Abstract
Background:
Pulmonary alveolar proteinosis (PAP) typically manifests as diffuse lung disease, while nodular PAP presenting as pulmonary nodules is extremely rare and often radiologically indistinguishable from early-stage lung adenocarcinoma (LUAD).
Objectives:
This study aims to establish an accurate diagnostic algorithm to prevent unnecessary invasive procedures in patients with nodular PAP.
Design:
This study was a retrospective, single-center study.
Methods:
This study enrolled 16 pathologically confirmed nodular PAP patients and 52 early-stage LUAD patients, systematically comparing their clinical characteristics and biochemical markers. A radiomics model was developed based on CT imaging features, followed by a multimodal model incorporating significant clinical and biochemical discriminators.
Results:
The nodular PAP cohort demonstrated a 3:5 male-to-female ratio, equivalent distribution of solitary versus multiple nodules, and nodule diameters spanning 3–28 mm. Nodular PAP patients were significantly younger (mean 51.4 vs 67.0 years, p < 0.001), had higher body mass index (BMI, 25.8 ± 2.5 vs 23.7 ± 2.8 kg/m², p < 0.01), and lower lactate dehydrogenase levels (LDH, 188.44 ± 26.71 vs 214.79 ± 56.84 U/L, p = 0.01) compared to LUAD patients. The radiomic model achieved an AUC (area under the curve) of 0.91, while the multimodal model combining clinical and biochemical features showed similar performance (AUC = 0.97).
Conclusion:
This study provides the characterization of nodular PAP and establishes radiomic and multimodal diagnostic models with similar accuracy, which are reliable tools to make a differential diagnosis between nodular PAP and LUAD and to prevent unnecessary surgery.
Plain language summary
A rare benign lung disease (nodular PAP) is highly similar with early-stage lung cancer on CT imaging, often leading to misdiagnosis and unnecessary surgeries. This study developed an innovative diagnostic method by analyzing data from 16 cases of benign nodules nodular PAP and 52 cases of lung cancer. The results showed that patients with nodular PAP tend to be younger, have a higher body mass index, and exhibit lower lactate dehydrogenase levels. Two models were established: an imaging-based model achieved an accuracy of 90%, while a comprehensive model combining imaging and clinical data reached an accuracy of 93%. This approach effectively distinguishes between the two diseases, providing a reliable new tool to prevent unnecessary surgeries caused by misdiagnosis.
Keywords
Introduction
Pulmonary alveolar proteinosis (PAP) is a rare pulmonary disorder characterized by progressive accumulation of surfactant material within the alveoli, leading to an increased risk of secondary infections and/or pulmonary fibrosis and hypoxemic respiratory failure. Based on its underlying pathogenesis, PAP is classified into three distinct subtypes: primary, secondary, and congenital. 1 Primary PAP results from disruption of granulocyte-macrophage colony-stimulating factor (GM-CSF) signaling, which impairs the function of alveolar macrophages and neutrophils. This subtype is further subdivided into autoimmune (representing ~90% of all PAP cases) and hereditary forms.2,3 Autoimmune PAP is caused by neutralizing antibodies against GM-CSF, while hereditary PAP arises from mutations in colony-stimulating factor 2 receptor subunit alpha (CSF2RA) or colony-stimulating factor 2 receptor subunit beta (CSF2RB) genes encoding the α-subunit and common β-subunit of the GM-CSF receptor, respectively.4–6 Secondary PAP develops due to reduced alveolar macrophage numbers and/or function, typically associated with hematologic malignancies, occupational dust exposure, or immunodeficiency disorders. 7 In contrast to primary PAP, this form reflects impaired surfactant clearance secondary to underlying systemic conditions rather than intrinsic GM-CSF pathway abnormalities. Congenital PAP results from mutations in surfactant metabolism-related genes (e.g., Surfactant Protein B gene (SFTPB), Surfactant Protein C gene (SFTPC), ATP-Binding Cassette Subfamily A Member 3 gene (ABCA3)), leading to dysfunctional production and clearance of pulmonary surfactant without GM-CSF pathway involvement. This subtype typically presents in infancy with distinct histopathological features.1,8
The hallmark imaging features of PAP typically include bilateral, symmetric ground-glass opacities (GGOs) with superimposed interlobular septal thickening, classically described as the “crazy-paving” pattern.9,10 Some patients may exhibit areas of consolidation with air bronchograms, representing surfactant-filled alveoli surrounding patent airways. 11 On the contrary, nodular PAP, which is presented as a single nodule or multiple nodules and confirmed through histopathological examination with characteristic periodic acid-Schiff (PAS)-positive proteinaceous material within alveolar spaces, is rarely reported in the medical literature. A Chinese retrospective study of secondary PAP cases identified scattered nodules in four of nine patients, though all cases concurrently exhibited typical diffuse GGOs. 12 To date, well-documented cases of nodular PAP without concomitant diffuse parenchymal abnormalities remain scarce.
