Abstract
Background:
There is uncertainty regarding the optimal next imaging modality for identifying likely malignant pulmonary lesions in patients with abnormal chest radiography, with or without respiratory symptoms.
Objectives:
We compared the diagnostic accuracy of chest contrast-enhanced computed tomography (CECT) and positron emission tomography or positron emission tomography-computed tomography (PET/PET-CT) for identifying malignant pulmonary lesions.
Design:
Systematic review and meta-analysis
Data sources and methods:
We searched the PubMed, Embase, Scopus, and Cochrane CENTRAL databases to identify head-to-head diagnostic accuracy studies comparing CECT and PET/PET-CT for their ability to differentiate between benign and malignant pulmonary lesions. The risk of bias of included studies was assessed using the Quality Assessment of Diagnostic Accuracy Studies – Comparative (QUADAS-C) tool. Meta-analysis was performed using the bivariate random effects model.
Results:
We included eight studies, comprising a total of 873 subjects. The pooled sensitivity and specificity of CECT (850 lesions) were 0.93 (95% CI, 0.89–0.96;
Conclusions:
Based on limited evidence with applicability concerns, CECT of the chest may have a higher sensitivity but lower specificity than PET/PET-CT for identifying malignant lesions among patients with suspected lung cancer.
Trial registration:
The protocol for the systematic review was prospectively registered on PROSPERO (CRD42024590904)
Keywords
Introduction
Lung cancer is the leading cause of cancer-related death worldwide. 1 Despite advances in the management of lung cancer, recent data reveal that the 5-year survival rate is only 27% in the United States. 2 The 5-year survival rates are likely lower in developing countries. 3 Diagnosis of lung cancer at an advanced stage is partly responsible for the high death rates, and the 5-year survival for metastatic lung cancer remains below 10%. 2 There has been some success in screening for lung cancer using low-dose computed tomography (LDCT) of the chest among high-risk groups such as current or former smokers in developed countries. 4 However, such screening programs are difficult to implement in developing countries, where most patients are still diagnosed at a relatively advanced stage after presenting with respiratory symptoms or incidentally detected lesions on a chest radiograph. 5
There are specific clues indicating the malignant nature of pulmonary nodules on a chest radiograph or a non-contrast computed tomography (CT) scan of the chest. For instance, malignant lesions are more likely to have a larger size, spiculated margins, pleural retraction or thickening, and vascular convergence. 6 However, the diagnostic accuracy of plain CT is unsatisfactory; therefore, most patients with abnormal chest radiography or LDCT undergo further investigation using other imaging modalities before proceeding to invasive procedures for histopathological testing. These imaging modalities can predict malignancy in lung lesions using features such as vascularity (e.g., contrast-enhanced computed tomography (CECT)) or metabolic activity (e.g., positron emission tomography (PET)). 7 While CECT is commonly employed in this setting, there is a lack of literature reviews comparing its effectiveness head-to-head with other imaging modalities in this context.
In this systematic review and meta-analysis, we compare the diagnostic accuracy of CECT of the chest with positron emission tomography or positron emission tomography-computed tomography (PET/PET-CT) for detecting malignancy among patients with suspected lung cancer based on abnormalities on chest radiography or non-contrast CT, with or without respiratory symptoms.
Methods
The protocol for the systematic review was prospectively registered on PROSPERO (CRD42024590904). The protocol was prepared in accordance with the standard operating procedure of the funding agency. The complete review protocol encompasses comparison of diagnostic accuracy of chest contrast-enhanced computed tomography (CECT) with other imaging modalities (i.e., positron emission tomography (PET) or positron emission tomography-computed tomography (PET-CT) or magnetic resonance imaging (MRI)) for biopsy-proven lung cancer. The study was conducted between June 2024 and March 2025. In the present study, we report the results of the comparison between the diagnostic accuracy of CECT and PET/PET-CT. This report has been prepared following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 8
Inclusion and exclusion criteria
In this review, we included English-language studies that compared the diagnostic accuracy of CECT of the chest with PET/PET-CT for differentiating benign and malignant pulmonary lesions. Only studies that included subjects with abnormalities on chest radiography and non-contrast CT of the chest suspicious of cancer, with or without respiratory symptoms, were considered. The permissible reference test was either histopathological diagnosis (for malignant or benign lesions) or 2-year radiological follow-up (for benign lesions only). We excluded review articles, case reports, and case series. We also excluded studies of dynamic CECT that did not report the diagnostic accuracy of a peak contrast enhancement parameter, as dynamic CECT protocols are more complex, less standardized, and less widely used in clinical practice than conventional CECT of the chest.
