Abstract
The renin–angiotensin system (RAS) affects vascular tone, cardiac output and kidney function. By these means the RAS plays a key role in the pathogenesis of arterial hypertension. As a result, RAS inhibition is highly effective not only in lowering blood pressure but also in reducing kidney disease progression (particularly when associated with proteinuria) and cardiovascular events.
Among RAS blocking agents, direct renin inhibitors have shown not only excellent efficacy in hypertension control but also pharmacologic tolerance that is comparable with other renin–angiotensin suppressors. Indeed, aliskiren, the only direct renin inhibitor available is effective in controlling blood pressure as monotherapy or in combination with other antihypertensive drugs, irrespective of patient’s age, ethnicity or sex. It is also effective in patients with metabolic syndrome, obesity and diabetes. Long-term studies comparing ‘hard endpoints’ of aliskiren therapy versus treatment with other RAS inhibitors, including cardiac and kidney protection, are currently ongoing. Combined with other antihypertensive agents, aliskiren not only improves their hypotensive response but may also lessen the adverse effects of other drugs. In high-risk patients, however, precautions should be taken when combining two or more renin–angiotensin inhibiting agents, as tissue perfusion may be highly renin-dependent in these patients and serious adverse side effects could take place.
Background
Lowering high blood pressure has long been a major goal in clinical practice, even before therapy-driven improvement in survival rates and outcomes were demonstrated. For years, however, therapy was intuitive and efficacy was low. The treatment goals were poorly defined, and while the better-tolerated agents had limited efficacy, the more effective drugs were poorly tolerated. These difficulties were largely resolved by a rational approach based on reducing one or both of the components of the blood pressure equation:
The method entailed the use of diuretics for volume reduction, or vasodilators to control high peripheral vascular resistance. In this manner, while high blood pressure could be lowered in some patients, a significant proportion could not achieve acceptable levels. In these cases, physicians could choose either a dose uptitration of the drug already in use (with a potential rise in adverse effects), or adding a second drug with alternate hypotensive actions that did not enhance adverse effects [Gifford, 1974]
Plasma renin activity levels and drug choice
Plasma renin activity (PRA) measures angiotensin generation in a predefined time and in the presence of an angiotensinogen excess. Under these conditions, the synthesis of angiotensin depends on the amount of renin available in the sample. Hence, as a rule, PRA is a good reflection of angiotensin II (Ang II) concentration. However, not being a direct measure of circulating Ang II, care should be taken when interpreting PRA levels. Anyhow, PRA has been used in clinical practice to estimate the volume of the extracellular compartment, as it correlates inversely with PRA.
Indeed, 40 years ago, Laragh and colleagues grouped hypertensive patients by their volume status using PRA levels [Brunner et al. 1972; Laragh, 1973]. The rationale behind this proposal was that patients with low PRA have volume excess and thus should respond better to diuretics. In contrast, high renin activity in hypertensive patients denoted vasoconstriction and therefore should respond better to RAS inhibitors [Man in′t Veld and den Meiracker, 1990]. This idea had limited acceptance among internists and cardiologists, mainly because no good correlation could be found in clinical practice between PRA and the hypotensive response to different kinds of drugs.
This lack of correspondence between PRA and drug response was confirmed after the introduction of angiotensin-converting enzyme inhibitors (ACEIs). Indeed, these drugs prevent the synthesis of Ang II and yet their therapeutic efficacy is PRA-independent. Thus, the antihypertensive actions of ACEIs, and angiotensin II–AT1 receptor blockers (ARBs) could be accounted for by Ang II dual action: vasoconstriction and renal sodium retention (due to direct tubular effects and to increased aldosterone synthesis). In other words, RAS inhibitors may affect not only the vascular tone but also fluid volume.
A second explanation for the lack of correlation between PRA and drug response is that circulating renin levels may not reflect tissue renin levels. Indeed, many organs (heart, blood vessels, adrenal glands, central nervous system, kidney and others) may not only synthesize RAS components, but also may store and build up such components to achieve levels that are much higher than plasma levels [Kifor and Dzau, 1987; Pendergrass et al. 2006; Navar and Nishiyama, 2001, 2004].
Unfortunately, despite numerous studies, it remains challenging to discriminate between the putative roles of circulating versus local RAS effects. The evidence available indicates that both effects are important functions by themselves or combined. For instance, while Ang I and Ang II are produced in endothelia and in vascular smooth muscle, renin synthesis in these tissues has never been demonstrated, suggesting that renin must be taken up from the circulation [Linz et al. 1995; Campbell, 1985; Hilgers et al. 2001; van del Wal et al. 2007]. Likewise, even though most of the RAS components are also synthesized in the heart, there is no evidence that renin can be synthesized by this organ [Danser et al. 1994, 1997; Dostal et al. 2000]. This suggests that circulating renin is needed for the synthesis of angiotensin in the myocardium. Moreover, a role for circulating renin is also suggested by increased aldosterone synthesis in response to intravenous angiotensin II infusions [Ganong et al. 1962; Mason et al. 1976; Blair-West et al. 1962]. In summary, a role for circulating renin seems indisputable and, thus, its inhibition may participate in putative beneficial effects.
