Abstract
Keywords
Introduction
Among the various biological rhythms contributing to normal bodily functions, disturbances in the 24 h day/night activity/rest cycle is considered a key chronobiologic factor predisposing to many medical conditions. In most healthy people, the blood pressure biological rhythm is characterized by people exhibiting the highest levels of arterial pressure from late morning to middle afternoon. A physiologic decline in blood pressure of at least 10% from daytime values occurs during sleep. The 24 h mean level and amplitude of the blood pressure rhythm may be altered in people with hypertension, including suppression of the normal decreases in arterial pressure during sleep. The loss of a nocturnal fall in blood pressure is associated with increased risk of sleep apnea, chronic kidney disease, insulin resistance, atherosclerosis, stroke, left ventricular hypertrophy, congestive heart failure, and myocardial infarction [Cuspidi et al. 2004; Della et al. 2005; Fagard et al. 2009; Higashi et al. 2002; Ingelsson et al. 2006; Ohkubo et al. 2002].
Although the brain regulates the mechanisms affecting the ‘biological clocks’, the neuronal pathways determining the intrinsic activity of these clocks can be altered by extrinsic hormonal and environmental factors. In human hypertension, alterations in sympathetic nerve activity and the renin–angiotensin system (RAS) contribute to loss of the normal pattern of the blood pressure fluctuations. An absence of a blood pressure fall during sleep (nondippers) has been recorded in African-American people with hypertension [Viera et al. 2011] while the presence of nondipping hypertension has been shown to correlate with target organ damage and increased cardiovascular morbidity and mortality [Cuspidi et al. 2004; Fagard et al. 2009; Hermida et al. 2010; Ohkubo et al. 2002]. Early studies in experimental models of hypertension demonstrated that sinoaortic denervation in dogs was associated with increases in nocturnal blood pressure [Ferrario et al. 1969] that in baroreceptor- denervated cats were linked to loss of the carotic chemoreceptor [Baccelli et al. 1976; Del et al. 1985; Guazzi et al. 1968; Kumazawa et al. 1969]. The neurogenic form of experimental hypertension induced by chronic administration of subpressor doses of angiotensin II (Ang II) in the dog was also associated with increased blood pressure variability and episodic rises in arterial pressure during sleep [McCubbin et al. 1965]. Although the data implicate an interaction among baroreceptor reflexes, Ang II, and sympathetic nerve activity in accounting for the disruption of the blood pressure circadian rhythm, limited information exists as to whether blockade of Ang II activity is associated with restoration of the alterations in the blood pressure rhythm.
In rodents, as in humans, the blood pressure circadian profile correlates with the activity periods but the peak changes in blood pressure and heart rate in this species occur at night given the nocturnal behavior and feeding characteristics of these animals. Studies in a transgenic model of renin-dependent hypertension created by insertion of the mouse Ren-2 gene into the rat genome (the [mRen-2]27 transgenic hypertensive rat) [Lee et al. 1996] showed a reversal of the blood pressure circadian rhythm characterized by higher blood pressure during the day compared with the night [Lemmer et al. 1993, 2005]. The inversion of the blood pressure circadian rhythm was associated with maintenance of higher heart rates and locomotive activity at night [Lemmer et al. 1993, 2005]. The dysregulation of the blood pressure circadian rhythm in these m[Ren2]27 transgenic hypertensive rats suggest a critical role of tissue RAS in affecting the neurohormonal mechanisms of the brain biological clock. The brain expresses all of the components of RAS, and Ang II exists within the suprachiasmatic and hypothalamic nuclei containing the central pacemaker for the biological clocks [Moriguchi et al. 1994a, 1994b; Senanayake et al. 1994; Thomas et al. 2004a, 2004b]. Thomas and colleagues concluded that the presence of Ang II immunoreactivity at the luminal face of brain suprachiasmatic parenchyma blood vessels in rats provides a route for actions of circulating Ang II to influence neuronal circuits regulating biological rhythms [Thomas et al. 2004a, 2004b]. The existence of sites where the blood–brain barrier is permeable to the actions of Ang II has been demonstrated previously (see Ferrario and colleagues for a review) [Ferrario et al. 1972, 1979].
