Abstract
Coronary vasospasm is an unusual cause of angina and myocardial ischemia, with the potential to provoke acute myocardial infarction, malignant cardiac arrhythmias, and sudden cardiac death. The diagnosis is largely clinical and requires a high index of suspicion. Provocation studies are rarely performed due to the risks of the procedure and the relatively low incidence of disease. A subset of patients does not respond to conventional medical therapy and a paucity of evidence exists to guide therapy. While generally believed a multifocal phenomenon, there have been reports of successful treatment of focal, refractory vasospasm with coronary stent implantation. Furthermore, consideration of an implantable cardioverter defibrillator is warranted when vasospasm is complicated by lethal ventricular arrhythmias.
Keywords
Introduction
Though most patients with coronary vasospasm respond favorably to treatment with calcium channel blockers and long-acting nitrates, a subset remains symptomatic despite appropriate medical therapy. We present a case of refractory coronary vasospasm with associated polymorphic ventricular tachycardia in a woman who was successfully treated with percutaneous coronary intervention (PCI) and an implantable cardioverter defibrillator, followed by a review of the literature.
Case presentation
A 60-year-old woman with coronary artery disease, ongoing tobacco abuse and well controlled hyperlipidemia presented to the emergency department following a syncopal episode preceded by chest pain. She reported a 2-week history of brief, intermittent substernal chest pain that radiated to the neck. The pain occurred during the day and awakened her from sleep. Her chest pain was evaluated 1 week earlier with pharmacological myocardial perfusion imaging and ambulatory electrocardiographic monitoring, both of which were unremarkable. She had undergone stenting of the right coronary artery 1 year earlier for chronic angina. Medications included atorvastatin and aspirin.
On arrival, she appeared comfortable and her vital signs, physical exam, and laboratory values, including troponin, were normal. An electrocardiogram (ECG) was normal (Figure 1), but 40 min later she developed acute severe chest pain with significant ST elevation in lead aVF that resolved with sublingual nitroglycerin (Figure 2). Emergent coronary angiography and intravascular ultrasound demonstrated nonobstructive atherosclerotic disease of the proximal and mid-right coronary artery with a patent proximal stent (Figure 3). Myocardial infarction was ruled out and, suspecting coronary vasospasm, she was discharged home the next day with the addition of amlodipine 5 mg daily and sublingual nitroglycerin to her current medications.

