Abstract
Objective:
To assess the efficacy and safety of once daily olmesartan medoxomil (OM)/amlodipine besylate (AM)/hydrochlorothiazide (HCTZ) 40/10/25 mg in patients with hypertension not at goal with mono, dual or triple drug therapy.
Methods:
This was a single-center, prospective, open-label, blinded-endpoint study. After a 1-week screening visit, 40 patients were enrolled into the study and given once daily treatment with OM/AM/HCTZ after the patients underwent baseline ambulatory blood pressure monitoring (ABPM) on their original therapy. The primary endpoint was changes from baseline in mean 24 h ABPM [systolic blood pressure (SBP)] after the first day of therapy with OM/AM/HCTZ 40/10/25 mg. Secondary endpoints were changes from baseline in mean 24 h ABPM [diastolic blood pressure (DBP)] after the first day of therapy with OM/AM/HCTZ 40/10/25 mg; mean changes from baseline in trough seated SBP (SeSBP) at day 1 and SeSBP at weeks 1, 2, 3 and 4; mean changes from baseline in trough seated DBP (SeDBP) at day 1 and SeDBP at weeks 1, 2, 3 and 4; and the percentage of subjects achieving mean 24 h, daytime and night-time ABPM BP goals.
Results:
The baseline paired t-test systolic ABPM was 134.0 ± 2.77 mmHg and day 1 was 128.6 ± 2.47 mmHg with a treatment difference of −5.55 ± 1.3 mmHg (p<0.0001). At week 1, paired t-test ABPM SBP reduction was 117.7 ± 2.0 mmHg with a treatment difference of −16.5 ± 1.8 mmHg (p < 0.0001). At week 2, paired t-test ABPM SBP reduction was 115.8 ± 1.8 mmHg with a treatment difference of −18.4 ± 2.0 mmHg (p < 0.0001). At week 3, paired t-test ABPM SBP reduction was 115.5 ± 1.9 mmHg with a treatment difference of −18.6 ± 2.0 mmHg (p < 0.0001). At week 4, paired t-test ABPM SBP reduction was 115.5 ± 1.8 mmHg with a treatment difference of −18.6 ± 2.2 mmHg (p < 0.0001). The baseline paired t-test SeSBP was 142 ± 2.43 mmHg and day 1 was 132 ± 2.59 mmHg with a treatment difference of −9.78 ± 1.51 mmHg (p < 0.0001). At week 1, paired t-test SeSBP reduction was 124.0 ± 1.6 mmHg with a treatment difference of −17.9 ± 1.8 mmHg (p < 0.0001). At week 2, paired t-test SeSBP reduction was 120.3 ± 1.7 mmHg with a treatment difference of −21.5 ± 2.1 mmHg (p < 0.0001). At week 3, paired t-test SeSBP reduction was 118.5 ± 1.8 mmHg with a treatment difference of −23.3 ± 1.7 mmHg (p < 0.0001). At week 4, paired t-test SeSBP reduction was 119.6 ± 1.7 mmHg with a treatment difference of −22.2 ± 1.9 mmHg (p < 0.0001).
Conclusion:
Treatment with OM/AM/HCTZ achieved superior (SBP) ABPM reductions compared with mono, dual or triple drug therapy, resulting in all patients achieving systolic ABPM goal without ABPM documented hypotension.
Keywords
Introduction
In recent years, improvement in the awareness and treatment of hypertension has resulted in somewhat higher rates of blood pressure (BP) control in the United States. Data from the 2009–2010 National Health and Nutrition Examination Survey (NHANES) estimated that BP was controlled in 64% of patients with hypertension who are treated, but only 47% of all patients with hypertension [Egan et al. 2010]. This is an increase of approximately 10% from the estimated 37% control rate of all patients with hypertension based on NHANES 2003–2004 data [Guo et al. 2012]. This improvement in the rate of BP control was aided by the availability of a wide range of therapeutic options. However, challenges remain due to increasing rates of obesity and other comorbidities that affect the treatment of hypertension [Guo et al. 2012].
