Abstract
Background
Inhibition of platelet activation is a cornerstone of adjunctive medical treatment during and after percutaneous coronary interventions (PCIs) with stent implantation (PCI-S) in order to prevent acute and long-term thrombotic complications. Antiplatelet therapy was shown to reduce the occurrence of nonfatal myocardial infarction (MI), nonfatal stroke, or vascular death [Antithrombotic Trialists’ Collaboration Collaborative, 2002]. Dual antiplatelet therapy with aspirin and clopidogrel provides incremental platelet inhibition (compared with either agent alone) and more effective suppression of adverse ischemic events and has been studied in the settings of medical therapy and PCI as well as in stroke prevention and treatment [Holmes et al. 2009]. However, dual therapy (DT) with aspirin and clopidogrel is associated with greater bleeding risks [Becker et al. 2008; McQuaid and Laine, 2006; Peters et al. 2003]. Current American College of Cardiology/American Heart Association/Society for Cardiac Angiography and Interventions (ACC/AHA/SCAI) as well as the European Society of Cardiology (ESC) guidelines recommend prolonged dual antiplatelet therapy for at least 12 months after placement of drug-eluting stents (DESs) [King et al. 2008; Silber et al. 2005].
Anticoagulation with warfarin is the recommended treatment for patients at risk of thromboembolic events due to atrial fibrillation (AF), mechanical heart valves, deep vein thrombosis, pulmonary embolism and left ventricular thrombi. Randomized clinical trials have shown that warfarin is superior to either ASA or clopidogrel for prevention of stroke in patients with AF, and observational data seem to have established it as the standard antithrombotic treatment in patients with mechanical valve prostheses. The number of patients who have an indication for both DT and warfarin is increasing, since more patients who are already on oral anticoagulation are scheduled for percutaneous coronary interventions and some patients who are on DT will develop a medical condition which requires warfarin changing regimen to triple antithrombotic therapy (TT).
Optimal antithrombotic therapy after PCI is currently undefined in patients requiring long-term anticoagulation. This question is particularly vexing among patients who have received DESs and who, with the indication for long-term treatment with dual antiplatelet therapy, have both a need for as well as prolonged exposure to the risk of triple therapy. Current guidelines for acute coronary syndrome (ACS) and PCI recommend selected use of the combination of low-dose aspirin, clopidogrel and warfarin using a more narrow target International Normalized Ratio (INR) of 2.0–2.5 [King et al. 2008; Anderson et al. 2007; Silber et al. 2005]. Studies comparing TT of warfarin, aspirin and clopidogrel with DT of aspirin and clopidogrel have yielded conflicting results. A meta-analysis of these studies was performed to evaluate safety and efficacy of TT
Methods
A protocol was developed prospectively, detailing the objectives, criteria for study selection, and approach to assessing the study quality, primary outcome, and methodology.
Literature search
We performed a computerized search to identify all relevant studies published in English language up to and including January 2009 in EMBASE, CINAHL, PubMed, and the Cochrane database. The keywords used for search were: ‘percutaneous coronary intervention’, ‘angioplasty’, ‘coronary stenting’, ‘bleeding complications’, and ‘major adverse cardiovascular events’.
Study selection
All titles and abstracts from the results of our computerized search were reviewed by the authors for potential inclusion in our study. We also went into the related links of all relevant articles. In addition to our computerized search, we manually reviewed the reference list of all retrieved articles to complete our search. The study selection process is outlined in Figure 1.