Lung adenocarcinoma (LUAD) demonstrates malignant cellular proliferation, with definitive immunohistochemical confirmation through markers such as thyroid transcription factor-1 (TTF-1) and Napsin A. 13 Clinically, lung cancer patients frequently show elevated tumor markers including carcinoembryonic antigen (CEA), cytokeratin 19 fragment (CYFRA21-1), and neuron-specific enolase (NSE), which are typically absent in PAP. 14 These differential features suggest that integrating multiple clinical parameters could enhance preoperative discrimination between nodular PAP and malignant pulmonary nodules.
The radiological similarity between nodular PAP and malignant pulmonary nodules, particularly LUAD, poses a significant diagnostic challenge. Misinterpretation often leads to unnecessary surgical resection, highlighting an urgent need for reliable discriminative tools. Recent advances in artificial intelligence (AI)-based radiomic analysis have demonstrated considerable potential in differentiating benign and malignant pulmonary lesions, with several studies reporting diagnostic accuracies exceeding 85% for conventional machine learning approaches and up to 92% for deep learning models when analyzing indeterminate pulmonary nodules.15–18 However, these developments have predominantly focused on distinguishing typical malignant nodules from granulomas or other benign etiologies, leaving a significant knowledge gap regarding their applicability in differentiating rare mimics such as nodular PAP.
This study aims to address the differential diagnosis between nodular PAP and early-stage LUAD by presenting a retrospective cohort of surgically resected nodular PAP cases in comparison with LUAD controls through an integrated analysis of clinical, biochemical, and radiomic profiles. Our findings are expected to provide the first comprehensive characterization of nodular PAP and establish a practical framework for differential diagnosis.
Methods
Study population
This study is a single-center retrospective study on nodular PAP. We systematically retrieved all medical records from Shanghai Chest Hospital between June 2016 and December 2024, and then identified and included 16 patients with pathologically diagnosed nodular PAP. A study cohort consisting of 16 PAP patients and 52 LUAD patients was ultimately formed by matching early-stage LUAD patients diagnosed by pathology with nodular PAP patients in a ratio of approximately 1:3.
The nodular PAP group included patients pathologically diagnosed with PAP according to the 2024 European Respiratory Society (ERS) guidelines for the diagnosis and management of PAP. 10 The LUAD group comprised patients diagnosed according to the 5th edition WHO Classification of Thoracic Tumors, including minimally invasive adenocarcinoma (MIA) and invasive adenocarcinoma (IAC). 13 All pathological specimens included in this study were independently evaluated by two senior pathologists, with a consensus diagnosis established for final pathological determination. Patients were excluded based on the following criteria: (1) concurrent pulmonary infections; (2) mixed pathological diagnoses; and (3) absence of high-resolution CT (HRCT) images within 4 weeks prior to surgery.
This study was conducted in accordance with the Declaration of Helsinki and with all relevant guidelines and regulations, and it adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental File 1). 19
Data collection
Demographic and clinicopathological characteristics were extracted from the hospital’s medical records database. Data collection and pre-processing were performed following standardized protocols to ensure consistency across all study subjects. Clinical data were collected from electronic clinical records, encompassing demographic characteristics, clinical parameters, pathological characteristics, and postoperative follow-up.
CT data acquisition
All CT scans were acquired in the supine position during end-inspiration breath-hold to minimize respiratory motion artifacts. Scanning parameters included 0.625–1.25 mm detector collimation, 0.64 pitch, 0.625–1.25 mm section thickness and interval, 512 × 512 or 1024 × 1024 matrix, 350–400 mm field of view (FOV), 120 kVp, and 220–300 mA.