An important component of ensuring successful treatment is the use of more aggressive treatment strategies. BP goals are reached in only one-third of patients receiving monotherapy [Oparil et al. 2005]. As a result, in the majority of patients with hypertension, particularly those with stage 2 hypertension, combination therapy is required to achieve recommended BP goals. Treatment guidelines emphasize the importance of starting antihypertensive combination therapy in patients with a BP level that exceeds the goal by more than 20/10 mmHg.
The calcium channel blockers (CCBs) are a chemically diverse drug class that has the common property of blocking the flow of calcium ions through voltage-gated L-type channels in vascular smooth muscle. This produces a vasodilating effect that results in a decrease of total peripheral resistance. There are several different types of CCBs which differ in their binding sites on the calcium channel.
Angiotensin II receptor blockers (ARBs), hydrochlorothiazide (HCTZ) and CCBs differ in their mechanisms of action but studies have found that they may have complementary actions, which could explain why the triple combination is effective and has a lower incidence of adverse effects (e.g. edema).
ARBs are a well tolerated drug class. Olmesartan medoxomil (OM) is a widely prescribed ARB and has been shown in some head-to-head studies to have greater BP-reducing efficacy than older ARBs such as losartan potassium [Oparil et al. 2001; Weir et al. 2011; Punzi et al 2012a], valsartan and irbesartan [Oparil et al. 2001; Giles et al. 2007].
The intent of this study was to demonstrate that the use of OM/AM/HCTZ 40/10/25 mg in patients not at goal BP would be both efficacious and safe in the management of hypertension in an outpatient clinic setting.
Materials and methods
Study design
This single-site study, conducted from 16 July 2012 to 25 March 2013 in Carrollton, Texas was a phase IV, prospective, randomized, open-label, blinded-endpoint study (PROBE Design) with a 2–9-day screening period, followed by 4–6 weeks of open-label treatment with a OM/AM/HCTZ 40/10/25 mg fixed dose combination.
Patients entering the study could be on one, two or three antihypertensive medications and not at goal BP, defined as less than 140/90 mmHg or less than 130/80 mmHg if they had diabetes or renal disease. Eligible patients underwent the initial 24 h ambulatory BP monitoring (ABPM) and Micro Mini-motion Logger (Ambulatory Monitoring, Inc., Ardsley, NY, USA) placement on their baseline antihypertensive therapy. If passed, patients had their baseline water displacement measured via the EDEMAT (Punzi Medical Center, Carrollton Texas, USA) immersion technique and were randomized to once daily OM/AM/HCTZ (40/10/25 mg) for 4 weeks. The EDEMAT (Punzi Medical Center, Carrollton Texas, USA) technique utilized in this study was designed to minimize operator errors and enhance reproducibility. The procedure requires a polyurethane tank that sits on top of a scale. The foot cradle is inserted into the polyurethane tank and then the tank is filled with water. The displacement of the water by the foot sitting on the cradle increases the weight on the scale and the measurement of the edema is in grams.
The initial in-office dose of OM/AM/HCTZ was immediately followed by a second 24 h ABPM measurement and Mini-motion Logger placement. The following day, patients had a repeat EDEMAT (Punzi Medical Center, Carrollton Texas, USA) procedure. The 24 h ABPM and Mini-motion Logger were then done an additional four times at weeks 1, 2, 3 and 4, as were the EDEMAT (Punzi Medical Center, Carrollton Texas, USA) measurements the day following the ambulatory measurements.
The investigators and patients were aware of the identity of study treatments, but blinded to the 24 h ABPM and EDEMAT (Punzi Medical Center, Carrollton Texas, USA) procedures.
Study medication was taken once daily at 7 a.m. ± 120 min, except when a clinic visit was scheduled. During clinic visits, study medication was taken after BP measurements and during ABPM placement. Patients with a mean seated systolic BP (SeSBP) greater than 180 mmHg or mean seated diastolic BP (SeDBP) greater than 115 mmHg at any visit, or with symptomatic hypotension, as determined by the investigator, exited the study.
