Abstract
Corynebacterium jeikeium is an uncommon but increasingly recognized cause of invasive infection, usually affecting immunocompromised patients. Empyema due to C. jeikeium has not been previously reported in an immunocompetent adult. We describe the first case and review the existing literature on non-diphtherial Corynebacterium-related empyema to highlight diagnostic and therapeutic challenges. A 26-year-old previously healthy man presented with 5 days of pleuritic chest pain, fever, and productive cough. Chest computed tomography revealed a large, loculated left pleural effusion with an air–fluid level. Initial thoracentesis yielded frank pus, confirming empyema, but the patient declined immediate drainage and was started on intravenous cefoperazone/sulbactam and moxifloxacin. His condition worsened, prompting repeat thoracentesis and catheter drainage. Culture of pleural fluid grew C. jeikeium, leading to a switch to intravenous vancomycin and meropenem. The patient improved rapidly, with defervescence, normalization of inflammatory markers, and complete radiographic resolution. He remained well at the 3-month follow-up. A literature review identified only four previous cases of non-diphtherial Corynebacterium-related empyema, all in patients with significant comorbidities. This case demonstrates that C. jeikeium can cause empyema even in immunocompetent hosts. Early microbiological diagnosis, timely pleural drainage, and appropriate antimicrobial therapy, particularly vancomycin, are critical for successful outcomes. Increased awareness and case reporting will help refine management strategies for this rare but clinically relevant pathogen.
Introduction
Empyema, defined as the presence of pus in the pleural cavity, represents a severe and potentially life-threatening condition. Despite advances in diagnostics and treatment, empyema still carries a substantial burden, with mortality rates of 9.0%–15.2% and a mean hospital stay of 14 days.1,2 Current management relies on prompt antibiotic therapy and pleural drainage, with evolving techniques favoring minimally invasive approaches that maintain therapeutic efficacy. 3 Optimal antibiotic selection requires awareness of the growing diversity in empyema-causing pathogens.
Empiric treatments may be ineffective when rare organisms cause empyema, increasing the risk of poor outcomes. We report a rare case of Corynebacterium jeikeium isolated from pleural pus in a previously healthy young patient with community-acquired infection. A literature review of empyema cases caused by Corynebacterium species was conducted to assist physicians in recognizing this rare yet clinically significant pathogen.
We adhered to the relevant CARE guidelines (see Supplemental Material). 4 These guidelines require the inclusion of essential components such as the title, keywords, abstract, case presentation, patient details, clinical findings, diagnostic evaluation, therapeutic interventions, outcomes and follow-up, discussion, patient perspective, informed consent, and an ethics statement.
Case presentation
A previously healthy 26-year-old man was admitted with a 5-day history of left pleuritic chest pain, fever, and productive cough with yellow sputum. He denied prior tuberculosis, pneumonia, immunodeficiency, or chest trauma. He had taken amoxicillin/clavulanic acid 875/125 mg twice daily, antipyretics, pain relievers, and mucolytic agents for 2 days. However, his symptoms did not improve, prompting him to be hospitalized. Upon admission, his temperature was 38°C, heart rate 96 beats/min, blood pressure 110/70 mmHg, respiratory rate 18 breaths/min with no use of accessory muscles, and SpO2 99% on room air. He appeared ill, with dry lips and a coated tongue. Breath sounds were diminished over the lower two-thirds of the left lung with dull percussion and reduced vocal fremitus. Assessment of other organ systems was normal.
Laboratory findings were summarized in Table 1 and showed leukocytosis with a neutrophilic predominance and elevated C-reactive protein (CRP). Chest computed tomography (CT) (Figure 1) demonstrated a loculated left pleural effusion containing an air–fluid level. Thoracentesis performed immediately upon admission yielded frank yellow pus (Figure 1(d)). Fluid analysis (Table 2) revealed a predominance of neutrophils, a very low glucose level, and a markedly elevated lactate dehydrogenase (LDH) level, consistent with empyema.
Laboratory results upon admission.

Abnormal values are highlighted in bold.