Alternatively, local renin production takes place in the kidney, ovary, brain, uterus and others organs [Re, 2001, 2003a, 2003b; Paul et al. 2006]. A putative role for this local RAS is sustained by a number of theoretical estimates and experimental evidence. For instance, the equilibrium dissociation constant (KD) of the Ang II reaction with AT1 receptors suggests that circulating RAS may be insufficient to stimulate AT1 receptors. Indeed, if KD (Ang II level needed to saturate 50% of the receptors) is of the order of 10−9 mol/l, the normal circulating Ang II concentrations are much lower and insufficient to saturate half of the receptors. Yet, Ang II infusions that increase the plasma levels from 10 × 10−15 to only 30 × 10−15 mol/ml are sufficient to increase mean blood pressure [Chinn and Dusterdieck, 1972]. To explain this notion, Schalenkamp and Danser have proposed a mechanism of amplification and facilitated diffusion [Schalenkamp and Danser, 2011]; through a mathematical model that considers the fluid barriers that Ang II has to cross to reach the AT1R at the vascular smooth muscle cell, the authors concluded that circulating Ang II concentration needs to be 20–30 times higher than normal plasma levels to maintain the vascular tone in resistance vessels. This means that abluminal rather than intraluminal Ang II is vital for maintaining vasoconstrictor tone. Moreover, the association rate amplification proposed, should minimize overflow of tissue Ang II into the circulation thus explaining why Ang II acts at levels far below KD, and why AT1R blockers are effective in hypertension even when PRA is low.
In addition, the internalization and binding of renin and prorenin to cellular receptors has been associated with increased cellular synthesis of angiotensin, subsequent generation of second messengers and induction of physiological effects. These observations not only reveal prorenin and renin to be hormones in their own right, they suggest that prorenin and renin are intracrines; that is, they are locally produced to exert local physiologic effects. Indeed, there is abundant evidence showing local synthesis of each of the components of the renin–angiotensin system (RAS) in various tissues [Kifor and Dzau, 1987; Pendergrass et al. 2006].
In the kidney for instance, RAS components reach exceedingly high concentrations in several specific compartments, suggesting regulatory actions and local production. The prototype is the proximal tubule, where all RAS components reach levels that are much higher than plasma levels (Figure 1). This is achieved by means of an augmentation mechanism, whereupon Ang II, increases renin synthesis, instead of decreasing it [Navar and Nishiyama, 2001; Pendergrass et al. 2006].
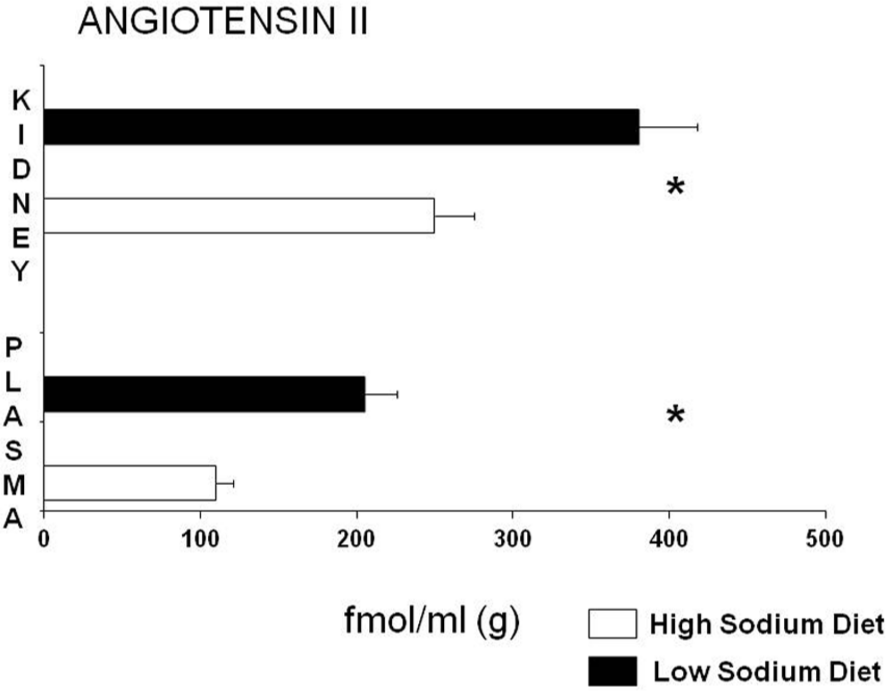
Plasma and renal angiotensin levels: low and high dietary sodium.
Because these tissue concentrations of renin and angiotensin are not mirrored in blood levels, it could explain while PRA fails to predict the blood pressure response in hypertensive patients.
