Abstract
Introduction
The dried root of Salvia miltiorrhiza, listed as Danshen in the Chinese Pharmacopoeia, is a widely used traditional Chinese medicine to treat coronary artery disease, cerebrovascular disease, hepatocirrhosis and bacterial infections [Lin and Chang, 2000]. Danshen is an important source of a large number of active natural compounds which are mainly classified as aqueous and lipid soluble (diterpenes) fractions [Zhou et al. 2005]. Salvianolic acid B (Sal B) is one of the major water-soluble extracts while Tanshinone IIA (Tan IIA) is the most active diterpenoid quinone pigment. Both have been studied extensively for their broad pharmacological activities.
Previous studies have shown that Danshen has cardioprotective effects [Ji et al. 2003]. The efficacy of Danshen in the treatment of coronary heart disease was observed in a double-blind study using phenolphthalein as a placebo [Lin and Chang, 2000]. Zhou and Ruigrok showed that Danshen increased coronary blood flow and mediated a negative ionotropic effect in isolated rat hearts. Furthermore, Danshen-treated hearts had better postischemic reperfusion recovery and less contracture than untreated hearts [Zhou and Ruigrok, 1990]. Kamata and colleagues revealed that Danshen was able to dilate the rat aorta in an endothelium-dependent manner [Kamata et al. 1993]. The cardioprotective effects of Danshen are considered to be associated with its function in regulating hypoxic factors, mitochondria function defects, endothelial cell dysfunction and angiogenesis factors. Kang and colleagues demonstrated that Danshen-induced antihypertensive effects may be in part due to the angiotensin-converting enzyme (ACE) inhibitor effect of the extract, lithospermic acid B (LSB) [Kang et al. 2003].
Although pharmacological studies indicate that Danshen has cardiovascular benefits, the mechanisms underlying its protective effects are not yet fully understood. Our results indicate that Tan IIA and Sal B can dilate C57/BL6 mice coronary arteries and that this effect can be inhibited by using a nitric oxide synthase (NOS) inhibitor (L-NAME) or PI3K inhibitor. We further utilize the myocardial infarction (MI) model of mice, ex vivo micro-artery system and endothelial cell culture system to explore the molecular mechanisms of Danshen extracts in vasodilatation.
Materials and Methods
Reagents
Salvianolic acid B (MW: 718.62) and tanshinone IIA (MW: 294.33) were purchased from the Chinese National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). L-NAME was purchased from Sigma Chemical Co. We initially used the metric system to quantify the weight of Sal B and Tan IIA and converted into molarities using the following conversion formula:
Cell culture
Human umbilical vein endothelial cells (HUVECs) were harvested from human umbilical vein by enzymatic dissociation [Baudin et al. 2007]. Cells were cultured in M199 (Gibco) supplemented with 10% fetal bovine serum (Hyclone) and antibiotics (0.1 mg/ml streptomycin, 100 U/ml penicillin). The experiments were performed on cells in four to six passages. HUVECs were incubated in serum-depleted medium for 20 hours prior to incubation with Sal B or Tan IIA, with or without 10 μM of compound C (AMP-activated protein kinase [AMPK] inhibitor), and 1.0 mM aminoimidazole carboxamide ribonucleotid (AICAR), an AMPK agonist. The viability of HUVECs was determined by trypan blue exclusion.
Animals
Male C57BL/6 mice, aged 6–8 weeks with body weights of 18-22 g, were used for this study. Endothelial nitric oxide synthase (eNOS) knockout mice were purchased from Jackson Laboratories. All animal procedures were carried out in accordance with the guidelines of the Institutional Animal Care and Use Committee of the University of Chinese Medicine, Beijing, China.
Myocardial infarction
After anesthesia with intraperitoneal sodium pentobarbital (Inactin Byk-Gulden, 50 mg/kg body weight), mice were artificially ventilated with a small animal volume-control ventilator (Harvard Apparatus; Holliston, MA). Ventilation was done with a tidal volume of 1 ml at a rate of 100 cycles/min. Thoracotomy was performed at the left third intercostal space, the heart was then exposed via a small retractor. A 7−0 suture was placed in the anterior myocardium to occlude the left anterior descending artery (LAD). The thorax and the skin incision were closed with 4−0 sutures. The endotracheal tube was gently retracted after spontaneous breathing was restored. Sham operated animals were subjected to similar surgery, except that no ligature was placed [Lutgens et al. 1999]. An olive oil sham group was set for Tan IIA as it is not soluble in water. The surviving mice were divided randomly into six groups: sham operation (IP 0.1 ml/10 g NS; 0.1 ml/10 g olive oil by a gavage); MI (IP 0.1ml/10 g NS; 0.1 ml/10g olive oil by a gavage); MI + Sal B (IP 40 mg/kg or 54.5 µmol/kg) and MI + Tan IIA (20 mg/kg or 66.65 µmol/kg by a gavage) beginning on the day following surgery, and vehicle was given to sham and MI groups once a day for 4 weeks. In another experiment, all surviving mice were grouped and treated the same except for receiving L-NAME in the drinking water (0.5 mg/ml, Sigma Chemical Co.).