DICOM series were converted to neuroimaging informatics technology initiative (NIfTI) format using SimpleITK (v2.4.1), preserving original Hounsfield units (HU) and spatial coordinates. Lung segmentation was performed using a pretrained U-Net model (R231 from lungmask v0.2.2), generating bilateral lung masks that were visually verified by two thoracic radiologists. 20 The volume of interest was defined by combining both masks with 5-slice cranial/caudal extensions to ensure complete lesion inclusion, followed by removal of extraneous thoracic structures. All images were resampled to an isotropic 512 × 512 × 128 matrix using B-spline interpolation (order = 3) and normalized to 0–256 grayscale range (−1200 to 600 HU window). Lesion segmentation was performed in ITK-SNAP (v4.2.0) by a board-certified thoracic radiologist with 10 years of experience, who manually delineated pulmonary nodules. All segmentations were independently verified by a second radiologist to ensure annotation accuracy. 21
Radiomic feature extraction and selection
Radiomic features were extracted from the segmented regions of interest (ROI) using the open-source PyRadiomics package (version 3.1.0, developed by the AIM Lab at Dana-Farber Cancer Institute/Harvard Medical School, Boston, USA). A comprehensive set of 1218 features was initially obtained, including first-order statistics, shape-based features, texture features (gray-level co-occurrence matrix, gray-level run-length matrix, gray-level size zone matrix), and higher-order wavelet features. 22 Feature selection was performed using a two-step approach. First, features demonstrating statistically significant intergroup differences were identified using independent samples t-tests with the significance level set at p < 0.05. These selected features were then standardized and subjected to principal component analysis (PCA). The optimal number of principal components was determined based on a cumulative variance threshold of 95%, which was subsequently used for further analysis.
Radiomic model construction and validation
In this step, statistically significant radiomics features were selected to train three distinct machine learning models: Random Forest (RF), Gaussian Naive Bayes (GaussianNB), and L2-regularized logistic regression. To address the substantial class imbalance (ratio 16:52), algorithm-specific countermeasures were implemented. For the RF and L2-regularized logistic regression models, we leveraged their inherent class-balancing capabilities by setting the class_weight parameter to “balanced,” which automatically adjusts weights inversely proportional to class frequencies. By contrast, GaussianNB, which lacks a native class_weight parameter and relies on the assumption of Gaussian-distributed features, required an alternative approach; conventional resampling techniques were deemed unsuitable as they would distort the underlying feature distributions. Instead, class balance was achieved by directly modifying the model’s prior probabilities based on the training set labels. To rigorously evaluate model performance under this imbalance, we employed a comprehensive set of metrics: the Matthews Correlation Coefficient (MCC), F1-score, area under the Precision-Recall curve (PR-AUC), and receiver operating characteristic (ROC) curves. In addition, performance was visualized using fivefold averaged confusion matrices. This multi-faceted evaluation strategy facilitated an unbiased comparative analysis and the identification of the optimal single-modality radiomics model.
Multimodal model construction and validation
First, statistically significant clinical variables were standardized using z-score normalization and then sequentially integrated into the PCA-reduced radiomics feature matrix. To ensure balanced contributions from multimodal data, the combined feature set underwent global re-standardization. Then, the RF classifier was used for training, and the internal verification was completed on the training data through fivefold cross-verification. Finally, the optimal cutoff value was determined by maximizing the Youden index. Subsequently, the per-fold area under the curves (AUCs) of the unimodal and multimodal models were compared using the Wilcoxon signed-rank test to assess whether the overall discriminative capacities of the two models differed significantly. In addition, the coefficient of variation and the range of the fivefold AUC values were calculated for each model to evaluate their relative stability. The model construction workflow is illustrated in Figure 1.

Model construction workflow.
Statistical analysis
Continuous and categorical variables were compared using the t-tests or Wilcoxon tests and χ² test or Fisher’s exact tests, respectively. Radiomic and multimodal models were constructed and assessed using RF, GaussianNB, and L2-logistic regression classifiers. For all analyses, p < 0.05 was considered statistically significant in all two-tailed tests. The statistical analyses were performed using Python version 3.12.7 (Python Software Foundation) and SPSS (v26.0 IBM, Armonk, NY, USA).
Results
Patient clinical characteristics
The study cohort included 16 patients with surgically confirmed nodular PAP and 52 patients with pathologically proven LUAD, all presenting as pulmonary nodules on CT imaging. The key clinicopathological characteristics of 16 patients with nodular PAP are presented in Table 1. The cohort consisted of 6 male (37.5%) and 10 female (62.5%) patients. Eight patients (50%) presented with multiple nodules, while the remaining patients had a solitary nodule, with 3–28 mm in diameter. Among nine patients who underwent postoperative CT surveillance, three (33.3%) developed new nodules during follow-up. One of these patients underwent repeat surgical resection, with pathology confirming recurrent PAP. None of the PAP patients reported respiratory symptoms and underwent bronchoalveolar lavage.
