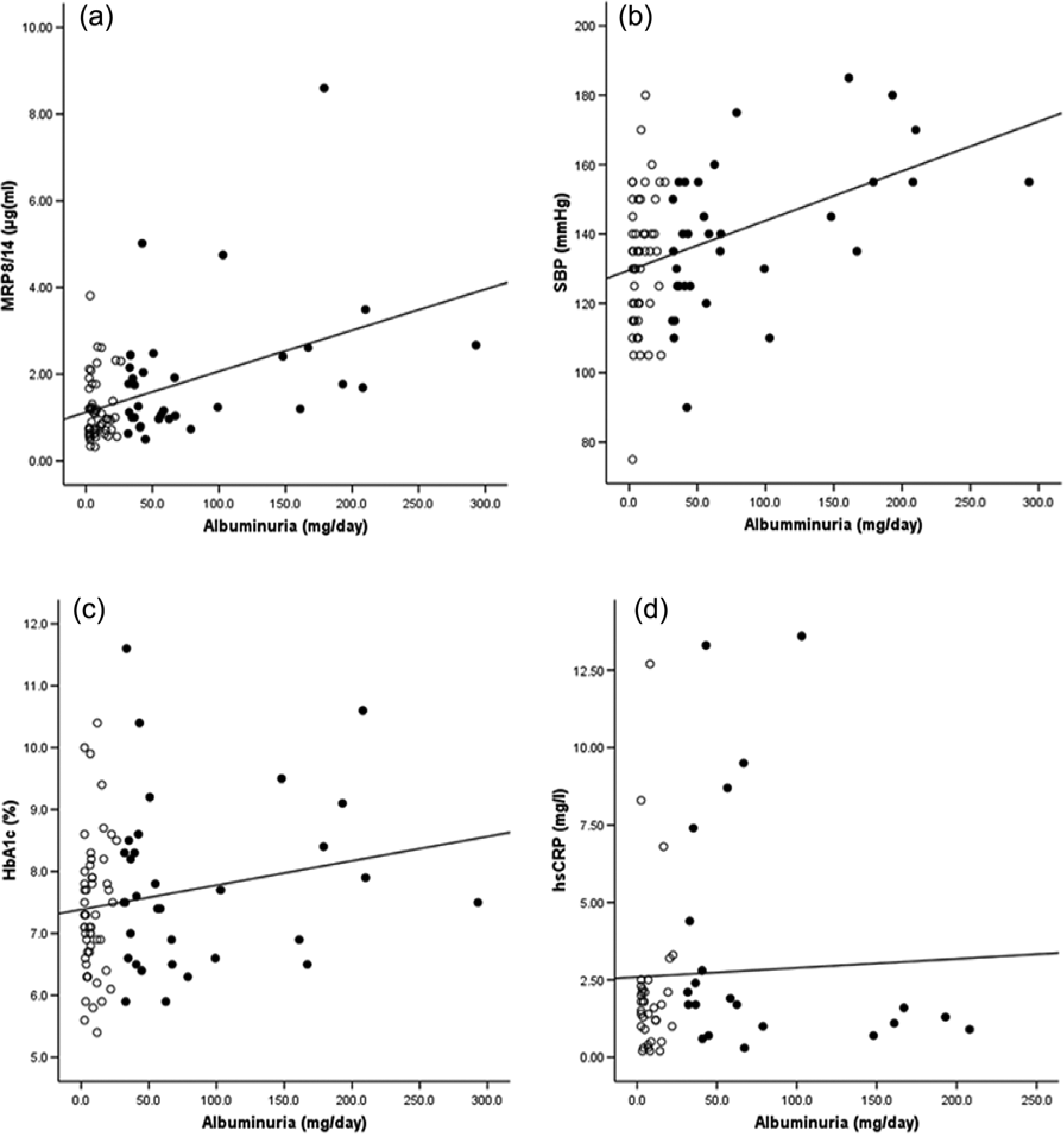
Bivariate correlations of albuminuria in patients with type 2 diabetes and (a) myeloid-related protein 8/14 complex (MRP8/14) (r = 0.34; p = 0.002); (b) systolic blood pressure (SBP) (r = 0.33; p = 0.002); (c) haemoglobin A1c (HbA1c) (r = 0.15; p = 0.16); and (d) high-sensitivity C-reactive protein (hsCRP) (r = 0.03; p = 0.8). Microalbuminuria is characterized by closed circles and normoalbuminuria by open circles. Bold p values indicate statistical significance (p < 0.05).
Discussion
This study demonstrated that albuminuria in patients with type 2 diabetes was positively associated with MRP8/14, a marker for the active process of transmigrating macrophages. Moreover, the microalbuminuric state predicted MRP8/14 levels in this study in patients with type 2 diabetes. We suggest that both MA and MRP8/14 reflect a state of local tissue inflammation. Based on the idea that chronic low-grade inflammation aggravates cardiovascular risk, we further speculate that transmigrating macrophages are linked to elevated cardiovascular risk in patients with type 2 diabetes and MA.
Previous studies demonstrated a predictive value of MA for the incidence of cardiovascular disease. Furthermore MA correlated with cardiovascular mortality and morbidity [Hillege et al. 2002; Arnlov et al. 2005]. MA was highly prevalent in patients with type 2 diabetes. Cohort studies showed that in 39% of patients with diabetes without known kidney disease the levels of albumin excretion were in the microalbuminuric range [Parving et al. 2006]. Moreover, MA was an independent predictive factor for the development of subsequent chronic kidney failure [Ishani et al. 2006].
