Abstract
Objectives
We aimed to explore the clinical manifestations, examination findings, diagnostic methods, and treatment efficacy of patients with pulmonary alveolar proteinosis and improve clinicians' understanding of this disease.
Methods
We retrospectively analyzed the clinical characteristics of 39 patients diagnosed with autoimmune pulmonary alveolar proteinosis admitted to Fuzhou Pulmonary Hospital, Fujian Province, from August 2013 to December 2024. We also analyzed the correlations between lactate dehydrogenase, arterial partial oxygen pressure, and pulmonary alveolar arterial oxygen differential pressure, lung function, tumor markers, blood lipids, and disease severity. Furthermore, the relevant literature was reviewed.
Results
Of the 39 patients, 23 were male, 16 were female, 18 were smokers, and 11 had pneumoconiosis. The main clinical symptoms were cough and shortness of breath. The typical manifestations of chest computed tomography were “map-like” changes or “crazy-paving pattern”. The main manifestations of pulmonary function were diffusion dysfunction and restricted ventilation dysfunction. pulmonary alveolar proteinosis disease severity score was positively correlated with lactate dehydrogenase, pulmonary alveolar arterial oxygen differential pressure and the tumor markers carcinoembryonic antigen, cytokeratin 19 fragment antigen 21-1, neuron specific enolase, and carbohydrate antigen 153 (r = 0.689, P < 0.01; r = 0.801, P < 0.01; r = 0.414, P < 0.05; r = 0.570, P < 0.01; r = 0.488, P < 0.01; and r = 0.415, P < 0.05, respectively). The disease severity score was negatively correlated with the percentage of arterial partial oxygen pressure and carbon monoxide dispersion to the predicted value (r = −0.952, r = −0.554, P < 0.01). Lactate dehydrogenase was negatively correlated with carbon monoxide dispersion to the predicted value (r = −0.522, P < 0.01). Arterial partial oxygen pressure was negatively correlated with carcinoembryonic antigen, cytokeratin 19 fragment antigen 21-1, neuron specific enolase, and carbohydrate antigen 153 (r = −0.479, r = −0.607, r = −0.598, and r = −0.425, respectively, P < 0.01). In contrast, arterial partial oxygen pressure was positively correlated with carbon monoxide dispersion to the predicted value (r = 0.616, P < 0.01). pulmonary alveolar arterial oxygen differential pressure was positively correlated with carcinoembryonic antigen, cytokeratin 19 fragment antigen 21-1, neuron specific enolase, and carbohydrate antigen 153 (r = 0.529, P < 0.01; r = 0.546, P < 0.01; r = 0.554, P < 0.01; and r = 0.348, P < 0.05, respectively). Pulmonary alveolar arterial oxygen differential pressure was negatively correlated with carbon monoxide dispersion to the predicted value (r = −0.641, P < 0.01).
Conclusion
Pulmonary alveolar proteinosis is a rare diffuse lung disease without specific clinical manifestations. Whole lung high-volume alveolar lavage is the most effective treatment for pulmonary alveolar proteinosis. Arterial partial oxygen pressure, pulmonary alveolar arterial oxygen differential pressure, lactate dehydrogenase, tumor markers, and carbon monoxide dispersion to the predicted value were correlated with the severity of pulmonary alveolar proteinosis.
Keywords
Introduction
Pulmonary alveolar proteinosis (PAP) was first described by Rosen and Castleman in 1958. 1 Due to alveolar macrophage dysfunction, surfactant proteins and lipids accumulate in the alveoli, thereby hindering gas exchange. PAP is an extremely rare disease, with an incidence of approximately seven per million.2,3
PAP can be classified into the following three types based on the underlying mechanism: congenital, autoimmune (primary or idiopathic), and secondary types. Congenital disease is primarily observed among children, as genetic mutations impair the production of alveolar surfactant. The autoimmune forms of PAP are most common in adults and are driven by the disruption of signaling by granulocyte/macrophage colony-stimulating factor (GM-CSF), leading to alveolar macrophage and neutrophil dysfunction and resulting in the accumulation of surface-active proteins and lipids in the alveoli. Approximately 90% of PAP cases are autoimmune PAP. 2 Secondary PAP may develop following infections, hematological malignancies, immunodeficiencies, inhalation of toxic substances, or connective tissue diseases.3–5 The diagnostic criteria for autoimmune PAP are as follows: (a) histopathological findings in specimens obtained via open, percutaneous lung puncture, or transbronchial lung biopsy, confirmed by amorphous Schiff-positive particles of periodic acid, or positive periodic acid-Schiff (PAS) staining of bronchoalveolar lavage fluid (BALF); (b) typical milky BALF appearance; (c) ground-glass opacities or crazy-paving patterns on high-resolution computed tomography; and (d) a positive test for serum GM-CSF antibodies. Patients with autoimmune PAP have antibodies against GM-CSF, which can help distinguish autoimmune PAP from congenital and secondary PAP. 2 In this study, 39 patients with PAP had positive serum levels of anti-GM-CSF antibodies.