The study was conducted in compliance with the Declarations of Helsinki and in accordance with the International Conference on Harmonization E6 Guidelines for Good Clinical Practices (GCP), Food and Drug Administration GCP guidelines, and the US Code of Federal Regulation Title 21 (parts 50, 54, 56 and 312).
Patients
Men and women were included if they were aged 18 years or over with a mean SeDBP of 90 mmHg or over, up to 115 mmHg, and a mean SeSBP of 140 mmHg or over, up to 180 mmHg on one, two or three antihypertensive medications at the screening visit. If the patients had diabetes or renal disease then the levels were a mean SeDBP of 80 mmHg or over, up to 115 mmHg and a mean SeSBP of 130 mmHg or over, up to 180 mmHg. Patients were excluded if they were pregnant or breastfeeding. Patients able to become pregnant and practicing protocol-approved birth control or who had been postmenopausal for over a year, had undergone a hysterectomy or had post-tubal ligation at least 6 months previously were eligible. In addition, patients were also excluded if they had type 1 or type 2 diabetes with hemoglobin A1c greater than 10.5%; had circumference of the nondominant arm (arm extended, upper arm midpoint circumference) of at least 45 cm; had serious cardiovascular, renal, pulmonary, hepatic, gastrointestinal, endocrine, metabolic, hematologic or psychiatric disease; had a history of myocardial infarction, unstable angina, coronary angioplasty, bypass surgery, heart failure, cerebrovascular accident or transient ischemic attack within the previous 6 months or had clinically significant cardiac conduction defects, hemodynamically significant valvular disease or secondary hypertension.
Efficacy and tolerability assessments
The efficacy cohort included all patients who received at least one dose of study medication, had a baseline assessment and at least one postbaseline efficacy assessment. The safety cohort included all patients who received at least one dose of study medication.
BP values were assessed at screening, Week 0 (day −1, day 0 and day 1) and weeks 1 (days 8–9), 2 (days 15–16), 3 (days 22–23) and 4 (days 29–30) of the active treatment phase. BP was measured prior to treatment administration using a calibrated mercury sphygmomanometer (Baumanometer WA Baum Co Inc., Copiague, NY, USA) on the nondominant arm, after the patient had been sitting for 5 min. Three separate seated BP (SeBP) measurements were taken at 2 min intervals with the mean of these three readings constituting the SeSBP and SeDBP for that visit. Pulse rate was obtained during the 2 min interval between the second and third SeBP readings by palpation.
Eligible patients had two consecutive 24 h ABPM measurements with the Spacelabs 90207 monitor (Issaquah, Washington DC, USA), the first placed on day −1 and the second on day 0. ABPM was repeated at weeks 1, 2, 3 and 4.
The Micro Mini-motion Logger is a wristwatch device that monitors motion. The faux-watch configuration encloses the Micro Mini in a watch type housing weighing 0.8 oz. The device was placed on the patient’s dominant wrist during the 24 h ABPM procedure to obtain the sleep–awake circadian rhythm. At the completion of the 24 h ABPM, the Mini-motion Logger was removed and placed on the interface unit to download the data. The human rest–activity pattern of the study patient, determined by the Mini-motion Logger, was used to estimate sleep–awake patterns to obtain accurate daytime–night-time ABPM data.
Study baseline for trough SeBP was defined as the mean SeBPs measured at study visit 1 (day −1). Study baseline for trough 24 h ABPM was defined as the 24 h ABPM average measured at visit 2 (day 0). This value was used to calculate the changes from baseline at day 1 and weeks 1, 2, 3 and 4 for the study patients. Study drug compliance was assessed by tablet count at each clinic visit. Safety variables were evaluated by monitoring adverse events, changes in vital signs, routine laboratory tests and physical findings. In addition, complete medical histories were performed at screening visit.
Endpoints
The primary efficacy variable was the mean change from baseline in 24 h SBP ABPM at day 1. Secondary efficacy variables included the mean change from baseline in 24 h DBP ABPM at day 1, the change from baseline in mean trough SeSBP at weeks 1, 2, 3 and 4, and the change from baseline in mean trough SeDBP at weeks 1, 2, 3 and 4. Other efficacy variables included the percentage of patients achieving a SeBP goal of less than 140/90 mmHg and the percentage of patients achieving a mean 24 h ambulatory BP target of less than 130/80 mmHg at weeks 1, 2, 3 and 4.
