Abstract
The use of 3-hydroxy-3-methylglutaryl coenzyme A reductase (statins) is associated with a marked reduction in morbidity and mortality in patients at high cardiovascular risk or with established cardiovascular disease. In the last decade, several randomized controlled studies have demonstrated the benefit of statins in patients with acute coronary syndrome (ACS). These studies showed that use of statins in patients with ACS is associated with a significant reduction of the risk of recurrent cardiovascular events. Current American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommend (Level of Evidence 1A) the use of statin therapy before hospital discharge for all patients with ACS regardless of the baseline low-density lipoprotein. Although there is no consensus on the preferable time of administration of statins during ACS, some clinical trials and pooled analyses provided substantial support for the institution of an early initiation to improve strategies that target the pathophysiologic mechanism operating during myocardial infarction. In particular, recent findings suggested that the earlier the treatment is started after the diagnosis of ACS the greater the expected benefit. Experimental studies with statins in ACS have shown several other effects that could extend the clinical benefit beyond the lipid profile modification itself. In particular, statins demonstrated the ability to induce anti-inflammatory effects, modulate endothelium and inhibit the thrombotic signaling cascade. Given these recognized potential benefit, statins should conceivably modulate the pathophysiological processes involved in the very early phase of plaque rupture and coronary thrombosis.
Keywords
Introduction
Acute coronary syndromes (ACS) have an estimated annual age-adjusted incidence in the general population ranging from 3 to 8 per 1000 subjects and are associated with high rates of in-hospital morbidity and mortality [Moustapha and Anderson, 2003; Granger and Weaver, 2004; Roger et al. 2012]. Despite treatment strategies, including drug therapies which modify platelet function and coagulation cascade, angiotensin-converting enzyme (ACE) inhibitors, beta-blockers and interventional procedures, ACS patients have a higher incidence of recurrent coronary events compared with those with stable coronary artery disease (CAD). The rate of recurrence is particularly high in the 30 days after an acute presentation [Paradiso-Hardy et al. 2003] and nearly 20% of men and 26% of women die within 1 year of having a first myocardial infarction (MI) [Roger et al. 2012].
On the basis of these observations, the need for improved strategies that target the pathophysiologic mechanism operating in ACS is mandatory.
In this regard, there has been increasing interest to the use of statins early in the course of ACS and quite recently the positive impact of initiating statin treatment immediately following the occurrence of ACS has emerged [Waters and Hsue, 2001; Pepine, 2003].
In the last few years, data from observational studies and randomized controlled trials (RCTs) supported the use of statin therapy during ACS for the reduction of death and cardiovascular (CV) events and, consequently, oriented the current American College of Cardiology/American Heart Association (ACC/AHA) guidelines to recommend (Level of Evidence 1A) the use of statin therapy before hospital discharge for all patients with ACS regardless of the baseline low-density lipoprotein (LDL) [Wright et al. 2011].
However, the results of a recent meta-analysis from the Cochrane collaboration group fuelled debate on the real benefit of statin treatment during ACS [Briel et al. 2011]. In particular, Briel and coworkers did not find a significant reduction of the composite primary endpoint (death, MI or stroke) or individual secondary endpoints from early (<14 days from onset of ACS) statin therapy. Only for unstable angina (UA) did the authors find a significant relative risk (RR) reduction of 24% at 4 months [Briel et al. 2011].
Our objective was to update the evidence on statin treatment during the acute phase of ACS. In particular, the main aim of our narrative overview was to assess whether early initiation of statin therapy is associated with relevant benefits in term of clinical outcome and to highlight the mechanism that may be responsible for these potential beneficial effects. For this purpose, we identified main observational and RCTs using research methodology filters [Haynes et al. 1994]. The following research terms were used: hydroxymethylglutaryl-CoA reductase inhibitors, myocardial infarction, acute coronary syndrome, intensive care, intensive care units, outcome, prognosis. We also performed hand-searching of conference proceedings, pharmaceutical industry files and personal communication from experts in the field [Mcauley et al. 2000], to identify any other relevant study. No language restriction was applied, to avoid discriminating against papers not written in the English language (‘tower of Babel bias’) [Gregoire et al. 1995].