PAP lacks specific clinical manifestations, and chest computed tomography (CT) scan shows the “crazy-paving pattern”. 6 Small interlobular and intralobular septa are thickened, superimposed on a background of ground-glass opacities (GGOs) and the “map sign” (the border between the GGOs and normal lung tissue is usually clear, exhibiting a “map sign”). PAP is diagnosed based on CT and bronchoalveolar lavage cytology or lung histology, 7 and whole lung lavage (WLL) is the most effective treatment. We retrospectively analyzed 39 cases of autoimmune alveolar proteinosis, including their clinical features and treatment, to prevent misdiagnosis.
Methods
General information
In total, we included 39 cases of autoimmune PAP (aPAP) diagnosed based on CT and pathological examination or bronchoalveolar lavage cytology in our hospital from August 2013 to December 2024, who were positive for serum GM-CSF antibodies based on the quantitative test results. One 10-year-old patient tested positive for the GM-CSF antibody and underwent genetic testing. No positive results were found for the GM-CSF receptor (CSF2RA or CSF2RB) or STAT5B, etc. There were 23 male and 16 female patients, with a male-to-female ratio of 1.44:1. Their age ranged from 10–71 years, with an average age of 47.26 ± 12.69 years. Their body mass index ranged from 15.09 kg/m2 to 30.10 kg/m2, with an average of 23.57 ± 3.94 kg/m2. Eighteen cases had a smoking history (46.15%), and 11 cases had a background of pneumoconiosis (28.20%). This study was conducted following the 1975 Revised Declaration of Helsinki (revised in 2024). This study was reviewed and approved by the Ethics Committee of Fuzhou Pulmonary Hospital, Fujian Province (Ethics Review 2025-010 (Research)- 01). Informed consent was obtained from all subjects involved in the study. We reported this study in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting checklist, 8 and all patient details were de-identified.
PAP disease severity score
The disease severity score (DSS) was assessed based on the arterial partial oxygen pressure (PaO2) obtained from a blood gas analysis after admission and the presence or absence of symptoms: 1 point for no symptoms and PaO2 ≥ 70 mmHg, 2 points for symptoms and PaO2 ≥70 mmHg, 3 points for 60 mmHg ≤PaO2 < 70 mmHg, 4 points for 50 mmHg ≤PaO2 < 60 mmHg, and 5 points for PaO2 < 50 mmHg. 9 There was 1 patient with a DSS score of 1, 15 patients with a DSS score of 2, 12 patients with a DSS score of 3, 6 patients with a DSS score of 4, and 5 patients with a DSS score of 5.
Tumor markers and blood lipids
The serum levels of tumor markers were measured using an automated electrochemiluminescence immunoassay system, and the upper limit of normal was 16.3 μg/L for neuron specific enolase (NSE), 5.0 μg/L for carcinoembryonic antigen (CEA), 25.0 kU/L for carbohydrate antigen 153 (CA153), 3.3 μg/L for cytokeratin 19 fragment antigen 21-1 (CYFRA21-1), and 27 KU/L for carbohydrate antigen 199 (CA199). The total cholesterol (TC) and triglyceride (TG) levels were measured based on an enzyme assay, and low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) levels were measured via direct selective inhibition, with the upper limit of normal for TC at 5.72 mmol/L, for TG at 1.7 mmol/L, for LDL-C at 4.1 mmol/L, and 1.58 mmol/L for HDL-C.
Pulmonary function test
Lung function tests can help measure respiratory diseases and are mainly used to detect the degree of airway patency, lung volume size, and severity and prognosis of respiratory diseases. The following are considered as normal: a forced vital capacity per predicted (FVC/pred%) greater than or equal to 80% and forced expired volume in one second per predicted (FEV1/pred%) greater than or equal to 80%. This value is used to assess the severity of restrictive ventilation dysfunction. Carbon monoxide diffusing capacity per predicted (DLCO/pred%) is considered normal when it is greater than or equal to 80% and is used to assess the diffusion dysfunction.