Echocardiography
Twenty eight days after LAD ligation, transthoracic echocardiography was performed using a Vevo 660 system (VisualSonics, Toronto, Canada) equipped with a 30 MHz real-time microvisualization scan head probe at a frame rate of 65 frames/s. Briefly, each mouse was anesthetized with 1.5% isoflurane and the chest was shaved. Then, echocardiographic parameters (left ventricular end-systolic volume [LVESV], left ventricular end-diastolic volume [LVEDV], left ventricular ejection fraction [LVEF] and left ventricular fractional shortening [LVFS]) were measured as described previously [Takagawa et al. 2007].
Vasorelaxation assay
The coronary artery was removed from wildtype (C57/BL6) and eNOS knockout mice (Jackson Laboratories) and placed in cold physiological saline solution (PSS) composed of (in mmol/l): NaCl 119, KCl 4.7, CaCl2·2H2O 2.5, MgSO4·7H2O 1.2, NaHCO3 25, KH2PO4 1.2, and glucose 5.5. The left anterior descending coronary artery was mounted onto a PSS-filled cannula in a myograph chamber as described previously [Charan Sahoo et al. 2009]. The vessel was visualized using a video camera (VM-902; Hitachi Denshi Ltd) and the internal diameter was determined using a video dimension analyzer (Living Systems Inc., MA). To standardize the data, the vascular tone of U46619-contracted (BIOMOL) vessels was set equal to 0% relaxation. The chemicals L-NAME (Alexis, MA) and LY294002 (Cayman Chemical, MI) were preperfused for 30 min before adding Sal B and Tan IIA in PSS (10 ml/min) through the artery.
L-arginine uptake assay
L-arginine uptake was determined in cells that had previously been depleted of intracellular free amino acids by incubation for 2 h in 1 ml of Krebs solution. Cells were then incubated in the presence or absence of different doses of Sal B or Tan IIA for 3 h. L-arginine uptake was measured by incubating the cells for 5 min at 37°C with Krebs bicarbonate solution containing [3H] L-arginine (GE healthcare, 50 mM, 1 mCi/ml) and L-arginine (Sigma, 64 µmol/l). L-arginine transport was also measured in Krebs solution in which NaCl was replaced by equimolar choline chloride [Geng et al. 2007]. Radioactivity of the ambient incubation buffer and solution was measured by beta-scintillation counting (Bechman FJ-2170, Mississauga, ON). To correct for nonspecific uptake and binding, cells were incubated in parallel wells with a buffer containing [3H] L-arginine. The fraction of the radioactivity associated with the cells was determined, and this fraction was then subtracted.
NO production assay
Various concentrations of Sal B and Tan IIA were prepared in phenol red-free Dulbecco’s Modified Eagle Medium (DMEM; to reduce assay interference by phenol red). Cell culture supernatants were collected after treatment with different compounds (or vehicle alone in the controls) for 3 hours. Nitric oxide (NO) production was detected spectrophotometrically by measuring its final stable equimolar degradation products, nitrite and nitrate, by using nitrate reductase (Assay Design Inc.) and the acid-catalyzed diazotation reaction by using sulfanilamide and naphtylethylenediamine (Griess reaction). Total nitrite was quantified after the reduction of all nitrates with nitrate reductase. Nitrite levels in culture supernatants were within the linearity range of calibration curves that were generated from a solution of sodium nitrite. Total nitrite concentration was calculated from a standard curve constructed over the linear range of the assay and expressed as 100 pmol per 105 cells.
Real-time RT-PCR

Amplification was followed by melting curve analysis to verify the accuracy of the amplicon. A negative control without cDNA was run with every PCR to assess the specificity of the reaction. Analysis of data was performed using Light Cycler software version 3.5.
Western blot analysis
The mouse hearts and HUVECs were lysed in RIPA buffer (Boston Bioproducts, MA) with protease inhibitors, and proteins were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS/PAGE) gel and transferred to a polyvinylidene fluoride (PVDF) membrane (Millipore, MA). After being blocked for 1 h in Tris-buffered saline/Tween 20 (TBST) with 5% nonfat milk, the PVDF membrane was then probed with the following primary polyclonal rabbit antibodies: anti-AMPK (1 : 1000, Cell Signaling), or anti-phosphorylated Thr172 AMPK(1 : 1000, Cell Signaling), rabbit anti-Akt (1 : 1000, Cell Signaling), or antiphosphorylated Ser473-Akt (1 : 1000, Cell Signaling), mouse anti-eNOS/NOSIII (1 : 2500) or antiphosphorylated Ser1177-eNOS (1 : 1000, B&D). Rabbit antitubulin β (1 : 3000, Sigma) served as the internal control, and the samples were incubated at 4°C overnight and then for 1 hour in TBST/0.2% bovine serum albumin (BSA) containing horseradish peroxidase-conjugated antirabbit or antimouse antibodies (1 : 5000). The proteins were visualized with an ECL detection system (Amersham). Semiquantifications were performed with densitometric analysis using Metamorph software.
Statistical analysis
All values are expressed as means ± SEM. Relaxation in response to herbs is presented as percentage change in tension from preconstriction. When multiple vessel rings were studied from one mouse, responses were averaged, and n represents the number of mice per group. All of the experiments and analyses were performed in a blinded fashion. Comparisons were made by using a one-way analysis of variance (ANOVA) with repeated measures, followed by the Student–Newman–Keuls test to detect individual differences. A value of p < 0.05 was considered significant.
