Abstract
Intraaortic balloon pump (IABP) is the most widely used left ventricular support device in a variety of indications. This review focuses on the current literature and discusses the evidence of IABP in ST-elevation myocardial infarction (STEMI) with and without cardiogenic shock. In high-risk STEMI patients without cardiogenic shock several randomized clinical trials have been performed. The majority of the studies could not demonstrate an efficacy benefit for IABP as adjunctive therapy in comparison to standard treatment alone. Hence, recent meta-analyses could not reveal diverging mortality rates at a higher incidence of stroke and major bleedings with IABP use independent of the type of reperfusion therapy. IABP in STEMI patients with cardiogenic shock is recommended according to current American College of Cardiology/American Heart Association (AHA/ACC) and European Society of Cardiology (ESC) guidelines. In recent meta-analyses, IABP in cardiogenic shock complicated by STEMI has been shown to be associated with decreased mortality. However, these beneficial effects are limited to patients treated with thrombolysis, whereas in patients undergoing mechanical revascularization IABP therapy is associated with an increase in mortality. Nevertheless, these data only arise from prospective and retrospective cohort studies, as up to date only one very small randomized clinical trial has been completed. In summary, in high-risk STEMI patients without cardiogenic shock, current data do not support the use of IABP and should only be considered as a standby and bailout strategy if patients develop haemodynamic instability. Current data on IABP in patients with cardiogenic shock complicated by STEMI are scarce and highly limited due to the nonrandomized design of previous trials. However, according to current AHA/ACC and ESC guidelines its use is recommended. Although recent meta-analyses challenge current AHA/ACC/ESC guidelines, adequately powered randomized studies are needed to elucidate the role of IABP in patients with acute myocardial infarction complicated by cardiogenic shock.
Introduction
Intraaortic balloon pump (IABP) has been shown to lead to augmentation of coronary blood flow, unloading of the left ventricle and subsequent increase in myocardial oxygen supply by improving the peak diastolic pressure and lowering the end-systolic pressure by means of diastolic inflation and rapid systolic deflation (Figures 1 and 2) [Kern et al. 1993; Williams et al. 1982]. Despite its favourable haemodynamics, IABP is associated with rare but relevant complications such as major bleedings, stroke, local and systemic infections and vascular complications [Davidson et al. 1998]. Owing to the ease of percutaneous implantation, the relatively low cost and the beneficial haemodynamic effects at a presumably low complication rate, IABP is the most widely used left ventricular assist device since its development in the 1960s. In daily clinical routine IABP is mainly used in patients with acute myocardial infarction complicated by cardiogenic shock. In addition, in patients with acute myocardial infarction at high risk for haemodynamic instability and in elective high-risk patients undergoing percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG), IABP therapy is frequently performed. Apart from a significant variance in indications, previous data on IABP and its outcomes report conflicting results. This review will focus on the current literature on IABP in ST-elevation myocardial infarction (STEMI) patients with and without cardiogenic shock and discuss the existing evidence.
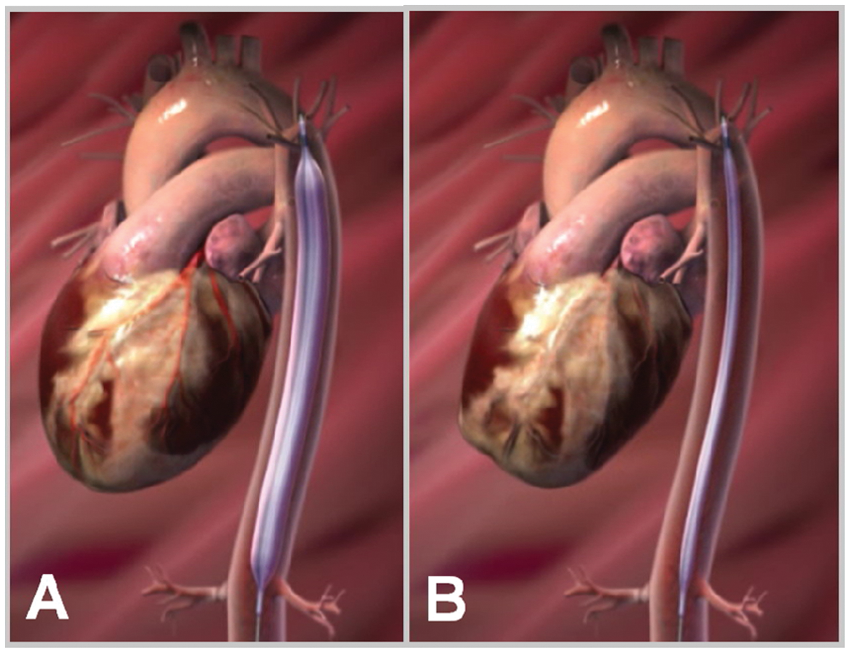
Schematic drawing of an intraaortic balloon pump: inflated during diastole (A) and deflated during systole (B).