Search strategy
We performed a literature search of PubMed, Embase, Scopus, and the Cochrane Central Register of Controlled Trials (CENTRAL) using the keywords “computed tomography,” “contrast,” and “lung cancer.” The exact search strategies are presented in Supplemental Tables 1 to 4. The search results were compiled in the CADIMA software, and duplicates were removed. Two authors (TS and AG) initially screened the titles and abstracts and then performed a full-text review of selected articles. Any conflicts were resolved through mutual discussion or with the involvement of a third investigator (AM).
Data extraction
Two authors (TS and AG) independently performed data extraction from the eligible full texts. Details recorded included the author’s name, year of publication, country, study design, imaging modalities used, imaging-specific cutoffs for labeling malignancy, sample size, and baseline demographics of the participants. Furthermore, for each imaging modality, we recorded the true positives, true negatives, false positives, and false negatives, using either histopathology (for benign or malignant diagnosis) or a 2-year multidisciplinary clinical follow-up (only for benign diagnosis) as the gold standard for lung cancer diagnosis. Thus, we obtained the sensitivity and specificity of each imaging modality for biopsy-proven cancer in the included studies. No attempt was made to contact the authors of individual studies for missing data.
Risk of bias assessment
The included studies were assessed for their risk of bias using the Quality Assessment of Diagnostic Accuracy Studies – Comparative (QUADAS-C) tool. 9 This tool is an extension of the QUADAS-2 tool and is specifically designed for reviews comparing the diagnostic accuracies of two index tests against a gold standard test. Accordingly, the risk of bias is assessed under the domains of population (P), index test (I), reference test (R), and flow and timing (FT) for each index test independently, as well as for the comparison of the two index tests separately. Applicability concerns are also addressed under the domains of population (P), index test (I), and reference test (R) to determine whether the study directly addresses the review question.
Statistical analysis
We performed the statistical analysis using the Stata software (StataCorp, 2023.
We had planned a pre-determined exploratory subgroup analysis based on age groups, sex, smoking status, and previous history of tuberculosis. Sensitivity analysis consisted of a leave-one-out analysis of the relative sensitivity and specificity of PET/PET-CT versus CECT to assess whether any of the included studies were influencing the results. Publication bias was evaluated using the Deeks’ funnel plot asymmetry test. 11
Results
Study selection and characteristics
The PRISMA flow diagram of study selection is shown in Figure 1. A total of 4979 records were identified through the database search, of which 4220 remained after duplicates were removed. Among these, 4160 studies were eliminated during the title and abstract screening. During the full-text screening, another 53 studies were excluded (Supplemental Table 5 lists the reasons for exclusion for each study at the full-text screening phase). A citation review revealed one additional study eligible for inclusion. Hence, a total of eight studies were included in the systematic review.

PRISMA flow diagram.
The characteristics of the included studies are summarized in Table 1. Among these, seven studies compared the diagnostic accuracy of CECT with PET-CT (832 subjects)12–18 and one study compared CECT with PET (41 subjects). 19 The studies were published between 2005 and 2022 and were conducted in Europe, South Korea, and the United States. All studies employed a prospective observational design, except for the study by Christensen et al., which compared CECT and PET and was retrospective. 19
Study characteristics and key findings.

CECT, contrast-enhanced computed tomography; HU, Hounsfield units; NA, not available; PET, positron emission tomography; PET-CT, positron emission tomography-computed tomography; SPN, solitary pulmonary nodule; SUV, standardized uptake value.
The thresholds for diagnosing malignancy varied between studies for each imaging modality. For CECT, seven out of eight studies used contrast enhancement cutoffs ranging between 15 and 25 Hounsfield Units (HU), with higher values used to identify likely malignancy. One study used a five-point criterion based on nodule characteristics on CECT, 14 whereas one study used either an HU cutoff or additional nodule characteristics. 16 For PET/PET-CT, seven out of eight studies used a standardized uptake value (SUV) cutoff ranging from 2.5 to 3.5, with higher values denoting likely malignancy. One study employed a five-point criterion, 14 whereas another used either an SUV cutoff or additional nodule characteristics. 16
Quality assessment
According to the QUADAS-C assessment, six out of the eight included studies had a high or unclear risk of bias in the population domain (Figure 2). Among these, one study employed an unpaired, non-randomized design, meaning that not all patients underwent both comparator index tests. 13 One of the studies employed a retrospective design, which could lead to selection bias. 19 Three of the studies had selection bias because PET/PET-CT was performed based on either CECT findings or if they were planned for surgical excision of the nodule.14,16,18 Most studies had a low risk of bias in both the index test and reference test domains. Across studies, the image assessors for CECT and PET/PET-CT were blinded to both the results of the alternative index test and to the results of the reference test. Most studies had a low risk of bias in the flow and timing domain, except for one report where concerns were raised regarding the interval between the index and reference tests. 13

Risk of bias assessment of included studies using the Quality Assessment of Diagnostic Accuracy Studies – Comparative (QUADAS-C) tool.
