Abstract
Background:
Limited data exist on idiopathic pulmonary fibrosis (IPF) incidence and risk factors in elderly populations, making early intervention and preventive strategies challenging. Further research is needed to address this gap and improve outcomes.
Objectives:
This study used the Korean National Health Insurance Service (NHIS) database to investigate factors associated with the development of IPF in elderly people.
Design:
A retrospective cohort study was conducted to evaluate risk factors for IPF in an elderly population.
Methods:
This study evaluated 1,250,901 participants aged 75 years and older with health screening examination records in the NHIS database from 2012 to 2015. Cox proportional hazards regression models were used to assess the associations between IPF risk and demographics, lifestyle factors, and comorbidities, and subgroup analyses were used to explore multifactor interactions.
Results:
During a median follow-up of 7.4 years, 3955 participants developed IPF, for an incidence rate of 0.38 per 1000 person-years. Elderly participants (aged 75–85 years) exhibited higher IPF incidence than extremely elderly participants (⩾85 years) (0.40 vs 0.17 per 1,000 person-years, p < 0.01), though the difference was not significant in the multivariable analysis. The factors independently associated with increased IPF risk were male sex (adjusted hazard ratio [aHR] = 2.56, 95% confidence interval [CI] = 2.35–2.78), high body mass index (BMI) (aHR=1.45, 95% CI = 1.34–1.56 ), smoking (aHR = 1.24, 95% CI = 1.13–1.37 for ever-smokers < 20 pack-years [PYs]; aHR = 1.28, 95% CI = 1.17–1.39 for ever-smokers ⩾ 20 PYs), dyslipidemia (aHR = 1.17, 95% CI = 1.08–1.26), chronic kidney disease (aHR = 1.08, 95% CI = 1.01–1.16), and chronic obstructive pulmonary disease (COPD) (aHR = 1.49, 95% CI = 1.37–1.63). Additive or synergistic effects were observed among sex, BMI, smoking, and dyslipidemia, notably among males, and interactions between COPD and smoking status modulated IPF risk.
Conclusion:
Male sex, high BMI, smoking, and dyslipidemia might additively or synergistically increase the IPF risk among elderly people, highlighting the need for targeted prevention strategies in those populations.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic interstitial lung disease characterized by progressive and irreversible pulmonary fibrosis. While IPF predominantly affects males and individuals with a history of smoking, recent epidemiological studies reveal regional and ethnic variations in its incidence. The typical age of onset is between 60 and 70 years, with incidence rising sharply after age 60.1,2 According to the Organization for Economic Cooperation and Development in 2022, the average life expectancy across member countries was 80.5 years. In the Republic of Korea, the average life expectancy is even higher, at 83.8 years, as reported in the same year. 3 As global societies transition into an ultra-aging era in which life expectancy is consistently increasing, 4 studies focused on aging-related chronic inflammatory lung diseases, such as IPF, are needed.
Elderly IPF patients tend to be frailer, have more comorbidities, experience more severe clinical symptoms such as shortness of breath, and are more prone to adverse drug reactions than young IPF patients.5,6 Therefore, the evaluation of this population is important; however, elderly people are rarely included in clinical studies. For example, previous antifibrotic trials have not included a sufficiently large proportion of patients older than 75 years. In the INPULSIS and TOMORROW studies, the average age of the randomized patients was 64.8–67.1 years, and the CAPACITY and ASCEND trials excluded patients older than 80 years.7–10
Furthermore, information on the epidemiology of this study population is scarce. One study, using data from US Medicare beneficiaries aged 65 years and older between 2000 and 2011, reported that the incidence and prevalence of IPF in that population were substantially higher than previously estimated, with prevalence increasing annually. 11 In that study, the annual incidence of IPF in the Medicare population remained stable between 2001 and 2011, with an overall estimate of 93.7 cases per 100,000 person-years during the study period. 11 Although that study used a large cohort and provided a decade-long perspective on IPF incidence and prevalence in an elderly population, it lacked information on factors such as smoking status and was limited by the fact that the population studied was from the early 2000s, rendering the data relatively outdated. Information on the incidence of IPF in the elderly population remains scarce. Along with IPF incidence, it is also important to evaluate risk factors for IPF to develop preventive strategies and provide early interventions. However, little such information is available for elderly populations.
Therefore, this study assesses the incidence of IPF and identifies its risk factors in individuals aged 75 years and older living in an ultra-aging society.