The introduction of an orally active direct renin inhibitor for the treatment of hypertension [Ferrario, 2010; Hollenberg, 2002; Ichihara et al. 2010] may confine the increase in circulating renin and its activity in response to monotherapy with an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II type 1 (AT1) receptor blocker. An additional benefit of renin inhibition results from the blunting of the increase in circulating Ang II due to monotherapy with an AT1 receptor blocker which reportedly limits the blood pressure lowering effects of these antihypertensive agents. The Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial (ONTARGET) study revealed that a combination of an ACE inhibitor and AT1 receptor blocker is not superior to monotherapy of each of them in reducing blood pressure and in preventing organ damage [Yusuf et al. 2008]. Therefore, it is important to find other effective combinations of RAS blockade.
We developed a congenic model of mRen2.Lewis hypertensive rats, a strain which, derived from the original parent m[Ren2]27 transgenic hypertensive rats, does not share in the genetic variability found in the parent hypertensive rats originally outbred from Sprague Dawley and Wistar Kyoto rats. We reported that the mRen2.Lewis rat exhibits hypertension and target organ injuries equivalent to those found in the original m[Ren2]27 transgenic hypertensive strain [Chappell et al. 2003; Jessup et al. 2006, 2009]. In this study, we describe the effects of combined direct renin inhibition and AT1 receptor blockade in the regulation of the blood pressure oscillations and the levels of Ang II in the blood and kidney of this new model of renin-dependent hypertension.
Methods
Animals
Experiments were conducted in 29 male mRen2.Lewis rats that were 10 weeks of age obtained from the Hypertension and Vascular Research Center Congenic colony of Wake Forest University. This congenic strain was developed by our program from the backcross of the original m[Ren2]27 transgenic hypertensive rat with normotensive Lewis rats obtained from Charles River (Wilmington, MA, USA) [Groban et al. 2008; Jessup et al. 2010]. All animal procedures were performed in accordance with National Institutes of Health (NIH) guidelines and were approved by the Wake Forest University animal care and use committee. Rats were housed in individual cages under a 12 h light (6:00–18:00), 12 h dark (18:00–6:00) cycle, at a constant humidity and temperature, with free access to standard laboratory rat chaw and drinking water.
Arterial pressure, heart rate and locomotive activity measurements
Systolic blood pressure, diastolic blood pressure, mean arterial pressure (MAP), pulse pressure, heart rate, and locomotive activity were assessed by chronically implanted telemetry probes in conscious rats moving freely in their home cages. Telemetry probes were inserted at 10 weeks of age, as described previously [Schnell and Wood, 1993]. Briefly, pressure transmitters were implanted into the peritoneal cavity under aseptic conditions and anesthesia (inhalation of 2% isoflurane). The sensor catheter was placed in the aorta below the renal artery pointing upstream. Before surgery rats were administered 150 mg/kg ampicillin, 0.05 mg/kg buprenorphine subcutaneously, and 0.3 mg/kg atropine intramuscularly. Rats were allowed to recover for 2 weeks prior to the start of experimentation. Rats were then transferred to the quiet telemetry monitoring room where they remained free to move around. Three days of control measurements (C1−C3) were followed by 14 days of treatment (T1−T14). During this period, blood pressure, heart rate and locomotive activity were measured and recorded every 15 min throughout the 24 h cycle.
Telemetry data were recorded and digitalized using the Dataquest A.R.T. System (version 4.3; Data Sciences International, St Paul, MN, USA). Each animal was sampled for 10 s at 15 min intervals for a period of 17 days. All of the recordings were averaged per 12 h and per 24 h. Baseline values were calculated using the data from C1 to C3. Post-treatment values were calculated using the data from T12 to T14. The changes in MAP were calculated as the difference between averages of C1−C3 and T12−T14. Awake/asleep ratio [O’Brien et al. 1988] was defined as the percentage decrease in MAP during the hours of daytime rest (6:00–18:00) relative to the mean MAP obtained during the hours of night time activity (18:00–6:00) and calculated as (awake MAP mean – asleep MAP mean) / (awake MAP mean) x 100.