Statistical methods
A priori sample size calculation for a Student t-test was determined as follows: for a desired statistical power level of 80% and probability level of 0.05%, a minimal sample size of 40 patients was needed. Secondary variables were analyzed using the same method as the primary variable analysis. Tertiary variables in SeBP change were analyzed in the same fashion as the primary variable without adjustment for multiple comparisons. The number and percentage of patients who reached defined SeBP goals were summarized for the entire active treatment period (overall cumulative goals).
Results
Patient disposition and baseline characteristics
Of 43 patients screened, 41 (95%) entered the screening phase and 40 were randomized to OM/AM/HCTZ 40/10/25 mg. No randomized patients discontinued the study. The demographic and baseline characteristics are shown in Table 1. Adherence to study medication for the entire randomized treatment phase was 97.9%.
Demographics and baseline characteristics of the randomized population.

ABPM, ambulatory blood pressure monitoring; BMI, body mass index; DBP, diastolic blood pressure; SBP, systolic blood pressure; SD, standard deviation; SeDBP, diastolic blood pressure; SeSBP, seated systolic blood pressure.
Efficacy
ABPM changes from baseline
At day 1, for the primary efficacy endpoint, treatment with OM/AM/HCTZ resulted in a significant mean reduction from baseline in ambulatory SBP reduction of 5.55 ± 1.3 mmHg (p < 0.0001) (Figure 1).
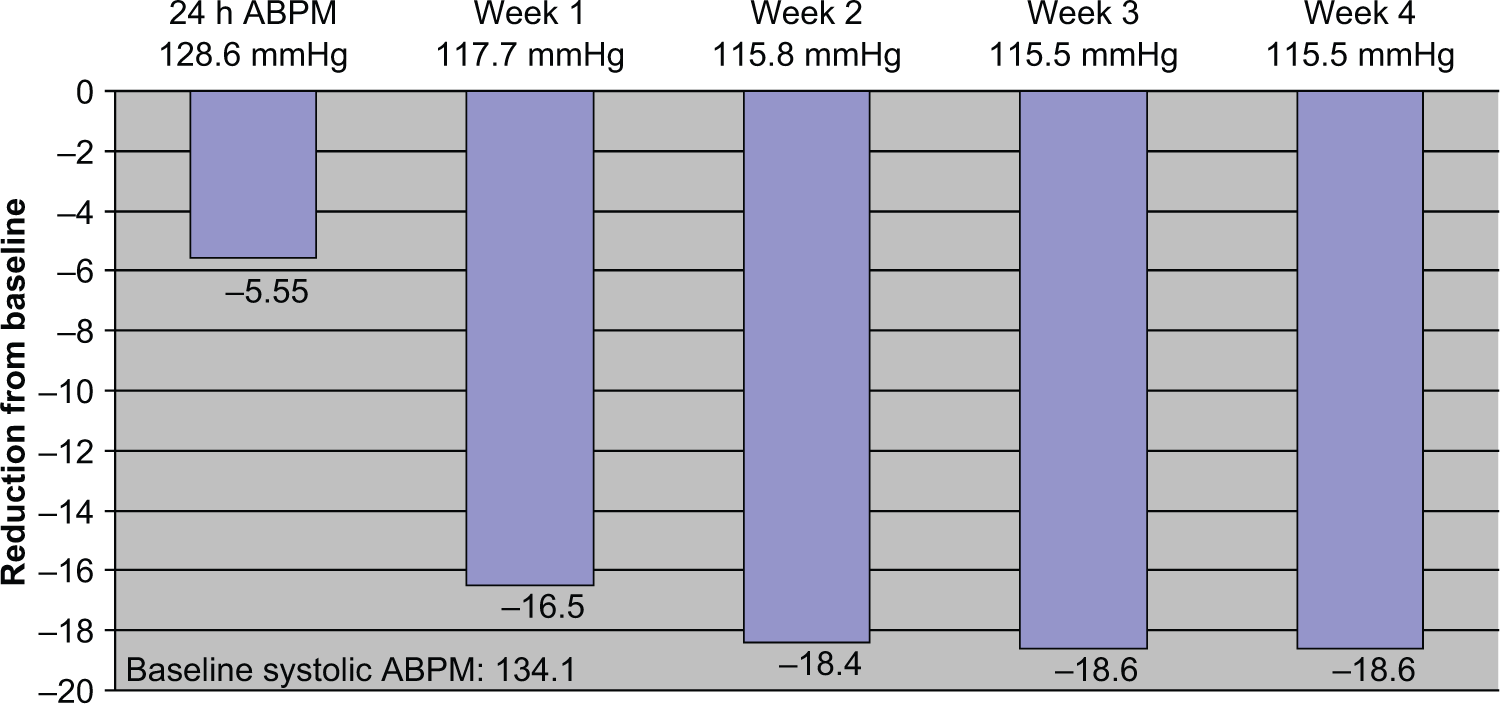
Mean systolic ambulatory blood pressure monitoring (ABPM) reductions.