Methods
Data source
This study utilized data from a retrospective cohort drawn from the Korean National Health Insurance Service (NHIS) database (NHIS-2025-09-1-055). Administered by the government, the NHIS serves as Korea’s universal health insurance system, covering approximately 97% of the population, around 50 million individuals. 12 The NHIS database offers comprehensive information, including socioeconomic and demographic details, healthcare utilization records, results from health screening examinations, disease diagnoses coded according to the 10th revision of the International Classification of Diseases (ICD-10), and data on medical treatments and procedures.12–20 A key component of the NHIS is its health screening database, which compiles data from nationwide screening programs provided free of charge to all adults by the Ministry of Health and Welfare.
These screenings, initiated in 2009 and conducted annually or biennially, include anthropometric assessments (e.g., body mass index [BMI]), lifestyle questionnaires (covering smoking, alcohol use, and physical activity), and blood tests such as lipid profiles. With a participation rate ranging from 70% to 80%, these screenings offer a representative snapshot of the Korean adult population.13,14 NHIS data have been extensively employed in research to investigate the incidence, risk factors, and clinical outcomes, including hospitalization and mortality, of various chronic pulmonary conditions, notably interstitial lung diseases.21–29
Study population
Among the 1,353,658 participants aged 75 years and older with records in the NHIS database from 2012 to 2015, we prioritized 1,343,621 participants by excluding 10,037 who were diagnosed with IPF before 2012. We further excluded participants with missing health screening examination data (n = 60,835) and those diagnosed with IPF within one year after the index date (n = 31,885), leaving 1,250,901 participants for the final analysis (Figure 1). We divided the participants into two groups: the elderly group (75–85 years old) and the extremely elderly group (⩾85 years old). 30
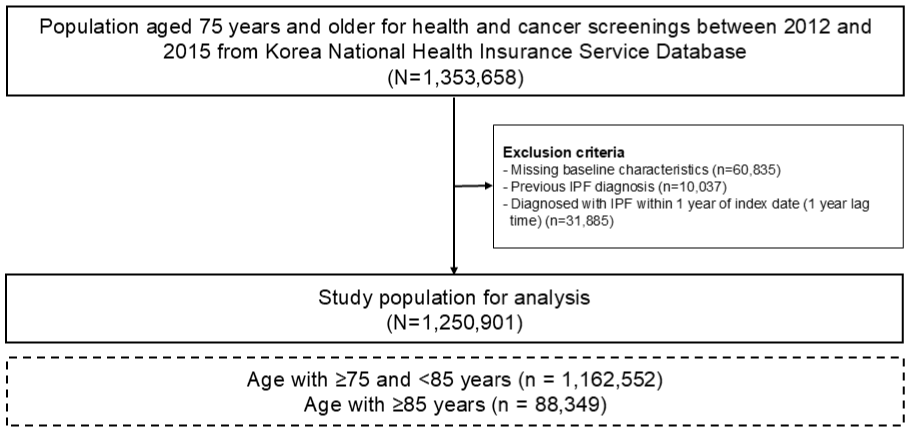
Flow chart of the study population.
Study outcome
The outcome of this study was the incidence of IPF. The NHIS operates the Rare and Intractable Disease (RID) program, which provides beneficial cost reductions for relevant medical expenses for patients with diseases such as IPF. Therefore, the validity of the RID code for IPF (V236) is strictly reviewed by the Health Insurance Review and Assessment Service. In this study, IPF is defined as claims under the ICD-10 diagnosis code J84.1 and RID code V236.21–25 The study participants were followed from 1 year after the index date to the date of an IPF diagnosis, censored date, or 31 December 2022.