Treatment protocol
Rats were randomized into three groups at 12 weeks of age: vehicle group, monotherapy with valsartan (30 mg/kg/day, orally, via drinking water); and combination therapy with valsartan (30 mg/kg/day) and the direct renin inhibitor (aliskiren fumarate, 50 mg/kg/day, subcutaneously). Aliskiren was delivered by osmotic mini-pumps (ALZET® model 2M2L; DURECT Corp., Cupertino, CA, USA) implanted in anesthetized rats (inhalation anesthesia, 2% isoflurane) under aseptic conditions on day T1. The pumps containing saline as vehicle were implanted in the vehicle and valsartan groups. Rats were put in metabolic cages during C2−C3 and T13−T14 to collect urine. Urine was collected at day C3 and T14. After 2 weeks of treatment, animals were euthanized by decapitation, and trunk blood and kidney cortex were collected and processed as previously described [Ferrario et al. 2005a, 2005b; Jessup et al. 2006]. The dose of aliskiren employed in these experiments has previously been shown to reverse hypertension and cardiac hypertrophy in m[Ren2]27 transgenic hypertensive rats [Habibi et al. 2008; Whaley-Connell et al. 2008, 2010, 2011]. Valsartan and aliskiren were a kind gift from Novartis, Inc.
Biochemistry
Plasma renin activity (PRA), defined as the rate of angiotensin I (Ang I) generation from endogenous substrate, was measured in incubated plasma treated with ethylenediaminetetraacetic acid (EDTA) and phenylmethylsulfonyl fluoride (PMSF) to prevent the degradation of the generated peptide. The Ang I was quantified by RIA (Diosarin Corp., Stillwater, MN, USA). Plasma renin concentration (PRC) was defined as the rate of Ang I generation from renin in the sample incubated at pH 6.5 for 90 min with excess exogenous substrate obtained from nephrectomized rat plasma. The Ang I generated in the sample was quantified by RIA (Diosarin Corp). Ang II levels in plasma and renal tissue were measured by RIA as described elsewhere [Ferrario et al. 2005a, 2005b; Jessup et al. 2006]. The minimum detectable level of the Ang II assay was 0.76 fmol/mL. The intra- and interassay coefficients of variability for Ang II were 12% and 22%, respectively.
Statistical analysis
All data are expressed as mean ± standard error of the mean (SEM). Laboratory data were analyzed by one-way analysis of variance (ANOVA) followed by the Tukey’s post hoc test. Changes in MAP, heart rate and awake/asleep ratio were analyzed by two-way ANOVA followed by the Bonferroni post test. Other hemodynamic data were analyzed by one-way ANOVA followed by the Tukey’s post hoc test. Values of p < 0.05 were considered statistically significant.
Results
Hemodynamics
Hemodynamic variables at baseline in each of the treatment groups are presented in Table 1. There are no significant differences in the MAP, systolic and diastolic blood pressure, pulse pressure, and heart rate among the three groups prior to initiation of the treatment regimens. Figure 1(a–c) shows the circadian pattern of the MAP, heart rate, and locomotive activity in all mRen2.Lewis hypertensive rats prior to initiation of treatments (C3). Heart rate and locomotive activity showed normal circadian pattern associated with activity levels (higher at night time and lower in the daytime) [Figure 1(b, c)]. However, MAP showed an inverse circadian pattern, higher in the daytime and lower at night time in all congenic mRen2.Lewis hypertensive rats [Figure 1(a)]. Figure 2(a) shows the average 24 h MAP at baseline (C1−C3) and at the end of the study (T12−T14) in mRen2.Lewis rats medicated with vehicle, valsartan, or combination therapy. There were no significant differences in 24 h MAP among the three groups at baseline. However, after treatment, MAP was reduced by valsartan (107 ± 3 mmHg versus 177 ± 3 mmHg in the vehicle, p < 0.001) and further reduced in rats receiving the combination therapy (91 ± 1 mmHg, < 0.001).
Baseline hemodynamic values prior to commencement of treatment in mRen2.Lewis hypertensive rats.

Values are mean ± standard error of the mean. Diurnal averages are values between 6:00 and 18:00 h. Nocturnal averages are values between 18:00 and 6:00 h.

Hourly averages in mean arterial pressure, heart rate, and locomotive activity document a desynchronization of the arterial pressure rhythm – higher in daytime and lower in night time in mRen2. Lewis hypertensive rats at baseline (C3). Heart rate and locomotive activity maintain a normal circadian pattern associated with activity levels (higher in night time and lower in daytime).