Results
Sal B and Tan IIA attenuate heart to body weight ratio and infarction sizes in myocardial infarcted mice via the eNOS/ NO pathway
To identify the effect of Sal B and Tan IIA on the ischemia tissue in vivo, we induced MI in C57 mice and measured the heart to body weight ratio and infarction sizes by triphenyltetrazolium chloride (TTC) staining. The observed increase in the heart to body weight ratio after the onset of ischemia is attributed to the cardiac muscle hypertrophy as shown earlier [Lutgens et al. 1999]. The treatment with either Sal B (40 mg/kg) or Tan IIA (20 mg/kg) was able to attenuate the post infarction hypertrophy as evident by their observed effects on the heart to body weight ratio at 4 weeks (Figure 1A). The concentrations selected in our studies were based on previous publications that showed effective doses for cardioprotective activity of Sal B [Jiang et al. 2008] and Tan IIA [Dong et al. 2009]. Interestingly, the pretreatment of mice with L-NAME diminished the effects of both Sal B and Tan IIA (Figure 1A). The treatment with either Sal B or Tan IIA was also able to reduce the infarction size significantly, as measured at 4 weeks in the C57 mice (Figure 1B), while in the mice pretreated with L-NAME water, both Sal B and Tan IIA had little effect (Figure 1B). The concomitant use of treatment with vehicle as a control was also done to minimize any scope of error.
Sal B and Tan IIA attenuate the heart to body weight ratio and infarction sizes in myocardial infarcted mice through the eNOS/NO pathway.
Sal B and Tan IIA improve the cardiac function of the myocardial infarcted mice through the eNOS/ NO pathway
Echocardiographic data for C57 mice 4 weeks after myocardial infarction with or without L-NAME.

p<0.05 as compared with corresponding sham group. LVESV, left ventricular end-systolic volume;
p< 0.01 as compared with corresponding sham group. LVEDV, left ventricular end-diastolic volume;
p<0.05 as compared with corresponding myocardial infarction group. LVEF, left ventricular ejection fraction;
p<0.01 as compared with corresponding myocardial infarction group. LVFS, left ventricular fractional shortening.
Sal B and Tan IIA have a vasodilator action via the eNOS/ NO pathway
It is known that the effects of eNOS/NO pathway are one of the major mediators of vascular dilatation in coronary arteries. This prompted us further to examine whether either Sal B or Tan IIA used the same mechanism for its effects. The mouse coronary arteries with intact endothelium were pre-contracted by U-46619 (a thromboxane mimic), and then treated with either Sal B or Tan IIA. Interestingly, both components of Danshen induced the vasodilatation effect (Figure 2A). The pretreatment of coronaries with eNOS inhibitors (L-NAME) and PI3K inhibitor (LY294002), 30 minutes before exposure to either Tan IIA (Figure 2B) or Sal B (Figure 2C) markedly decreased their vasodilator actions, hinting at the downstream involvement of eNOS/NO and PI3K pathways in Sal B and Tan IIA mediated effects.