Haemodynamic effects of intraaortic balloon pumping on the aortic pressure cycle.
IABP in STEMI without cardiogenic shock
Preclinical animal studies observed an association of IABP with smaller infarct size and larger amount of salvaged myocardium mainly if applied before reperfusion [Achour et al. 2005; Azevedo et al. 2005; LeDoux et al. 2008]. Thus, several randomized clinical trials were performed analyzing the effect of IABP in high-risk STEMI patients without cardiogenic shock (Table 1) [Flaherty et al. 1985; Kono et al. 1996; O’Rourke et al. 1981; Ohman et al. 1994; Ohman et al. 2005; Patel et al. 2011; Stone et al. 1997; van ‘t Hof et al. 1999; Vijayalakshmi et al. 2007]. Irrespective of the variety of definitions of high-risk STEMI patients such as patients with suboptimal PCI results, poor ST-segment resolution, failed thrombolysis, Killip-class ≥1 or large area at risk, the majority of these studies could not demonstrate an efficacy benefit for IABP as adjunctive therapy in comparison to standard treatment alone. Evidently, the standard treatment of STEMI evolved from thrombolysis to primary PCI in the last decades. A study by Ohman and colleagues performed in the pre-PCI era did observe a significantly lower rate of reocclusion of the infarct-related artery during follow up compared with control patients (8%
Randomized controlled trials on IABP in STEMI without cardiogenic shock.

IABP = intraaortic balloon pump, STEMI = ST-elevation myocardial infarction; PCI = percutaneous coronary intervention, < 12/6 h = less than 12/6 h from symptom onset, ΔSTT = ST-segment deviation, TT = thrombolytic therapy, sysRR = systolic blood pressure (mmHg), HR = heart rate, bpm = beats per minute, CAG = coronary angiography, TIMI = thrombolysis in myocardial infarction, LVEF = left ventricular ejection fraction, IRA = infarct related artery, SVG = saphenous vein graft, VT = ventricular tachycardia, CHF = chronic heart failure, MACE = major cardiac events, FUP = follow up, ECG = electrocardiography.
In line with the results of the individual studies, recent meta-analyses could not demonstrate a significant mortality reduction in high-risk STEMI patients treated with or without IABP independent of the type of reperfusion therapy. In addition, no significant effect could be observed concerning left ventricular ejection fraction (LVEF), recurrent ischaemia or reinfarction. Notably, the use of IABP was associated with a significant increase in stroke rate and higher incidence of major bleedings [Bahekar et al. 2011; Sjauw et al. 2009].
Most importantly, current guidelines of the American College of Cardiology and American Heart Association (ACC/AHA) and the European Society of Cardiology (ESC) do not address the topic of IABP in high-risk STEMI patients. In summary, according to current data routine prophylactic IABP use in stable high-risk STEMI-patients is not supported and should only be considered as a standby strategy if patients develop haemodynamic instability.