Changes in mean arterial pressure and heart rate in the vehicle, valsartan and combination groups. There are no significant differences in the mean arterial pressure among the three groups at baseline (C1−C3). However, after treatment (T12−T14), the mean arterial pressure was reduced by valsartan and further reduced by the addition of aliskiren to the valsartan treatment. There were no significant differences in heart rate among the three groups at baseline (C1−C3) and after treatment (T12−T14). ***p < 0.001 versus C1–C3; +++p < 0.001 versus vehicle; ###p < 0.001 versus valsartan.
Figure 2(b) shows the 24 h heart rate at baseline (C1−C3) and at the end of the study (T12−T14). There were no significant differences in average 24 h heart rate in the three groups at baseline and at the end of the study. These findings indicate that valsartan and combination therapy with valsartan and aliskiren reduced blood pressure without affecting heart rate and locomotive activities, although the combination therapy was significantly more effective in reducing blood pressure than valsartan alone in mRen2.Lewis rats.
Urinary sodium and potassium excretion
Table 2 shows the 24 h urinary sodium and potassium excretion of each group at baseline and after treatment. There were no significant differences in these variables in the three groups at baseline and after treatment.
Urinary sodium and potassium excretion.
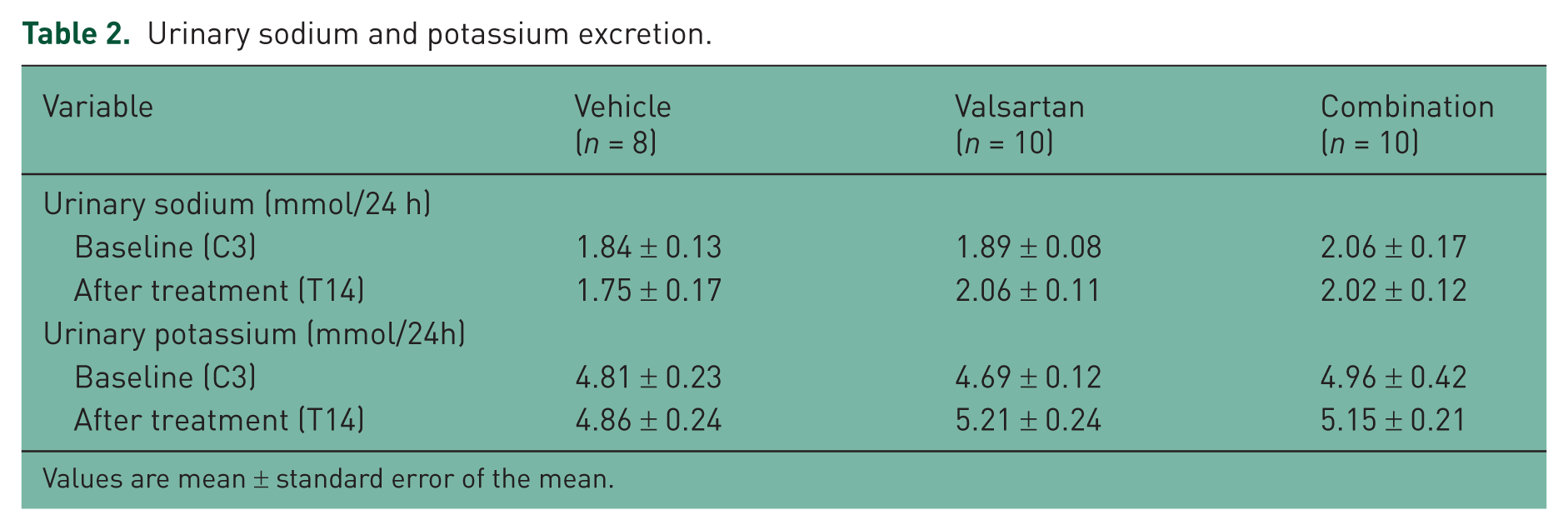
Values are mean ± standard error of the mean.
Effects of treatment on circadian rhythms in mRen2.Lewis rats
Figure 3(a–c) shows the circadian patterns of MAP, heart rate and locomotive activity in three groups at day T14. The antihypertensive effect of valsartan and valsartan combined with aliskiren in mRen2.Lewis rats was associated with a marked diminution of the circadian blood pressure variability found before treatment and the disappearance of the inverse blood pressure rhythm [Figure 3(a)]. The circadian patterns of heart rate and locomotive activity in the three groups were similar to those at baseline and no different among the three groups [Figure 3(b, c)].