Vasodilatation induced by Tan IIA and Sal B in mouse coronary arteries. The mouse coronary arteries were precontracted by U-46619 (a thromboxane mimic), and then treated with Tanshinone IIA (Tan IIA) and Salvianolic acid B (Sal B). Both Tan IIA and Sal B relaxed the microvessels in a dose-dependent manner (A). Prior administration of endothelial nitric oxide synthase (eNOS) inhibitors (L-NAME) or PI3K inhibitor (LY294002) markedly diminished the vasodilator action of Tan IIA (B) and Sal B (C).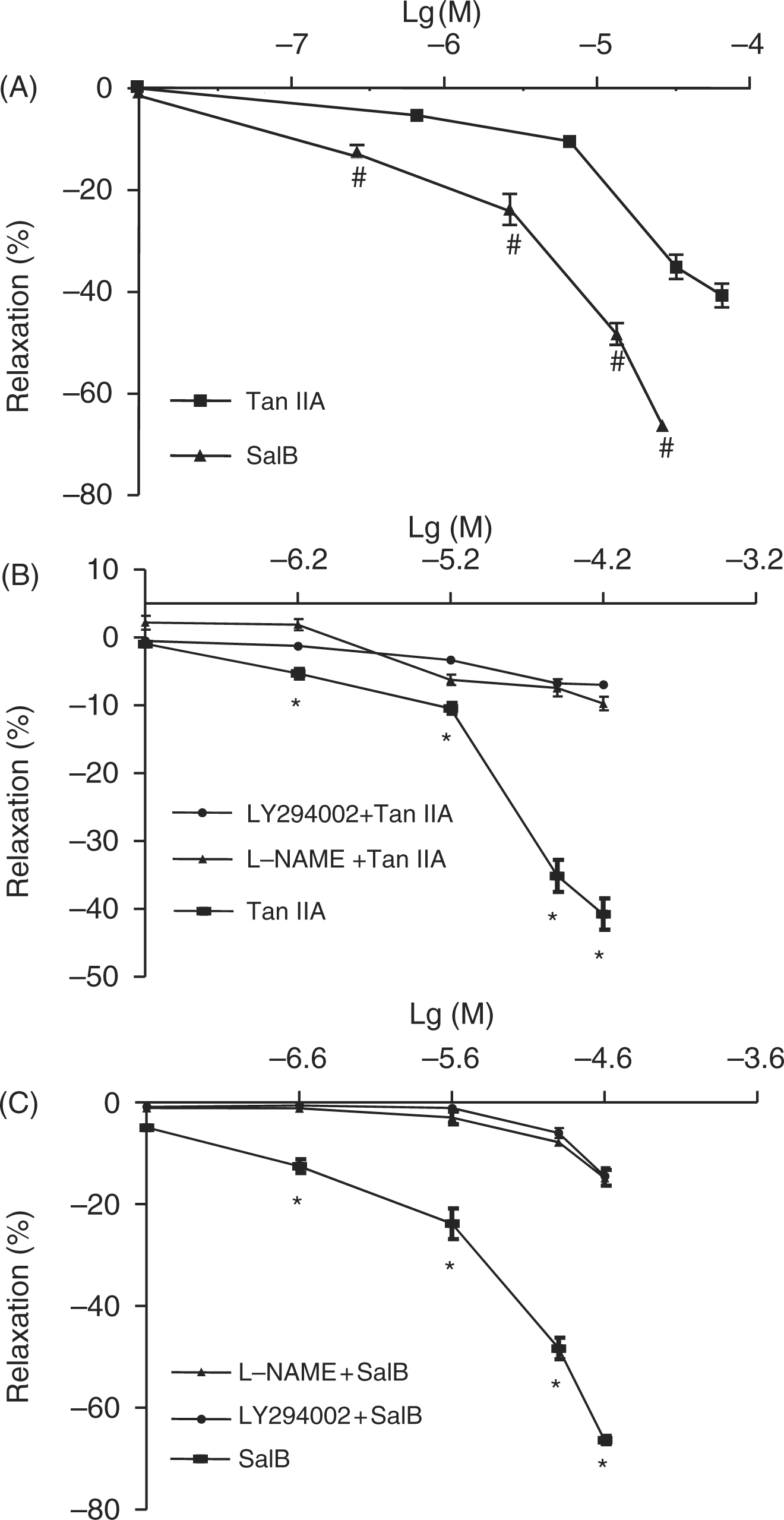
Sal B- and Tan IIA-induced vasodilatation is endothelial dependent
NO-induced vasodilatation can occur by its effect on either endothelium or the smooth muscle in vessel walls. We used eNOS-/- mice, as their endothelia do not generate NO and rely on the smooth muscle component for the same, in order to investigate the exact mechanism of coronary vasodilatation by Sal B and Tan IIA. Examination of the coronaries of eNOS -/- mice revealed that Tan IIA in all dosages (Figure 3A) and Sal B in high dosages (Figure 3B) caused significant vessel relaxation as observed in the wildtype controls. These results further suggest that eNOS-related Sal B- and Tan IIA-induced vasodilatation may be endothelium dependent.
The vasodilator effect of Tan IIA and Sal B in eNOS-/- mouse coronary arteries. The mouse coronary arteries from C57 mice or eNOS-/- mice were precontracted by U-46619 (a thromboxane mimic), and then treated with Tanshinone IIA (Tan IIA) or Salvianolic acid B (Sal B). Results shows that Tan IIA induced vasodilatation in coronary arteries of eNOS-/- mice (A). Sal B-induced vasodilator action in coronary arteries of eNOS-/- mice was no different to the wildtype controls (B). # *p < 0.05.
Sal B and Tan IIA activate phosphorylation of eNOS via the AMPK/PI3K/Akt pathway
In the cultured endothelial cells, both Tan IIA (Figure 4A) and Sal B (Figure 4B) stimulated eNOS phosphorylation in a concentration and time-dependent manner beginning at 5–10 minutes after incubation and peaking at approximately 30 minutes. After 30 minutes, cells were lysed and the lysates were subjected to immunoblot analysis of phosphorylation. When the endothelial cells were pretreated with an inhibitor of PI3K (LY294002), the Tan IIA-induced (Figure 4C) or Sal B-induced (Figure 4D) phosphorylation of eNOS was significantly diminished suggesting the involvement of the PI3K/Akt pathway in eNOS phosphorylation. Tan IIA and Sal B also rapidly increased phosphorylation of AMPK (Thr172) (Figure 4E and F) and Akt (Ser473) (Figure 4G and H). Cultured cells were then pretreated with AICAR (1 mM) or compound C (30 µM) for 30 min, prior to treatment with Sal B or Tan IIA. When we used AMPK antagonist-compound C, the phosphorylation of Akt (Ser473) was decreased significantly.