Mode of actions
The benefit of statins in primary and secondary prevention is based on the reduction of blood levels of LDL cholesterol. Currently, new benefits of statins, not dependent on the reduction of LDL cholesterol, have been proposed in the setting of ACS. These effects, which are not dependent on the reduction of LDL cholesterol, are called pleiotropic [Liao, 2005].
Some of the possible effects of statins, which may participate in the benefits that they represent for the ACS, are depicted in Figure 1.

The role of statin therapy in the pathogenesis of acute coronary syndrome. Main pleiotropic effects not dependent on the reduction of low-density lipoprotein cholesterol are depicted.
Previous studies in experimental animals have shown that statins increase the production of nitric oxide (NO) and fibrous cap thickness in association with collagen accumulation [Fukumoto et al. 2001], and reduce the number of inflammatory cells as well as various inflammatory mediators in atheromatous plaques [Bustos et al. 1998]. In particular, the pleiotropic effects of statins, possibly involved in the benefits that these drugs offer during the ACS, include the ability to decrease activity of the metalloproteinase matrix (MMP) in atherosclerotic plaques, interleukin-1, interferon gamma, interleukin-6 and soluble CD40 ligand, circulating rates of C-reactive protein (CRP), plasminogen activator inhibitor-1 (PAI-1) and levels of tissue factor (TF) that limits the onset and progression of intracoronary thrombus [Bustos et al. 1998]. As an example, an experimental study of apolipoprotein E knockout mice has demonstrated that statin treatment reduces the incidence of plaque rupture in the brachiocephalic arteries [Johnson et al. 2005].
In humans, a serial angioscopic study evaluated changes in coronary plaque color and morphology by statin therapy. Briefly, 31 patients with CAD were divided into either the comparison group (
A grayscale intravascular ultrasound (IVUS) study revealed that statins increase plaque hyperechogenicity without significantly decreasing plaque volume [Schartl et al. 2001].
A reduction in the lipid component of coronary plaques after statin treatment was also demonstrated by a three-dimensional integrated backscatter IVUS study [Kawasaki et al. 2005].
Another IVUS study has also reported that long-term statin treatment before the onset of ST elevation (STE) ACS is significantly associated with a lower incidence of plaque rupture as compared with the absence of statin treatment [Otsuka et al. 2010].
In addition, an optical coherence tomography (OCT) study has demonstrated that 9 months of statin treatment increases fibrous cap thickness in nonculprit coronary plaques of patients with acute MI [Takarada et al. 2009].
Observational studies
A number of observational studies conducted in patients with ACS have analyzed the impact of statin therapy on the risk reduction of recurrent coronary events and death.
A retrospective analysis of the Platelet Receptor Inhibition in Ischemic Syndrome Management (PRISM) study was designed to test the potential benefit of statins in 1616 patients with ACS [Heeschen et al. 2002]. The administration of statins was associated with a significant 51% reduction in the risk of events and the discontinuation during hospitalization was associated with an increased risk of developing CV events (odds ratio [OR] 2.93).
In a pooled analysis of the Global Use of Streptokinare or t-PA for Occluded Coronary Arteries (GUSTO IIb) and the Platelet Glycoprotein IIb/IIIa in Unstable Angina: Receptor Suppression Using Integrilin Therapy (PURSUIT) studies [Aronow et al. 2001], all-cause death in 3653 patients that were prescribed statin therapy was compared with 17,156 who were not. Lipid-lowering therapy was associated with a smaller proportion of deaths at 30 days (hazard ratio [HR] 0.44; 95% confidence interval [CI] 0.27–0.73;
More recently, the Global Registry of Acute Coronary Events (GRACE) study recruited 20,000 patients with ACS and compared those who had no started statins during hospitalization with those who discontinued this therapy and those who continued using during this period [Spencer et al. 2004]. The latter (Figure 2) had significantly lower combined risk of MI or death (OR 0.66; 95% CI 0.56–0.77). Notably, early use of statins at hospital admission was also beneficial (OR 0.87; 95% CI 0.78–0.97) and patients who discontinued statin therapy upon admission had a similar risk to those who have never received statins (OR 1.02; 95% CI 0.74–1.41).