Study selection process.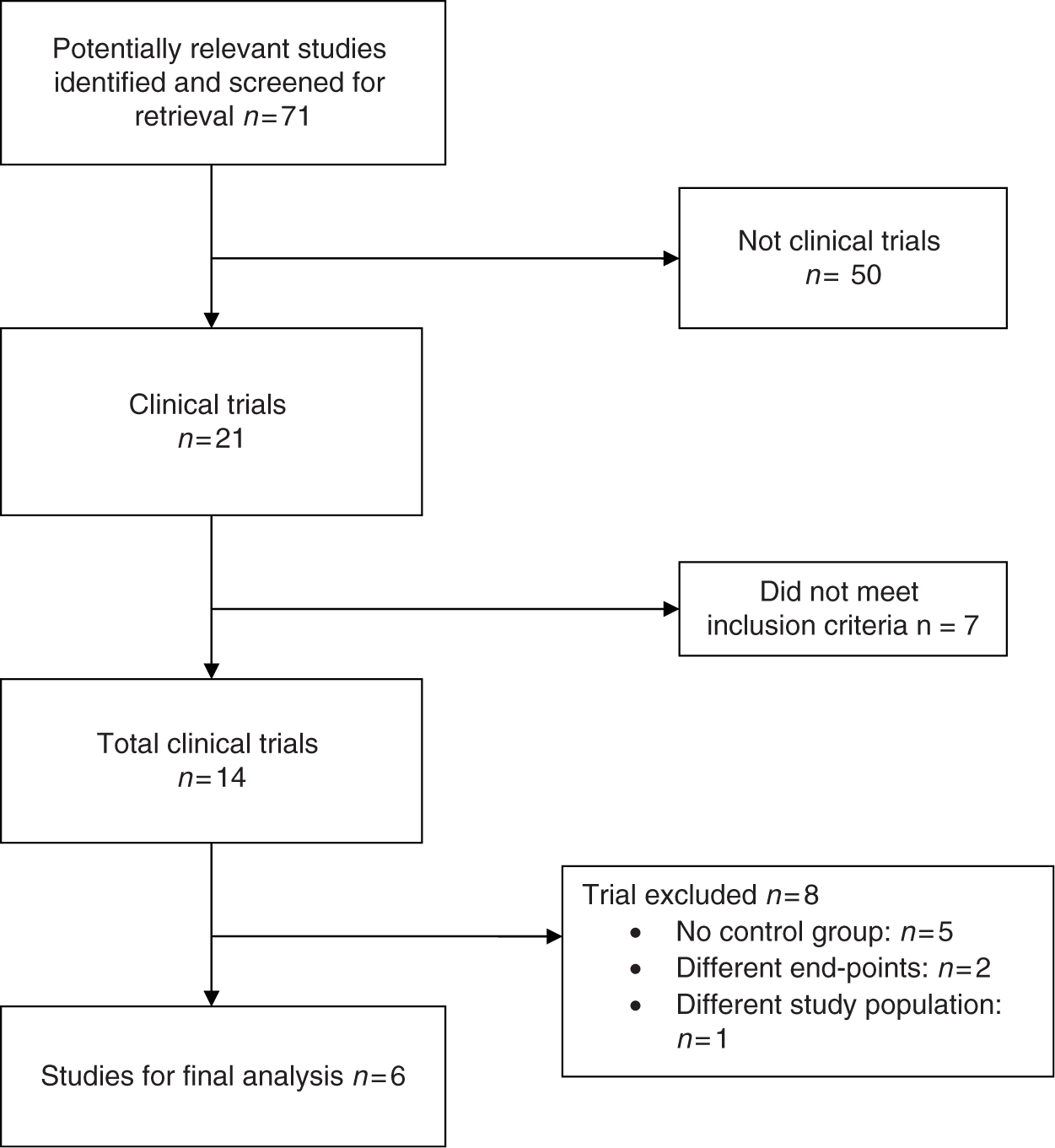
Inclusion criteria
All studies had to meet all of the following criteria to be included in the analysis: Include patients undergoing PCI. Compare TT with DT group. Report at least one of the cardiovascular outcomes: all cause mortality, nonfatal MI, coronary artery revascularization, stroke/transient ischemic event (TIA). Report bleeding complications.
Exclusion criteria
Studies that did not meet the above criteria were excluded.
Data abstraction
After identifying all relevant articles, we extracted characteristics of the study (author, year, design, duration, sample size, antithrombotic regimen use, total mortality, cardiovascular mortality, bleeding complications, and follow-up percentage) and participants (age, gender). Two reviewers independently extracted data and assessed outcomes. The interrater agreement was 90%, and disagreements were resolved by consensus.