Statistical analysis
All data were analyzed using SPSS 27.0. Measurement data with a normal distribution are expressed as mean ± standard deviation (x̄ ± s). Measurement data without a normal distribution are expressed as median (M) and interquartile range (Q1, Q3). Count data are expressed as constituent ratio (%). A Pearson correlation analysis was used to assess the correlation of normally distributed continuous variables, and a Spearman rank correlation analysis was used to assess the correlation of the ranked data. The r value is used to quantify the degree of linear correlation between two variables. The range of values includes (−1, 1). The larger the absolute value of the correlation, the stronger the correlation. Positive and negative signs indicate the direction of correlation. A P-value <0.05 was considered statistically significant, and a P-value <0.01 was considered highly significant.
Results
Clinical manifestations
Among a total of 39 patients, 36 exhibited shortness of breath, 35 exhibited cough, and 31 had expectoration. In addition to infection, 6 patients had fever, and 7 patients suffered from chest tightness. The other rare symptoms included chest pain (2 cases), palpitation (1 case), and blood-tinged sputum (1 case). One case was observed to be asymptomatic. Notably, 8 patients exhibited cyanosis (20.51%), 7 exhibited crackles (17.95%), and 6 presented with clubbing (15.38%).
Imaging and color Doppler ultrasound examinations
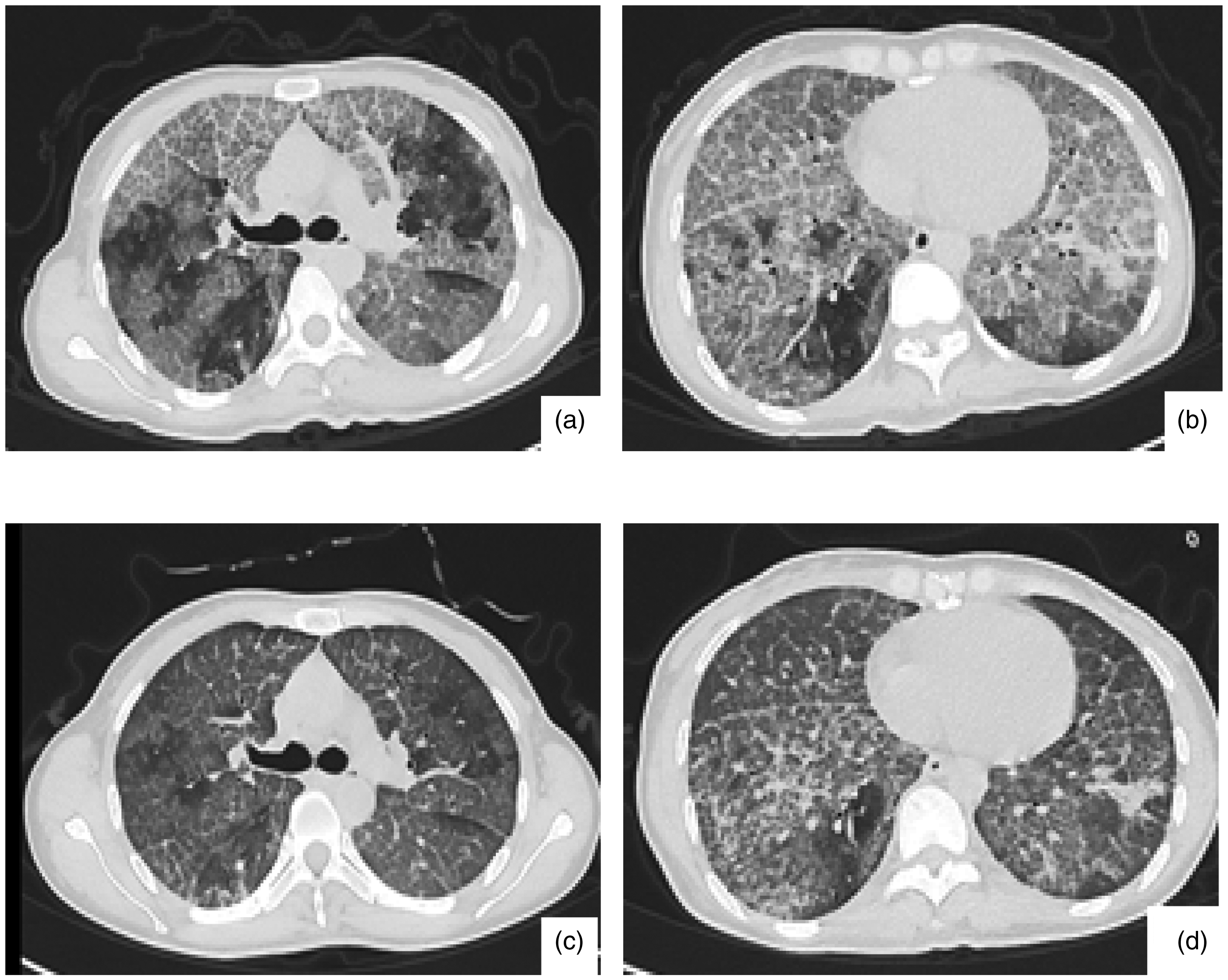
Chest CT: All patients showed bilateral lung lesions. Thirty-two patients presented with a crazy-paving pattern (82.05%, Figure 1(a)), 30 patients showed map-like changes (76.92%, Figure 1(b)), 32 patients exhibited GGOs (82.05%), 16 patients had consolidation shadows (41.03%), 7 cases presented with grid-like or honeycomb-like shadows (17.95%), 28 patients had mediastinal lymph node enlargement (71.79%), 5 patients had hilar lymph node enlargement (12.82%), 8 patients had pleural effusion (20.50%), and 3 patients had pericardial effusion (7.69%). A total of 11 patients with pneumoconiosis combined with alveolar proteinosis presented with chest CT. Diffuse centrilobular nodular shadows and patchy ground-glass shadows were observed in both lungs, with thickening of some interlobular septa, and shadows of lymph nodes were seen in part of the mediastinum, with calcification of some lymph nodes (Figures 1(c) and (d)).