There were concerns regarding applicability in the population domain in six out of eight studies, as these studies included patients with pulmonary nodules only, and did not consider respiratory symptoms as an inclusion criterion. However, our review was aimed toward the broader population of suspected lung cancer based on symptoms and/or equivocal chest radiography or non-contrast CT. Only two studies (viz. Harders et al. and Quaia et al.) directly addressed this group.14,16 One study had applicability concerns in the index test domain because it used a unique five-point criterion for defining the likelihood of malignancy in CECT and PET-CT, rather than cutoffs based on HU or SUV parameters. 14
Study outcomes
To identify malignant pulmonary lesions, the sensitivity of CECT in the included studies ranged from 0.84 to 1.00, whereas the specificity of CECT ranged from 0.29 to 1.00 (Table 2). For PET/PET-CT, the sensitivity ranged from 0.76 to 0.97, and the specificity ranged from 0.18 to 1.00.
Diagnostic performance of CECT and PET/PET-CT for the diagnosis of malignant pulmonary lesions in the included studies.

Across the eight studies, there were a total of 850 lesions that were tested by both CECT and reference standard, while there were 851 lesions that were tested by both PET/PET-CT and reference standard.
CECT, contrast-enhanced computed tomography; FN, false negative; FP, false positive; PET, positron emission tomography; PET-CT, positron emission tomography-computed tomography; TN, true negative; TP, true positive.
On performing a meta-analysis of eight studies, including 873 participants, that compared the diagnostic accuracy of CECT and PET/PET-CT, the pooled sensitivity and specificity of CECT (n = 850 lesions) were 0.93 (95% CI, 0.89–0.96;

Pooled sensitivity and specificity of contrast-enhanced computed tomography (CECT) of chest (

Pooled sensitivity and specificity of positron emission tomography/positron emission tomography-computed tomography (PET/PET-CT;

Summary receiver operating characteristics curve for pooled sensitivity and specificity of (a) contrast-enhanced computed tomography (CECT) of chest, and (b) positron emission tomography/positron emission tomography-computed tomography (PET/PET-CT) for the diagnosis of malignant pulmonary lesions.
Relative sensitivities and specificities of PET/PET-CT in comparison to CECT of the chest for the diagnosis of malignant pulmonary lesions.

The table represents the marginal summary measures of test accuracy (relative measures) using the metadta command on Stata software.
CECT, contrast-enhanced computed tomography; CI, confidence interval; PET/PET-CT, positron emission tomography/positron emission tomography-computed tomography.
None of the pre-planned exploratory subgroup analyses were performed because the primary data in the included studies were not stratified according to age groups, sex, smoking status, or history of tuberculosis. To better understand the clinical implications of our results, we created a summary of findings (SoF) table for the pooled diagnostic accuracy results using three different levels of pre-test probability (Supplemental Table 6).
Sensitivity analysis
We performed a leave-one-out analysis for the comparison between the sensitivities and specificities of CECT and PET/PET-CT (Table 4). After excluding the study with the largest sample size (Gilbert et al.),
12
PET/PET-CT no longer had a lower relative sensitivity (RR, 0.99; 95% CI, 0.94–1.04;
Leave-one-out analysis of relative sensitivity and specificity of PET/PET-CT compared to CECT for diagnosis of biopsy-proven lung cancer.