Statistical analysis
The statistical analysis was performed by the Comprehensive Meta-Analysis software package (version CM 2.2, Biostat, Englewood, NJ). Heterogeneity of the studies was analyzed using Cochran’s
Results
Literature search
A total of 113 articles were identified of which 60 were potentially relevant studies and screened for retrieval. After title and abstract screening, 46 studies were excluded and the remaining 14 studies were retrieved for a more detailed evaluation. Out of these 14 studies, 8 were excluded as they did not meet inclusion criteria. Five studies did not have control groups [Anand et al. 2007; Karjalainen et al. 2007; Rubboli et al. 2007; Porter et al. 2006; Orford et al. 2004], two studies had different end points [Konstantino et al. 2006; Lip and Karpha, 2006] and the study population was different in one study [Nguyen et al. 2007]. Thus, six studies were included in the final analysis [Manzano-Fernandez et al. 2008; Ruiz-Nodar et al. 2008; Sarafoff et al. 2008; DeEugenio et al. 2007; Khurram et al. 2006; Mattichak et al. 2005].
Overview of study and patient characteristics
Study characteristics.
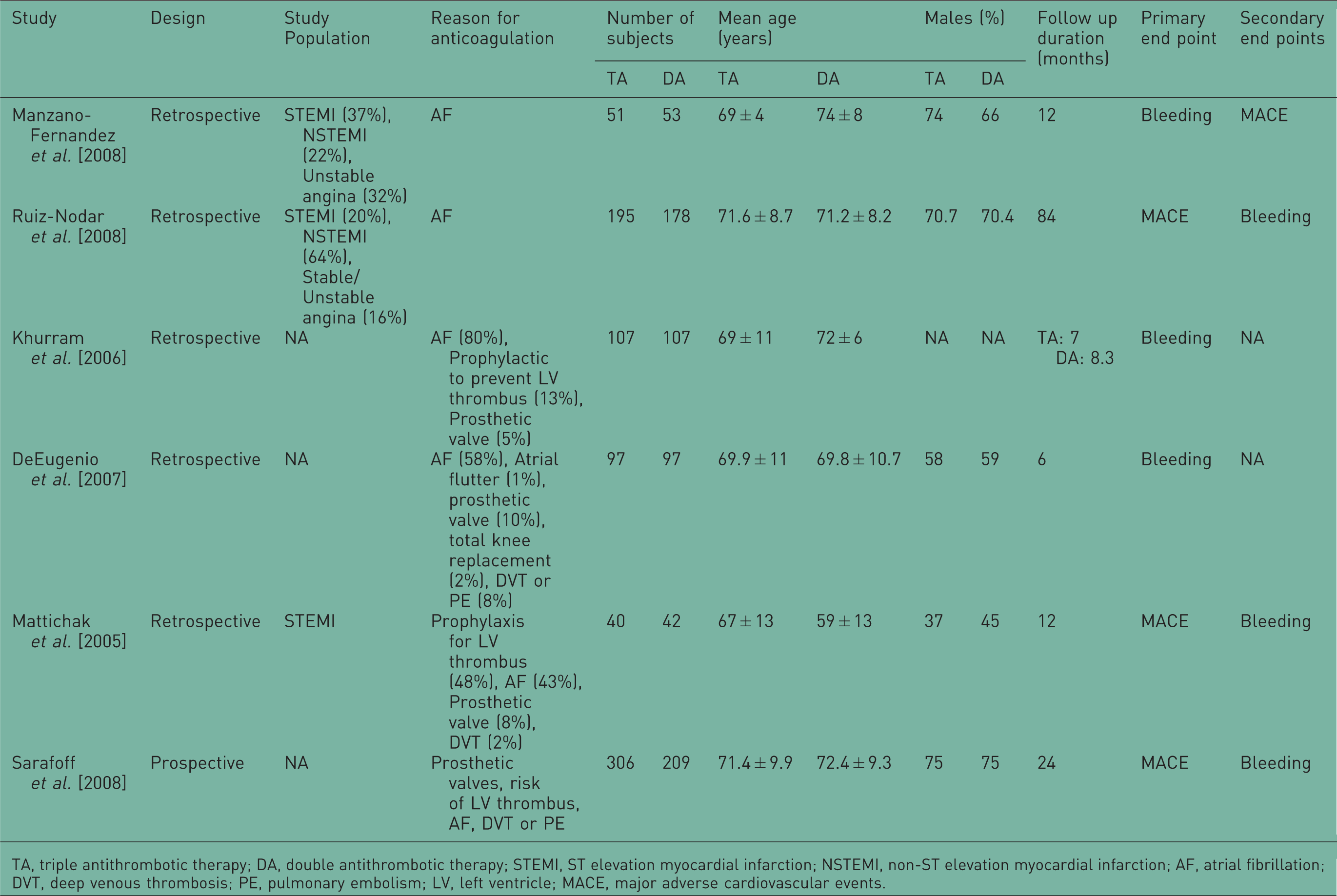
TA, triple antithrombotic therapy; DA, double antithrombotic therapy; STEMI, ST elevation myocardial infarction; NSTEMI, non-ST elevation myocardial infarction; AF, atrial fibrillation; DVT, deep venous thrombosis; PE, pulmonary embolism; LV, left ventricle; MACE, major adverse cardiovascular events.