Chest computed tomography at admission. (a–c) Demonstrate a loculated left-sided pleural effusion measuring 9.3 cm × 6.2 cm, with an air–fluid level and pleural thickening. No surrounding consolidation was observed. (d) Shows the appearance of pleural pus obtained following thoracentesis.
Pleural fluid analysis results.
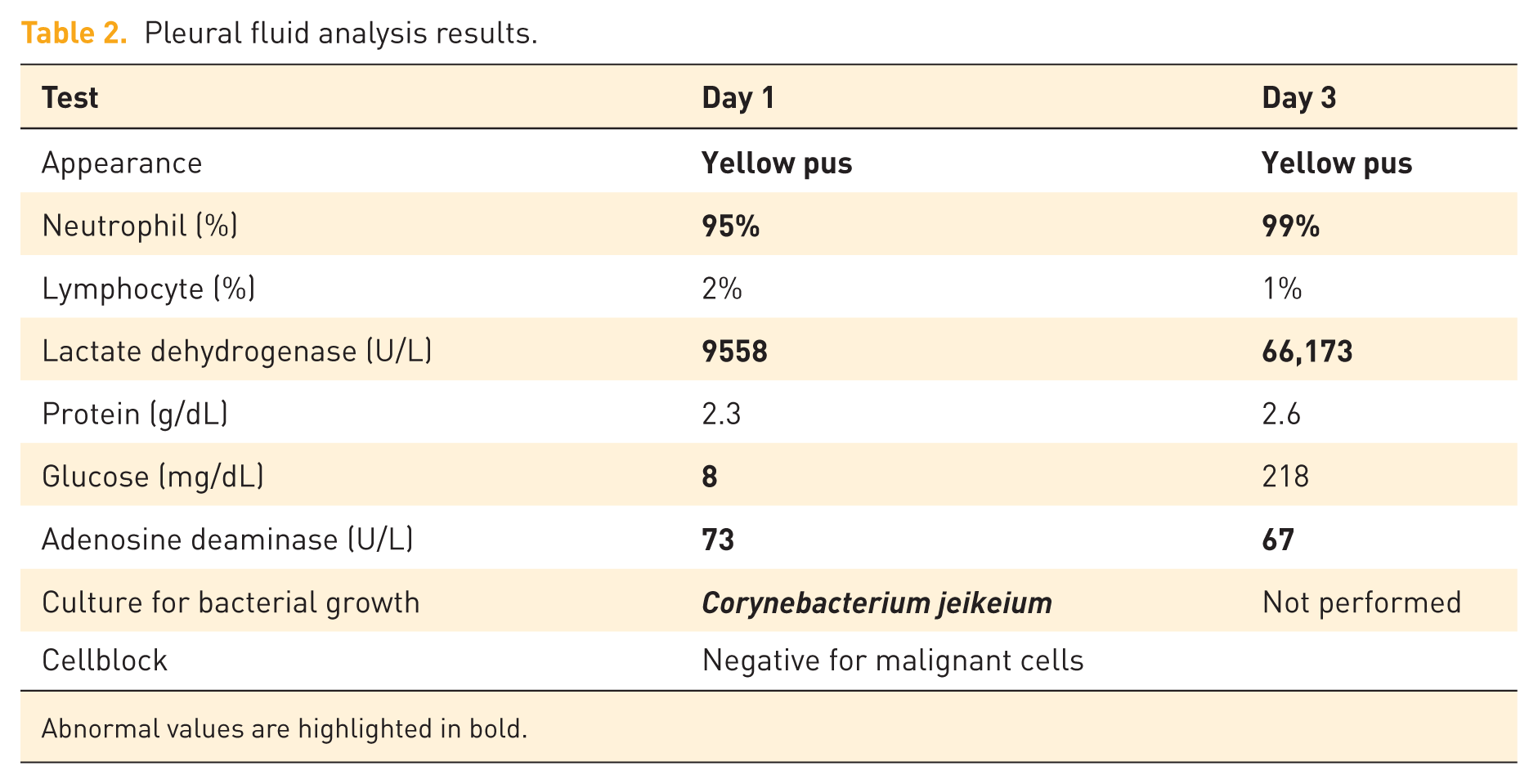
Abnormal values are highlighted in bold.