Typical CT manifestations of PAP. Figure 1(a) “crazy-paving pattern”: Small interlobular and intralobular septa were thickened, superimposed on a background of ground-glass opacities; Figure 1(b) “Map Sign”: The boundary between the ground-glass opacity and normal lung tissue is usually clear, exhibiting a “map sign.” Figure 1(c) and Figure 1(d): A patient with pneumoconiosis combined with pulmonary alveolar proteinosis. Chest CT: Diffuse centrilobular nodular shadows and patchy ground-glass shadows were observed in both lungs. Some of the interlobular septa were thickened. Multiple groups of lymph node shadows were visible in the mediastinum, and some of them were calcified. CT: computed tomography; PAP: pulmonary alveolar proteinosis.
Overall, 25 patients underwent abdominal color Doppler ultrasound or abdominal CT examination, among whom 16 patients exhibited fatty liver in abdominal color Doppler ultrasound or abdominal CT (64.00%), and 9 patients had no fatty liver (36.00%). Notably, 14 patients did not undergo abdominal color Doppler ultrasound or abdominal CT for confirmation.
Pulmonary function tests: the correlation of pulmonary function with DSS, lactate dehydrogenase, PaO2, and P(a-a)O2
Among the 39 patients, only 27 underwent pulmonary function tests. Among them, 2 patients had only small airway dysfunction, without restrictive ventilation dysfunction or diffusion dysfunction. One patient had normal pulmonary function, 24 patients had restrictive ventilation dysfunction (88.89%), and 23 patients had diffusion dysfunction (85.19%). One patient's diffusion function could not be measured. The percentage of forced vital capacity per predicted (FVC/pred%) was 65.29% ± 14.88%, the percentage of forced expired volume in one second per predicted (FEV1/pred%) was 68.89% ± 15.22%, and the percentage of carbon monoxide diffusing capacity per predicted (DLCO/pred%) was 43.76% ± 15.18%.
The carbon monoxide dispersion to the predicted value (DLCO/pred%) was negatively correlated with DSS, lactate dehydrogenase (LDH), and pulmonary alveolar arterial oxygen differential pressure (P(A-a)O2) (r = −0.554, r = −0.522, and r = −0.641, respectively, P < 0.01). PaO2 was positively correlated with DLCO/pred% (r = 0.616, P < 0.01). The FVC/pred% and FEV1/pred% were not linearly correlated with DSS, LDH, P(A-a)O2, or PaO2 (Table 1).
Correlation between pulmonary function and DSS, LDH, PaO2, and P(A-a)O2.

DSS: disease severity score; LDH (U/L): lactate dehydrogenase; PaO2 (mmHg): arterial partial oxygen pressure; P(A-a)O2 (mmHg): pulmonary alveolar arterial oxygen differential pressure; FVC/pred%: forced vital capacity per predicted; FEV1/pred%: forced expired volume in one second per predicted; DLCO/pred%: carbon monoxide diffusing capacity per predicted.
Laboratory tests
Characteristics of LDH, PaO2, and P(a-a)O2, and the correlation between LDH, PaO2, and P(a-a)O2 and DSS
The LDH levels were 278.8 (216, 346) U/L among 39 patients (upper limit of normal: 220 U/L), with 25 cases exhibiting elevated LDH levels (64.10%). The average PaO2 was 66.38 ± 12.31 mmHg, and alveolar arterial oxygen difference (P(A-a)O2) was 44.62 ± 13.59 mmHg, with 23 patients having P(A-a)O2 more than 40 mmHg (58.97%).