Pathogenic importance of RAS
Even though PRA does not predict the hypotensive response to RAS inhibition, its pathogenic relevance is undeniable. The worse outcomes of patients with high PRA have been long demonstrated. Moreover, such increased activity is currently considered a risk factor for myocardial infarction in hypertensive patients [Alderman et al. 1991; 1997; Muhlestein et al. 2010] suggesting that PRA inhibition could be a therapeutic goal by itself.
Certainly, renin-associated risks arise through Ang II effects. Therefore, preventing the conversion of Ang I to Ang II, or blocking the AT1 receptor should avoid the risk. However, ACEIs block the synthesis of Ang II only partially and conversion of Ang I to Ang II can be achieved by mean of other enzymes [Hollenberg et al. 1998]. Moreover, parallel activation pathways generate angiotensin peptides capable of stimulating the AT1 receptors. These phenomena have been proposed as an explanation for the alleged ACE ‘escape’ [Hollenberg et al. 1998]. Likewise, ARBs efficacy can be hampered in the presence of very high Ang II levels resulting from negative feedback (first-order kinetics). Since renin and prorenin are the exclusive entrance to the RAS, their inhibition should prevent any potential escape (Figure 2). Actually, Gradman and colleagues found that ACEIs increase PRA by 116.3%, ARBs by 110.6% and hydrochlorothiazide by 35.4%. In contrast, aliskiren decreases PRA by 70.3%. In fact, aliskiren markedly inhibits both renin and activated prorenin [Gradman et al. 2006]. In brief, Ang II escape has been reported with both ACEIs and ARBs but never with renin inhibitors [Wolf and Ritz, 2005].

Generation and feedback pathways of the renin–angiotensin system. APA, aminopeptidase A; APN, aminopeptidase N.
Another possible indication for direct RAS inhibition is that both renin and activated prorenin seem to have direct intracellular effects (before being released) that may induce hypertrophy, proliferation and fibrosis [Nguyen and Danser, 2008]. For these reasons, and because diuretics, ACEIs and ARBs increase PRA, adding aliskiren may be a proper way to neutralize the intracellular effects [O’Brien et al. 2007].
Renin-dependence versus volume-dependence
Interpreting high blood pressure as either renin-dependent or volume-dependent led to unjustified doubts on the role of RAS in high blood pressure and cardiovascular disease. In fact, based on PRA levels, renin would play a pathogenic role only in about 17% of the hypertensive patients [Brunner et al. 1972; Laragh, 1973]. This notion did not match clinical experience and was rebutted when Gorbea-Oppliger and colleagues showed that subpressor doses of Ang II caused persistent hypertension that was reversed by AT1 receptor antagonists, even though angiotensin levels remained unchanged [Gorbea-Oppliger et al. 1994] (Figure 3). Currently, there is no doubt that the RAS plays a key role in the development of hypertension.
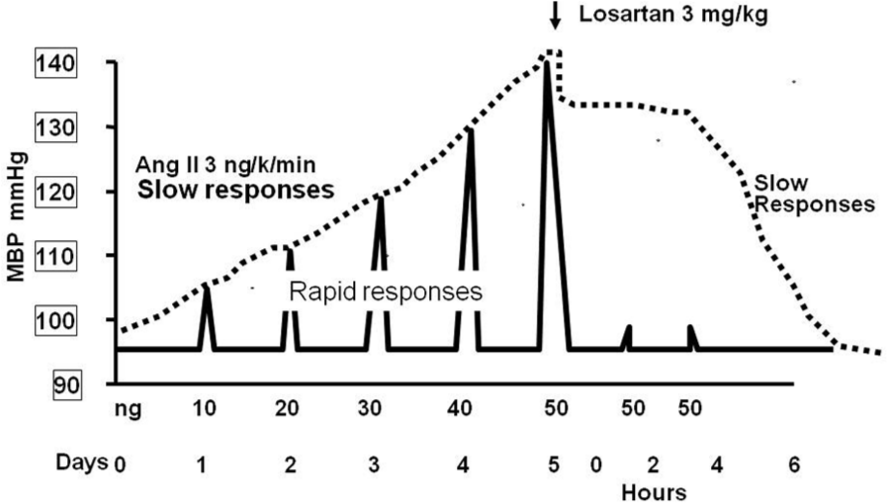
Changes in mean blood pressure following angiotensin II boluses (rapid responses to pressor doses in bolus and slow responses to ‘subpressor’ doses administered by continuous infusion).
Precisely, and based on this crucial pathogenic role of the RAS, drugs that inhibit its cascade show great effectiveness as isolated or combined therapies [Wing et al. 1997; Kjeldsen et al. 2002]. Other advantages that should be added to these benefits are: tolerability, antioxidant effects and the ability to offset other drugs undesired effects (edema and flashing of vasodilators, hypokalemia and insulin resistance of diuretics, etc.) [Drummond et al. 2007]. Their well-known protective effects on the heart, blood vessels, kidney, and central nervous system, among others, could be considered additional benefits [Dell’Italia, 2011; Schrader et al. 2005].