Empirical therapy with cefoperazone/sulbactam 1g/1g intravenous (IV) every 12 h plus moxifloxacin 400 mg IV daily was started, and chest drainage was initially deferred at the patient’s request. By day 3, he remained febrile and dyspneic. Repeat thoracentesis showed high LDH, and clindamycin 600 mg IV every 8 h was added. Pleural fluid culture result obtained on day 5 grew C. jeikeium, resistant to clindamycin and ciprofloxacin but susceptible to vancomycin (MIC 1 µg/mL). Antibiotic therapy was switched to vancomycin 1 g IV every 12 h plus meropenem 1 g IV every 8 h because a coinfection could not be ruled out. After further counseling, the patient consented to pleural drainage; a pigtail catheter was inserted under ultrasound guidance, and the pleural cavity was irrigated four times daily with 250 mL saline 0.9%.
After 10 days, the patient improved significantly: he was afebrile, chest pain diminished, chest CT showed resolution, CRP level dropped to 38.7 mg/L, and white blood cells normalized to 7.26 × 109 cells/L. The chest tube was removed, and he was transferred to the local hospital to complete a 6-week course of antibiotics. Clinical and radiographic recovery was confirmed at 3-month follow-up.
Discussion
We report a rare case of community-acquired empyema in a previously healthy 26-year-old man, characterized by a pleural air–fluid level and isolation of C. jeikeium. Clinical condition improved following susceptibility-guided therapy, with vancomycin and catheter drainage resulting in rapid clinical and radiographic resolution.
Empyema frequently arises as a complication of bacterial pneumonia. 5 Among patients hospitalized with pneumonia, pleural effusion develops in 20%–57%, and 5%–10% of these cases advance to empyema. 5 Consequently, guidelines emphasize two cornerstones of care: early broad-spectrum antibiotics and prompt evacuation of the infected space, escalating to thoracoscopy or decortication when necessary. 3 Despite high bacterial loads, cultures remain negative in 44% of parapneumonic pleural effusions. 6 A systematic review of 56 studies showed that, in tropical regions, pneumococci are the most common pathogens, followed by Staphylococcus aureus and viridans streptococci. Other notable organisms include Pseudomonas, Enterobacteriaceae, and Klebsiella, while Acinetobacter and coagulase-negative staphylococci are less frequently isolated. 6 Corynebacterium is a very rare cause of pleural infection. To investigate this further, we performed a PubMed search using the following criteria: (1) articles published from 2000 to June 2025; (2) the keywords “empyema” AND “Corynebacterium”; (3) full-text articles in English; and (4) detailed clinical descriptions of Corynebacterium-related empyema. Table 3 summarizes a total of five reported cases of empyema, including our case.7–10 Among these, two cases were caused by C. jeikeium, two by Corynebacterium striatum, and one by Corynebacterium afermentans subsp. lipophilum. Most patients had significant underlying comorbidities, and empyema was occasionally associated with concomitant osteomyelitis. 8 Antibiotics to which C. jeikeium remained susceptible included vancomycin, clindamycin, and erythromycin. Notably, all patients had favorable outcomes.
Reported cases of Corynebacterium-related empyema.

AMC, amoxicillin–clavulanate; CD4, cluster of differentiation 4; HTN, hypertension; TB, tuberculosis; TMP-SMX, trimethoprim–sulfamethoxazole; VATS, video-assisted thoracoscopic surgery.