DSS was positively correlated with LDH and P(A-a)O2 (r = 0.689 and r = 0.801, respectively, P < 0.01), and DSS was negatively correlated with PaO2 (r = −0.952, P < 0.01, Table 2).
Correlation between DSS and LDH, PaO2, P(A-a)O2.

DSS: disease severity score; LDH (U/L): lactate dehydrogenase; PaO2 (mmHg): arterial partial oxygen pressure; P(A-a)O2 (mmHg): pulmonary alveolar arterial oxygen differential pressure.
Characteristics of tumor markers, and the correlation between tumor markers and DSS, PaO2, and P(A-a)O2
Among the 39 patients, 2 patients did not undergo tumor marker measurement, 37 patients had an average CEA level of 9.46 (5.25, 18.82) ng/mL, and 29 patients had elevated CEA levels (78.37%). The average CYFRA 21-1 level was 11.65 (7.88, 25.04) ng/mL, and 33 patients had positive CYFRA 21-1 (89.19%). The average NSE level was 24.044 ± 11.94 ng/mL, and 24 patients had elevated NSE levels (64.86%). The average CA153 level was 48.54 (29.06, 149.20) KU/L, and 28 patients had elevated CA153 (75.68%) levels. The average CA199 level was 8.25 (6.11, 15.85) KU/L, and 4 patients had elevated CA199 levels (10.81%).
The values of CEA, CYFRA 21-1, NSE, and CA153 were positively correlated with DSS (r = 0.414, P < 0.05; r = 0.570, P < 0.01; r = 0.488, P < 0.01; and r = 0.415, P < 0.05, respectively). CEA, CYFRA 21-1, NSE, and CA153 were positively correlated with P(A-a)O2 (r = 0.529, P < 0.01; r = 0.546, P < 0.01; r = 0.554, P < 0.01; and r = 0.348, P < 0.05, respectively). The CEA, CYFRA 21-1, NSE, and CA153 levels were negatively correlated with PaO2 (r = −0.479, r = −0.607, r = −0.598, and r = −0.425, respectively, P < 0.01, Table 3).
Correlation between tumor markers and DSS, PaO2, and P(A-a)O2.

DSS: disease severity score; PaO2 (mmHg): arterial partial oxygen pressure; P(A-a)O2 (mmHg): pulmonary alveolar arterial oxygen differential pressure; CEA (μg/L): carcinoembryonic antigen; CYFRA21-1 (μg/L): cytokeratin 19 fragment antigen 21-1; NSE (μg/L): neuron specific enolase; CA153 (KU/L): carbohydrate antigen 153; CA199 (KU/L): carbohydrate antigen 199.
Characteristics of serum lipids, and the correlations between serum lipids and DSS, LDH, PaO2, P(a-a)O2
Among the 39 patients, the average serum level of TC was 5.12 ± 1.74 mmol/L, and 11 patients had increased TC levels (28.21%). The average LDL-C level was 3.19 ± 1.73 mmol/L, and 6 patients had increased LDL-C levels (15.38%). The average triglyceride (TG) level was 1.29 (1.00, 1.73) mmol/L, and 16 patients had increased TG levels (41.03%). Five patients had increased levels of both TC and TG (12.82%). The average HDL-C level was 1.16 ± 0.432 mmol/L, and 11 patients had increased HDL-C levels.
There was no linear correlation between the values of TC, TG, LDL, HDL-C, TC/HDL-C, TG/HDL-C, and LDL-C/HDL-C and DSS, LDH, PaO2, and P(A-a)O2 (Table 4).
Correlation between blood lipids and DSS, LDH, PaO2, and P(A-a)O2.