Tan IIA- and Sal B-activated phosphorylation of eNOS, Akt and AMPK. Tanshinone IIA (Tan IIA) and Salvianolic acid B (Sal B) stimulated endothelial nitric oxide synthase (eNOS) phosphorylation in human umbilical vein endothelial cells (HUVECs) in a time- and concentration-dependent manner peaking at approximately 30 minutes of incubation (A, B). eNOS phosphorylation was induced with Sal B or Tan IIA. Pretreatment with LY294002 (2 × 10−5 M), significantly abolished the Tan IIA- or Sal B-induced phosphorylation of eNOS (C, D). Tan IIA and Sal B (10 µg/ml or 33.3 μM/13.6 μM) stimulated phosphorylation of AMPK and Akt, respectively (E, F). The addition of AMPK inhibitor (compound C, CC) mitigated phosphorylation of Akt by Tan IIA and Sal B (G, H). *p < 0.05, **p < 0.01 versus con, #p < 0.01 versus column before it.
Sal B and Tan IIA stimulates production of NO and L-arginine transport and uptake
We examined whether Sal B- and Tan IIA-induced eNOS activation results in enhancing the NO production. The NO level in cultured medium was measured in endothelial cells after 3 hours of exposure to either Sal B or Tan IIA at varying concentrations (1, 5, 10 and 20 µg/ml). The results were analyzed using the NO production assay, in which NO production was detected by measuring nitrite and nitrate. The total nitrite concentration was calculated from a standard curve constructed over the linear range of the assay and expressed as 100 pmol per 105 cells (Supplementary Figure 1). The NO production was significantly increased in a concentration-dependent manner with both Sal B and Tan IIA. Interestingly, the exposure to 20 µg/mL of Tan IIA yielded NO equal to an exposure to 5 µg/ml of Tan IIA (Figure 5A and B) which might be related to the toxicity/apoptotic activity of Tan IIA at higher concentrations to the HUVECs [Yang et al. 2005].
Tan IIA and Sal B stimulate production of NO. HUVECs were incubated with Tanshinone IIA (Tan IIA; 5, 10, and 20 µg/ml or 3.3 μM, 16.6 μM, 33.3 μM, and 66.6 μM) or Salvianolic acid B (Sal B; 1, 5, 10, and 20 µg/ml or 1.4 μM, 6.8 μM, 13.6 μM, and 27.3 μM) for 3 hours. The medium was collected for NOX detection. The nitric oxide (NO) production was significantly increased by both (A) Tan IIA and (B) Sal B.
In order to examine whether Sal B and Tan IIA are also involved in modulating the transport of L-arginine into the endothelial cells, [3H] L-arginine (1 mCi/mL) and L-arginine were added to Sal B- and Tan IIA-treated endothelial cells. The results showed that both Tan IIA and Sal B significantly increased L-arginine transport into endothelial cells in a dose-dependent manner (Figure 6A and B). To further elucidate whether the [3H]-L-arginine uptake induced by Sal B and Tan IIA occurred independently of extracellular sodium and by the Na+-independent system y+ carrier, we replaced extracellular Na+ by equimolar choline and measured [3H]-L-arginine uptake. [3H]-L-arginine uptake induced by Sal B and Tan IIA was unchanged in the presence of equimolar choline than Na+, suggesting that Sal B- and Tan IIA-related L-arginine uptake is mediated by the Na+-independent system y+ carrier (Figure 6C).
Tan IIA and Sal B stimulate transport of L-arginine. Amino acid depleted human umbilical vein endothelial cells (HUVECs) when incubated with different concentrations of Salvianolic acid B (Sal B) or Tanshinone IIA (Tan IIA) for 3 hours shows increased L-arginine uptake as measured by [3H]-L-arginine uptake (A, B). When we replaced extracellular Na+ by equimolar choline, [3H]-L-arginine uptake induced by Sal B and Tan IIA was unchanged (C). *p < 0.05, **p < 0.01 versus con.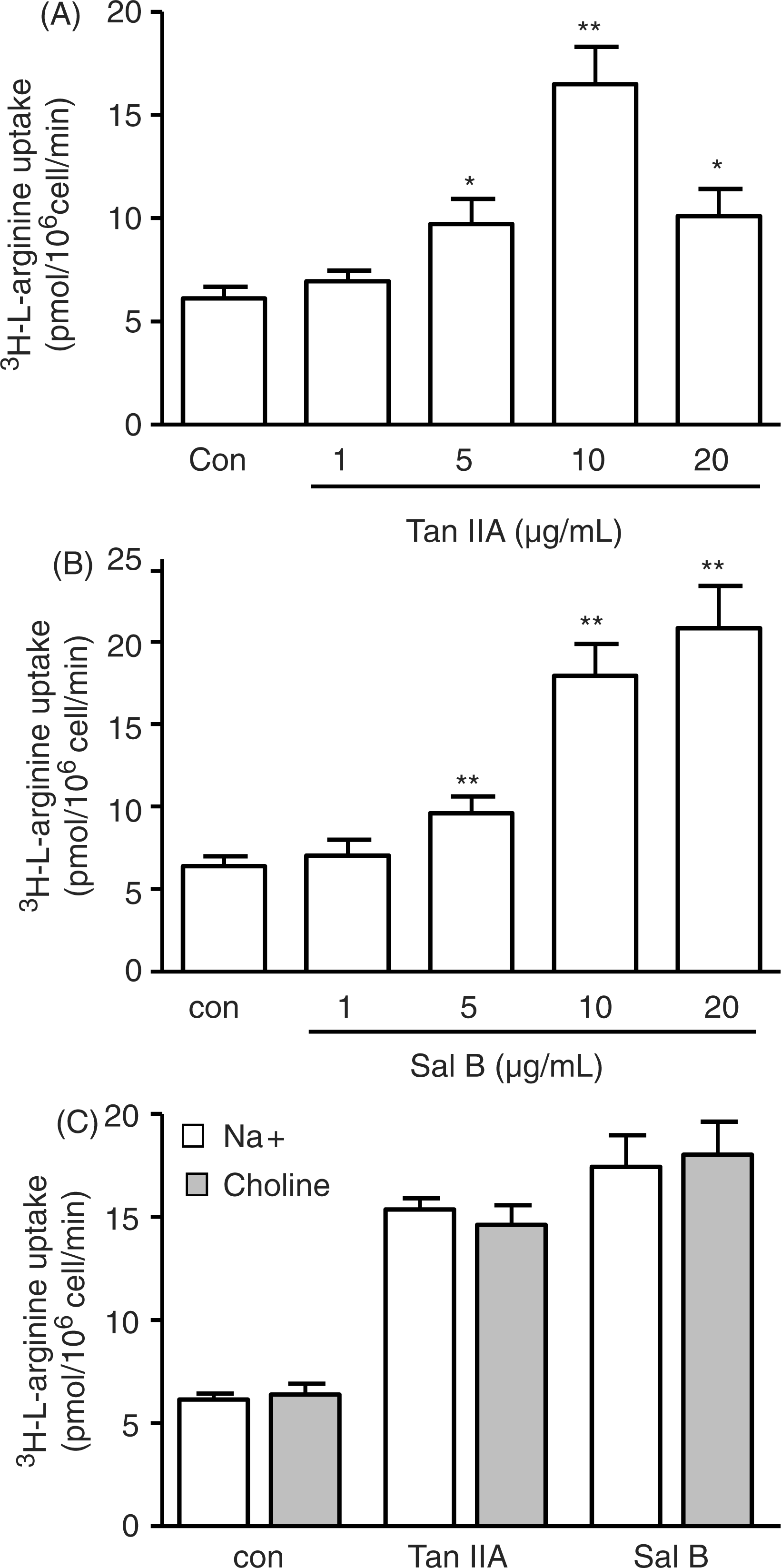