Attainment of SeBP goals (secondary)
A significant proportion of patients (90%) receiving OM/AM/HCTZ achieved the SeBP goal of less than 140/90 mmHg at week 4, with 97% achieving less than 140 mmHg.
Attainment of ambulatory and cuff BP targets (secondary)
The proportion of patients achieving the 24 h ambulatory BP target of less than 130/80 mmHg was 84% at week 4. At day 1, for the secondary endpoints, treatment with OM/AM/HCTZ resulted in a significant mean reduction from baseline in ambulatory DBP of 2.55 ± 1.0 (p < 0.0052) (Table 3, Figure 2), seated cuff SBP reduction of 9.78 ± 1.5 (p < 0.0001) (Table 3, Figure 3) and seated cuff DBP reduction of 4.13 ± 1.4 (p < 0.0052) (Table 3, Figure 4).

Mean diastolic ambulatory blood pressure monitoring (ABPM) reductions.

Mean systolic seated cuff reductions.

Mean diastolic seated cuff reductions.
Safety and tolerability
Treatment-emergent adverse events (TEAEs) occurred in 28 patients (Table 2). No discontinuations due to TEAEs occurred. No patients reported experiencing a serious TEAE. No patient experienced an adverse event leading to death. Drug-related TEAEs occurred in five patients. The most common TEAEs were body aches, fatigue and dizziness (Table 2).
Number and percentage of patients experiencing treatment-emergent adverse events during 4 weeks of treatment with olmesartan/amlodipine/hydrochlorothiazide (OM/AM/HCTZ).

TEAE, treatment-emergent adverse event.
Secondary endpoint mean reductions from baseline for ABPM and seated cuff.

ABPM, ambulatory blood pressure monitoring; DBP, diastolic blood pressure; SBP, systolic blood pressure; SE, standard error; SeDBP, seated diastolic blood pressure; SeSBP, seated systolic blood pressure.
Discussion
The current study determined that all patients, approximately 42% of whom had stage 2 hypertension (despite antihypertensive therapy), receiving OM/AM/HCTZ had statistically significant greater ambulatory SBP reductions from baseline. To our knowledge, this is the first study that evaluated both cuff and ambulatory BP consecutively on current antihypertensive therapy and subsequent OM/AM/HCTZ. The reductions from baseline in mean 24 h ambulatory SBP were significant at day 1. Moreover, a significant proportion of patients achieved the SeBP goal of less than 140/90 mmHg, as well as achieving the 24 h ambulatory BP target of less than 130/80 mmHg.
Although most patients with hypertension will require two or more antihypertensive agents to achieve BP control [Kereiakes et al. 2007], the current findings of this study are important because they demonstrate that for our patients not at goal, switching from prior therapy to a fixed dose triple drug therapy, 60% attained the BP goal on the initial dose, 77 % at week 1, 89% at week 2, 90% at week 3 and 85% at week 4. In addition, maximum BP lowering is important because studies have shown that even small BP reductions can significantly affect outcomes [Lewington et al. 2002].