Circadian rhythms in the vehicle, valsartan and combination groups after treatment (T14). The circadian rhythm of mean arterial pressure in the vehicle-treated group exhibits an inverse rhythm (a) which is essentially normalized in the mRen2.Lewis rats medicated with valsartan or the combination of valsartan/aliskiren (b). The circadian rhythms of heart rate (c) and locomotive activity (d) are no different than vehicle-treated rats.
As shown in Figure 4(a), valsartan reduced mean blood pressure more during the daytime (−78 ± 2 mmHg) than at night time (−62 ± 13 mmHg, p < 0.05) while in the animals medicated with a combination of valsartan and aliskiren the reduction in MAP averaged −93 ± 2 mmHg versus −77 ± 27 mmHg, respectively (p < 0.01). However, there was no significant difference in the change in MAP between daytime and night time in vehicle-treated rats (3 ± 2 mmHg versus 2 ± 2 mmHg). Changes in awake/asleep ratios in the three groups are shown in Figure 4(b). At baseline (C3), awake/asleep ratios in all groups were negative. The ratios in the valsartan and combination groups turned positive at day T14, although the ratio in the vehicle group was still negative. These differences in the awake/asleep ratio between vehicle and mRen2.Lewis rats medicated with valsartan or valsartan and aliskiren were revealed at day T5 (data not shown). Furthermore, the awake/asleep ratio of the combination group is significantly higher than that of the valsartan monotherapy group (6.19 ± 0.33% versus 3.74 ± 0.55%, p < 0.05).

Day–night difference in changes in mean arterial pressure and the awake/asleep ratio in the vehicle, valsartan and combination groups. (a) There was no significant difference in changes in mean arterial pressure between day and night in the vehicle group. However, valsartan and combination treatments reduced mean blood pressure more in the daytime than at night time. (b) At baseline (C3), awake/asleep ratios in all groups were negative. The ratios in the valsartan and combination groups turned positive at day T14, although the ratio in the vehicle group was still negative.
Effects of treatment on renin activity and angiotensin II levels
Table 3 and Figure 5 show the effects of the treatments on PRA and Ang II plasma and renal tissue concentrations. Valsartan monotherapy or the combination of valsartan and aliskiren increased PRA compared with vehicle (7.54 ± 1.22 ng/ml/h in the valsartan group versus 1.82 ± 0.33 ng/ml/h in the vehicle group, p < 0.001 and 9.64 ± 4.40 ng/ml/h in the combination group, p < 0.0001 versus vehicle). There was no significant difference in PRA between the valsartan and the combination groups. Although valsartan monotherapy tended to increase PRC compared with vehicle (56.60 ± 12.50 ng/ml/h versus 18.80 ± 1.71 ng/ml/h), this difference was not statistically significant. PRC in mRen2.Lewis rats medicated with the combination therapy was much higher than the values found in vehicle-treated or valsartan-treated rats (260.2 ± 23.6 ng/ml/h, p < 0.0001 versus vehicle, p < 0.0001 versus valsartan). In contrast, the PRA/PRC ratio was no different in mRen2.Lewis rats given valsartan (0.14 ± 0.02) and vehicle (0.10 ± 0.02) while the PRA/PRC ratio was significantly lower in mRen2.Lewis rats given valsartan and aliskiren (0.04 ± 0.01, p < 0.05 versus vehicle, p < 0.0001 versus valsartan). Likewise, the addition of aliskiren to valsartan blunted the increases in plasma Ang II levels (74.9 ± 9.2 fmol/ml versus 28.8 ± 4.9 fmol/ml in the vehicle group, p < 0.01) induced by blockade of AT1 receptors. Figure 5 also shows the effect of the treatments on kidney cortex Ang II. As shown in Figure 5(b), monotherapy with valsartan and combination therapy significantly reduced renal Ang II levels. Figure 6 shows that there was a robust and negative correlation between awake/asleep ratio and kidney Ang II (r2 = 0.64, p < 0.0001).
Effects of treatment on plasma renin activity (PRA) and concentration (PRC), and PRA/PRC

Values are mean ± standard error of the mean. *p < 0.05 versus vehicle; **p < 0.01 versus vehicle; ***p < 0.001 versus vehicle; +++p < 0.001 versus valsartan.