Outcome definitions
Major adverse cardiovascular events (MACEs) was defined as any or all of cardiovascular death, MI, need of new revascularization, stent thrombosis, or thromboembolic complications during the follow up. Myocardial infarction was diagnosed when periprocedural levels of creatine kinase (CK) or its MB isoenzyme (CK-MB) increased at least two to three times above than the upper limit or when new pathological Q waves developed on postprocedural ECG.
Major bleeding was defined differently in different studies. Ruiz-Nodar and colleagues defined major bleeding as a decrease in the blood hemoglobin level of more than 5.0 g/dl (including the period around the coronary interventional procedure), the need for the transfusion of 2 or more units of blood, the need for corrective surgery, the occurrence of an intracranial or retroperitoneal hemorrhage, or any combination of these events [Ruiz-Nodar et al. 2008]. DeEugenio and colleagues defined major bleeding as fatal bleeding, bleeding at a critical site (retroperitoneal, intracranial, or intraocular), bleeding that led to hemodynamic compromise requiring treatment, bleeding that required intervention (surgical or endoscopic) or decompression of a closed space to stop or control the event, bleeding that required transfusion of one or more units of packed red blood cells or whole blood, or bleeding that led to a decrease in hemoglobin of 3 g/dl or more [DeEugenio et al. 2007]. Sarafoff and colleagues defined major bleeding as intracranial (confirmed by computed tomography or magnetic resonance imaging of the head) or clinically significant overt signs of hemorrhage associated with >50 g/l decrease in hemoglobin level or an absolute decrease in hematocrit >15% (when hemoglobin level was not available) [Sarafoff et al. 2008]. Minor bleeding was defined as blood loss and a decrease in hemoglobin level of 30–50 g/l (or a decrease in the hematocrit between 9% and 15%) or a decrease in hemoglobin level >40 g/l (or >12% in hematocrit) if no bleeding site was identifiable. Khurram and colleagues defined major bleeding as bleeding that was significantly disabling, intraocular or required greater than two units of blood transfusion [Khurram et al. 2006]. Minor bleeding was defined as other bleeding that led to an interruption of the antiplatelet or anticoagulant medication. Mattichak and colleagues defined major bleeding as gastrointestinal (GI) bleeding or bleeding requiring blood transfusion [Mattichak et al. 2005]. Manzano-Fernandez and colleagues defined major bleeding complications as fatal bleeding, a decrease in the blood hemoglobin level ≥4 g/dl, need for transfusion of at least two units of blood, need for corrective surgical intervention, the occurrence of intracranial or retroperitoneal bleeding, or any combinations of these events [Manzano-Fernandez et al. 2008].
End points
All of the six trials included in the study had bleeding events as their end point with three trials having it as a primary end point [Manzano-Fernandez et al. 2008; DeEugenio et al. 2007; Khurram et al. 2006] and three trials as a secondary end point [Ruiz-Nodar et al. 2008; Sarafoff et al. 2008; Mattichak et al. 2005]. Three trials out of six had ischemic events as their primary end point [Ruiz-Nodar et al. 2008; Sarafoff et al. 2008; Mattichak et al. 2005] while it was the secondary end point in one trial [Manzano-Fernandez et al. 2008]. The definitions of end points were as used in individual trials.
Heterogeneity testing
Results of heterogeneity testing are shown in Table 2. Studies were heterogeneous for major bleeding outcome and homogeneous for ischemic outcomes. So, the Mantel–Haenszel random effect model was used for the analysis of major bleeding events (Figure 1), while the Mantel–Haenszel fixed effect model was used for analyzing ischemic outcomes (Figure 2).