Lewington and colleagues conducted a meta-analysis of 61 prospective studies that assessed cardiovascular outcomes in over 1 million participants and showed that BP reductions of as little as 2 mmHg can reduce the risk of ischemic heart disease mortality by 7.0% and the risk of stroke by 10.0% [Lewington et al. 2002]. Thus, it is important to uptitrate antihypertensive therapy with the most efficacious treatment possible.
Combination therapy should comprise different classes of agents with complementary mechanisms of action, which may provide an antihypertensive effect greater than either component alone, and with a favorable tolerability profile. Blockade of the renin–angiotensin system (RAS) pathway by ARBs provides an antihypertensive effect [Le et al. 2007] and can be enhanced by the addition of HCTZ. HCTZ acts in the kidney by blocking the reabsorption of sodium and chloride in the distal portion of the kidney tubule. Additionally, HCTZ is believed to have direct (vasodilation) or indirect effects on the blood vessel itself, although the exact mechanism explaining this is not known. Use of HCTZ alone causes volume contraction, which has been shown to cause an increase in RAS pathway activity to compensate. The synergistic addition of a RAS inhibitor to HCTZ blunts this physiological response to diuresis, thereby achieving volume contraction with decreased RAS activity [Punzi 2009, 2011; Punzi et al. 2010].
An advantage of the present study is that it used seated cuff, ABPM and wrist actigraphy methods to determine BP-lowering efficacy. A particularly useful benefit of ABPM is that it can be utilized to evaluate how well an agent maintains BP control throughout the 24-hour dosing interval, which is a key determinant of an antihypertensive agent’s efficacy [Punzi, 1998]. The actigraph allows unbiased determination of the sleep–awake cycle that allows accurate measurement of daytime versus night-time ABPM. In the current study, patients switched to OM/AM/HCTZ experienced significantly greater reductions from baseline in mean 24 h ambulatory SBP/DBP compared with day 1, week 1 and week 2 (5.55 ± 1.3, 16.5 ± 1.8, 18.4 ± 2.0; p < 0.0001) (Figures 1 and 2). Although there are currently no guideline-recommended ambulatory BP goals, the American Heart Association suggests normal 24 h, daytime and night-time ambulatory BP targets for adults of less than 130/80 mmHg, less than 135/85 mmHg and less than 120/70 mmHg respectively [Pickering et al. 2005].
ARBs have excellent tolerability with an adverse event profile similar to that of placebo [Israili, 2000]. The use of the CCB amlodipine has shown similar morbidity and mortality reduction compared with chlorthalidone in the ALLHAT study, but with an 18% lower incidence of new-onset diabetes [Punzi and Punzi, 2004]. In this study, treatment with OM/AM.HCTZ 40/10/25 mg was well tolerated with no patients discontinuing the study. Body aches, fatigue and dizziness, were the most common drug-related TEAEs
Other benefits of a combination of an ARB or angiotensin-converting enzyme (ACE) with a CCB is the reduction of bipedal edema [Punzi et al. 1997], the product of a bias of CCBs toward arteriolar vasodilation, which causes a pressure gradient between the arteriolar and venular capillaries. This results in exudation of fluid into the interstitial tissue, an effect that is exaggerated by gravity upon standing. The use of an ARB with a CCB stimulates both arteriolar and venodilatation, reducing the pressure gradient and thereby enhancing the absorption of interstitial fluid and reversing the edema. The use of amlodipine (10 mg) monotherapy is associated with a high degree (33%) of peripheral edema. Only three patients complained of swelling.
In the COACH study, there was a significant reduction in the incidence of edema with increasingly higher doses of olmesartan (10, 20, then 40 mg) added to amlodipine (10 mg) (p = 0.009) [Chrysant et al. 2008]. Similar to our study findings that OM/AM/HCTZ 40/10/25 mg at the higher doses was efficacious with the added benefit of a reduced incidence of edema of 5%, Punzi and Punzi used the EDEMAT (Punzi Medical Center, Carrollton Texas, USA) technique in patients with hypertension receiving CCB/ACEI combination therapy. After 4 weeks of therapy, patients were force-titrated to high-dose CCB/ACE inhibitor therapy, either amlodipine/benazepril (10/40 mg) or verapamil/trandolapril (480/8 mg).