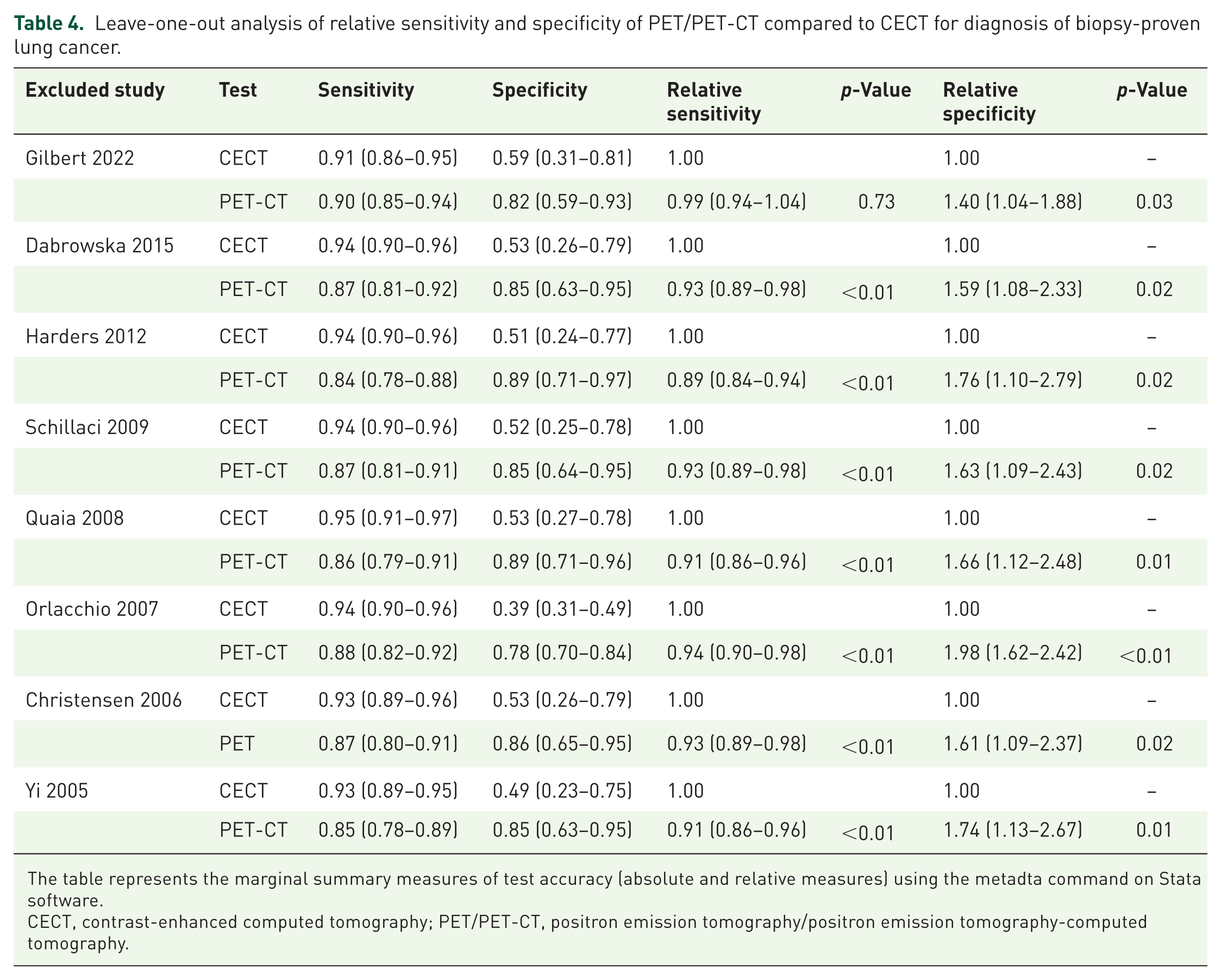
The table represents the marginal summary measures of test accuracy (absolute and relative measures) using the metadta command on Stata software.
CECT, contrast-enhanced computed tomography; PET/PET-CT, positron emission tomography/positron emission tomography-computed tomography.
The exclusion of the study that compared with CECT with PET instead of PET-CT and used a retrospective design (Christensen et al.), 19 did not alter the findings of relatively lower sensitivity and higher specificity of PET/PET-CT compared to CECT. Similarly, the exclusion of study that used higher cutoffs for SUV (3.5) and HU (25), that is, Yi et al, did not alter these findings. 18 Finally, the exclusion of the study that performed histopathology on all subjects with a benign diagnosis (Quaia et al.) also did not alter these findings (Table 4). 16
Publication bias
Upon applying Deeks’ funnel plot asymmetry test, we did not find any publication bias in the pooled analyses of diagnostic accuracy for either CECT (

Publication bias assessment using Deeks’ funnel plot asymmetry test for pooled diagnostic accuracy of (a) contrast-enhanced computed tomography (CECT) of chest, and (b) positron emission tomography/positron emission tomography-computed tomography (PET/PET-CT) for the diagnosis of malignant pulmonary lesions.