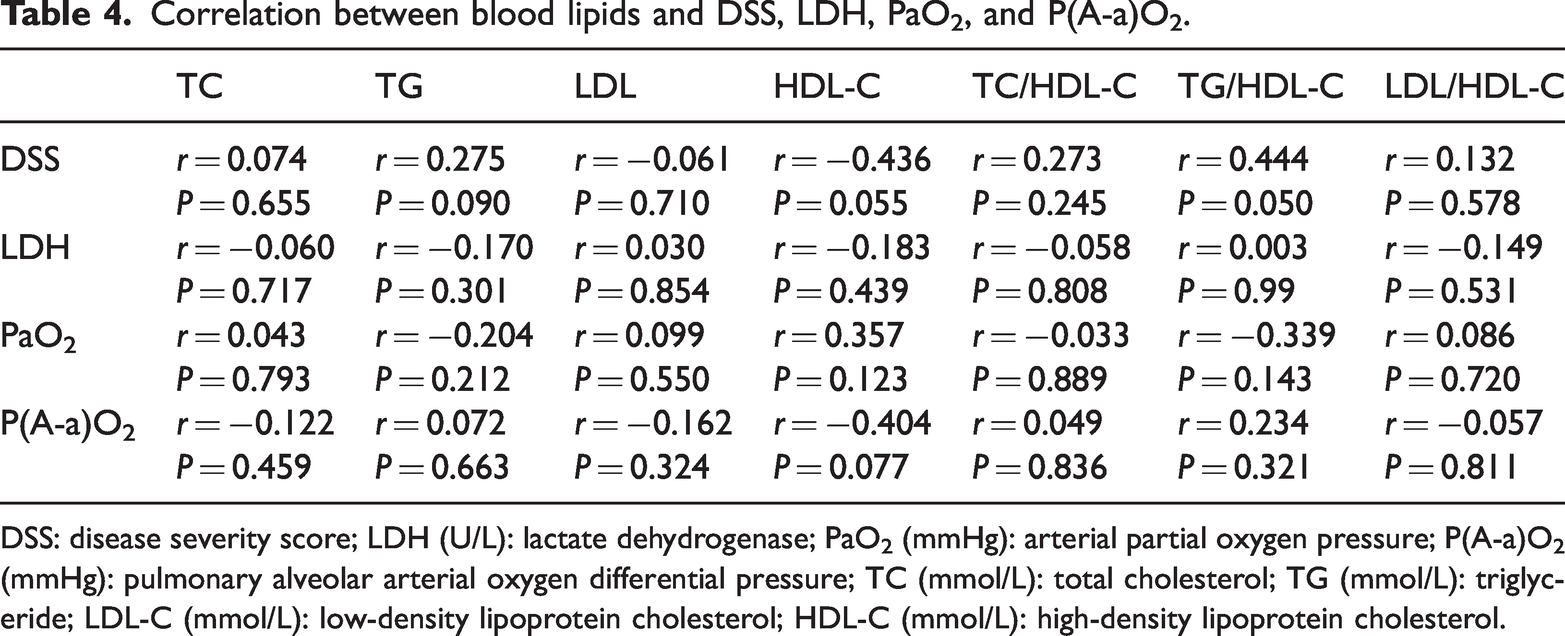
DSS: disease severity score; LDH (U/L): lactate dehydrogenase; PaO2 (mmHg): arterial partial oxygen pressure; P(A-a)O2 (mmHg): pulmonary alveolar arterial oxygen differential pressure; TC (mmol/L): total cholesterol; TG (mmol/L): triglyceride; LDL-C (mmol/L): low-density lipoprotein cholesterol; HDL-C (mmol/L): high-density lipoprotein cholesterol.
Co-infection
Among the 39 patients, 9 patients suffered from co-infections (23.08%), including 4 cases of tuberculosis, 2 cases of Klebsiella pneumoniae infection, 1 case of Nocardia infection, 1 case of Trichosporon asahii infection, and 1 case of Malassezia sp. infection.
Treatment and prognosis
Among the 39 patients, 30 patients underwent WLL, 6 patients refused to undergo WLL, and 3 patients did not meet the criteria for WLL. In total, 39 WLL procedures were performed. After WLL, the shortness of breath significantly improved in 30 patients, partial pressure of oxygen increased, arteriovenous carbon dioxide partial pressure difference decreased, and repeated chest CT scans showed a significant absorption of lesions in both lungs (Figure 2). Ten patients with a DSS score of 2 had a DSS score of 1 after WLL. There were 9 patients with a DSS score of <3, including 6 patients with a DSS score of 2 after WLL and 3 patients with a score of 1 after WLL. There were 6 patients with a DSS score of 4, 5 patients with a score of 2 after WLL, and 1 with a score of 1 after WLL. There were 5 patients with a DSS score of 5, 4 with a score of 2 after WLL, and 1 with a score of 3 after WLL (Figure 3).
Comparison of lung CT before and after PAP whole lung lavage. Figure 2(a) and 2(b) show the lung CT of the patient before whole lung lavage, where the typical crazy-paving pattern and map sign were observed; Figure 2(c) and 2(d) show the lung CT of the patient after whole lung lavage. Bilateral ground-glass opacities and thickening of the interlobar septa were improved significantly compared with that before treatment. CT: computed tomography; PAP: pulmonary alveolar proteinosis.

Number of patients with DSS scores before and after WLL. The horizontal axis represents the preoperative DSS score for WLL, and the vertical axis denotes the number of patients. Blue represents the number of patients with a postoperative DSS score of 1 after WLL. Green represents the number of patients with a postoperative DSS score of 2 after WLL. Purple represents the number of patients with a postoperative DSS score of 3 after WLL. DSS: disease severity score; WLL: whole lung lavage.