Electrocardiogram obtained on presentation without any ST segment abnormality.
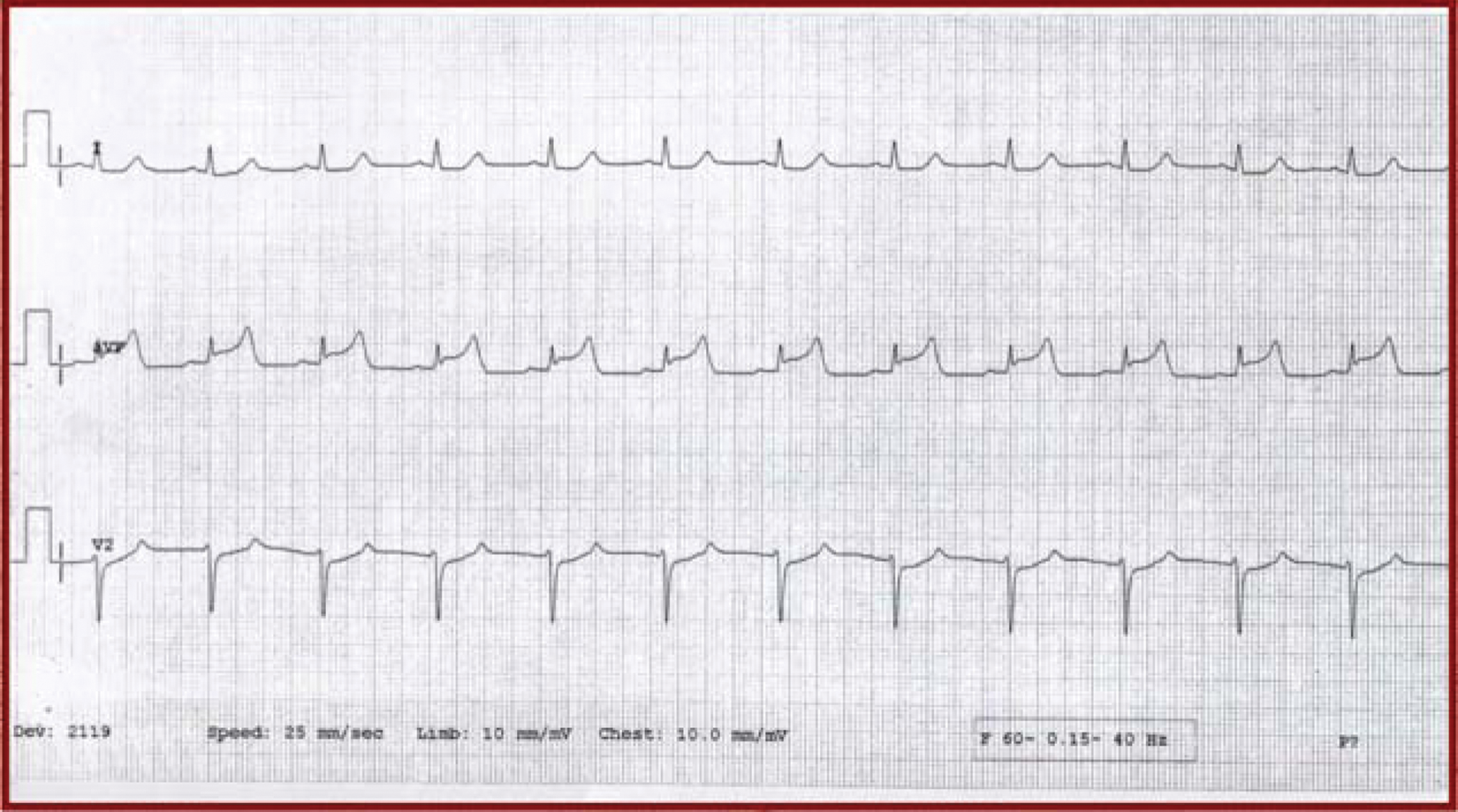
A rhythm strip obtained during an acute episode of angina, with ST segment elevation in lead aVF.

Cardiac catheterization demonstrating diffuse, nonobstructive disease of the proximal and mid right coronary artery with a patent proximal stent (arrow).
Twelve hours later, the patient experienced a second syncopal event. On presentation to the emergency department there were no ischemic ECG changes; she was admitted for observation and treated with amlodipine 5 mg daily and topical nitrates. The next morning, the patient’s chest pain recurred; telemetry demonstrated inferior ST elevation followed by nonsustained polymorphic ventricular tachycardia. Emergent coronary angiography demonstrated no significant changes from the prior study. Two overlapping drug-eluting stents were placed in the diseased segment of her proximal and mid right coronary artery (Figure 4). An implantable cardioverter defibrillator (ICD) was placed 4 days later.

Angiography after drug-eluting stent placement to the proximal and mid right coronary artery.
Background
In 1959 Prinzmetal first described a variant form of angina in patients with ST segment elevation seen on the resting ECG [Prinzmetal et al. 1959]. Confirmation of pathologic coronary vasospasm as the mechanism for Prinzmetal’s clinical observation occurred in the early 1970s when it was witnessed during cardiac catheterization. Worldwide, the incidence of clinically recognizable coronary vasospasm is waning, due in part to the widespread use of calcium channel blockers and nitrates, declining tobacco use, and the general lack of confirmatory provocative testing in North America [Yasue et al. 2008]. Historically, provocative testing has shown the incidence to be approximately 50% in patients presenting with presumed ischemic heart disease without a culprit lesion seen on angiography [Ong et al. 2008]. Racial differences exist, with a threefold increased incidence in Japanese populations [Kusama et al. 2011].