IABP in STEMI with cardiogenic shock
Despite advances in treatment strategies including aggressive therapeutic interventions such as early revascularization by PCI and/or CABG, mortality of cardiogenic shock complicating STEMI remains high with mortality rates approaching 50% [Goldberg et al. 2009; Thom et al. 2006; Zahn et al. 2001]. Next to an early revascularization strategy and optimal drug therapy including administration of vasopressors and positive inotropic agents, mechanical support is considered as an additional option to achieve initial haemodynamic stabilization and improve clinical outcome. In this setting, IABP is the most often used left ventricular support device. Currently, IABP use in patients with cardiogenic shock complicating acute myocardial infarction is recommended with a class 1B recommendation according to the AHA/ACC guidelines and a class 1C recommendation according to the ESC guidelines [Antman et al. 2008; Van de Werf et al. 2008]. As indicated by the level of evidence, no randomized clinical trial investigating the impact on outcome of IABP use in patients with STEMI and cardiogenic shock has been completed at the time of guideline publication. Thus, this recommendation is based on prospective and retrospective observational studies (Table 2)[Anderson et al. 1997; Barron et al. 2001; Bengtson et al. 1992; Kovack et al. 1997; Moulopoulos et al. 1986; Sanborn et al. 2000; Stomel et al. 1994; Vis et al. 2007a, 2007b; Waksman et al. 1993; Zeymer et al. 2011].
Observational studies on IABP in cardiogenic shock complicated by STEMI.

IABP = intraaortic balloon pump, STEMI = ST-elevation myocardial infarction; PCI = percutaneous coronary intervention, sysRR = systolic blood pressure (mmHg), PCWP = pulmonary capillary wedge pressure, CI = cardiac index, HR = heart rate, bpm = beats per minute.
Recent meta-analyses including these prospective and retrospective cohort studies could demonstrate a mortality reduction using IABP [Bahekar et al. 2011; Sjauw et al. 2009]. However, the beneficial effects of IABP were strongly dependent on the type of reperfusion therapy. The results were mainly driven by the studies performed in the prethrombolytic and thrombolytic era, i.e. the pre-PCI era: in patients undergoing thrombolysis or no reperfusion IABP use resulted in 18% and 29% mortality risk reduction, whereas IABP support in patients undergoing PCI led to a 6% mortality risk increase [Sjauw et al. 2009].
The mortality reduction of IABP in conjunction with thrombolysis might support the hypothesis that IABP and thrombolytic therapy have synergistic effects as IABP may increase the efficacy of thrombolysis by increasing coronary perfusion [Prewitt et al. 1994]. However, according to the results of these meta-analyses, patients treated with IABP were younger and more often of male gender, both baseline characteristics known to be associated with improved outcome. In addition, rescue-PCI or additional CABG was significantly more frequently performed in patients receiving IABP therapy which is also known to be associated with reduced mortality as previously demonstrated in the Should We Emergently Revascularize Occluded Coronaries for Cardiogenic Shock (SHOCK) trial [Hochman et al. 1999]. Here a significant mortality reduction at 6 and 12 months, as well as in long-term follow up after 6 years, was observed with an early revascularization-based management. Thus, these factors might explain the poor outcome in patients treated without IABP after thrombolysis. However, as recommended by current guidelines of the ACC/AHA and ESC, early mechanical revascularization is of paramount importance in the therapy of cardiogenic shock complicated by STEMI, whereas initial stabilization including thrombolysis with or without additional IABP should play only a secondary role.