There was less edema at the target dose compared with baseline (1603 g baseline versus 1655 g target dose) with the higher dose CCB/ACE inhibitor combination therapy, as opposed to the lower dose CCB/ACE inhibitor therapy (1517 g baseline versus 1663 g target dose). In our study the baseline EDEMAT (Punzi Medical Center, Carrollton Texas, USA) measure was 1488.2 g and after 4 weeks of therapy, 1516.8 g, with a nonsignificant difference of 28.6 g (Figure 5).

Mean difference in Edema water displacement.
The findings of the current study support the results of the TRINITY trial, in which the efficacy and tolerability of a triple-combination treatment with OM 40/AML 10/HCTZ 25 mg was compared with the component dual-combination treatments [Oparil et al. 2010]. At 6 weeks (i.e. 2 weeks after patients were assigned to either triple- or dual-combination treatment), 64.3% of patients in the triple-combination treatment group had reached the BP goal compared with 39.7–47.8% of patients in the dual-combination treatment groups (p < 0.001 for all treatment comparisons). Our current study bridged the 2-week gap in the TRINITY study. It demonstrated that 60% of our patients attained the BP goal 24 h after the initial dose, 78% after 1 week of therapy and 89% after 2 weeks of therapy. Our higher control rate may be related to our use of Mercury Sphygmomanometry and 24 ambulatory measures compared with the automated Omrom device (Omron Corporation, Bannockburn, IL, USA) used in the latter study [Oparil et al. 2010].
The design, efficacy and safety results of our study are similar to the much larger Blood Pressure Control in All Subgroups with Hypertension (BP CRUSH) study that assessed the efficacy and tolerability of combination therapy with OM and AML, with or without HCTZ, in patients with hypertension uncontrolled on monotherapy [Punzi et al. 2012b]. At the end of the study, uptitration to OM 40/AML 10/HCTZ 25 mg allowed 90.3% of patients to achieve the cumulative BP goal of less than 140/90 mmHg. These results are similar to our study, both for cuff and ABMP, but we were able to determine that the BP reduction started on the first dose and continued to decline for 4 weeks.
A limitation of this study was that we enrolled only 40 patients; however, the protocol was designed to demonstrate the BP-lowering efficacy in a real world setting. A combination such as OM/AM/HCTZ that can provide 24 h BP coverage as a single dose may potentially increase adherence compared with agents requiring multiple doses per day [Iskedjian et al. 2002]. This can be beneficial in helping patients to achieve BP control.
Another limitation was that ambulatory BP was not an entry criterion, but the similarity in baseline office SeBP and ABPM populations suggests the results of the study are unbiased.
Conclusion
At maximum once-daily doses, the fixed dose combination of OM, AM and HCTZ (40/10/25 mg) resulted in reductions from baseline in mean 24 h ambulatory SBP and DBP compared with their initial therapies. The antihypertensive effect commenced with the first dose with no hypotensive episodes in any of our 40 patients. This underscores the efficacy and safety of the treatment regimen. BP control continued to improve through the first and fourth weeks of therapy. The seated cuff BP was also significantly improved with excellent tolerability. Treatment with the triple fixed dose of OM, AM and HCTZ also enabled 89% of patients to achieve the recommended ambulatory BP goal of less than 130/80 mmHg.
Footnotes
Acknowledgements
Kathy Cassidy, RN, Kristin Schumacher, RN, and Laura Rivera, MA are acknowledged for their dedication and professionalism in assisting to complete the clinical trial.
Funding
This work was supported by Daiichi-Sankyo, Inc.
Conflict of interest statement
Daiichi-Sankyo, Forest Pharmaceuticals, Takeda Pharmaceuticals, National Institute of Health (NIH). Speakers Bureau: Daiichi-Sankyo, Forest Pharmaceuticals.