Discussion
In this systematic review and meta-analysis, we compared the diagnostic accuracy of contrast-enhanced computed tomography (CECT) of the chest versus positron emission tomography/positron emission tomography-computed tomography (PET/PET-CT) for identifying malignant pulmonary lesions among patients with suspected lung cancer who presented with an abnormal chest radiography or non-contrast CT with or without respiratory symptoms. We found that the pooled sensitivity and specificity of CECT were 0.93 and 0.54, while the pooled sensitivity and specificity for PET/PET-CT were 0.87 and 0.83, respectively. Furthermore, while CECT was relatively more sensitive, PET/PET-CT was relatively more specific for identifying malignancy in this setting.
Cancerous tissues have high vascularity and increased glucose metabolism. These properties can be leveraged through the use of imaging techniques such as CECT (which achieves higher contrast enhancement in more vascular tissues) or fluorodeoxyglucose F18 (FDG)-PET (which detects higher metabolic activity by increased FDG uptake).20,21 Currently, FDG-PET scanners are typically integrated with computed tomography (CT) to provide both anatomical and metabolic information about lesions in the form of a PET-CT scan. There are relative advantages and disadvantages of each of these modalities. CECT is widely available and less expensive than PET-CT, offering better anatomical resolution for characterizing smaller lesions (below 8–10 mm) and part-solid lesions (e.g., adenocarcinoma). 22 On the other hand, PET-CT may be superior to CECT in identifying the stage of the tumor by detecting mediastinal and distant metastases. 23
Previous meta-analyses of the diagnostic accuracy of CECT and PET-CT for identifying malignant lung nodules have shown similar findings to our study. For instance, Zhang et al. performed a meta-analysis of 32 studies, finding a pooled sensitivity and specificity of 0.89 and 0.70, respectively, for chest CT. 24 On the other hand, Ruilong et al. conducted a meta-analysis of 12 studies and found that the pooled sensitivity and specificity of PET-CT for identifying malignant lung nodules were 0.82 and 0.81, respectively. 25 Thereafter, Jia et al. performed a meta-analysis providing an indirect comparison of CECT and PET-CT for differentiating malignant and benign pulmonary nodules. They found there was no significant difference in the pooled sensitivity and specificity of CECT (0.94 and 0.73) and PET-CT (0.89 and 0.78) in this setting. 26 However, Jia et al. did not include studies that directly compared the diagnostic performances of CECT and PET/PET-CT performed in the same patients.
In this context, a key strength of our review is the inclusion of studies that directly compared the use of CECT and PET/PET-CT for differentiating benign and malignant pulmonary lesions. In our head-to-head comparison, we found that CECT had a higher sensitivity than PET/PET-CT, which enabled it to detect more cases of malignancy upfront, albeit with an increased number of false positives. On the other hand, PET/PET-CT had a significantly higher specificity but may miss identifying many cases of cancer. While false positives on CECT can cause anxiety and lead to more invasive testing, it is essential not to miss identifying any potentially malignant lesions for appropriate invasive tissue sampling. In this context, sequentially performing CECT upfront followed by PET-CT in suspicious lesions may improve the diagnostic accuracy of imaging for identifying malignancy at potentially lower cost, although such an approach needs to be explored in prospective studies. Gilbert et al. have shown that the combined testing approach improves diagnostic accuracy compared to either PET-CT alone or CECT alone, and is cost-effective provided that there is a willingness to pay over £15,500 incremental cost per correctly treated malignancy. On the other hand, CECT alone may be preferred if the willingness to pay was under £9,000. 12
Our systematic review primarily compared the diagnostic accuracies of CECT and PET-CT for detecting lung malignancy. One included study (Christensen et al.) compared CECT with PET, the latter being obsolete in current clinical practice. 19 Nonetheless, upon excluding this study in a leave-one-out analysis, the results did not alter, and PET-CT was significantly less sensitive and more specific than CECT for detecting lung malignancy. Furthermore, while the pooled specificity of PET/PET-CT was 0.83, the study by Quaia et al. was an outlier, with a PET-CT specificity of only 0.18. 16 This study was unique in using either an SUV cutoff or additional nodule characteristics (spiculation, invasion, ground-glass opacity, and nodal metastasis) to denote malignancy on PET-CT. Additionally, it performed histopathology in all benign cases (as opposed to radiological follow-up for benign lesions). Whether the liberal PET-CT positivity definition, combined with the stringent reference standard for malignancy diagnosis, led to a lower specificity for PET-CT in this study is unclear. Nonetheless, excluding this study in a leave-one-out analysis did not alter our overall results.