The observed increase in mortality in patients treated by IABP in addition to early PCI or CABG was mainly influenced by the National Registry of Myocardial Infarction-2 (NRMI-2) registry results [Barron et al. 2001]. In the NRMI-2 cardiogenic shock cohort, IABP therapy was independently associated with mortality after multivariate adjustment for age, several clinical risk factors and revascularization therapy. Nevertheless, selection bias cannot be ruled out due to the nonrandomized design of the studies. Thus, one might argue that in general IABP might have been more frequently used in patients with extremely poor prognosis. In line with NRMI-2 results, data from the Amsterdam Medical Center Cardiogenic Shock (AMC CS) registry found IABP use to be associated with higher mortality rates [Vis et al. 2007a, 2007b], whereas only one other registry reported mortality rates of 38% with
In summary, the body of evidence to support or dismiss IABP in patients with cardiogenic shock complicating STEMI is scarce due to the nonrandomized design of previous studies and their inconclusive results with respect to clinical endpoints. Thus, further properly powered, randomized, clinical trials are needed to analyze the impact of IABP on outcome in patients with cardiogenic shock in the setting of acute myocardial infarction and early revascularization. Based on a small randomized pilot study [Prondzinsky et al. 2010], the prospective, randomized, open-label, multicentre, controlled Intraaortic Balloon Pump in cardiogenic shock 2 (IABP-SHOCK II [ClinicalTrials.gov identifier: NCT00491036]) trial is ongoing and will hopefully give us the answer to whether IABP treatment is beneficial for the treatment of cardiogenic shock in addition to PCI.
Discussion
Since its development in the 1960s, IABP is the most commonly used left ventricular support device in clinical routine. By means of diastolic inflation and rapid systolic deflation in the aorta, the peak diastolic pressure is augmented and end-systolic pressures are lowered. This results in unloading of the left ventricle, augmentation of coronary perfusion and reduction in myocardial oxygen demand [Kern et al. 1993; Williams et al. 1982]. Nevertheless, effects on cardiac output are limited [Scheidt et al. 1973]. Despite the potential beneficial haemodynamic effects, the use of IABP is associated with rare but relevant complications such as major bleedings, stroke, local and systemic infections and vascular complications [Davidson et al. 1998]. To reduce mechanical vascular complications such as limb ischaemia, distal embolization or dissection, techniques for sheathless insertion and catheters with smaller diameters were developed. Nevertheless, the clinical benefit of these new developments could not been demonstrated convincingly so far [Patel et al. 1995]. In addition to these local complications, a higher rate of stroke has been reported in patients treated with IABP. This might be explained in part by haemorrhagic events due to the need of full systemic heparinization, or by ischaemic events induced by peripheral embolization. Apart from intracranial haemorrhagic events, IABP use has been shown to be associated with overall major bleedings which again can in part be attributed to the need for full systemic heparinization and the need for prolonged arterial access. Evidently, the complications of full systemic heparinization might be even more pronounced after administration of glycoprotein IIb/IIIa-inhibitors in the setting of high-risk acute myocardial infarction. Apart from the adverse effect on clinical outcome induced by major bleedings itself, they further trigger transfusion which might increase the mortality risk in patients with acute coronary syndromes [Rao et al. 2004]. Evidently, the use of IABP may also lead to local and systemic infections. Although these infections may often be of minor clinical relevance, in patients with pronounced comorbidities they potentially influence clinical outcome and might lead to a further progress of the systemic disease of cardiogenic shock [Stegman et al. 2012].
Certainly, the potential benefits of IABP have to be weighed against its complications. This is especially important in STEMI patients without cardiogenic shock, since in this setting IABP is used as a prophylactic intervention. Although individual studies did report a beneficial impact of IABP use in high-risk STEMI patients reperfused by thrombolysis [Kono et al. 1996; O’Rourke et al. 1981], trials including patients reperfused by primary PCI could not observe any improvement in outcome due to IABP [Flaherty et al. 1985; Ohman et al. 2005; Patel et al. 2011; Stone et al. 1997; van ‘t Hof et al. 1999; Vijayalakshmi et al. 2007]. In line with these results, recent meta-analyses did not observe a 30-day survival benefit or an improvement in LVEF at increased rates of stroke and major bleedings independent of the type of reperfusion therapy [Bahekar et al. 2011; Sjauw et al. 2009]. Thus, the significant higher complication rates were not outweighed by any clinical benefit. Notably, mortality after STEMI in patients without cardiogenic shock could be reduced to <10% in the last decades [Schiele et al. 2010]. This is mainly attributable to optimal interventional and drug treatment in the acute and subacute phase. Thus, much less invasive procedures such as manual thrombectomy or administration of glycoprotein-IIb/IIIa inhibitors or bivalirudin further challenge invasive therapeutic strategies such as IABP use.