ACEIs, ARBs and direct renin inhibitors: differential features
Having reviewed the favorable features of RAS inhibition, it is important to define potential differences in terms of effectiveness and tolerability exhibited by the different classes of RAS inhibitors: ACEIs, ARBs and DRIs. The few comparative studies with different types of RAS inhibitors are not conclusive due to large variations in study populations [Pfeffer et al. 2003; Mann et al. 2008; Pitt, 2000]. However, the excellent efficacy and tolerability profiles of ACEIs and ARBs are shared with aliskiren [Gradman et al. 2005; Andersen et al. 2007]. Moreover, aliskiren does not affect substance P synthesis or kinin metabolism and, therefore, it is not expected to cause dry cough or angioneurotic edema.
Aliskiren has shown therapeutic advantages in some trials [Andersen et al. 2007; Solomon et al. 2009] (Table 1). However, should ‘superiority’ exist for one kind of inhibitor over the other, the emergent question would be: how could an ACEI, an ARB or a direct renin inhibitor (DRI) produce different results if pharmacologically equivalent doses are used?
Studies comparing aliskiren with an angiotensin-converting enzyme inhibitor or an angiotensin II receptor antagonist. Values indicate systolic blood pressure (SBP) and diastolic blood pressure (DBP) reductions versus baseline values. (Doses are indicated as maximum allowed by protocol). Two studies showed no inferiority, whereas the remainder showed better blood pressure lowering by aliskiren.

The evaluation of potential differences in efficacy requires the consideration of changes induced by each drug in all levels of the RAS cascade. For instance, angiotensin-converting enzyme (ACE) inhibition decreases Ang II synthesis and by negative feedback it increases the syntheses of pre-prorenin, prorenin, renin and angiotensin I [Brunner et al. 1972; Laragh, 1973]. If ACE were the only enzyme capable of converting Ang I into Ang II, the process would end at this point. However, many tissues have enzymes other than ACE that are capable of converting angiotensin I into Ang II (Figure 4). For instance, the heart expresses tonins, cathepsins, chymases and trypsin that can synthesize Ang II from Ang I [Hollenberg et al. 1998; Neves et al. 1996; Kramkowski et al. 2006]. As a result; attempts to inhibit the RAS could fail to induce the desired effects on vasoconstriction and on renal sodium excretion. In brief, converting enzyme plays no exclusive role on Ang I. Alternative enzymatic pathways could generate Ang II from Ang I [Wolny et al. 1997].
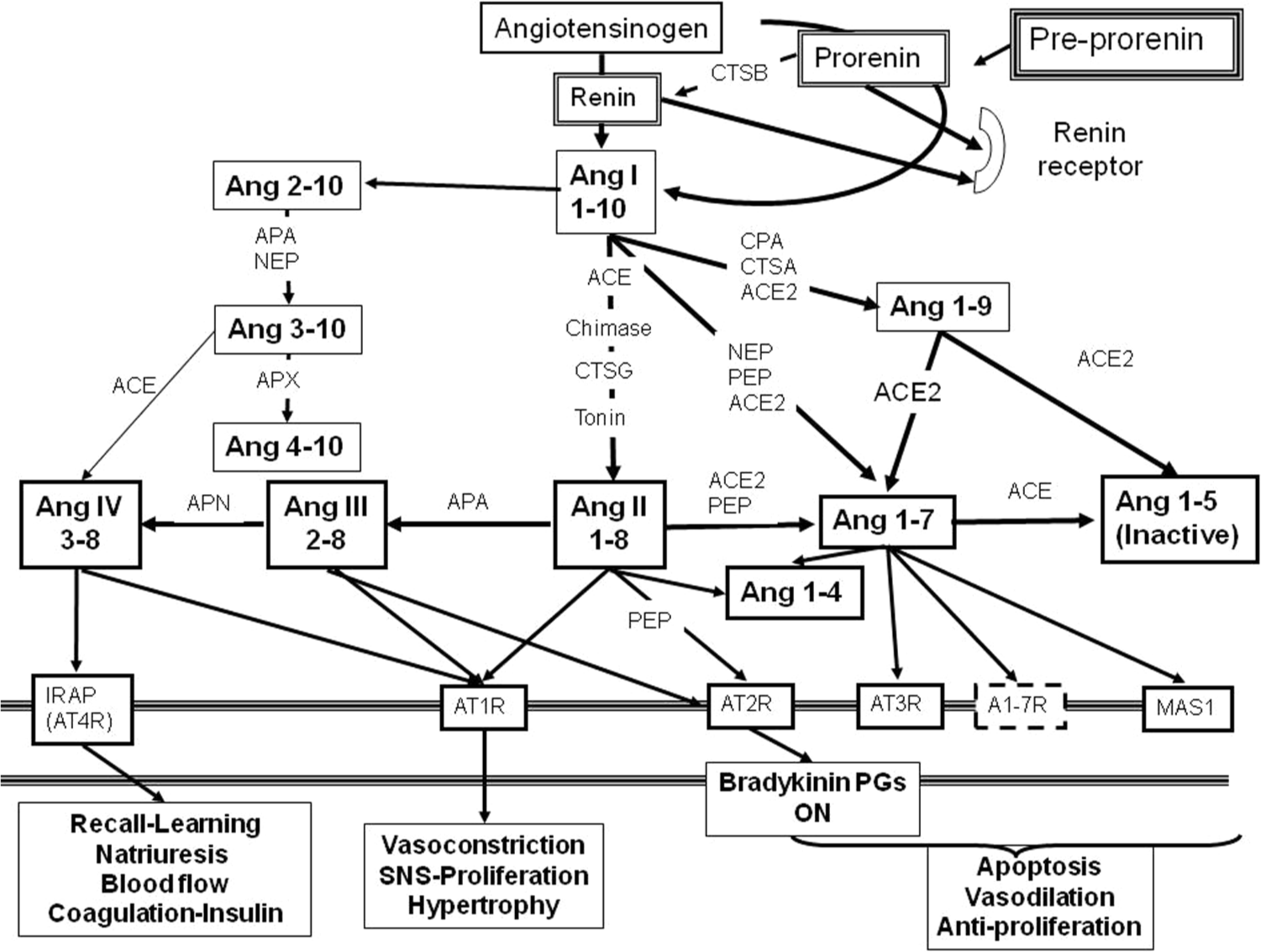
Carboxipeptidases and aminopeptidases involved in the activation of the renin angiotensin system. The multiple pathways provide routes of “escape” for the synthesis of various angiotensin peptides.