The clinical characteristics of nodular PAP patients.

BMI, body mass index; LUL, left upper lobe; mm, millimeter; PAP, pulmonary alveolar proteinosis; RLL, right lower lobe; RUL, right upper lobe.
PAP patients were significantly younger than the LUAD patients (51.4 ± 10.1 vs 67.0 ± 8.5 years, p < 0.001) but had higher body mass index (BMI) values (25.8 vs 23.7 kg/m², p = 0.01). No significant differences were observed in gender distribution or smoking history between the groups (Table 2).
The clinical and surgical characteristics of nodular PAP patients and LUAD patients.

Boldface indicates statistical significance (p<0.05).
BMI, body mass index; LUAD, lung adenocarcinoma; PAP, pulmonary alveolar proteinosis; VATS, video-assisted thoracic surgery.
All patients in our study were managed with VATS, without any diagnostic interventions such as cryo-biopsy, transbronchial lung biopsy, or other minimally invasive procedures. The surgical information for the two groups is shown in Table 2. The extents of resection (lobectomy, segmentectomy, and wedge resection) were similar between the nodular PAP and LUAD groups (p = 0.465). Regarding nodule distribution, the majority of PAP lesions (57.1%) were found in the right upper lobe, with the rest in the left upper and right lower lobes. However, LUAD cases were identified in all five pulmonary lobes. This difference in anatomical distribution did not reach statistical significance (p = 0.143). Furthermore, the number of nodules (single, two, or multiple) did not differ significantly between the two groups (p = 0.227), with single nodules being the most common presentation in both groups.
The nodular PAP group showed lower serum lactate dehydrogenase (LDH) levels compared to the LUAD group (188.44 ± 26.71 vs 214.79 ± 56.84U/L, p = 0.01). Although NSE levels tended to be lower in the nodular PAP group (14.25 ± 4.24 vs 17.16 ± 7.07 U/mL, p = 0.05), this difference did not reach statistical significance. No notable variations were found in liver function, lipid profiles, or other tumor markers between the two groups (Table 3). These findings suggest that the combination of younger age at presentation, elevated BMI, and decreased LDH may serve as clinically useful discriminators to distinguish nodular PAP from malignant pulmonary nodules during diagnostic evaluation.
The clinical laboratory parameters of nodular PAP patients and LUAD patients.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; CA125, Cancer Antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; GGT, γ-glutamyl transpeptidase; LDH, lactate dehydrogenase; LUAD, lung adenocarcinoma; NSE, neuron-specific enolase; PAP, pulmonary alveolar proteinosis; ProGRP, pro-gastrin-releasing peptide; SCC, squamous cell carcinoma antigen; TBIL, total bilirubin; TC, cholesterol; TG, triglycerides.
For LUAD patients, the pathological examination revealed 30 cases of MIA and 45 cases of IAC. The nodule size was predominantly less than 10 mm (36.0%) or between 10 mm and 20 mm (49.3%). For CT image features, half of the LUAD patients presented with pure ground-glass nodules (pGGNs), while mixed ground-glass nodules (mGGNs) and solid nodules accounted for 36.0% and 12.0%, respectively (Supplemental Table 1).
Radiomic model construction and validation
Representative CT images of selected nodular PAP cases are presented in Figure 1. A total of 182 radiomic features with statistically significant differences were identified. The radiomic feature analysis revealed clear separation between cohorts in the two-dimensional PCA projection space (e.g., principal component 1(PC1) vs PC2, Supplemental Figure 1). After class rebalancing, among the single-modality radiomic models, the RF classifier achieved a mean AUC of 0.91, compared with 0.90 for GaussianNB and 0.87 for L2-regularized logistic regression (Figure 2). Inspection of the fivefold averaged confusion matrices revealed identical true-positive (9) and true-negative (3) counts across the three algorithms (Figure 2). Furthermore, RF yielded a false-positive rate of 0.8771, while GaussianNB yielded 0.8659, and L2-regularized logistic regression yielded 0.8550 (Supplemental Table 2). In addition, RF attained PR-AUC, F1-score, and MCC at 0.8145, 0.8262, and 0.7757, respectively, underscoring its superior robustness to class imbalance (Supplemental Table 2). Although no statistically significant differences were observed across the metrics among the three single-modality radiomic models, RF demonstrated numerically superior performance in all the aforementioned indicators. Consequently, RF was selected as the optimal single-modality radiomic model.