Risk factors
Among the traditional risk factors for coronary disease, cigarette smoking is most commonly associated with coronary vasospasm [Yasue et al. 2008]. Despite conventional wisdom to the contrary, coronary vasospasm occurs more commonly in men, though arrhythmogenic complications appear equally amongst men and women. There is no association with significant (>50%) coronary stenosis or previous myocardial infarction [Takagi et al. 2011]. Other risk factors include elevated high-sensitivity C reactive protein, increasing age and remnant lipoprotein levels [Yasue et al. 2008]. Associated conditions include Raynaud’s phenomenon, migraine headaches, obstructive sleep apnea, and a low glomerular filtration rate [Nakamura et al. 2000; Tamura et al. 2010; Koga et al. 2011]. Precipitants are numerous, including but not limited to the use of cocaine and other illicit drugs, alcohol, and early morning exercise [Yasue et al. 2008].
Mechanism of disease
The pathogenesis of coronary vasospasm is complex and not fully understood, but prominently features endothelial and autonomic dysfunction, oxidative stress, and hypercontractility of coronary smooth muscle. A circadian component exists, with most attacks occurring at night or during the early morning hours. Endothelium-dependent vasodilators that work via nitric oxide (NO), including acetylcholine and ergonovine among others, paradoxically cause vasoconstriction, possibly due to a lack of NO synthase in patients with coronary vasospasm [Yasue et al. 2008]. Oxidative stress creates free radicals that damage the endothelium and degrade NO. Other proposed mechanisms include chronic low-grade inflammation and hypomagnesemia [Kusama et al. 2011]. An emerging pathway involves Rho-kinase, a modulator of myosin light chain phosphorylation involved in vascular smooth muscle contraction and atherosclerosis [Shimokawa, 2002]. In addition, polymorphisms of the endothelial NO synthase gene (e-NOS), paraoxonase gene, and the Rho-family GTPases have been implicated in Japanese populations [Kusama et al. 2011; Takefuji et al. 2010].
Coronary vasospasm traditionally manifests in segmental areas of nonobstructive atherosclerosis. Even in angiographically normal vessels, vasospasm was associated with occult atherosclerosis when assessed by intravascular ultrasound [Khatri et al. 2002]. When compared with patients with unstable angina, the atherosclerotic lesions involved had less plaque burden and calcification, but more constrictive remodeling [Hong et al. 2010]. In many patients, vasospasm involves multiple coronary vessels and has been shown to migrate within the vasculature over time [Yasue et al. 2008].
The pathogenesis of ventricular arrhythmias attributed to coronary vasospasm is thought to involve both ischemia from transient vessel occlusion and subsequent reperfusion injury. Arrhythmias secondary to vessel occlusion occur after a delay of several minutes, whereas reperfusion arrhythmias tend to occur immediately after the re-establishment of blood flow and positively correlate with a prolonged period of ischemia [Previtali et al. 1983]. The degree of ischemia, as measured by the amplitude of the ST segment deviation, correlates most closely with the propagation of ventricular arrhythmias, with ST segment elevation indicating more profound ischemia than ST segment depression, whereas the extent of coronary atherosclerosis and left ventricular function have not been associated with arrhythmia propagation. ST segment elevation not only reflects the degree of ischemia, but also the change in intracellular potential and underlying metabolic derangements, including increased lactic acid and decreased adenosine triphosphate, all of which may contribute towards arrhythmia induction [Kerin et al. 1979].
ECG changes, arrhythmias, and clinical events
Manifestations of coronary vasospasm vary, ranging from silent ischemia to angina at rest, various arrhythmias, infarction, or sudden death, with a 50-fold increase in risk of out-of-hospital cardiac arrest (OHCA) [Takagi et al. 2011]. The classic ECG finding is transient ST elevation in the leads corresponding to the myocardial territory affected by the artery in spasm. Involvement of the right coronary artery is more apt to trigger bradyarrhythmias, while the spasm of multiple vessels or the left anterior descending is associated with ventricular tachyarrhythmias. Other ECG findings may include ST depression, widening of the R wave, T-wave abnormalities, or reverse U waves [Yasue et al. 2008]. In patients presenting with vasospasm-provoked acute coronary syndrome, the most common arrhythmias were complete atrioventricular block, ventricular fibrillation, and sinus arrest with junctional escape rhythm [Hung et al. 2007]. Ventricular fibrillation and ventricular tachycardia are the most common arrhythmias present at the time of vasospasm-induced sudden cardiac death [Myerburg et al. 1992]. Importantly, the majority of ischemic episodes occur without symptoms [Kusama et al. 2011].