Four patients required another WLL 1–5 years after the first WLL. Among them, one patient underwent 6 WLL procedures during 7 years after the disease due to recurrent exacerbations, and the length of remission after WLL was gradually decreased from 28 months to 2 months. After the 6th WLL, the patient also received GM-CSF nebulization, 150 μg/dose, once/day, for 6 months. After 6 months of treatment, the patient's shortness of breath significantly improved, PaO2 increased, P(A-a)O2 decreased, and chest CT suggested that the lesions in both lungs were basically absorbed. However, 6 months after stopping the medication, the patient's condition worsened again, and the patient underwent GM-CSF nebulization again, 150 μg/dose, once/day for 2 years. After 2 years, there was no shortness of breath on follow-up, and a chest CT showed complete absorption of the lesions and improved lung function. There were no side effects during the treatment with nebulized GM-CSF. In addition, WLL was no longer needed during the treatment period and during the 2 years of follow-up. Due to the difficulty in obtaining GM-CSF, in addition to the one patient above, three other patients received nebulized GM-CSF after WLL at a dose of 150 µg once daily for 6 months. These patients were reviewed for elevated PaO2, decreased P(A-a)O2, and DSS scores, and lesion resorption based on chest CT. At 2–5 years after treatment, all three patients no longer required WLL.
Among 9 patients with co-infection, 5 patients with non-tuberculous PAP underwent WLL after controlling infection. Among 4 patients with tuberculosis, 3 patients underwent WLL 2 weeks after anti-tuberculosis treatment, and 1 patient underwent standard anti-tuberculosis treatment for half a year before WLL. The symptoms improved post-treatment, and no tuberculosis dissemination was found. Among the 30 patients who received WLL, the BALF was milky white and turbid in 20 patients, brown in 10 patients, and reddish due to alveolar hemorrhage in 2 patients. The fluid could be separated into a clear supernatant and a thick precipitate after standing (Figure 4).

Perfusion fluid after whole lung lavage. The lavage fluid was opaque and milky white in Figure 4(a) and turbid brown and reddish due to alveolar hemorrhage in Figure 4(b). After standing still, the lavage fluid was separated into a translucent upper layer and a thick sediment.
Discussion
PAP is more common in men, with most cases diagnosed in patients of age 40–50 years. Smoking and dust exposure may be risk factors for PAP. 2 In this study, 46.15% of the patients had a history of smoking, and 28.20% had a history of pneumoconiosis. The clinical manifestations of PAP are non-specific and include fever, cough, progressive dyspnea, etc. Even one-third of the patients remain asymptomatic. 10 In this study, the main symptoms were shortness of breath, cough, and expectoration, and one patient remained asymptomatic. Chest CT is crucial for diagnosing PAP. The typical manifestations are “crazy-paving pattern” 8 and “map sign”; however, they are not specific to PAP and can be seen in other diseases such as Yersinia pestis pneumonia, cardiogenic pulmonary edema, alveolar hemorrhage, bronchioloalveolar carcinoma, and acute respiratory distress syndrome. In this study, 82.05% of the patients had a “crazy-paving pattern” changes on chest CT, and 76.92% had “map-like” changes. The pulmonary function is of limited value in diagnosing PAP. In pulmonary function tests, patients with PAP mainly exhibit a decreased diffusion function and restrictive ventilatory dysfunction. A decreased diffusion function is more obvious than restrictive ventilatory dysfunction, which may be related to the nature of the disease. Specifically, the accumulation of phospholipid protein substances in the alveoli impairs gas exchange. Moreover, a decrease in the DLCO/pred% of the lung is associated with disease severity.2,11,12 Similarly, among patients who underwent pulmonary function tests in this study, the main manifestations were restrictive ventilatory dysfunction and decreased diffusion function. Furthermore, DLCO/pred% was negatively correlated with DSS and exhibited a significant statistical difference, while FVC/pred% and FEV1/pred% were not linearly correlated with DSS.