Fivefold cross-validation ROC curves for the unimodal model. (a) RF, (b) Gaussian naive Bayes, and (c) L2-regularized.
Multimodal model construction and validation
The multimodal RF model achieved a mean AUC of 0.97 (Figure 3(a)), demonstrating comparable diagnostic performance to the single-modality approach. In addition, unimodal models were separately constructed using radiomics and clinical data, achieving mean AUCs of 0.91 and 0.92, respectively (Figure 3(b) and (c)). The SHapley Additive exPlanations (SHAP) summary plot of the multimodal model indicates that the three added clinical features all contributed substantially to the model (Figure 4). Elevated LDH values and older age consistently suppressed model output, whereas higher BMI promoted it—patterns that align well with clinical diagnostic expectations. The clinical-only model and the multimodal model were evaluated using fivefold cross-validation, with the performance metrics for each fold in Supplemental Table 3. Wilcoxon signed-rank tests (n = 5-fold) indicated that there were no statistically significant differences in cross-validated AUCs, neither between the radiomics-only and clinical-only models (p = 0.9366), nor between the radiomics-only and multimodal modal models (p = 0.2437, Supplemental Table 4). Although the Wilcoxon test revealed no significant difference, the multimodal model exhibited a trend of stability, with a lower coefficient of variation (5.12% vs 10.13% and 10.26%) and a smaller range, 0.0909 vs 0.2 and 0.2424, Supplemental Table 4).
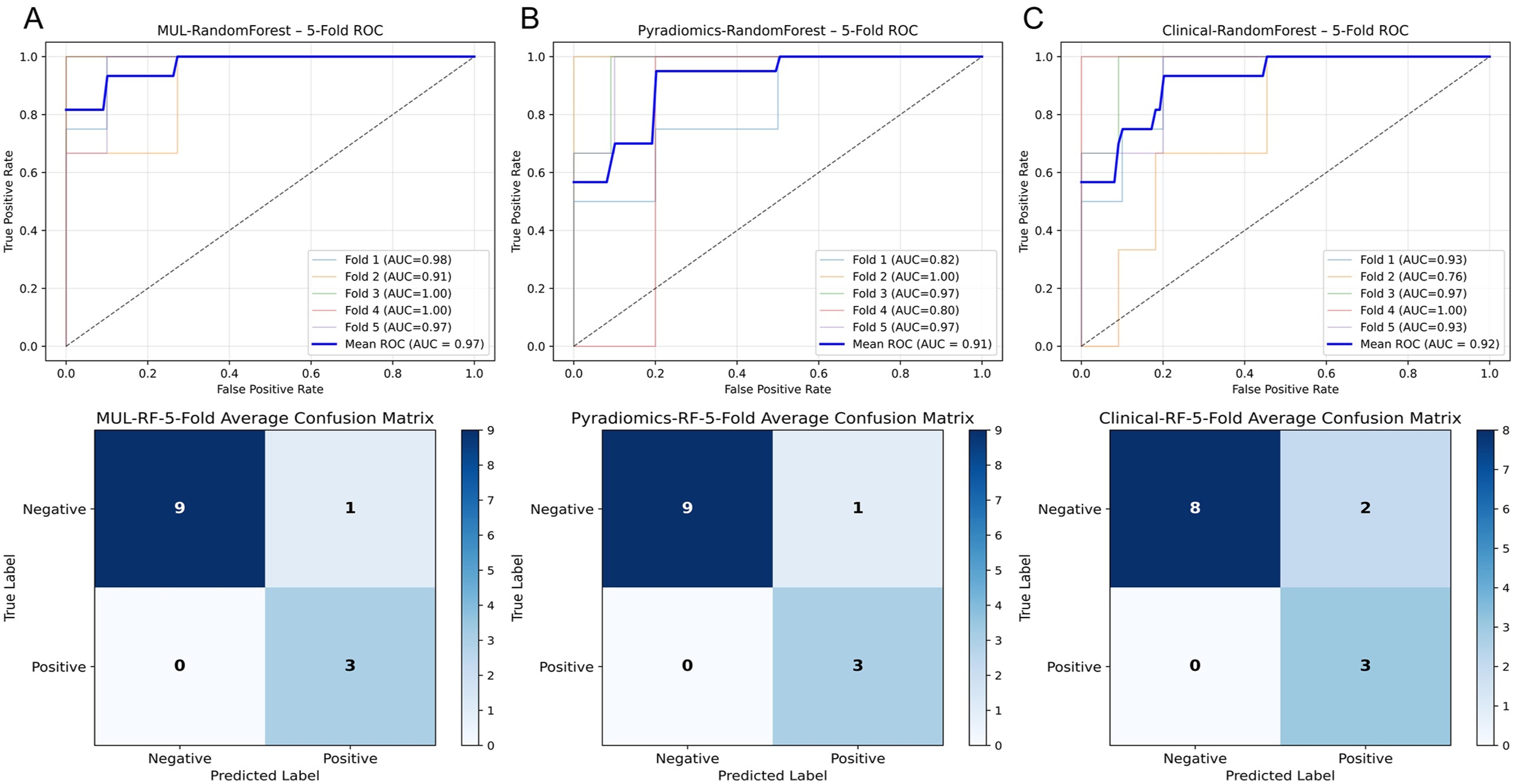
ROC curves of unimodal and multimodal model. (a) Fivefold cross-validation ROC curve of the multimodal model integrating radiomics and clinical data, (b) Fivefold cross-validation ROC curve of the unimodal radiomics-based model, (c) Fivefold cross-validation ROC curve of the unimodal clinical-data-based model.
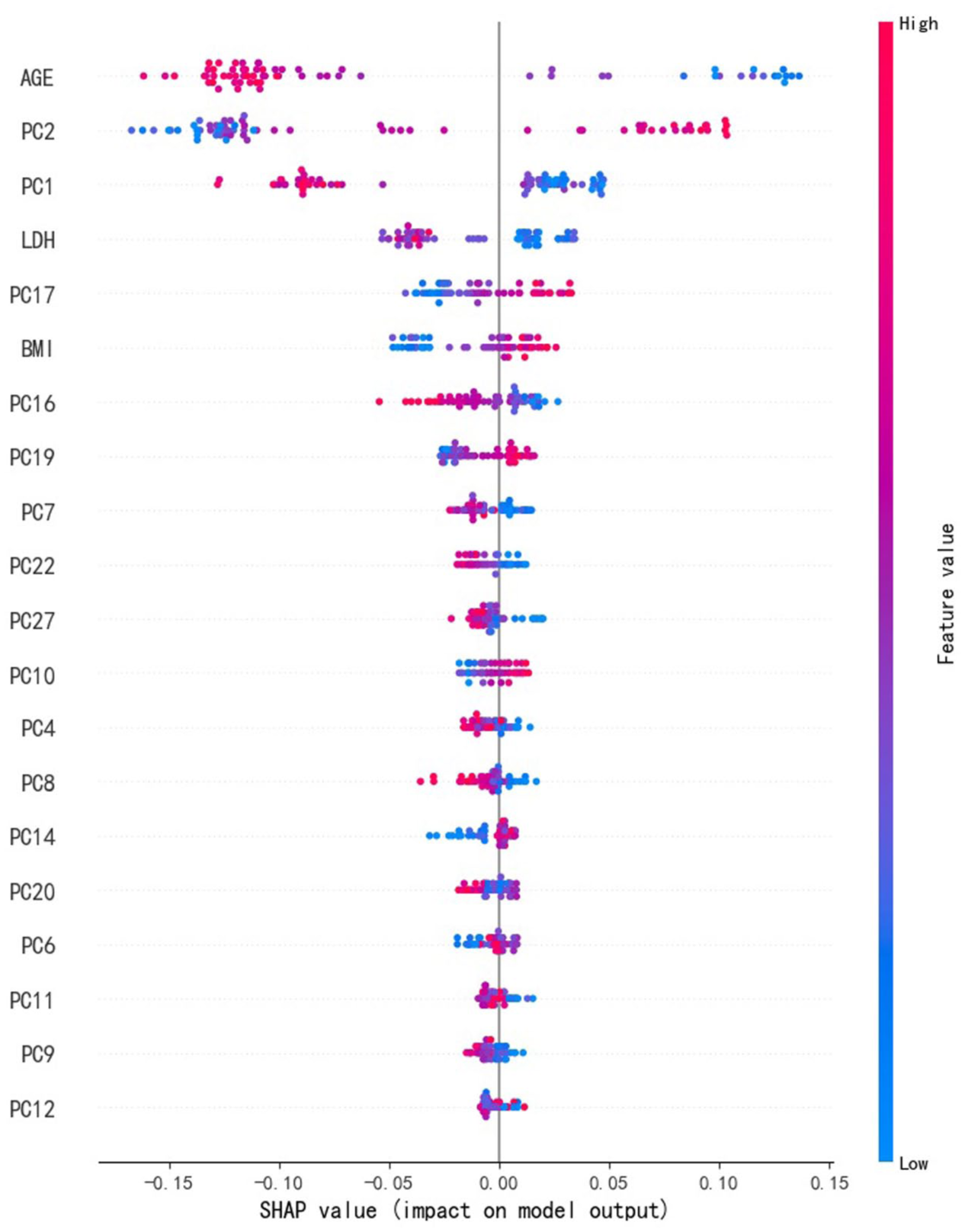
The performance verification of multimodal models. SHapley Additive exPlanations (SHAP) plot of the multimodal model.