Prognostic implications of statin therapy during acute coronary syndromes in the Global Registry of Acute Coronary Events (GRACE). (Data from Spencer et al. [2004].)
Similarly, a report from the Euro Heart Survey on ACS [Lenderink et al. 2006] tested the hypothesis that initiation of statin therapy within 24 h after admission was associated with improved outcome. As chronic use of statins before hospitalization might influence its early effects, 2099 patients already receiving statin treatment before hospitalization were excluded. Overall, 8197 patients who survived the first 24 h were available for analysis. A total of 1426 patients received statin therapy within 24 h after admission. All-cause mortality at 7 days was significantly lower in the group of patients receiving early statin therapy (0.4%
At variance, in the Sibrafan
It is worth mentioning that in these observational studies, there were significant differences in the baseline characteristics between the statin users and nonusers. As an example, in the Euro Heart Survey report [Lenderink et al. 2006], statin users were younger and more often men than nonusers. Traditional risk factors for CAD, including diabetes mellitus, hypercholesterolemia, a family history of coronary disease, and cigarette smoking as well as a prior history of coronary disease were also more prevalent in the group of statin users.
In addition, multivariate and propensity adjustments did not suppress a number of residual confounders.
Despite these limitations, the concordance of some large studies seems to supports a potential positive influence of an early treatment of statin therapy in patients with ACS. Moreover, these observational studies clearly remarked the safety of statin when administered within complex therapeutic strategies including anticoagulants, antiplatelet and anti-ischemic drugs and blood pressure (BP)-lowering agents.
Randomized clinical trials
In the last few years, several RCTs were conducted to establish the benefit of early statin therapy in ACS patients. However, they showed mixed results.
Among available RCTs, 5 trials recruited more than 500 patients with ACS and deserve mention.
In the Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) trial [Schwartz et al. 2001], 3086 patients with non-ST elevation (NSTE) ACS were randomized within 4 days of the event to atorvastatin 80 mg/day or to placebo and followed for 16 weeks. The primary endpoint, a composite of death, MI, resuscitated cardiac arrest, and recurrent symptomatic myocardial ischemia with objective evidence requiring emergency hospitalization, occurred in 14.8% of atorvastatin patients and 17.4% of the placebo patients (HR 0.84, 95% CI 0.70–1.00,
The Pravastatin or Atorvastatin with Aggressive Cholesterol Lowering (PROVE-IT) trial [Cannon et al. 2004] has extended the findings of the MIRACL trial, while also demonstrating the greater benefits of intensive lipid-lowering therapy with atorvastatin 80 mg, compared with conventional lipid-lowering therapy with pravastatin 40 mg. In total, 4162 patients hospitalized with an ACS within the preceding 10 days were randomized to either standard therapy or intensive therapy and were followed for a mean of 24 months. The primary endpoint consisted of death, MI, documented UA requiring rehospitalization, stroke, and revascularization (at least 30 days after randomization). The primary event rate was 22.4% in the atorvastatin group and 26.3% in the pravastatin group (HR 0.84, 95% CI 0.74–0.95,
In the small Fluvastatin On Risk Diminishment after Acute Myocardial Infarction (FLORIDA) trial [Liem et al. 2002], patients presenting with STE-ACS were randomized to fluvastatin 80 mg or placebo within 14 days of acute MI. The median time to the initiation of study medication after the onset of symptoms was 8 days and the primary endpoint was a composite of either ischemia on ambulatory electrocardiographic (ECG) monitoring at 12 months or the occurrence of a major clinical event including death, recurrent MI, or revascularization. Fluvastatin therapy did not affect the incidence of major adverse cardiac events when compared with placebo over the period of the study. However, the trial was underpowered and the results are therefore difficult to interpret.
The larger Phase Z of the Aggrastat to Zocor (A to Z) trial [De Lemos et al. 2004], which included NSTE-ACS and STE-ACS patients, showed a favorable, but statistically nonsignificant trend towards reduction of major CV events during 6-24 months follow up in patients receiving an intensive simvastatin regimen (40 mg/day for 1 month followed by 80 mg/day thereafter) when compared with those receiving a less intensive regimen (placebo for 4 months followed by 20 mg/day of simvastatin). None of the components of the primary endpoint was reduced except for CV death (HR 0.75; 95% CI 0.57–1.00). However, study medication in the A to Z trial was started after patients were stabilized for at least 12 h.