In contrast to the clinical outcome in STEMI patients with haemodynamic stability, mortality of cardiogenic shock complicating STEMI remains high with rates of approximately 50% [Goldberg et al. 2009; Thom et al. 2006; Zahn et al. 2001]. The current concept of cardiogenic shock implies the development of a shock spiral induced by progressive left ventricular dysfunction leading to impaired peripheral perfusion and hypoxemia. Apart from the direct haemodynamic effects, the systemic inflammation response syndrome (SIRS) is further promoted leading to paradoxical reduced systemic vascular resistance [Reynolds and Hochman, 2008; Thiele et al. 2010]. In theory IABP use might beneficially influence haemodynamics and improve myocardial recovery which might translate into more rapid haemodynamic stabilization and disruption of the shock spiral. However, by means of insertion of a foreign body with relatively large intravascular surface area, SIRS and thus the shock spiral may further be promoted. In addition, bleeding complications and subsequent transfusions both also negatively contribute to inflammation and subsequently further trigger the shock spiral [Rao et al. 2004]. Local or systemic infections also negatively influence the complex pathophysiology of cardiogenic shock despite its potential limited relevance in stable patients. Although cardiogenic shock can be understood as a systemic illness, early revascularization is of paramount importance and can be understood as the cornerstone for the treatment of cardiogenic shock complicating acute myocardial infarction: as demonstrated by the SHOCK trial, early mechanical revascularization leads to a significant decrease in mortality in comparison to haemodynamic stabilization and optimal medical therapy including thrombolysis [Hochman et al. 1999]. These findings help to interpret the results of current meta-analyses: in patients undergoing thrombolysis or no reperfusion which represents an inferior treatment strategy, IABP use resulted in a significant mortality risk reduction, whereas IABP support in patients undergoing PCI led to a significant mortality risk increase [Bahekar et al. 2011; Sjauw et al. 2009]. However, these data are solely based on registries as up to date only one small randomized clinical trial investigating the impact of IABP use in patients with STEMI and cardiogenic shock has been completed. Certainly registries suffer from multiple potential sources of bias and confounding, thus data arising from such need to be interpreted with caution. Nevertheless, current guidelines of the AHA/ACC and ESC highly recommend IABP use in patients with cardiogenic shock complicated by STEMI (class 1B and 1C recommendation, respectively) [Antman et al. 2008; Van de Werf et al. 2008]. In line with the international ESC/AHA/ACC guidelines, further national directives such as the German–Austrian guidelines also recommend IABP use in patients treated with thrombolysis and clearly state that IABP may be used in patients treated by primary PCI despite lacking evidence [Werdan et al. 2011]. In spite of these recommendations IABP is currently only used in one quarter of patients with cardiogenic shock complicated by STEMI [Thom et al. 2006; Zeymer et al. 2011], which might be in part explained by the lack of evidence and reimbursement policies. The IABP-SHOCK II trial, the first adequately powered randomized controlled study, will further elucidate the current role of IABP in cardiogenic shock. Finally, if a positive impact of IABP on outcome should eventually be demonstrated further large-scale clinical trials are needed to compare the potential beneficial effects of IABP to other left ventricular assist devices. However, data from several small trials could not indicate superiority of left ventricular assist devices over IABP [Arias et al. 2005; Burkhoff et al. 2006; Seyfarth et al. 2008; Thiele et al. 2005; Unverzagt et al. 2011].
Conclusion
In summary, according to current data IABP use in stable high-risk STEMI patients cannot be recommended and should only be considered as a standby and bailout strategy if patients develop haemodynamic instability. In contrast, in patients with cardiogenic shock complicated by STEMI current guidelines strongly recommend the use of IABP. Although data are scarce, based on registries only and challenged by recent meta-analyses, clinical practice should not be changed until further evidence from randomized, controlled, clinical trials is available.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Holger Thiele has received research funding and speaker honoraria from Maquet Cardiopulmonary AG, Hirrlingen, Germany and Teleflex Medical, Everett, MA, USA.