A significant limitation of our review was that applicability concerns arose for six out of eight included studies. These studies were confined to patients with pulmonary nodules, which represent only a subset of patients with suspected lung cancer. Respiratory symptoms were not considered for inclusion in most studies. Second, many studies had a mixed population of lung cancer, carcinoid tumors, and pulmonary metastases from extrapulmonary tumors. However, this is reflective of real-life clinical practice, wherein it may be difficult to predict the nature of a lung tumor before histopathological diagnosis. Third, there was considerable inter-study heterogeneity in the pre-specified thresholds of contrast enhancement (>15, 20, or 25 HU) for CECT and SUVs (2.5 or 3.5) for PET/PET-CT for determining the risk of malignancy. Hence, it is not possible to estimate ideal cutoffs for these parameters based on this study. Furthermore, it was not feasible to perform subgroup or meta-regression analyses to examine the impact of varying diagnostic cutoffs due to the small number of included studies. Fourth, the use of histopathology as a reference standard in all malignant cases and the choice of either histopathology or radiological follow-up for benign cases could introduce bias in the study. However, this is also reflective of standard clinical practice, wherein a lack of growth in pulmonary nodules over two or more years is taken as a sign of a benign process. We also did not include dynamic CECT studies based on contrast wash-in or wash-out characteristics to predict malignancy, unless they explicitly reported the diagnostic performance of a peak post-contrast enhancement level. Dynamic CT protocols are more complex, less standardized, time-consuming, and less widely used in clinical practice than conventional CECT.
Our review included only English language studies due to logistical reasons and may have missed relevant non-English language studies. Furthermore, although no publication bias was detected, the Deeks’ funnel plot asymmetry test may be unreliable in the presence of only eight included studies. Finally, a leave-one-out analysis found that the sensitivity of CECT and PET/PET-CT did not differ after excluding the study with the highest sample size, which limits our confidence in this finding.
Conclusion
In conclusion, we performed a meta-analysis of diagnostic accuracy studies providing a head-to-head comparison of CECT and PET/PET-CT for identifying malignant pulmonary lesions among patients with suspected lung cancer. We found that CECT had a significantly higher pooled sensitivity than PET/PET-CT (0.93 vs 0.87), but a lower pooled specificity (0.54 vs 0.83) for identifying biopsy-proven lung tumors. However, after excluding the study with the largest sample size, CECT was no longer more sensitive than PET/PET-CT. Furthermore, there were concerns about the high risk of bias and applicability in most studies. Further research is needed to clarify the diagnostic performance and cost implications of a sequential or combined imaging approach with CECT and PET-CT for determining the presence of malignancy in suspicious lung lesions.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251395432 – Supplemental material for Contrast-enhanced computed tomography versus positron emission tomography/positron emission tomography-computed tomography in suspected lung cancer: a systematic review and meta-analysis of diagnostic accuracy studies
Supplemental material, sj-docx-1-tar-10.1177_17534666251395432 for Contrast-enhanced computed tomography versus positron emission tomography/positron emission tomography-computed tomography in suspected lung cancer: a systematic review and meta-analysis of diagnostic accuracy studies by Tejas Menon Suri, Ayush Goel, Maroof Ahmad Khan, Pawan Tiwari, Saurabh Mittal, Vijay Hadda, Karan Madan and Anant Mohan in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251395432 – Supplemental material for Contrast-enhanced computed tomography versus positron emission tomography/positron emission tomography-computed tomography in suspected lung cancer: a systematic review and meta-analysis of diagnostic accuracy studies
Supplemental material, sj-docx-2-tar-10.1177_17534666251395432 for Contrast-enhanced computed tomography versus positron emission tomography/positron emission tomography-computed tomography in suspected lung cancer: a systematic review and meta-analysis of diagnostic accuracy studies by Tejas Menon Suri, Ayush Goel, Maroof Ahmad Khan, Pawan Tiwari, Saurabh Mittal, Vijay Hadda, Karan Madan and Anant Mohan in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