Ischemic outcomes. Results of heterogeneity testing.

Clinical outcomes
Effect of on ischemic events
Overall there were a total of 173 (19.39%) MACEs of which 78 (15%) were in the TT group while 95 (27.9%) were in the DT group. Compared with the DT group, risk of MACEs was significantly lower in the TT group (RR: 0.72; CI: 0.56–0.98;
Effect on bleeding complications
There were a total of 98 bleeding events with 71 (8.91%) events in the TT group and 27 (3.93%) events in the DT group. Compared with patients receiving DT, the risk of major bleeding was significantly higher in the TT group (RR: 2.74, CI: 1.08–6.98; Major bleeding outcomes.
Discussion
Our meta-analysis shows that TT is beneficial in reducing the incidence of ischemic cardiovascular events (MI, stroke, and need for coronary revascularization) but is associated with a higher incidence of major bleeding complications. Overall there were a total of 173 (19.39%) major adverse ischemic events of which 78 (15%) were in the TT group while 95 (27.9%) were in the DT group and this difference was statistically significant (
The principal concern associated with TT is the risk of bleeding or transfusion. In our meta-analysis, there were a total of 98 bleeding events with the rate of bleeding complications in TT group as 8.91% and it was significantly higher than the DT group (3.93%). The frequency of such events in reported series varies, with up to 21% of patients needing a transfusion and bleeding events typically involve the GI tract. This frequency might increase with longer durations of TT, which has been shown to directly correlate with bleeding risk and might influence mortality in follow up after PCI [Holmes et al. 2009]. Major bleeding following hospital discharge for PCI or ACS was shown to be increased in patients receiving aspirin and a thienopyridine in combination with warfarin, with 3.2- to 6.6-fold increased risk compared with DT alone [DeEugenio et al. 2007; Khurram et al. 2006]
Options to reduce the incidence of bleeding complications include using lowest possible dose of aspirin (75 or 81 mg), standard dose of clopidogrel (75 mg), and close monitoring of INR to keep it within the 2.0–2.5 range [Holmes et al. 2009]. Although recent data have identified a specific interaction with proton pump inhibitors (PPIs), which might decrease platelet inhibition by clopidogrel [Gilard et al. 2008], the clinical significance of this interaction remains to be determined. The Clopidogrel and the Optimization of Gastrointestinal Events (COGENT) [Bhatt et al. 2010] trial looked at the specific issue of the effect of PPI on clopidogrel. The specific question addressed was the link between changes in platelet function testing in the laboratory
Current guidelines for ACS and PCI recommend selected use of the combination of low-dose aspirin, clopidogrel, and warfarin using a more narrow target INR of 2.0–2.5 [King et al. 2008; Anderson et al. 2007]. On the other hand, the most recent ACC/AHA atrial fibrillation guidelines [Fuster et al. 2006] suggest no indications for using warfarin plus DT and recommend that aspirin be omitted in patients undergoing PCI, despite the fact that no randomized trials of warfarin plus thienopyridine in PCI have been conducted.
As with any meta-analysis, one of the limitations of our study is the difference in the definitions of the end points in the component trials, such as the definition of CAD/MI, was different in various studies. Similarly, baseline characteristics between the two groups cannot be compared completely in most meta-analyses because of differences in the study protocols across the component trials. Moreover, all studies have been nonrandomized and most retrospective. The major limitation of the data to date is that most have not compared the TT group with a comparable control group of patients with the same indication for Coumadin. Making this even more complex, each of the studies used different definitions for each of the end points (particularly bleeding) and the control groups also varied from consecutive patients to random to matched patients. Also, there is a potential for publication bias but the trials in our analysis had different results and it should reduce this potential risk. Lastly, ischemic events, stroke, MI, or stent thrombosis, are less well studied than are bleeding events, with only three of the six trials reporting those outcomes. Also many of these trials were small trials with limited ability to assess clinical outcomes. The best and perhaps only way to answer these shortcomings is a randomized trial.
Conclusion
In patients requiring long-term anticoagulation after PCI, TT may be superior to DT in reducing the incidence of MACEs; however, the risk of major bleeding complications is increased significantly (Figure 4). TT should be individualized on the appropriateness, duration, and safety of such a regimen.
Efficacy and safety of dual 
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
None declared.