Similarly AT1 receptor blockade leads to a feedback response that increases the synthesis of all components of the RAS cascade, including Ang II. For this, AT1 receptor blockers are compelled to compete for the receptor with the higher Ang II levels (first-order kinetics), thus decreasing their specific blocking effects. In addition, other recently described receptors could be activated by Ang II-or Ang I-derived peptides excess (Figure 4) [Mogi et al. 2007; de Gasparo et al. 2000; Kostenis, 2005].
In contrast, although DRIs also increase renin synthesis, PRA ‘activity’ is powerfully blocked and, thus, the entire RAS cascade is inhibited [Nussberger et al. 2002]. Moreover, aliskiren (the available information on enalkiren, remikiren or zankiren is insufficient) has the ability to enter the cell where it could inhibit not only renin (before its release), but also activated prorenin [Krop et al. 2008]. The latter exerts cellular and systemic effects that are similar to those of renin. Thus, aliskiren could have beneficial intracellular effects [Re, 2003; Saris et al. 2006], preventing renin from inducing apoptosis, fibrosis and cell degeneration [Singh et al. 2008; Baker and Kumar, 2006]. Truly, intracellular renin activation is unexpected, since renin (like a myriad of proteins produced by cells) is retained within cytoplasmic vacuoles or stored in cell membrane ‘vesicles’ until its release [Castrop et al. 2010; Sealey and Laragh, 2007; Mendez et al. 2011].
Nevertheless, the finding of a second transcription start site within the renin gene predicted the synthesis of a shorter form of prorenin. This ‘truncated’ form of prorenin lacks the ‘signal peptide’ that blocks its enzymatic activity [Lee-Kirsch et al. 1999]. Normally, by gluing the prosegment to the rest of the molecule, the signal peptide suppresses renin enzymatic action [Reudelhuber, 2009; Kumar and Boim, 2009]. Consequently, ‘active’ truncated prorenin could play harmful effects within the cell [Re, 2003]. Aliskiren’s special ability to enter the cells [Krop et al. 2008] suggests that these deleterious intracellular effects from a truncated form of prorenin could also be inhibited. Such putative action however has not been tested so far.
Paradoxical effect of direct renin inhibition
Direct renin inhibition largely accounts for its therapeutic effectiveness. However, it has been suggested that increased renin synthesis due to negative feedback could lead to a paradoxical rise in blood pressure [Sealey and Laragh, 2007, 2009]. This rationale does not take into account the fact that regardless of how high renin concentration is, renin activity is inhibited with effectiveness close to 100% [Stanton et al. 2003]. Resistant cases showing trivial blood pressure rises are expected in any large hypertension trial, a phenomenon probably related to individual variations in compliance, dietary salt intake and others. The ‘prohypertensive’ hypothesis has not been shown in trials reporting on aliskiren efficacy in various populations [Menard and Azizi, 2007; Stanton et al. 2010]. In contrast, aliskiren monotherapy has been shown to reduce blood pressure regardless of the PRA levels [Schmieder et al. 2009; Stanton et al. 2009].
