Abstract
Peripartum cardiomyopathy is defined as heart failure secondary to left ventricular (LV) dysfunction with a left ventricular ejection fraction <45% occurring towards the end of pregnancy or in the months following delivery without other identifiable cause. Pathogenesis of peripartum cardiomyopathy and the role of genetic variants are unknown. We present a previously healthy 33-year-old woman in her first pregnancy with acute onset of heart failure postpartum. She developed cardiogenic shock four days after caesarean section and was treated with levosimendan, cabergoline, ramipril and bisoprolol. She used a wearable cardioverter/defibrillator for 3 months. After 8 months, she was free of symptoms with normal left ventricular function and brain-natriuretic-peptide-levels. Genetic analysis was carried out due to a positive family history and disclosed a heterozygous variant c7627dupA in the TTN gene. Genetic analysis in patients with a positive family history should be carried out since that may provide insights in the pathogenesis of heart failure due to systolic dysfunction including peripartum cardiomyopathy.
Introduction
The leading cause of postpartum heart failure (HF) and cardiogenic shock is peripartum cardiomyopathy (PPCM), defined as HF secondary to left ventricular (LV) systolic dysfunction with an LV ejection fraction (LVEF) < 45%, occurrence towards the end of pregnancy or in the months following delivery and no other identifiable cause of HF.1,2 In PPCM, recovery of systolic function after 6 months occurs only in 46%. 3 The role of genetic variants, especially in recovery of PPCM, is largely unknown. 4 Thus we report a patient with acute HF postpartum with rapid recovery of systolic dysfunction and a likely pathogenic variant of the Titin gene (TTN).
Case report
A 33-year-old Caucasian woman developed cough, dyspnoea and orthopnea, starting suddenly 4 days after the caesarean delivery of her first child. The indications for caesarean section were breech position of the child. The patient was healthy before and during pregnancy except for gestational diabetes, which was treated with insulin. She did not take any other medication. Three years earlier a missed miscarriage was identified at 12 weeks. Her father suffers from HF due to systolic dysfunction.
She was discharged from the hospital but returned on the next day because of increasing dyspnoea. Blood pressure was 100/60 mmHg, the electrocardiogram (ECG) showed sinus tachycardia, 149/min, peripheric low voltage, poor R-wave progression, and negative T-waves in I, aVL and V4-V6. Chest X-ray showed an enlarged cardiac silhouette and bilateral shadows in both lungs. Chest computed tomography showed bilateral densities, pleural effusions, and no pulmonary embolism. Pneumonia was suspected because of leukocytes 18.2 x 109/L (normal 3.9–10.2), C-reactive protein 24.6 mg/L (normal −5.0), Interleukin-6 34.4 p/mL (normal −7.00), why antibiotic therapy with meropenem was initiated. Despite nasal oxygen, the dyspnoea worsened, and the oxygen saturation decreased to 88%. Emergency echocardiography showed systolic dysfunction with an LVEF 25%, moderate mitral regurgitation and 5 mm pericardial effusion (Table 1). Because of cardiogenic shock due to suspected PPCM, she was transferred to a cardiologic intensive care unit (ICU) three days after symptom onset.
Echocardiographic findings, QTc and NT-Pro BNP levels.
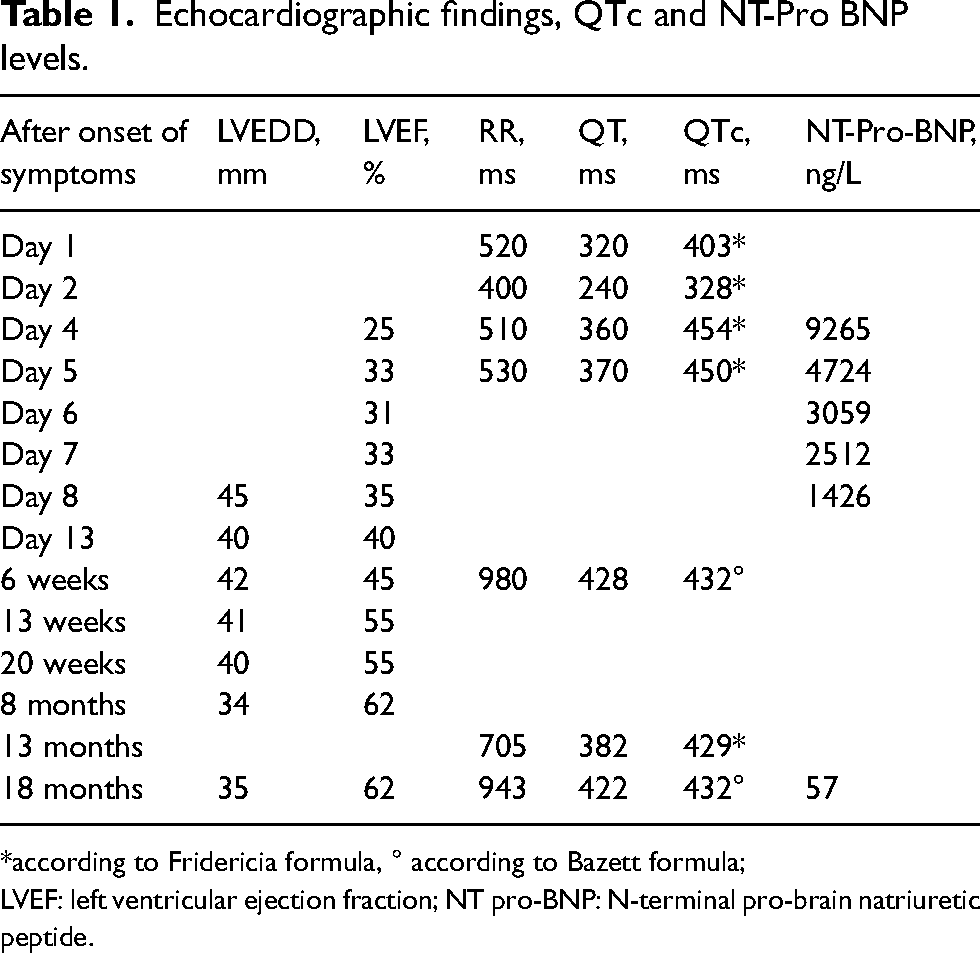
*according to Fridericia formula, ° according to Bazett formula;
LVEF: left ventricular ejection fraction; NT pro-BNP: N-terminal pro-brain natriuretic peptide.