Covariates
Data for the basal characteristics of age, income, BMI, specific smoking status, alcohol consumption, regular exercise, and comorbidities were collected. Income status was categorized into quartiles based on insurance premium levels, with those covered by medical aid (poorest 3%) being merged into the lowest income quartile.31–33 BMI was calculated as body weight divided by the square of height (kg/m2) and classified into four groups: <18.5, 18.5 to <23.0, 23 to <25.0, and ⩾ 25.0 kg/m2.33–36 Smoking status, alcohol consumption, and regular exercise were determined based on a self-reported questionnaire. Smoking status was divided into never-, ex-, and current smokers. 33 Alcohol consumption was divided into none, mild (average daily alcohol consumption less than 30 g in males and 20 g in females), and heavy (average daily alcohol consumption more than 30 g in males and 20 g in females). Regular exercise was defined as moderate-intensity exercise on five or more days per week or vigorous exercise on three or more days per week.33,37–39 The presence of comorbidities (diabetes mellitus, hypertension, dyslipidemia, and chronic pulmonary obstructive disease [COPD]) was assessed using ICD-10 codes, as previously described.40–48 Chronic kidney disease (CKD) was defined as an estimated glomerular filtration rate <60 mL/min/1.73 m2 by the 2009 CKD-EPI equation or records of ICD-10 codes V001, V003, or V005. Dyslipidemia was defined as total cholesterol ⩾ 240 mg/mL or the use of dyslipidemia medication. 49 The detailed definitions of each variable are summarized in Supplemental Table 1.
Statistical analysis
Descriptive statistics are presented as the number (percentage) for categorical variables and the mean ± standard deviation for continuous variables. We compared two groups using the χ2 test for categorical variables and t-tests for continuous variables. The incidence rates of IPF were calculated by dividing the number of incident events by the total follow-up period (1000 person-years). A cumulative incidence plot was used to compare the incidence of IPF, and a log-rank test was used to evaluate significant differences between groups.
The risk of incident IPF was estimated using univariable and multivariable Cox proportional hazards regression analyses. The multivariable models were adjusted for baseline characteristics and potential variables that could affect the development of IPF (age, sex, income level, BMI, smoking status, alcohol consumption, regular exercise, and comorbidities). To examine multifactor interactions influencing the development of IPF in the elderly participants, subgroup analyses were performed according to sex, BMI, smoking status, dyslipidemia, and COPD. A two-sided p-value < 0.05 was considered statistically significant. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA. http://www.sas.com).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 50