Discussion
In this study, we characterized a rare nodular manifestation of PAP, that is, nodular PAP, and developed a diagnostic framework to differentiate it from early-stage LUAD. Systematic analysis of 16 cases of nodular PAP revealed distinct clinical, radiological, and pathological features, challenging the conventional perception of PAP as a predominantly diffuse disease. Comparative analysis demonstrated significant differences in clinical parameters (e.g., age, BMI), biochemical markers (e.g., LDH levels), and radiological characteristics between nodular PAP and LUAD, suggesting their potential utility as diagnostic adjuncts. Both the radiomic model based on imaging features and the multimodal model incorporating clinical, biochemical, and radiological parameters demonstrated similar discriminatory performance, with AUC values of 0.91 and 0.97, respectively.
Conventional PAP typically manifests on CT as diffuse GGOs with interlobular septal thickening, demonstrating the characteristic “crazy-paving” pattern, as extensively documented in previous studies.1,10 By contrast, our study of 16 nodular PAP cases demonstrated well-circumscribed solitary nodules or multiple nodules instead of the classic diffuse pattern. These atypical radiographic findings are consistent with limited case reports in the literature.23–26 One case report described a 74-year-old Japanese female patient with a solitary pulmonary nodule (maximum diameter 10 mm) on chest CT, which was pathologically confirmed as PAP following surgical resection. 23 Subsequently, the other case report described a 71-year-old Japanese male patient whose 12 mm pulmonary nodule showing increased density during 9-month follow-up, with postoperative pathology confirming PAP. This patient had no family history of PAP and tested negative for serum GM-CSF autoantibodies, with no recurrence observed on 3-month postoperative CT. 25 An Indian study reported a 73-year-old male patient presenting with multiple bilateral mixed nodules (including GGOs and solid nodules) on CT, with CT-guided biopsy confirming PAP. During 8-year follow-up, the lesions never progressed to the characteristic “crazy-paving” pattern, with some nodules spontaneously resolving while others developed calcifications. 26 In addition, a Korean case report described a 21-year-old male patient with a 12 mm solitary nodule confirmed as PAP after surgical resection, with no new GGOs during 1-year postoperative follow-up. 24 Notably, our study represents the first systematic investigation to confirm the existence of nodular PAP in a relatively large cohort.
Our study revealed nodular PAP patients were younger, with higher BMI and lower LDH than LUAD. The significantly younger age of nodular PAP patients underscores fundamental etiological differences. While LUAD arises from accumulated genetic damage over decades, PAP centers on GM-CSF signaling disruption, which may occur independently of aging.1,27–29 Previous large-scale retrospective studies focusing on diffuse PAP patients have reported disease onset age of 50–58 years, which corresponds with our findings.2,30 The mean BMI in our nodular PAP cohort (mean 25.8 kg/m²) aligned with existing reports of PAP patients (mean 25.37 and 24.7 kg/m²) and was significantly elevated compared to LUAD cases (mean 23.7 kg/m²; p = 0.01).31,32 While GM-CSF signaling defects in PAP have been mechanistically linked to systemic lipid dysregulation, we observed only marginal, statistically insignificant increases in serum total cholesterol (5.70 ± 0.54 vs 5.43 ± 0.62 mmol/L, p = 0.11) and triglycerides (2.13 ± 0.70 vs 2.07 ± 0.70 mmol/L, p = 0.75), possibly reflecting the less pulmonary involvement in the nodular PAP subtype compared to diffuse PAP.33,34 The nodular PAP cohort exhibited markedly reduced serum LDH levels (mean 188.44 ± 26.71 U/L) relative to both LUAD controls (214.79 ± 56.84 U/L; p = 0.01) and historical data for diffuse PAP (257.16–295.85 U/L).34,35 The decrement in LDH, which is a known biomarker of disease burden in PAP, likely reflects the limited alveolar involvement and earlier disease stage characteristic of nodular presentation.7,35 A parallel decrease in NSE levels (14.25 ± 4.24 vs 17.16 ± 7.07, p = 0.05) further supports this pattern of milder biochemical disturbance in localized disease.