At admission to the ICU the patient was awake and oriented, 156 cm height and 50 kg weight (BMI 20.5 kg/m2). She was severely distressed with a heart rate of 130/min, respiratory rate 40/min and blood pressure 95/70 mmHg. Crackling sounds were found on both lungs. Results of echocardiography, ECG and N-terminal pro-brain natriuretic peptide levels are listed in Table 1. Since oxygen saturation was only 93% with 6 l/min nasal oxygen, she received respiratory support from a nasal high-flow system. Levosimendan 0,1 µg/kg/min was started. Furosemide, esmolol and low-molecular weight heparin in therapeutic dosage were initiated. Hypalbuminemia (27.6 g/L, normal 35–52) and hypokalaemia (2.9 mmol/L, normal 3.4–4.5) were corrected. For ablactation 2 mg cabergoline was given. After thoracentesis and continuous positive airway pressure ventilation, the respiratory situation improved. On day 7, levosimendan was discontinued, bisoprolol and ramipril were started. Echocardiography on day 8 showed an improved LVEF of 35% and the mitral regurgitation resolved. She declined cardiac magnetic resonance imaging (cMRI). She was discharged on day 13 with bisoprolol 3.75 mg/d, spironolactone 25 mg/d, ramipril 2.5 mg/d and recommended to use a wearable cardioverter/defibrillator (WCD) for the following 3 months.
Follow-up showed an improvement of the clinical condition and systolic function (Table 1). No arrhythmias were detected by the WCD. Since LV function remained excellent, ramipril and spironolactone were discontinued after 18 months and she remained on bisoprolol 1.25 mg/d. Twenty-two months after delivery, she became pregnant again, bisoprolol was changed to metoprolol 23.75 mg/d. In the tenth week of gestation she suffered from a missed miscarriage. Thirty-two months after delivery she is free of any cardiac symptoms and wishes to become pregnant again. Unfortunately, she still declines cMRI.
Because of the family history, genetic analysis was carried out which showed a likely pathogenic heterozygous variant c.7627dupA (p.Thr2543AsnfsTer11) in the TTN gene. No analysis, so far, was carried out in the patient's father.
Discussion
This woman with a novel gene-truncating variant of the titin gene regained full systolic function within 8 months after postpartum cardiogenic shock. Most probably, the cause of systolic dysfunction was PPCM. The aetiology of PPCM is uncertain. Prolactin fragments and placental angiogenetic factors seem to be involved. 1 Additionally, it is assumed that in women with a genetic predisposition, hemodynamic and hormonal changes during pregnancy may induce HF. 2
Pulmonary embolism, acute coronary syndrome, Takotsubo syndrome and myocarditis were largely excluded by history and absence of preceding infections, although cMRI and coronary angiography were not performed. 2 A pre-existing cardiomyopathy manifesting only after delivery cannot be excluded because no previous ECG or echocardiographic investigations were carried out.3,5,6
A family history of cardiomyopathy is found in 17% of PPCM patients. 7 Gene variants in PPCM comprise TTN, TNNC1 and LMNA.4,8 The variant c.7627dupA has not yet been described and is not known in the common population. 9
Pharmacotherapy according to the European Society of Cardiology (ESC) was combined with levosimendan and cabergoline. 2 Since patients with PPCM are sensitive to the toxic effects of beta-adrenergic agonists, levosimendan may be the preferred inotrope and may be considered in PPCM with cardiogenic shock according to the ESC.10–12 Bromocriptine, which is recommended by the ESC as class IIb, level B to enhance recovery in PPCM, was not available in the hospital. 10 Cabergoline, a synthetic ergoline that affects the dopamine D2 receptor, is recommended as an alternative.2,13 The use of WCD is recommended due to an increased risk of malignant arrhythmias in PPCM. 14
Gestational diabetes associated with PPCM is reported, but the pathogenesis remains unknown. 15
The duration of HF therapy in PPCM after LV-recovery remains uncertain. Reassessment of LV function is advised after drug discontinuation, followed by annual clinical and echocardiographic assessment. 10
Regarding subsequent pregnancies, patients with remaining LV dysfunction showed more recurrences of PPCM and increased mortality during a subsequent pregnancy than patients with recovered LV function. Multidisciplinary monitoring is recommended for subsequent pregnancies.14,16
The cause of systolic dysfunction could not be definitively assessed since coronary angiography and cMRI were not performed. In similar cases, these investigations and genetic analysis in patients with positive family history should be carried out to elucidate the pathogenesis of HF, including PPCM.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The Medical University of Vienna does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained for the publication of this case report.
Guaranteeing author
Eva Karner.
Contributorship
EK wrote the first draft of the manuscript. CS reviewed and edited the manuscript. All authors reviewed and approved the final version of the manuscript.