C. jeikeium is commonly regarded as a skin commensal and is often overlooked. 11 However, it has been reported in diverse anatomical sites, including blood, cardiac valves, implanted medical devices, bone marrow, and the respiratory tract.8,11–13 While Corynebacterium diphtheriae is associated with diphtheria, infections caused by non-diphtherial Corynebacterium species are increasing.11,14 C. jeikeium, first described in 1976, is a Gram-positive, non-motile bacillus that may exhibit a coccobacillary appearance and produces distinctive white, metallic colonies within 24–48 h of culture. 12 C. jeikeium can cause sepsis, endocarditis, peritoneal dialysis-related peritonitis, bacteremia, keratitis, skin, and joint infections.8,9,11,13,15,16 In the respiratory system, C. jeikeium can lead to fulminant pneumonia accompanied by empyema or pulmonary necrosis. 9 These infections predominantly occur in individuals with underlying pulmonary disease, hematologic malignancies, immunosuppression, or recent invasive procedures. For community-acquired empyema, current guidelines recommend a penicillin with β-lactamase inhibitor combination or a second- or third-generation cephalosporin plus anaerobic cover with metronidazole or clindamycin.3,17 Alternative regimens may include monotherapy with moxifloxacin or levofloxacin. 17 Vancomycin is reserved for nosocomial infection, culture-proven susceptibility, or clinical non-response to empiric therapy.3,17 Given the rarity of C. jeikeium, there are currently no established treatment guidelines for pleural infections caused by this organism. C. jeikeium is often resistant to multiple classes of antibiotics, including β-lactams, aminoglycosides, and tetracyclines.12,13 Vancomycin remains an effective antibiotic against C. jeikeium and is commonly used in clinical practice.9,18 However, in vitro studies have demonstrated that C. jeikeium may retain susceptibility to doxycycline and daptomycin.18–20 A recent study analyzing 20 clinical Corynebacterium isolates (excluding C. jeikeium) reported high levels of multidrug resistance. 21 Specifically, 85% of isolates were resistant to penicillin G, cefuroxime, ceftriaxone, ciprofloxacin, and chloramphenicol; 80% to erythromycin; 75% to clindamycin and rifampin; and 70% to gentamicin. Notably, all isolates (100%) remained susceptible to linezolid, vancomycin, and teicoplanin, highlighting the continued reliability of glycopeptides against Corynebacterium species. 21 A systematic review analyzing 42 studies comprising 85 individual cases of invasive Corynebacterium striatum infections found that all isolates tested were susceptible to vancomycin (n = 50), linezolid (n = 30), teicoplanin (n = 16), and piperacillin–tazobactam (n = 8). 22 However, data on the antimicrobial resistance of C. jeikeium remain limited.
Table 3 also highlights that empyema caused by Corynebacterium often has a complicated clinical course, requiring prolonged antibiotic treatment, with some cases experiencing empyema recurrence as late as 7 months post-therapy. 9 These findings underscore the importance of individualized decisions regarding surgical versus medical management, extended antibiotic regimens, and careful follow-up even after clinical recovery. Given the documented multidrug resistance among Corynebacterium species, clinicians should consider early escalation to agents like vancomycin or linezolid when initial broad-spectrum β-lactams prove ineffective and culture results are pending or unavailable. While oral step-down therapy with trimethoprim–sulfamethoxazole or rifampin has been successful in selected cases,7,10 studies indicate that C. jeikeium exhibits only 20.4% susceptibility to trimethoprim–sulfamethoxazole, and no robust data are available regarding rifampin efficacy.23,24 Linezolid and doxycycline are potential oral alternatives, but their effectiveness in real-world settings and the risk of late relapse remain poorly defined.23–25 Additional reports involving larger patient cohorts are crucial to guide optimal antimicrobial selection and long-term management strategies for infections caused by this rare pathogen.
Conclusion
C. jeikeium can cause severe empyema even in otherwise healthy individuals. Empiric β-lactam therapy is often ineffective. Early culture and susceptibility testing are essential for accurate diagnosis and management. Vancomycin remains the most reliable empiric option when Corynebacterium is isolated. In our patient, targeted vancomycin therapy combined with pleural drainage led to complete recovery. Increased recognition and reporting will help optimize management of this rare pathogen.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666251412720 – Supplemental material for Corynebacterium jeikeium empyema in an immunocompetent adult: first case report and literature review
Supplemental material, sj-pdf-1-tar-10.1177_17534666251412720 for Corynebacterium jeikeium empyema in an immunocompetent adult: first case report and literature review by Khoa Nguyen-Dang, An Le-Hoang, Quoc-Khanh Tran-Le, Ngoc Duong-Minh and Hanh-Duyen Bui-Thi in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