Sal B and Tan IIA increased cationic amino acid transporter (CAT-1 and CAT-2B) mRNA expression
L-arginine transported into endothelial cells can be mediated by several different classes of cationic amino acid (CAA) transporters (CATs), such as CAT-1, CAT-2, CAT-3 or CAT-4, the vast majority of which are CAT-1 and CAT-2B transporters. Quantitative PCR was performed, and results showed that Tan IIA increased the CAT-1 and CAT-2B mRNA levels after 4 and 8 hours of incubation as compared with controls (Figure 7A and B). Sal B also increased the CAT-1 and CAT-2B mRNA levels after 8 hours of incubation (Figure 7C and D).
Tan IIA and Sal B stimulate CAT-1 and CAT-2B mRNA expression. Human umbilical vein endothelial cells (HUVECs) were incubated with Tanshinone IIA (Tan IIA; 10 µg/ml or 33.3 μM) or Salvianolic acid B (Sal B; 10 µg/ml or 13.6 μM) for 8 hours. Real-time PCR detection showed that Tan IIA increased CAT-1 and CAT-2B mRNA levels significantly after 4 and 8 hours of incubation compared with controls (A, B). A similar effect was seen with Sal B on mRNA levels after 8 hours of incubation (C, D). *p < 0.05, **p < 0.01 versus con.
Discussion
Salvia miltiorrhiza (Danshen), a popular Chinese herb, has been widely and successfully used for the treatment of angina pectoris, MI and stroke [Ji et al. 2000]. It has revolutionized the management of these diseases in Chinese societies. Danshen has multiple beneficial effects in coronary artery disease patients by virtue of its sedative, antioxidant, vasodilator, and platelet inhibitory effects as well as the enhancement of angiogenesis [Ji et al. 2003]. It has been of immense significance in preventing ischemia–reperfusion related microcirculatory disturbances by using additional mechanisms such as inhibition of apoptosis, inhibiting platelet degranulation, inhibiting mast cell degranulation, downregulating expression of adhesion molecules on leucocytes and inhibition of multiple cytokines such as tumor necrosis factor alpha (TNF-α), interleukin (IL)-1β, IL-6, and IL-12 [He et al. 2008]. Although numerous studies regarding Danshen’s clinical benefits have been performed [Zhou et al. 2005], the biochemical information available to demonstrate the mechanisms of Danshen’s cardioprotective effects in MI is still very limited.
In the present study, we have utilized the MI model of mice, an ex vivo micro-artery system and a HUVEC cell culture system to explore the molecular mechanisms of two most active components of Danshen, Sal B and Tan IIA. The major findings include the following. Sal B and Tan IIA limit the infarction sizes and increase in heart to body weight ratio while improving the cardiac function of mice at 4 weeks postinfarction, which is in agreement with previous studies [He et al. 2008; Wu et al. 1993]. The cardioprotective effect of Sal B and Tan IIA is endothelial dependent through vasodilatation, using signal mechanisms through the phosphorylation of eNOS via the AMPK/PI3K/Akt pathway and the subsequent generation of NO. Sal B and Tan IIA stimulate L-arginine transport and uptake to endothelium cells, which is linked with increased CATs (CAT-1 and CAT-2B) mRNA expression (Figure 8).