The Pravastatin in Acute Coronary Treatment (PACT) trial [Thompson et al. 2004] tested the effects of administering pravastatin within 24 hours of the onset of symptoms in patients with UA, NSTE-ACS, or STE-ACS. A total of 3408 patients were randomly assigned to treatment with pravastatin (1710 patients) or matching placebo (1698 patients). The primary endpoint of the study was a composite of death, recurrence of MI, or readmission to hospital for UA within 30 days of random assignment. A relative risk (RR) reduction of 6.4% in the occurrence of primary endpoint favored allocation to pravastatin but was not statistically significant (95% CI −13.2% to 27.6%).
Perspectives in clinical practice
High dose
Among the mega-trials [Schwartz et al. 2001; Cannon et al. 2004; De Lemos et al. 2004] which investigated intensive therapy compared with moderate therapy with statins during ACS, the results of the PROVE-IT trial [Cannon et al. 2004] are particularly noteworthy because high-dose atorvastatin was compared with pravastatin 40 mg/day, the drug and dose that significantly reduced the primary endpoint in older large, randomized, placebo-controlled trials. The benefits of intensive statin treatment as compared with standard therapy were consistent over time and across the prespecified subgroups, which included men and women and patients with or without diabetes mellitus. In other words, the results of the PROVE-IT trial support the evidence that an intensive lipid-lowering statin regimen during ACS provides greater protection against death or major CV events than does the standard regimen. In addition, the higher dose of atorvastatin had a safety profile that was comparable to that observed with lower dose of pravastatin: the rates of discontinuation of treatment were 21.4% in the pravastatin group and 22.8% in the atorvastatin group at 1 year (
Another clinical trial which compared intensive therapy
At least two recent meta-analysis of high-dose statin trials have been able to quantify the benefit during ACS [Cannon et al. 2006; Bavry et al. 2007]. In particular, a recent pooled analysis [Bavry et al. 2007] included 7 clinical trials that randomized patients with ACS to intensive statin therapy compared with less intensive lipid reduction for a total of 9553 patients who started statin therapy within 12 days of hospital presentation. The incidence of all-cause mortality was 3.4% in the intensive statin group
Very early treatment
Clinical trials are not designed to demonstrate when the benefit of statin treatment begins. However, the cumulative HR for the primary endpoint in the PROVE-IT trial was reduced by the same amount from 15 days to 4 months, with the difference becoming significant at the latter point. Using a composite triple endpoint of death, MI, and rehospitalization for ACS, the difference between the treatment groups is already statistically significant at 30 days and remain so throughout the follow-up period [Ray et al. 2005].
On the basis of these considerations, we supposed that the relative late start of statin treatment is one of the reasons for the lack of benefit observed in some trials, as the beneficial non-lipid-lowering effects might be optimal very early in the process of plaque destabilization. At this purpose we conducted a meta-analysis [Angeli et al. 2012] of clinical trials evaluating the prognostic impact on 1-month mortality of statins therapy initiated in the first 72 hours of ACS (Figure 3). A total of 10 trials [Kesteloot et al. 1997; Den Hartog et al. 2001; Schwartz et al. 2001; Kayikcioglu et al. 2002; Serruys et al. 2002; Okazaki et al. 2004; Thompson et al. 2004; Macin et al. 2005; Ren et al. 2009; Ostadal et al. 2010] were included in the analysis (Table 1). The 10 trials were allocated to three different subgroups defined by the mean time of initiation of statin therapy: the very early treatment (24 hours) included 5 RCTs, in 3 RCTs the mean time of statin therapy administration was 48 hours, while 2 RCTs were conducted in patients treated with statin during a mean time of 3 days from the onset of ACS (Table 1).

Impact of statin therapy on all-cause mortality in randomized clinical trials comparing early statin therapy with placebo or usual care in patients with acute coronary syndrome. Trials are allocated to three different subgroups defined by the mean time of initiation of statin therapy during acute coronary syndrome. (Data from Angeli et al. [2012].)