Some biological markers are associated with the severity of PAP, such as PaO2, P(A-a)O2, LDH, CEA, CYFRA 21-1, NES, and CA199,4,7,11–15 which is consistent with the findings of this study. In this study, the severity of PAP was positively correlated with P(A-a)O2 and LDH and negatively correlated with PaO2. DSS was positively correlated with CEA, CYFRA 21-1, and NSE; however, there was no linear correlation between DSS and CA199. We found that CA153 is positively correlated with DSS. Some studies have found an increase in the PaO2, a decrease in P(A-a)O2 and tumor markers after WLL or GM-CSF inhalation,13,16,17 which may be useful in assessing the efficacy of treatment. Previous studies have reported that patients with autoimmune PAP have elevated levels of TC, TG, and LDL, and dyslipidemia is associated with disease severity to some extent.18,19 Statins, as 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) reductase inhibitors, reduce the endoplasmic reticulum cholesterol levels and prevent cholesterol accumulation in alveolar macrophages. 20 Statins can reduce the cholesterol levels in macrophages and are promising for the treatment of PAP. 21 In this study with 39 patients, there was no linear correlation between the DSS, LDH, PaO2, or P(A-a)O2, and TC, TG, LDL, HDL-C, TC/HDL-C, TG/HDL-C, or LDL/HDL-C. This finding is inconsistent with that reported by previous studies. There is a paucity of research in this area, which needs further exploration.
PAP is diagnosed based on CT and bronchoalveolar lavage cytology or lung histology. 7 Pathological examination mainly shows the deposition of amorphous pink granular substances in the alveolar cavities, and PAS staining is positive in the alveolar cavities. In this study with 39 patients, 35 cases were diagnosed through bronchoscopic biopsy, among which 3 cases developed pneumothorax after biopsy. Besides, two cases were diagnosed through paraffin-embedding PAS staining of BALF under bronchoscopy, and 2 cases were diagnosed through lung puncture biopsy.
WLL is currently the most effective treatment method for PAP.22–25 WLL is performed in PAP for decreased lung function, decrease in the resting PaO2 <70 mmHg, alveolar arterial O2 gradient ≥40 mmHg, or severe dyspnea and hypoxemia at rest or exercise.26,27 The treatment of PAP also includes GM-CSF injection and inhalation, plasma exchange, rituximab, statins, and lung transplantation. The choice of treatment relies on the etiology. Lung function, DSS scores, PaO2, P(A-a)O2, and imaging indices may be used to assess the treatment efficacy.17,28–30 This study was a retrospective paper, and most lung function indices were missing after treatment, recording only DSS, PaO2, P(A-a) O2, symptoms, and imaging indices. In this study, the patients were treated with WLL and inhaled GM-CSF. After treatment, PaO2 increased, P(A-a)O2 and DSS scores decreased, and imaging suggested lesion resorption. In congenital PAP, patients with mild disease only need symptomatic treatment. For severe diseases, WLL is still necessary. Lung transplantation has been conducted for congenital PAP. 31 For autoimmune PAP, exogenous GM-CSF is an alternative therapy to correct GM-CSF deficiency. Several studies in China and other countries have shown that GM-CSF inhalation can result in satisfactory therapeutic effects in patients with idiopathic PAP,16,28,32–35 and may reduce the need for WLL.33–35 Other options include depleting B cells using rituximab, removing autoantibodies through plasma exchange, and maintaining lipid homeostasis with statin therapy. However, the efficacy of these treatments remains largely unclear. Lung transplantation has been recently conducted for the treatment of severely ill patients with autoimmune PAP.36,37 Among the 39 patients included in this study, 30 patients underwent large-volume lung lavage under general anesthesia, and all achieved good results. Among them, a patient who needed repeated WLL was treated with nebulized recombinant human GM-CSF after the last WLL, achieving good results and reducing the need for WLL.
PAP is a rare disease with non-specific clinical manifestations, increasing the risk of missed diagnosis and misdiagnosis. This study analyzed the clinical characteristics of PAP to improve its diagnosis. The typical chest CT manifestations of PAP are the “crazy-paving pattern” and “map sign”. PAP is diagnosed based on CT and bronchoalveolar lavage cytology or lung histology. Large-volume WLL is the most effective treatment method for PAP. In this study, DSS was linearly correlated with PaO2, P(A-a)O2, and LDH. In addition, DSS, PaO2, and P(A-a)O2 were linearly correlated with the tumor markers CEA, CYFRA 21-1, NSE, and CA153 as well as DLCO/pred%. All of these correlations were statistically significant or exhibited significant statistical significance. However, this study has limitations because of its small sample size and retrospective nature.
Footnotes
Acknowledgments
We are grateful for the data support provided by Fuzhou Pulmonary Hospital in Fujian Province. We sincerely thank Xiao Lv for his invaluable guidance throughout the research process, for the professional assistance he offered, and for the insightful discussions he had. We did not use AI during the creation of the manuscript.
Authors’ contributions
Xiao Lv: Conceptualization, Supervision, Resources, Validation, Writing – review & editing. Yanfen Zhong: Formal analysis, Data curation, Software development, Visualization, Writing – original draft. Both authors contributed to Investigation, Methodology, and Project administration.
Data availability statement
The datasets generated and/or analyzed during the current study are not publicly available, but are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare no conflicts of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