Effects of treatment on plasma and kidney angiotensin (Ang) II. (a) The addition of aliskiren to rats medicated with valsartan blunted the increases in plasma Ang II levels induced by blockade of Ang II type 1 (AT1) receptors. (b) Both treatments reduced kidney Ang II levels.

Correlation between kidney or plasma angiotensin (Ang) II level and awake/asleep ratio. (a) There was negative correlation between the awake/asleep ratio and kidney Ang II. (b) There was no significant correlation between the awake/asleep ratio and plasma Ang II.
Discussion
This study demonstrated that the congenic mRen2.Lewis hypertensive rat derived from the backcross of the m[Ren2]27 transgenic hypertensive rat with the normotensive Lewis rat is characterized by a shift in the peak blood pressure elevation from the nocturnal activity phase of the diurnal cycle into the resting daylight phase. The inversion of the blood pressure rhythm in these congenic mRen2.Lewis hypertensive rats results in a desynchronization of the 24 h blood pressure variability compared with the cycling changes of the heart rate and locomotive activity. We further demonstrate that the shift in the blood pressure circadian rhythm is normalized by blockade of the actions of Ang II by monotherapy with the AT1 receptor blocker valsartan or by combination therapy with valsartan and aliskiren. Moreover, the data show that the combination therapy is superior to blockade of AT1 receptors in the normalization of blood pressure and in restoring the blood pressure circadian rhythm. The marked reductions in plasma Ang II achieved with the combination of valsartan and aliskiren are associated with more pronounced effects on correcting the inversion of the blood pressure circadian rhythm.
Although m[Ren2]27 transgenic hypertensive rats are an established model for hypertension studies [Brosnihan et al. 1994; Lee et al. 1996; Lemmer et al. 1993, 2005; Monosikova et al. 2007; Moriguchi et al. 1994a, 1994b, 1995; Pons et al. 1996; Senanayake et al. 1994; Witte and Lemmer, 1999], the development of fulminant hypertension at an earlier age and the nonhomogenous genetic background from which these rats were outbred has limited the usefulness of this model for the extrapolation of identified mechanisms to human hypertension. The creation of a congenic model of increased tissue renin transgene expression derived from the backcross of the original m[Ren2]27 transgenic hypertensive rat with Lewis normotensive rats has markedly reduced the occurrence of fulminant hypertension while retaining the main features of the model such as sex differences in the magnitude of blood pressure elevation, target organ damage and renal injury, normalization of blood pressure and reversal of cardiac hypertrophy by blockade of Ang II expression or activity, and changes in cardiac and renal tissue expression of RAS components [Chappell et al. 2008; Jessup et al. 2009; Pendergrass et al. 2006, 2008]. In the pursuit of the further characterization of this experimental model of hypertension we now show that mRen2.Lewis hypertensive rats retain the parent trait of an inverse blood pressure circadian rhythm, uncoupled from the circadian oscillations in heart rate and motility.
The inclusion of an experimental arm combining AT1 receptor blockade with suppression of renin activity was designed to exclude the possibility that the large increases in plasma Ang II associated with blockade of AT1 receptors may still be capable of maintaining the altered blood pressure rhythm through incomplete blockade of Ang II actions in the brain due to low penetrability of the antagonist or Ang II action on AT2 receptors. The demonstration that the antihypertensive response and the degree of restoration of the awake/sleep ratios were significantly greater in mRen2.Lewis rats medicated with valsartan and aliskiren demonstrates an incomplete suppression of Ang II activity in rats given valsartan monotherapy. Whether this greater effect of the combination therapy resulted from the decrease in circulating Ang II levels, as shown in the current experiments, or an additional effect of aliskiren in the brain remains to be investigated. Nevertheless, the more robust effect of the combination of valsartan and aliskiren in restoring the circadian rhythm of the blood pressure shows that the desynchronization of the blood pressure rhythm in terms of heart rate and locomotive activity is primarily driven by Ang II.