Main characteristics of the randomized clinical trials comparing early statin therapy with placebo or usual care in patients with acute coronary syndrome (ACS). In all trials statin therapy was administered within 3 days following the symptom onset of ACS.
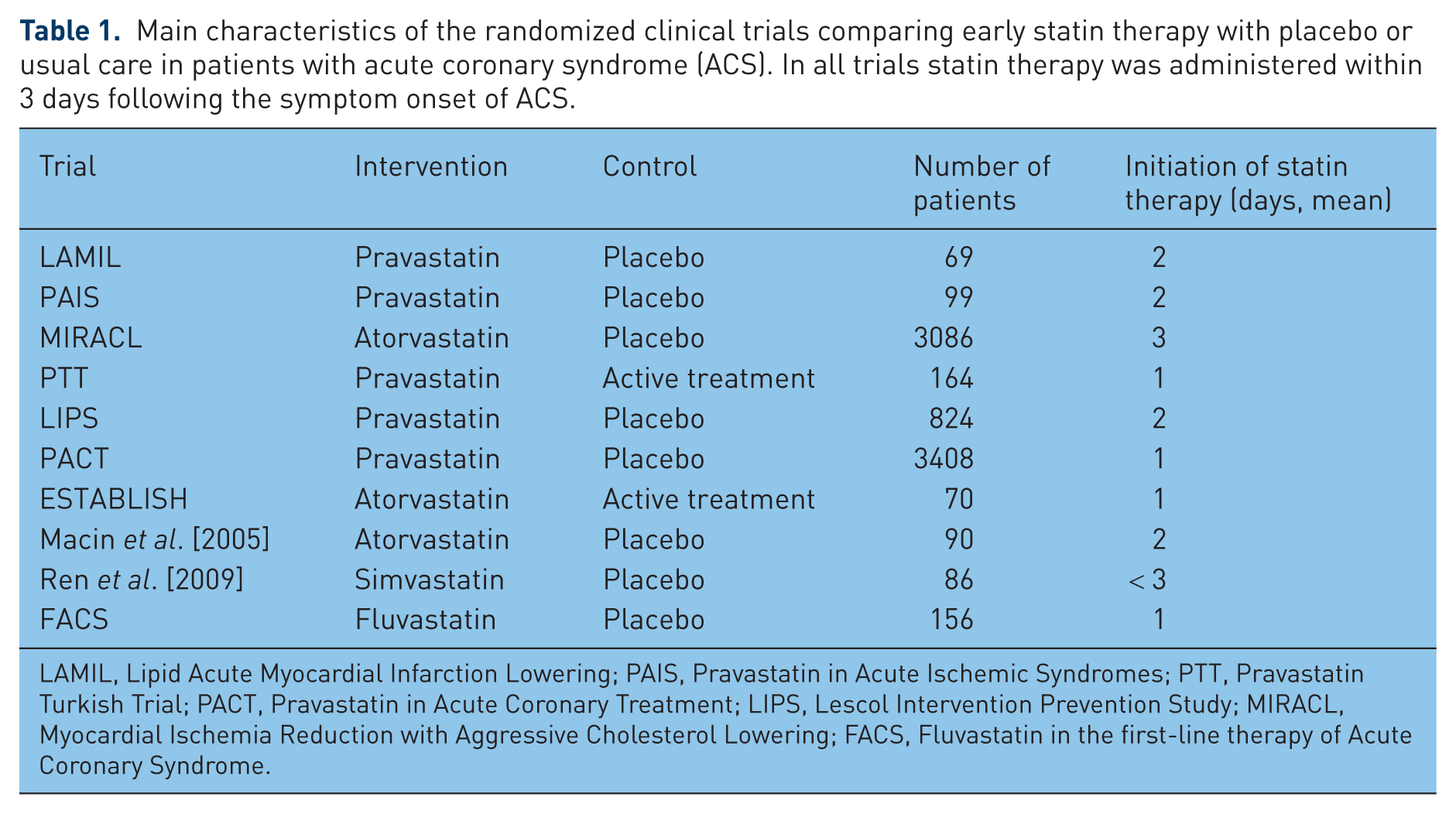
LAMIL, Lipid Acute Myocardial Infarction Lowering; PAIS, Pravastatin in Acute Ischemic Syndromes; PTT, Pravastatin Turkish Trial; PACT, Pravastatin in Acute Coronary Treatment; LIPS, Lescol Intervention Prevention Study; MIRACL, Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering; FACS, Fluvastatin in the first-line therapy of Acute Coronary Syndrome.