Our findings demonstrate that nodular PAP primarily presents as ground-glass opacity nodules (GGNs) with internal density heterogeneity and spiculated margins on CT imaging (Figure 1), features that show significant overlap with early-stage LUAD, posing considerable diagnostic challenges. Histopathological analysis reveals that the spiculations in PAP nodules result from localized inflammatory reactions induced by intra-alveolar proteinaceous material deposition, rather than the infiltrative growth characteristic of malignant tumors. 1 Given that PAP is a benign condition with markedly superior prognosis compared to adenocarcinoma, accurate identification of PAP nodules carries substantial clinical significance for preventing unnecessary surgical interventions.
Regarding model development, we initially established a radiomic model based on CT features, which achieved a diagnostic performance of AUC = 0.91. We also incorporated statistically significant clinical characteristics (including age, BMI, and LDH levels) to construct a multimodal prediction model, ultimately improving the AUC to 0.97. However, this difference did not reach statistical significance, potentially reflecting either (1) the already high performance of the radiomic model approaching diagnostic ceilings or (2) limited statistical power due to our modest sample size of this rare condition. Notably, while radiomic and multimodal models have been widely applied for benign–malignant pulmonary nodule differentiation, our study represents the first application of this approach specifically for distinguishing between PAP nodules and early-stage LUAD nodules, providing a novel solution to this unique clinical dilemma.
There were some limitations in our study. First, the 16 PAP patients did not undergo GM-CSF autoantibody testing or genetic sequencing, precluding precise subtype classification. Second, although this represents the largest reported cohort of nodular PAP patients to date, the sample size remains relatively small, necessitating future multicenter validation studies. Finally, additional research incorporating molecular profiling techniques would help elucidate the underlying pathogenesis of nodular PAP.
Conclusion
This study represents the first relatively large-scale case series analysis of nodular PAP, systematically characterizing its distinctive features and establishing a diagnostic framework for differentiating it from LUAD. The nodular PAP patients exhibited significantly younger age, higher BMI, and lower LDH levels compared to LUAD patients. Both the radiomic model and multimodal model demonstrated similar diagnostic performance, indicating their potential as clinical decision-support tools for differential diagnosis.
Supplemental Material
sj-doc-1-tar-10.1177_17534666261436223 – Supplemental material for Discriminating nodular pulmonary alveolar proteinosis from lung adenocarcinoma with radiomic model and multimodal model
Supplemental material, sj-doc-1-tar-10.1177_17534666261436223 for Discriminating nodular pulmonary alveolar proteinosis from lung adenocarcinoma with radiomic model and multimodal model by Qing Chang, Huaizhe Zhang, Ning Xu, Ruolin Mao, Mengnan Li, Hai Zhang, Yuqing Chen, Jianqi Sun and Feng Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666261436223 – Supplemental material for Discriminating nodular pulmonary alveolar proteinosis from lung adenocarcinoma with radiomic model and multimodal model
Supplemental material, sj-docx-2-tar-10.1177_17534666261436223 for Discriminating nodular pulmonary alveolar proteinosis from lung adenocarcinoma with radiomic model and multimodal model by Qing Chang, Huaizhe Zhang, Ning Xu, Ruolin Mao, Mengnan Li, Hai Zhang, Yuqing Chen, Jianqi Sun and Feng Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-png-3-tar-10.1177_17534666261436223 – Supplemental material for Discriminating nodular pulmonary alveolar proteinosis from lung adenocarcinoma with radiomic model and multimodal model
Supplemental material, sj-png-3-tar-10.1177_17534666261436223 for Discriminating nodular pulmonary alveolar proteinosis from lung adenocarcinoma with radiomic model and multimodal model by Qing Chang, Huaizhe Zhang, Ning Xu, Ruolin Mao, Mengnan Li, Hai Zhang, Yuqing Chen, Jianqi Sun and Feng Li in Therapeutic Advances in Respiratory Disease