Causal relationship of Danshen components and endothelial NO production. Salvianolic acid B (Sal B) and Tanshinone IIA (Tan IIA) use phosphorylation signal mechanisms via the AMPK/PI3K/Akt pathway leading to the generation of nitric oxide (NO). Sal B and Tan IIA also stimulate L-arginine transport and uptake to endothelium cells linked with increase in CAT-1 and CAT-2B mRNA expression.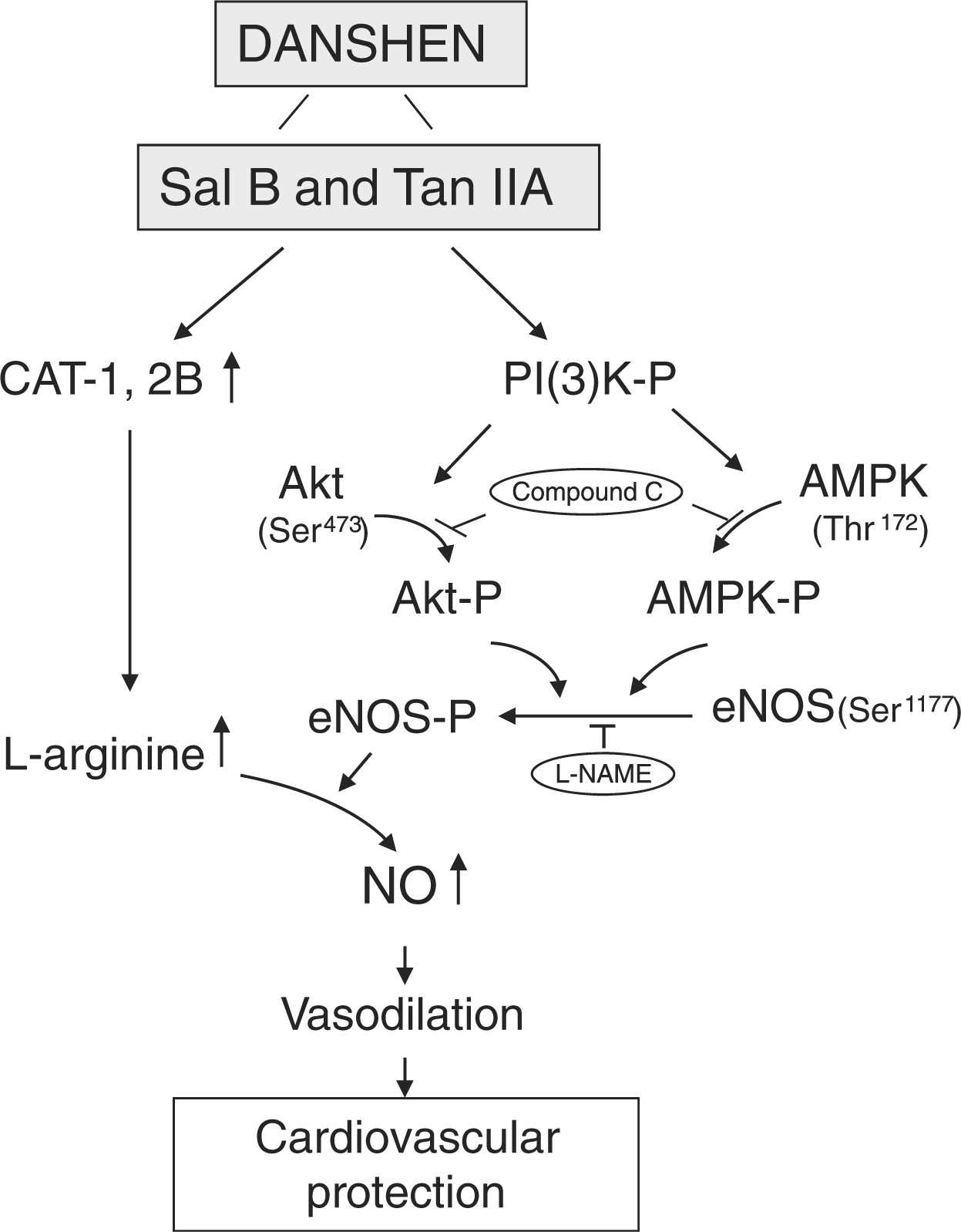
The pathophysiology of acute coronary syndromes involves phenomenon-like platelet activation, thromboxane A2 activation, vessel spasm, and coronary vessel blockade. This is associated with increased collagen deposition, interstitial DNA synthesis, and manifested by numerous structural changes in the heart including left ventricular dilatation, left ventricular hypertrophy and left ventricular wall thinning [Lutgens et al. 1999]. Modern cardiology clinics use a multipronged approach to limit the infract size including platelet stabilization, beta blockers, ACE inhibitors, rapid reperfusion, etc. The results of the present study show that both Sal B and Tan IIA mediate cardioprotection at clinically useful dosages (Figure 1A and 1B and Table 1) due to their ability to generate NO (Figures 1A and B, and 5A and B). Danshen extract is commonly used in clinics at the dosage of 6.56 mg/kg without any clinically significant side effects [Zhou et al. 2005]. The accuracy of these observations is strengthened by the previous reports of success of compound salvia pellet (CSP), in the management of angina pectoris. CSP consists of active herbal ingredients extracted from Danshen (Salvia miltiorrhiza), Sanqi (Panax notoginseng), and Borneol (Cinnamomum camphora) and has shown a significant effect on the improvement of angina symptoms and ECG results at least in comparison with nitrates [Wang et al. 2006].