Results
Baseline characteristics
During the median follow-up period of 7.4 years (interquartile range: 6.0–8.7 years), 3,955 participants developed IPF, for an incidence rate of 0.38 per 1,000 person-years. The cumulative incidence plot for the development of IPF in the entire cohort is shown in Figure 2(a).

Cumulative incidence plot. (a) Cumulative incidence of IPF in the total population. (b) Cumulative incidence of IPF in elderly populations according to age groups.
Among the elderly participants, those who developed IPF (IPF group) had a higher proportion of individuals aged 75–85 years, males, and individuals who were obese (p < 0.001 for all) compared with those who did not develop IPF (non-IPF group). Compared with the non-IPF group, the IPF group also had a significantly higher proportion of smokers (40.5% vs 22.6%, p < 0.001), particularly those with a smoking history of more than 20 pack-years (PYs) (23.8% vs 12.8%, p < 0.001). In addition, the IPF group had a greater proportion of heavy alcohol drinkers (4.3% vs 2.7%, p < 0.001) and a higher prevalence of diabetes mellitus (p = 0.010) and COPD (p < 0.001) than the non-IPF group (Table 1).
Baseline characteristics of the study population.
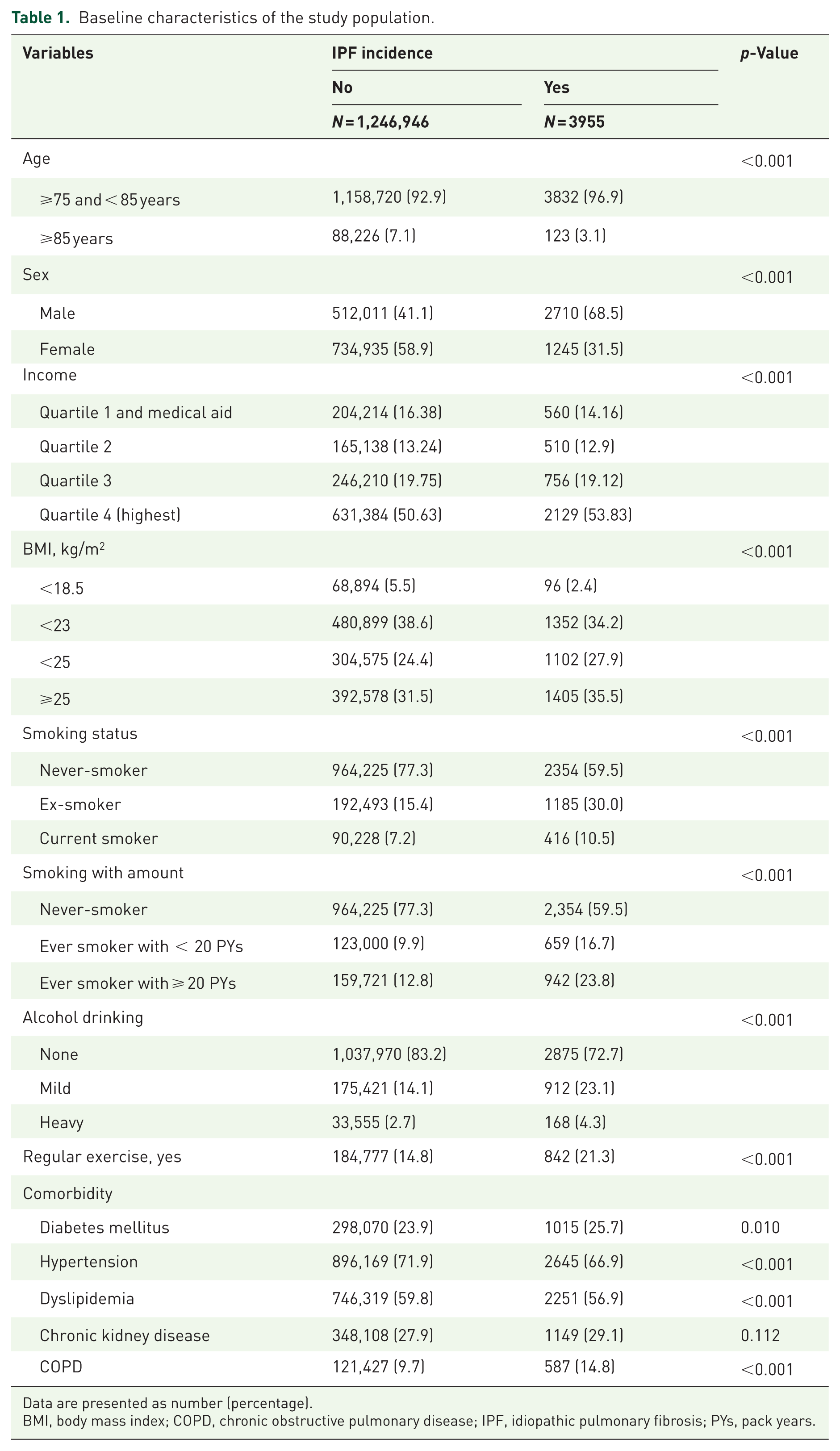
Data are presented as number (percentage).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; IPF, idiopathic pulmonary fibrosis; PYs, pack years.
Incidence of IPF according to age group
During the median follow-up period of 7.4 years (interquartile range: 6.0–8.7 years), the incidence rate of IPF was higher in the elderly group than the extremely elderly group (0.40 per 1000 vs 0.17 per 1000 person-years), with an unadjusted hazard ratio (HR) of 0.42 (95% confidence interval (CI): 0.35–0.51). However, the association was not statistically significant in the multivariable analysis. The cumulative incidence plot for the development of IPF in the two age groups is shown in Figure 2(b).
Factors associated with IPF
As shown in Table 2, factors associated with an increased risk of IPF were male sex (adjusted HR (aHR) = 2.56, 95% CI = 2.35–2.78), high BMI (aHR = 1.45, 95% CI = 1.34–1.56 for overweight compared with normal BMI), ever-smoking status (aHR = 1.24, 95% CI = 1.13–1.37 for ever-smokers < 20 PYs; aHR = 1.28, 95% CI = 1.17–1.39 for ever-smokers ⩾ 20 PYs), dyslipidemia (aHR = 1.17, 95% CI = 1.08–1.26), CKD (aHR = 1.08, 95% CI = 1.01–1.16), and COPD (aHR = 1.49, 95% CI = 1.37–1.63).
Risk of IPF according to baseline characteristics.

Adjusted for age, sex, income, BMI, smoking status, alcohol drinking, regular exercise, diabetes mellitus, hypertension, dyslipidemia, chronic kidney disease, and COPD
BMI, body mass index; COPD, chronic obstructive pulmonary disease; IPF, idiopathic pulmonary fibrosis; IR, incidence rate; PYs, pack years.