In summary, both renin and prorenin play a pathogenic role in high blood pressure. It would appear then that the main advantage of direct renin inhibition rests on effects involving both renin and prorenin within the cell and in the circulation. The intracellular inhibition could reverse direct intracellular effects, including those caused by truncated prorenin. These concepts are supported by several efficacy and tolerability studies.
Clinical efficacy of aliskiren
Effectiveness by gender and age
Several clinical studies have shown that direct renin inhibition by aliskiren effectively lowers systolic and diastolic blood pressure and that this reduction is dose-dependent (Table 1). For instance, the AGELESS Trial (Aliskiren for Geriatric Lowering of Systolic Hypertension) showed that aliskiren (either as monotherapy or combined with hydrochloro-thiazide) is more effective than ramipril at reducing systolic and diastolic blood pressure in patients older than 65 years with systolic hypertension [Duprez et al. 2010].
Blood pressure reduction in hypertensive patients in several populations. Values indicate systolic blood pressure (SBP) and diastolic blood pressure (DBP) reductions (in mm/Hg) versus baseline values (*p < 0.05) and reductions (in mm/Hg) versus lower dose (#p < 0.05).

Long-term effectiveness
A very interesting pharmacological aspect of aliskiren is that direct renin inhibition produces a long-term pharmacological effect, particularly for treatments lasting several weeks [Andersen et al. 2009; Sica et al. 2008] (Figure 5). Indeed, the vascular effects of aliskiren last longer than expected based on its half-life. This is particularly true in patients on long-term therapy who remain under control for several days and in some cases for several weeks after drug discontinuation. Krop and colleagues have shown that aliskiren accumulates in renin granules in the kidney, thus allowing long-lasting RAS blockade beyond the half-life of this drug [Krop et al. 2008]. These lasting effects from aliskiren could be of key importance, bearing in mind the extremely low compliance reported in the treatment of hypertension.
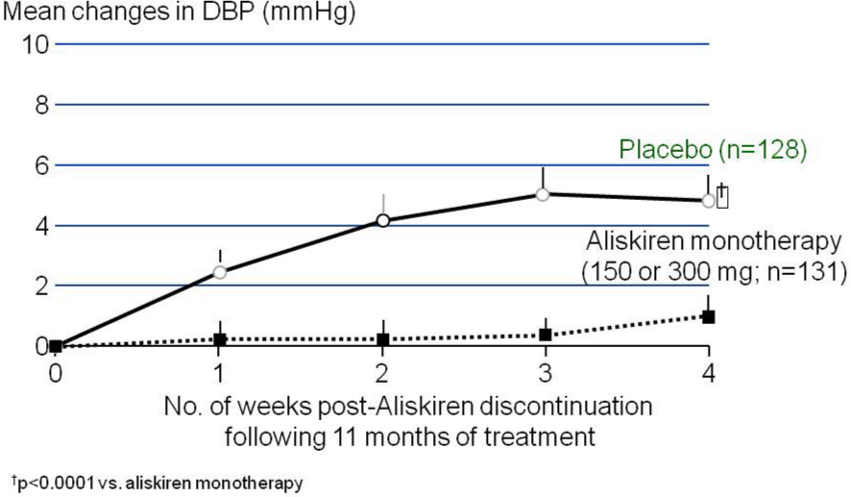
Antihypertensive effect of aliskiren following the discontinuation of long-term treatment.
Effectiveness in clinical groups
Essential hypertension
For aliskiren, the largest evidence regarding hypertension control comes from patients with primary hypertension (Table 1). In these patients, aliskiren reduces blood pressure with similar or higher effectiveness compared with drugs commonly used as first-line treatment [Frampton and Curran, 2007; Villamil et al. 2007].
High blood pressure and congestive heart failure
Aliskiren has also shown great efficacy when arterial hypertension and heart failure coexist. Indeed, the ALOFT trial [McMurray et al. 2008], comparing aliskiren with placebo added to standard of care therapy in heart failure, showed an excellent tolerability profile with clinical recovery and hemodynamic improvement in terms of left ventricular function (as assessed by echocardiography), along with reduction of brain natriuretic peptide (BNP) and NT-proBNP (n-terminal pro-brain natriuretic peptide) levels [Latini et al. 2002] (Figure 6). These parameters correlate with the hemodynamic status. Briefly, adding aliskiren to standard of care therapy in patients with heart failure produces comparable tolerability, reduction of BNP, NT-proBNP, aldosterone and renin activity levels and improvement in left ventricular function by ultrasound. These beneficial effects of aliskiren were confirmed in the ALLAY trial. Indeed, aliskiren was as effective as losartan in terms of blood pressure control and in reversing left ventricular hypertrophy [Solomon et al. 2009] (Figure 7).

Brain natriuretic peptide (BNP) levels in several clinical studies. Aliskiren versus other antihypertensive agents.