Overall, 4030 patients were randomized to statin therapy and 4022 patients were assigned to the control group (placebo or standard care): 66 and 83 died of any cause in the statin and control groups, respectively. Although treatment with statins had no significant effect on mortality (OR 0.80, 95% CI 0.58–1.12;
Coronary interventions and stent thrombosis
The pleiotropic effects of statins potentially provide a clinical benefit in the setting of percutaneous coronary intervention (PCI) by preventing postprocedural myocardial damage and CV events [Mihos et al. 2010].
Basic research studies have been conducted to elucidate the involvement of statins on thrombus formation and in stent complication pathophysiology (Figure 4).
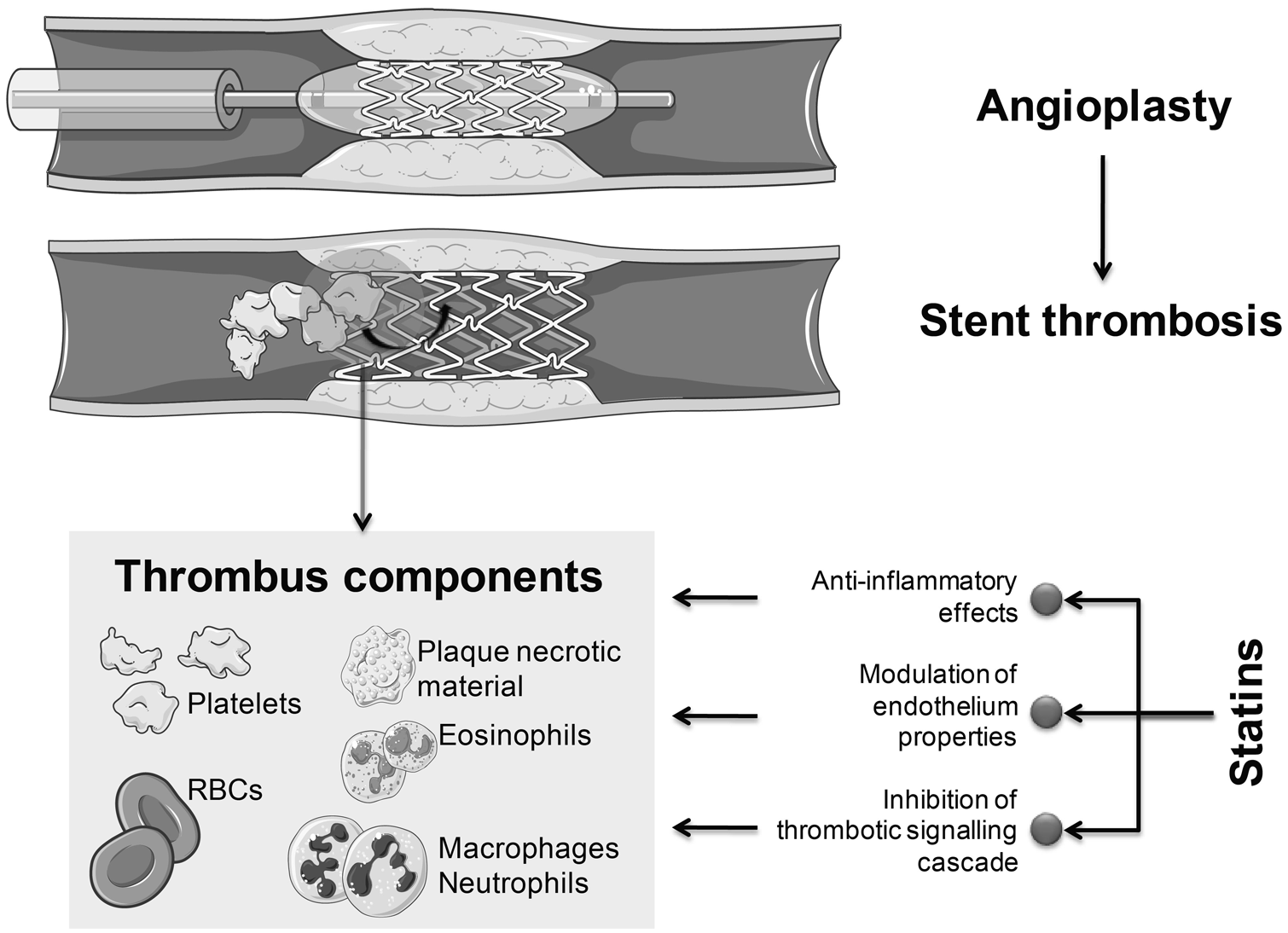
Potential beneficial effects of statins during and after percutaneous coronary intervention.