Similar results were shown in the subgroup analyses by sex, except for dyslipidemia and CKD, which were significant only among males. When the data were analyzed by age group, similar results were shown only in the elderly group (age < 85 years), not in the extremely elderly group, except for the subgroup analyses by sex (Table 2).
Multifactor interactions
As shown in Table 3, BMI and dyslipidemia had a significant interaction in the association between sex and IPF development (p < 0.05 for both), showing a relatively lower risk of IPF development in underweight males (aHR = 0.36, 95% CI = 0.28–0.47) and a higher risk in males with dyslipidemia (aHR = 1.26, 95% CI = 1.16–1.38). In the significant interaction between COPD and smoking status, the effect of smoking status on the development of IPF was significant only among individuals without COPD (p = 0.017).
Risk of IPF according to multifactor interaction analysis.

Adjusted for age, sex, income, BMI, smoking status, alcohol drinking, regular exercise, diabetes mellitus, hypertension, dyslipidemia, chronic kidney disease, and COPD.
BMI, body mass index; COPD, chronic obstructive pulmonary disease; IPF, idiopathic pulmonary fibrosis; IR, incidence rate; PYs, pack years.
Sex had additive or synergistic effects with BMI, smoking status, and dyslipidemia in the development of IPF, with the highest risk found among normal-to-obese males (aHR = 2.55, 95% CI = 2.34–2.77), males with ⩾ 20 PYs of smoking (aHR = 3.29, 95% CI = 2.99–3.61), and males with dyslipidemia (aHR = 2.98, 95% CI = 2.62–3.39). BMI also had significant additive or synergistic effects with smoking status and dyslipidemia for the development of IPF, with the highest risk of IPF among normal-to-obese participants with a smoking history of ⩾20 PYs (aHR = 1.28, 95% CI = 1.17–1.39) and those with dyslipidemia (aHR = 1.20, 95% CI = 1.11–1.29). Regarding COPD and smoking status, the highest risk of IPF development was in individuals with COPD and a smoking history of <20 PYs (aHR = 1.75, 95% CI = 1.41–2.18).
Discussion
This study used a comprehensive analysis of the Korean NHIS database to investigate the incidence and risk factors for IPF in elderly and extremely elderly people. Among 1,250,901 participants aged 75 years and older, the incidence rate of IPF was 0.38 per 1000 person-years during a median follow-up of 7.4 years, with elderly participants (aged 75–85 years) showing a higher incidence than extremely elderly participants (⩾85 years), although that difference was not significant in the multivariable analyses. Male sex, high BMI, smoking status, dyslipidemia, CKD, and COPD were identified as significant risk factors for IPF. Furthermore, analysis of multifactor interactions revealed additive or synergistic effects between the development of IPF and sex, BMI, smoking, and dyslipidemia, particularly among males, and highlighted a complex interplay between smoking and COPD status.
A prior study examined the incidence of IPF in the US population aged 65 years and older from 2001 to 2011. During that period, the annual incidence of IPF in the Medicare population remained stable, with an overall estimate of 93.7 cases per 100,000 person-years. 11 Age-specific incidence rates were reported as 63.3 cases per 100,000 person-years for the 66–69 age group, 95.2 cases per 100,000 person-years for the 70–74 age group, 118.7 cases per 100,000 person-years for the 75–79 age group, and 129.8 cases per 100,000 person-years for the ⩾ 80 age group, demonstrating a significant increase in incidence with age. In our study, the incidence rate was 0.38 per 1000 person-years (38 per 100,000 person-years) in the population aged 75 years and older. Inconsistent with the findings of the US study, the incidence in the elderly group (75–85 years old) was higher than in the extremely elderly group (⩾85 years old). The reduced incidence rate in the extremely elderly group likely reflects the competing risks of death and IPF development. Additionally, compared to studies conducted in the United States, a Western society, our study focused on an Asian population, and various socio-environmental factors, such as differences in study timing, may have contributed to the observed disparities.