Diagnosis
Definitive diagnosis of coronary vasospasm occurs during catheterization, with provocation via acetycholine, ergonovine, or hyperventilation [Yasue et al. 2008]. The acetylcholine challenge test is highly efficacious, with 90% sensitivity for detecting coronary vasospasm [Okumura et al. 1988]. Acetylcholine testing is safe, with serious adverse reactions such as sustained ventricular tachycardia or shock, occurring in less than 1% of patients tested [Sueda et al. 2000]. Similar results were seen with ergonovine testing, with serious nonfatal adverse reactions occurring in less than 1% of patients tested [Bertrand et al. 1982]. However, death due to refractory vasospasm has been reported with ergonovine testing in other analysis and resultantly provocation testing is only undertaken in specialized centers [Buxton et al. 1980]. In the future, Rho-kinase activity may serve as a noninvasive biomarker of coronary vasospasm [Kikuchi et al. 2011]. Today, coronary vasospasm is often a clinical diagnosis, seen in patients with an episodic history of nonexertional chest pain, associated ST segment elevation, and nonobstructive coronary disease seen on catheterization.
Medical therapy
Standard treatment of coronary vasospasm includes calcium channel blockers, long-acting nitrates and smoking cessation if applicable. Both dihydropyridine and nondihydropyridine calcium channel blockers have been used, and there may also be a role for statins and angiotensin-converting enzyme (ACE) inhibitors, given their effects on endothelial function [Yasue et al. 2008]. While all calcium channel blockers are effective in reducing major cardiovascular events in patients with coronary vasospasm, benidipine, a relatively new, more cardio-selective calcium channel blocker with anti-atherosclerotic properties not available in the USA, has shown greater efficacy than amlodipine or diltiazem in preventing such events [Fukumoto et al. 2008]. Vitamin E, magnesium, and estrogen in postmenopausal women may also be useful adjunctive treatments [Yasue et al. 2008]. The Rho-kinase inhibitor fasudil has been shown to suppress acetylcholine-induced coronary vasospasm and resultant ischemia, thus becoming a potential future therapy [Masumoto et al. 2002]. β Blockade is largely contraindicated due to prolonged vasospasm-induced ischemia, presumably from unopposed α-mediated constriction within the coronary vasculature [Yasue et al. 2008].
Therapy for refractory coronary vasospasm
Resistance to conventional medical therapy occurs frequently, with estimates approaching 20% of appropriately treated patients with coronary vasospasm [Bott-Silverman and Heupler, 1983]. Unfortunately, electrophysiological testing to identify those at highest risk for sudden cardiac death has been unreliable and cannot be used to guide therapy [Myerburg et al. 1992]. Coronary artery bypass grafting and balloon angioplasty have been unsuccessful; coronary stent implantation is sometimes used in practice but is controversial [Khatri et al. 2002]. In addition, the endothelin antagonist bosentan has demonstrated efficacy in a single case report [Krishnan et al. 2010].
Coronary stent placement
While coronary vasospasm is generally believed to be a multifocal phenomenon, there have been reports of successful treatment of focal, refractory vasospasm with coronary stent implantation. In a series of nine patients, stent placement relieved symptoms in two-thirds, including silent ischemia by Holter monitoring [Gaspardone et al. 1999]. In a subsequent series, PCI alleviated symptoms of ischemia in five of nine patients with refractory coronary vasospasm [Khatri et al. 2002]. In addition, numerous case reports have detailed symptomatic improvement with PCI, including one with successful stenting of 70 mm in the right coronary artery [Burns et al. 2007].