Walter and coworkers investigated a potential interrelation among statin therapy, serum evidence of inflammation, and the risk for recurrent coronary events in 388 consecutive patients undergoing coronary stent implantation. Patients were grouped according to the median CRP level (0.6 mg/dl) and to the presence of statin therapy. Recurrent events occurred significantly more frequently in patients with elevated CRP levels without statin therapy; in the presence of statin therapy the risk for recurrent events was significantly reduced in the patients with elevated CRP levels to about the same degree as in patients with CRP levels below 0.6 mg/dl [Walter et al. 2001].
Moreover, Zhu and coworkers showed that fluvastatin induced antithrombotic properties in human vascular smooth muscle cells. In particular, they demonstrated that this drug reduced the sirolimus-induced expression of tissue factor, a primary initiator of the coagulation cascade [Zhu et al. 2005].
Statins treatments have been also investigated in the improvement of CV outcomes after stent implantation in human beings. In this context, several studies showed that statin treatment before and after PCI reduces the incidence of coronary events [Serruys et al. 1999, 2002; Chan et al. 2002; Briguori et al. 2004; Pasceri et al. 2004; Mihos et al. 2010].
In particular, a recent clinical study in 455 patients undergoing primary coronary stenting for STE-ACS followed up for at least 1 month after PCI, showed a quite high incidence of definite stent thrombosis of 3.7%. The use of a statin was found significantly more frequent in the group without stent thrombosis as compared with patients affected by this complication (39.5%
Unfortunately, RCTs are still missing to evaluate the clinical benefit of statin administration before PCI in patients with ACS.
Conclusions
In the last few years, a great revolution from the therapeutic point of view was observed in the setting of ACS, including UA, STE-ACS, and NSTE-ACS. Despite this progress, patients with ACS still have a high risk of recurrent coronary events and death when compared with patients with stable CAD.
In this scenario, statin use during ACS has proven to significantly reduce recurrent CV events and death. Although there is no consensus on the preferable time of administration of statins during ACS [Wright et al. 2011], the evidence from clinical trials and pooled analyses discussed in this review provides substantial support for the institution of an early treatment [Schwartz et al. 2001; Cannon et al. 2004; Lenderink et al. 2006; Angeli et al. 2012].
In this context, a recent meta-analysis of 10 RCTs showed that a very early initiation of treatment provides near-immediate benefit (a reduction of 37% for the risk of all-cause death after the onset of ACS) [Angeli et al. 2012]. In other words, it seems that the earlier the treatment is started after the diagnosis of ACS the greater the benefit that is expected.
The early use of statins during ACS is also supported by the good safety and tolerability profile of statins and by experimental models which analyzed the effects of statins on the vulnerable plaque.
Recent evidence suggests that statins posses several pleiotropic effects besides their lipid-lowering effects: statins have been shown to induce anti-inflammatory effects, modulate endothelium, and inhibit the thrombotic signaling cascade [Mihos et al. 2010]. In conclusion, we believe that treatment with statins can be considered as a useful drug strategy to prevent recurrent CV events and death in the early phase of ACS.
Footnotes
Acknowledgements
The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE) and were fully responsible for all content and editorial decisions, and were involved at all stages of manuscript development. The authors received no compensation related to the development of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare there is no conflict of interest.