Although the epidemiology of IPF in elderly populations was previously evaluated, 11 risk factors for the development of IPF in such populations remain largely unknown. This study, therefore, provides valuable insights into risk factors (male sex, high BMI, smoking status, dyslipidemia, CKD, and COPD) in this population. Beyond revealing risk factors for IPF, our study explored multifactor interactions in the development of IPF, particularly the additive or synergistic effects of sex, BMI, smoking status, and dyslipidemia. Male sex was identified as a major risk factor, with higher risks observed among overweight or obese males and those with a history of smoking or dyslipidemia. These interactions likely stem from overlapping biological mechanisms, such as chronic systemic inflammation and oxidative stress, which are exacerbated by smoking and obesity and further compounded by the metabolic dysregulation associated with dyslipidemia. In addition, the interplay between smoking status and COPD highlights significant variations in IPF risk based on disease presence, suggesting that targeted interventions for smoking cessation and COPD management could effectively mitigate the IPF risk. However, given the limitations of ICD-1 codes, future studies are warranted to investigate the relationship between those factors and IPF risk.
Interestingly, in our study, regular physical activity was lower in individuals who did not develop IPF compared to those who did develop IPF. Considering regular physical activity is associated with reduced risk of various pulmonary diseases, this finding is unexpected. However, since our study is observational, it should be considered that this relationship is likely confounded by age distribution and sex differences between groups. The IPF group in our cohort had a higher proportion of males and fewer individuals aged 85 years or older, which may account for the higher reported rates of regular exercise rather than a direct protective effect. Future studies should collect detailed measures of activity intensity, duration, and changes over time to determine causality and potential mechanisms.
The clinical relevance of our study is that our results have valuable implications for the prevention and management of IPF, particularly in elderly populations. Identifying individuals with heightened risk profiles, such as elderly males with a history of smoking, obesity, or dyslipidemia, and those with COPD and a history of smoking, can guide early screening efforts and targeted preventive measures. Addressing modifiable risk factors, including smoking cessation programs, maintaining a healthy weight, and managing comorbid conditions such as dyslipidemia and COPD, could reduce IPF incidence in vulnerable groups. Moreover, the observed interactions between risk factors suggest that integrated strategies addressing multiple factors simultaneously might be more effective than isolated interventions.
Our study has several limitations. First, we included only participants who underwent health screening examinations, which could have introduced a healthy user bias. Second, the diagnosis of IPF and other comorbidities relied on ICD-10 codes, which could potentially lead to overestimations or underestimations of diagnoses. Therefore, some caution is warranted when interpreting the findings of this study. However, the ICD-10 codes used in this study have been validated in previous IPF studies.21,51 Specifically, we rigorously applied the operational definition of IPF using RID codes. Third, because our study was based solely on data from the Korean population, caution is needed when applying our results to other ethnic groups or populations in different countries. Finally, a limitation of our study is that information such as occupational exposure risk could not be included in the statistical analysis, as it was not available in the dataset. Another limitation is the absence of a sample size calculation or justification for the chosen cohort in the study’s methodology.
Conclusion
In conclusion, this study highlights key risk factors for IPF in elderly populations, including male sex, high BMI, smoking, dyslipidemia, CKD, and COPD, and demonstrates their multifactorial interactions. These findings advance the understanding of IPF in aging populations and offer a foundation for targeted prevention and management strategies.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251406024 – Supplemental material for Incidence and risk factors for idiopathic pulmonary fibrosis in individuals aged 75 years and older in an ultra-aging society
Supplemental material, sj-docx-1-tar-10.1177_17534666251406024 for Incidence and risk factors for idiopathic pulmonary fibrosis in individuals aged 75 years and older in an ultra-aging society by Bo-Guen Kim, Kyungdo Han, Ju-Yeong Park, Bongseong Kim, Dong-Woo Han, Sung Jun Chung, Hyung Koo Kang, Sang-Heon Kim, Moo Suk Park, Jang Won Sohn, Ho Joo Yoon and Hyun Lee in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251406024 – Supplemental material for Incidence and risk factors for idiopathic pulmonary fibrosis in individuals aged 75 years and older in an ultra-aging society
Supplemental material, sj-docx-2-tar-10.1177_17534666251406024 for Incidence and risk factors for idiopathic pulmonary fibrosis in individuals aged 75 years and older in an ultra-aging society by Bo-Guen Kim, Kyungdo Han, Ju-Yeong Park, Bongseong Kim, Dong-Woo Han, Sung Jun Chung, Hyung Koo Kang, Sang-Heon Kim, Moo Suk Park, Jang Won Sohn, Ho Joo Yoon and Hyun Lee in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
Not applicable.
Declarations
Supplemental material
Supplemental material for this article is available online.
Artificial intelligence use declaration
We hereby declare that no artificial intelligence tools or technologies were used in the creation, drafting, or editing of this work.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