Caution must be advised, however, as stenting is not the ultimate panacea for refractory coronary vasospasm. Focal areas of spasm have been shown to occur in different segments of the same vessel or in different coronary vessels altogether in up to 75% of cases, thus limiting the efficacy of coronary stenting [Tanabe et al. 2002]. Furthermore, diffuse coronary vasospasm has been reported after drug-eluting stent placement, though its pathophysiology is not yet understood [Brott et al. 2006].
Implantable cardioverter-defibrillators for secondary prevention
With an increased risk of sudden cardiac death, patients with coronary vasospasm are potential candidates for ICD placement, but current guidelines do not address their use. ICD use was first described in two patients with coronary vasospasm-induced ventricular arrhythmias in 1994 [Lacroix et al. 1994]. Later, Meisel and colleagues demonstrated appropriate ICD discharge in four of seven patients with coronary vasospasm and associated ventricular fibrillation [Meisel et al. 2002] and more recently this was demonstrated by Takagi and colleagues in 2 of 14 patients who underwent ICD placement for vasospasm-induced OHCA [Takagi et al. 2011].
Prognosis
Patients with coronary vasospasm have a favorable prognosis when maintained on calcium channel blockers, with 5-year survival estimates as high as 98% [Yasue et al. 2008; Takagi et al. 2011]. Most cardiac events occur within 1 month of symptom onset [Kusama et al. 2011]. Notably, the risk of cardiac death or nonfatal myocardial infarction increases tenfold when medical therapy is reduced or withdrawn. Even among those resuscitated from OHCA, a 97% 5-year survival is seen, though cardiac event-free survival is significantly lower [Takagi et al. 2011].
Discussion
The diagnosis of coronary vasospasm is often clinical and carries a markedly increased risk for sudden cardiac death. Accordingly, misdiagnosis may result in lethal consequences, particularly in a patient who presents with chest pain and syncope. Though generally responsive to medical therapy, guidelines do not exist for those patients with refractory coronary vasospasm and their management remains problematic. Though nitrates, calcium channel blockers, and abstinence from smoking remain cornerstones of therapy, symptoms may persist and consideration of device therapy (i.e. PCI and ICD) may be warranted, particularly in the setting of syncope due to ventricular arrhythmia. When possible, aggressive medical management should be pursued as a focal area of spasm is required for PCI to be effective, and even then the site of spasm has been known to wander. In this case, however, the patient’s rapid escalation of symptoms and potentially lethal arrhythmic complications precluded the attempt at medical optimization, which would have included maximizing her dose of a calcium channel blocker and long-acting nitrate, along with the consideration of magnesium, vitamin E, or estrogen replacement therapy. With relatively little known about the incidence of spasm associated with drug-eluting stent placement, it was the opinion of the treating interventional cardiologist (JAM) that the benefits of drug-eluting stent placement, particularly the decreased incidence of restenosis and need for target lesion revascularization, outweighed the risks in a relatively young, functional woman with known coronary artery disease.
As demonstrated in this case and in others, stenting for coronary vasospasm may demonstrate absolute efficacy when the area of refractory spasm is focal and unchanging. Furthermore, appropriate ICD discharge has repeatedly been observed amongst patients with spasm-induced ventricular tachyarrhythmias, justifying their use in selective patients. Randomized controlled trials would help to elucidate the utility and cost effectiveness of these advanced measures, but given the relatively low prevalence of coronary vasospasm, such trials are unlikely and will force clinicians to continue to rely on their clinical judgment and case reports to guide treatment.
Case follow up
The patient quit smoking at the time of ICD placement. Repeat catheterization 27 months later for an episode of angina demonstrated 30% in-stent restenosis to her proximal right coronary stent and was otherwise unchanged. At 3-year follow up she was free of angina and without further episodes of syncope. No defibrillator shocks have been delivered or significant arrhythmias detected. Intolerant of ACE inhibitors, her current medical therapy includes aspirin, atorvastatin, amlodipine, isosorbide dinitrate, and sublingual nitroglycerin as needed.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Potential conflicts of interest by Dr John McPherson include acting consultant to Abbot Vascular, Gilead, and CardioDx as well as a research grant from CardioDx. To the best of our knowledge, no further conflicts of interest, financial or other, exists for the remaining authors.