The precise pathophysiology of MA remains unknown, however we suggested in this study a role of activated macrophages in patients with type 2 diabetes. In this line several authors demonstrated a link between macrophages and diabetic nephropathy. Chow and colleagues demonstrated an association of kidney macrophages with albuminuria, plasma creatinine and tissue damage, suggesting a role for macrophages in diabetic nephropathy [Chow et al. 2004]. A recent work demonstrated that macrophages induce glomerular injury and promote proteinuria and mesangial proliferation [Ikezumi et al. 2003]. Glomerular and interstitial macrophage infiltration was recognized in human samples of renal biopsies and rodent models of diabetic nephropathy [Bohle et al. 1991; Furuta et al. 1993; Sassy-Prigent et al. 2000]. Evidence from renal biopsies in animal models revealed that macrophages were the major immune cells, which infiltrated the kidney in diabetic nephropathy and obviously contributed to the development of diabetic glomerulosclerosis [Tesch, 2010]. Our study demonstrated that serum MRP8/14, a heterodimeric protein complex expressed by activated transmigrating monocytes/macrophages and neutrophils, was significantly associated with albumin excretion in patients with type 2 diabetes. Moreover, MA predicted MRP8/14 level.
Mechanistically, MRP8 and MRP14 form the MRP8/14 complex during macrophage activation. During the transmigration process the MRP8/14 translocates to the cytoskeleton and plasma membrane, where it is secreted [Teigelkamp et al. 1991; Rammes et al. 1997]. This process represents an early event of transendothelial migration [Altwegg et al. 2007]. The pathogenesis of MA in patients with type 2 diabetes involves endothelial dysfunction and chronic or low-grade inflammation [Stehouwer et al. 1992, 1997; Schalkwijk et al. 1999]. Therefore MA and reduced glomerular filtration rate represent the renal manifestation of systemic microvascular disease in which albuminuria reflects the result of a global abnormality of endothelial function in the glomerulum [Abdelhafiz et al. 2011]. Transmigrating macrophages across vessel walls may be an initial factor for this condition. MRP8/14 was associated with systemic inflammation in stable coronary atherosclerosis in men [Baumann et al. 2011]. Experimental studies in animal models as well as human disease found that macrophage accumulation in diabetic nephropathy is associated with the degree of renal injury and declining renal function [Chow et al. 2004; Nguyen et al. 2006]. Our findings of elevated MRP8/14 levels in patients with diabetes and MA and the significant correlation of MRP8/14 and albumin excretion are in line with these findings and suggest that transmigration of macrophages could be a driving force for MA in diabetic nephropathy. In our study we had a predictive value of the range of albuminuria by MRP8/14 levels more than renal function (described by serum creatinine). Concerning the predictive power of MRP8/14 serum levels for the development of MA the early rise of MRP8/14 might represent the initial step of the pathological progression to diabetic nephropathy before measurable damage in kidney function occurs. Transmigration of macrophages across vessel walls induces an early state of inflammation and could be a trigger for the development of MA and a causative factor for microalbuminuria at an early stage of diabetic nephropathy, because at this early time point of diabetic nephropathy there is no association of MA with renal function (as described by serum creatinine).
We found a significant difference in SBP as well as incidence of hypertension between the normo- and microalbuminuric group. Hypertension contributes to MA and is associated with systemic inflammation and declining renal function [Murai et al. 2014]. Hypertension could be a real confounding factor for MA in our investigations and participates in the development of chronic nephropathy. This might indicate that low-grade inflammation induced by transmigrating macrophages could also increase cardiovascular risk in patients with nondiabetic hypertension. This remains to be determined in further studies.
Controversial results for high-sensitivity CRP (hsCRP) were reported [Kshirsagar et al. 2008]. In this study no association between MA and CRP was observed. As none of the previous studies investigated hsCRP and MRP8/14 simultaneously in diabetic nephropathy it remains speculative whether MRP8/14 and hsCRP represent different stages of inflammation in diabetic nephropathy.
The inhibition of the renin–angiotensin–aldosteron system by ACE inhibitors can prevent MA and decelerate the progression of diabetic nephropathy [Ruggenenti et al. 2010]. In our cohort 79% of patients with MA and 43% with normoalbuminuria were treated with ACE inhibitors. There was no significant difference in the serum MRP8/14 levels whether patients were treated with an ACE inhibitor or not. So it seems that MRP8/14 is independently associated with MA and not confounded by inhibition of the renin–angiotensin–aldosteron system.
A limitation of this study is its cross-sectional character and its small patient number. However, this study confirmed the previously reported association of albuminuria with systemic blood pressure, CAD and renal function [Hillege et al. 2002; Vedovato et al. 2004; Arnlov et al. 2005; Parving et al. 2006]. This suggests that this study reflects a typical cohort of patients with type 2 diabetes.
In summary, this study gave indirect hints that transmigration of macrophages measured by elevated serum MRP8/14 levels might be an initial step in the pathophysiology of microvascular disease in diabetic nephropathy. The presence of a systemically elevated level of MRP8/14 as a specific macrophage product confirms the idea of chronic low-grade inflammation in the early stage of diabetic nephropathy. We further speculate that the active process of transmigrating macrophages reflects a pathophysiological step causing increased cardiovascular risk in patients with type 2 diabetes. Diabetic animal models investigated therapeutic strategies targeting immune cells to reduce kidney and aortic macrophage accumulation [Ricote et al. 1998; Lee et al. 2004; Rodriguez-Iturbe et al. 2005]. However, more observational studies are necessary on this topic to strengthen the role of transmigrating macrophages in patients with type 2 diabetes before immunosuppressive regimens can be discussed as therapeutic options.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