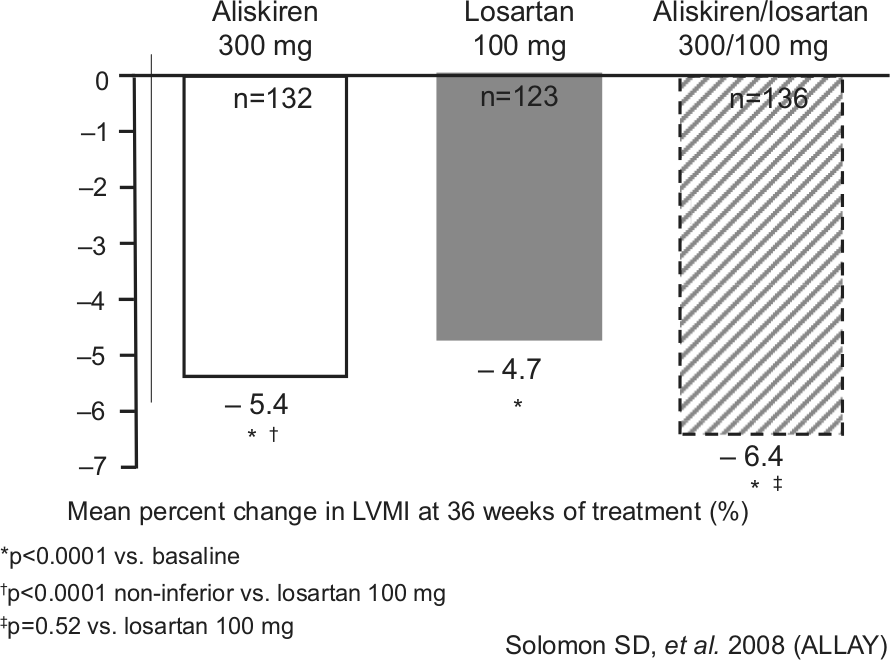
Aliskiren/losartan combination reduces left ventricular mass index ~20% more than losartan monotherapy (ALLAY [Solomon et al. 2008]).
High blood pressure and diabetes
Preliminary studies with aliskiren strongly suggest a dose-dependent efficacy in the treatment of hypertension in diabetic patients [Deinum et al. 1999; Kawazu et al. 2004]. The AVOID study showed a remarkable reduction in proteinuria in patients treated with aliskiren, beyond that achieved with losartan at maximal doses [Parving et al. 2008].
High blood pressure and metabolic syndrome
Krone and colleagues compared the effects of aliskiren 300 mg/day with irbesartan 300 mg/day in hypertensive patients with metabolic syndrome [Krone et al. 2011]. A larger proportion of aliskiren patients showed blood pressure reduction to 135/85 mmHg compared with irbesartan (29.2% aliskiren versus 16.7% irbesartan). Both drugs exhibited similar effects in terms of blood glucose, lipid profile, inflammation biomarkers levels and cardiovascular risk. Likewise, aliskiren efficacy has also been shown in obese populations [Jordan et al. 2007].
High blood pressure and chronic kidney disease
Aliskiren should be an excellent choice in patients with chronic kidney disease (CKD) [Campbell et al. 2003; Andersen et al. 2008]. Expectations are particularly high in patients with diabetic nephropathy in whom their frequent high prorenin levels may play an important pathogenic role [Jennings et al. 2007; Mogensen et al. 2000]. Aliskiren inhibits both renin and prorenin activity [Nussberger et al. 2002]. Unfortunately, the ALTITUDE study [Parving et al. 2012] approached renal protection from a double blocking standpoint in patients at high cardiovascular risk and had to be discontinued due to adverse effects considered to be the result of the vigorous RAS suppression. Regrettably, a long-term trial comparing renal function effects of direct renin inhibition with ACEIs or ARBs is not available. Be that as it may, the dual RAS blockade has fallen in disregard but the question remains open, in particular in patients with CKD and evidence of progressive renal injury.
Dual RAS blockade
The few studies intended to assess dual RAS blockade have been largely contradictory [Mogensen et al. 2000; Nakao et al. 2004; Kunz et al. 2008; Yusuf et al. 2008]. While seemingly beneficial in the ALLAY study, where the combination with losartan showed clear advantages over monotherapy [Solomon et al. 2009], the results where negative in the ONTARGET trial [Mann et al. 2008] and disappointing in the ALTITUDE study [Parving et al. 2012]. A possible explanation for these contradictions could be that dual blockade has been used nonspecifically; omitting situations where double or even triple RAS blockade could be most useful. One example is the ONTARGET trial, where it was concluded that telmisartan combined with ramipril had no added benefit and could be even detrimental [Mann et al. 2008]. However, it is difficult to support the use of the dual blockade approach in patients with normal or near-normal baseline blood pressure. Indeed, 142/82 mmHg was the mean baseline value in ONTARGET patients and 137/74 mmHg in the ALTITUDE study. Patients treated with dual RAS inhibition and whose basal blood pressure values were below this average could have suffered episodes of critically low coronary, brain or kidney perfusion.