As discussed above, the antihypertensive effect of valsartan in mRen2.Lewis rats was further potentiated in the presence of aliskiren. These data agree with the observation in humans of a greater antihypertensive action of the combined administration of these agents [Oparil et al. 2007]. Although aliskiren is associated with decreases in PRA in humans [Azizi et al. 2007; Krone et al. 2011; Moriyama et al. 2011; Nussberger et al. 2007; Stanton et al. 2009], we found that in mRen2.Lewis rats blood pressure normalization with the dual combination therapy was associated with increases in PRA and PRC. Although aliskiren is less specific in inhibiting rat renin, the dose of aliskiren used here has previously been shown to elicit significant antihypertensive effects in m[Ren2]27 transgenic hypertensive rats that were associated with reversal or cardiac hypertrophy, improved insulin resistance, and reduced renal oxidative stress [Habibi et al. 2008; Lastra et al. 2009; Whaley-Connell et al. 2008, 2010, 2011]. The finding that the addition of aliskiren resulted in decreases in blood pressure greater than those obtained with valsartan monotherapy provided further evidence that aliskiren neutralized the reactive increase in renin produced by blockade of AT1 receptors. These data agree with the concomitant finding that the addition of aliskiren to valsartan was associated with blunting of the increases in plasma Ang II concentrations resulting from the administration of valsartan. Previous studies in normal healthy volunteers and people with essential hypertension showed that aliskiren is accompanied by large increases in PRC [Jordan et al. 2007; Smith et al. 2007; Verdecchia et al. 2008], a measure of the amount of renin protein present in the sample [Nussberger et al. 2007; Sealey and Laragh, 2007]. However, PRA measures the rate of conversion of angiotensinogen to Ang I by renin [Sealey, 1991]. The characteristics of the procedures used in the assessment of PRA in the rat assay may cause dissociation of aliskiren from renin in the sample. This will need further investigation.
In spontaneously hypertensive rats, chronic elevations in arterial pressure do not result in changes in the circadian rhythm of blood pressure, heart rate, and motility compared with normotensive Wistar Kyoto and Sprague Dawley rats [Lemmer et al. 1993]. Importantly, an inversion of the blood pressure circadian rhythm but not of heart rate and locomotive activity results from the chronic administration of Ang II in Sprague Dawley rats [Baltatu et al. 2001] while administration of enalapril in m[Ren2]27 transgenic hypertensive rats is associated with normalization of the blood pressure circadian rhythm [Lemmer et al. 1994] underlining the important contributory role of RAS in the desynchronization of the blood pressure rhythm.
An important factor influencing the circadian rhythm of blood pressure is the daily fluctuation in renal function, such as renal blood flow, glomerular filtration rate (GFR), and the excretion of electrolytes [Stow and Gumz, 2011]. In this regard, Pons and colleagues reported that m[Ren2]27 transgenic hypertensive rats have a normal circadian pattern of urinary water and electrolytes excretion, and of GFR despite having an inverse circadian rhythm of the blood pressure [Pons et al. 1996]. In agreement with those studies we found that the 24 h sodium and potassium excretion rates in mRen2.Lewis rats were not changed by the treatments.
The first demonstration of a reversal of the awake/sleep ratio associated with reduced renal Ang II content by valsartan or the valsartan and aliskiren combination provides new evidence for the unraveling of the mechanism accounting for the control of the circadian blood pressure oscillations. We have previously shown increased content of Ang II associated with downregulation of AT1 receptors in the kidneys of mRen2.Lewis hypertensive rats [Pendergrass et al. 2006]. These data suggest that the reduction of renal Ang II content may be a contributing mechanism to the normalization of the blood pressure circadian rhythm most likely related to a decrease in neural input from renal afferent nerves reaching the brainstem or the hypothalamus. Renal afferent nerves provide information for hypothalamic structures and the nucleus of the solitary tract (nTS) region of the dorsomedial medulla oblongata known to be involved in the regulation of arterial pressure and fluid balance [Calaresu and Ciriello, 1981; Ciriello and Calaresu, 1980; Felder, 1986]. Moreover, selective removal of renal afferent nerves attenuates the development of deoxycorticosterone acetate-salt hypertension [Oparil et al. 1987] while bilateral rhizotomy delayed development of two-kidney two-clip (2K-2C) hypertension and prevented vascular hypertrophy; norepinephrine and epinephrine content in the medulla oblongata, adrenal gland, and plasma were all decreased, whereas hypothalamic catecholamine content was increased [Wang et al. 1995]. One possible site for the inversion of the blood pressure circadian rhythm may be the suprachiasmatic nuclei (SCN) of the hypothalamus, the site for the generation of neuronal and hormonal regulation of 24 h body cycles. Although ablation of the SCN abolished the inversion of the blood pressure rhythmicity in m[Ren2]27 transgenic rats, the loss was also associated with disappearance of the heart rate and locomotive day/night oscillations [Witte et al. 1998; Witte and Lemmer, 1999]. These data agree with the findings of Janssen and colleagues who showed that the circadian rhythmicity in blood pressure, heart rate, and food intake were abolished in SCN-lesioned rats [Janssen et al. 1994]. These findings indicate that other anatomical sites within the central nervous system may contribute to the selective desynchronization of the blood pressure rhythm.