The integrity of the vascular endothelium has been known to be pivotal for cardioprotective activity by virtue of its capacity to constitutively generate NO since the early 1980s. Today NO is regarded as one of the most important mediators of biological processes in the heart and blood vessels due to its vasodilator, antithrombotic and anti-inflammatory effects [Strijdom et al. 2009]. eNOS is the primary physiological source of NO regulating cardiovascular homeostasis. eNOS activity has been shown to be regulated by various mechanisms such as the interaction of eNOS with proteins including caveolin-1, calmodulin (CaM), heat shock protein 90, the B2 receptor, dynamin-2, as well as enzyme translocation and membrane phosphorylation [Strijdom et al. 2009]. Phosphorylation and dephosphorylation networks complement acylation and calmodulin as major posttranslational regulatory influences on eNOS activity [Dudzinski and Michel, 2007]. AMPK is an important intracellular signaling pathway in heart. Mutations in AMPK have been shown to cause an inherited syndrome of hypertrophic cardiomyopathy and ventricular pre-excitation [Kim et al. 2009]. Vascular endothelial growth factor (VEGF), shear stress and estrogen activate a signaling pathway involving phosphatidyl-inositol-3-kinase (PI3K) to cause eNOS phosphorylation via protein kinase B (Akt) phosphorylation at Ser473. Akt is a well-established survival signal inside the heart. It has also been demonstrated that PI3K and Akt lie upstream and can activate eNOS [Dimmeler et al. 1999; Fulton et al. 1999]. In this report, we demonstrated that the AMPK-PI3K-Akt pathway is composed of important signaling cascades that mediate eNOS activation in vascular endothelial cells induced by Sal B and Tan IIA (Figure 4). Our findings are consistent with previous in vivo studies with remodeling in rats demonstrating that eNOS expression-increasing effects of Danshen extracts play significant roles in hypoxic pulmonary hypertension [Chen et al. 2003], and Tan IIA increased eNOS expression and phosphorylation which induces vasodilatation and blood pressure reduction [Kim et al. 2007].
Arginine is an important substrate for metabolic pathways such as NO, agmatine, creatine, and urea synthesis. Its supply may become rate limiting for any of these enzymatic reactions [Closs et al. 2004]. Endothelial cells generate NO from L-arginine via the catalytic action of eNOS. We demonstrated here that Sal B and Tan IIA significantly stimulated L-arginine uptake mediated by the Na+-independent system y+ carrier, which shows that arginine transport through the plasma membrane is not energized by coupling to the Na+ gradient (Figure 5). Thus, a single Na+-independent transport system termed system y+ is postulated to be the major entry route for CAAs [Closs et al. 2004].
System y+ activity can be mediated by at least three different CAT proteins: CAT-1, CAT-2B, and CAT-3 [Closs et al. 2004]. Our work also shows the role of Sal B and Tan IIA in the upstream regulation of the y+ system by CAT-1 and CAT-2B mRNA for the generation of additional NO whenever needed by the cells (Figure 7). CAT-1–CAT-3 all mediate Na+-independent transport of CAAs, but differ in their substrate affinities and sensitivities to transstimulation. CAT-1 exhibits a broad expression pattern which is highly regulated on the transcriptional and posttranscriptional level. Among the factors reported to increase CAT-1 mRNA (and system y+ activity) are IL-1, insulin, glucocorticoids, angiotensin II, platelet-derived growth factor, and TNF-α. CAT-2B is generally co-expressed with CAT-1 and can be induced in many cell types by treatment with cytokines or lipopolysaccharide (LPS), often together with inducible nitric oxide synthase (iNOS). CAT-2B might be necessary for efficient arginine transport in cells with low intracellular levels of CAAs. In addition, another CAT protein, CAT-2A (an alternative splice variant of the CAT-2 transcripts), exhibits transport properties.
In summary, our work has demonstrated the cardioprotective function of the two most active components of Danshen, Sal B and Tan IIA, through multiple targets concerned with NO production. We observed for the first time that Sal B and Tan IIA stimulated L-arginine uptake by increasing CAT expression in addition to eNOS phosphorylation through the AMPK–PI3K–Akt pathway. Our findings establish that NO is the key factor that mediates the cardioprotective effects of Sal B and Tan IIA. Danshen may thus play a pivotal role in the management of myocardial ischemia in future. However, further study of the mechanism of Danshen-mediated NO generation and the observed cardioprotection is needed.
Footnotes
Acknowledgment
The authors thank Ms Melissa Philbrick for editing the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China NNSFC-30672740.
Conflict of interest statement
None declared.