The ONTARGET investigators concluded that drug combination did not exhibit any benefit in terms of kidney function. This conclusion seems unjustified for several reasons: (a) only a few patients had proteinuria (damage prevention could hardly be demonstrated where no active damage seemed evident, in a short-term study and with near-normal blood pressure levels; (b) kidney function was normal on average (mean value: 73.6 ml/min); (c) only 36.7% of the population were diabetics and 68.5% were hypertensive.
In addition to these serious issues, other questions arise. For instance, the ONTARGET study was not powered to detect changes in different renal outcomes (∼600 of >3000 were specifically renal outcomes). Mortality rate and acute hemodialysis (HD), not always related to kidney disease progression, were endpoints added to give statistical power to renal outcomes. In this regard, if acute HD is excluded, no difference persists in these kidney-related outcomes. Nonetheless, the rates of HD and increased serum creatinine were similar between groups (glomerular filtration rate [GFR] drop is expected and even desired, since it indicates glomerular pressure reduction). Indeed, in several studies, RAS inhibitors have shown to decrease glomerular filtration rate via a hemodynamic mechanism that is not overtly detrimental, provided that serum electrolyte levels are not markedly changed. In fact, once treatment is initiated, patients with higher serum creatinine levels obtain the highest cardiovascular benefits [Apperloo et al. 1997; Bakris and Weir, 2000]. This notion has been recently confirmed in a post hoc analysis of the RENAAL study. Indeed, patients with larger falls in GFR showed better long-term preservation of kidney function [Holtkamp et al. 2011].
Other known limitations in the ONTARGET trial include: (a) albuminuria was not assessed on a yearly basis; (b) serum creatinine was measured with nonstandardized or centralized methods; (c) doubling of serum creatinine was not confirmed; and (d) indication of HD was arbitrary and nonprotocol-specified. In 98 patients the reason for chronic HD was undefined.
Be that as it may, in both ONTARGET and ALTITUDE most of the serious adverse effects could have been the result of poor perfusion pressure, a predictable complication in high-risk patients. For instance, in ALTITUDE, hypotensive episodes occurred more frequently when aliskiren was added to a therapy based on an ACEI or an AR1T receptor antagonist, in patients who were older than 65 years old, with lower pulse pressure or taking loop diuretics.
Certainly, falls, fainting, hyperkalemia and incre-ased serum creatinine are well-known complications of poor organ perfusion and therefore avoidable. In the ALTITUDE trial, patients with serum potassium concentrations greater than 5 mMol/l were randomized to dual drug treatment. This is important, bearing in mind that trial definitions may not reflect the clinical significance of a side effect. For instance, combination therapy is associated with an increased risk of moderate hyperkalemia (serum potassium >5.5 mEq/l) but not of clinically significant hyperkalemia (serum potassium >6 mEq/l) compared with monotherapy [Harel et al. 2012]. Be that as it may, these studies clearly show that combined RAS inhibitors should be avoided in patients with basal normal or nearly normal blood pressure and in those with basal serum potassium concentration greater than 5 mEq/l.
The fact remains that aliskiren in combination with an ARB or an ACEI offers potential cardiorenal benefits, and its utilization has demonstrated a good tolerability profile compared with placebo, particularly in patients at high risk for congestive heart failure, diabetes mellitus or CKD [Solomon et al. 2009; McMurray et al. 2008; Deinum et al. 1999; Kawazu et al. 2004; Freiberger et al. 2009]. In CKD, it should be kept in mind that the goal is not only blood pressure control, but also proteinuria reduction. High protein excretion rate is a risk factor for kidney disease progression, and its reversion delays progression of CKD [Brenner et al. 2001; Jafar et al. 2001].
Tolerability and safety
Aliskiren has shown an excellent tolerability profile that is comparable with or higher than that of other RAS inhibitors [Gradman et al. 2005; Verdecchia et al. 2010]. For example, in the AGELESS study, aliskiren therapy was associated with a lesser incidence of adverse effects compared with ramipril (70% less undesired effects with aliskiren, compared with ramipril) [Duprez et al. 2010]
In summary, in light of new knowledge on RAS components, the approach to high blood pressure treatment continues developing encouragingly. Among all available agents, those inhibiting the RAS show clear pathogenic and pharmacokinetic advantages, as well as an excellent tolerability profile. Particularly, direct renin inhibition appears to be not only the most recent but also a promising advance in the search of an effective control for high blood pressure, in particular, when accompanied by CKD.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr Juncos has no relation or business interests with any pharmaceutical company, and does not receive or has received any (direct or indirect) research fund from any company that produces medications or medical devices. The author is not a consultant in any pharmaceutical company, and holds no shares or investment of any nature in any pharmaceutical company.
Dr Juncos participated as an investigator in the ALTITUDE study (a randomized, double-blind, placebo-controlled, parallel group study to determine whether, in patients with type 2 diabetes at high risk for cardiovascular and renal events, aliskiren, on top of conventional treatment, reduces cardiovascular and renal morbidity and mortality).