Master switches that in the brain regulate the activity of biological clocks are influenced by the intrinsic and extrinsic activities of neurotransmitters and neuromodulators expressed by the autonomic system and RAS. The interplay between RAS and the sympathetic nervous system, peripherally and centrally, is recognized to play a critical role in the evolution of hypertension in part due to the fact that Ang II has a major influence on the regulation of central and peripheral vasomotor tone [Ferrario et al. 1972, 1990; Ferrario, 1983] and acting within the nTS to inhibit baroreceptor reflexes [Averill et al. 1987; Diz et al. 1997]. An age-dependent altered brain and adrenal gland norepinephrine/epinephrine content and turnover have been reported in m[Ren2]27 transgenic rats with loss of increased cardiac norepinephrine turnover rate in the dark phase [Lemmer et al. 2005]. Since the rhythm of the circadian variations in heart rate and locomotive activity do not occur in m[Ren2]27 transgenic rats [Lemmer et al. 1993] or in our experiments in mRen2.Lewis hypertensive rats, a direct loss of sympathetic nerve activity may not explain our findings. However, normalization of the circadian blood pressure rhythm in mRen2.Lewis rats following blockade of Ang II receptors or the combined inhibition of Ang II activity and renin suggest a deterministic role of the peptide in the desynchronization of the central control of arterial pressure, heart rate, and locomotive activity. While our experiments did not explore the site and specific mechanism accounting for these changes, increased brain and circulating Ang II may contribute. All RAS components exist in the brain and circulating Ang II exerts powerful modulatory influences on brain vasomotor centers through its actions on circumventricular organs [Ferrario, 1983; Ferrario et al. 1987, 1990]. Therefore, increased plasma Ang II in response to valsartan monotherapy was abolished by the addition of aliskiren, uncovering the impact of elevated plasma Ang II on the blood pressure lowering effect and on reversal of the awake/sleep ratio in valsartan-treated rats. Further studies are needed to examine whether the superior restoration of the awake/sleep ratio by the combined administration of Ang II receptor blockers and renin inhibitors correlates with better control of target organ damage and reduced cardiovascular morbidity and mortality.
In summary, we showed that blockade of the actions of Ang II in a renin-dependent model of experimental hypertension results in the resynchronization of the circadian rhythm of arterial pressure with respect to the accompanying circadian variations in heart rate and locomotive activity. The observation that this reversal of the blood pressure circadian rhythm was associated with decreases in the renal content of Ang II suggests that the local tissue actions of Ang II may modulate renal afferent traffic activity to the brainstem and hypothalamic circuits regulating the activity of the circadian blood pressure rhythm. Moreover, the greater effects of the combined approach to blockade of Ang II actions, as demonstrated by blood pressure normalization, restoration of the blood pressure circadian rhythm, and the corresponding awake/sleep ratio, when compared with valsartan monotherapy is new evidence that needs to be further investigated. This may lead to better understanding of the mechanisms associated with blood pressure dysregulation and ultimately improved management of hypertensive target organ injuries and reduced cardiovascular morbidity and mortality.
Footnotes
This research was supported by an unrestricted research grant provided by Novartis, Inc. and the National Heart, Lung and Blood Institutes of the National Institutes of Health (2PO1 HL-051952 and R01 HL-56973). We also acknowledge partial support provided by the Farley-Hudson Foundation, Jacksonville, NC, USA.
The work reported here was supported in part by an unrestricted research grant provided by Novartis, Inc. to Carlos M. Ferrario. The sponsor had no direct input in the design of the study, data analysis and interpretation, or the writing of the paper.
