Abstract
Objective
To evaluate the clinical usefulness of preoperative positron emission tomography–computed tomography (PET–CT) in primary papillary thyroid cancer (PTC).
Methods
Preoperative PET–CT scans of patients with biopsy-confirmed PTC who were undergoing thyroidectomy were examined and the maximum standardized uptake value (SUVmax) of 2-deoxy-2-(18F)fluoro-
Results
The study included 194 patients. Multivariate analysis indicated that patients were significantly more likely to be PET positive if they were female, had larger tumours (>1 cm), coexisting pathology (nodular hyperplasia or Hashimoto’s thyroiditis) or nonfollicular variant PTC. SUVmax <2.0 indicated possible follicular variant PTC.
Conclusions
PET-positive results were unrelated to extrathyroidal extension and lymph node metastasis. PET positivity was related to larger tumour size and implied coexisting pathology. PET negativity or low SUVmax suggested possible follicular variant PTC.
Introduction
Positron emission tomography (PET) using 2-deoxy-2-(18F)fluoro-
Preoperative PET–CT is not universally performed in patients with thyroid cancer because 18F-FDG uptake depends on the degree of tumour cell differentiation, such that uptake is higher in poorly differentiated than in well-differentiated thyroid tumours. 5 In addition, well-differentiated thyroid tumours with iodine avidity have low glucose metabolism, limiting the diagnostic value of PET–CT in thyroid cancer. 6 PET is more useful in follow-up, for detecting recurrent or metastatic differentiated thyroid cancer in patients with negative radioiodine scans and elevated thyroglobulin levels. 7
The aim of the present study was to evaluate preoperative PET–CT 18F-FDG uptake in a group of patients with primary papillary thyroid cancer (PTC) with a wide range of clinicopathological features, in order to determine the usefulness of preoperative PET–CT in PTC.
Patients and methods
Study Population
This study involved consecutively recruited patients who underwent preoperative PET–CT and surgery for PTC at the Department of Surgery, Chung-Ang University Hospital, Seoul, Republic of Korea, between March 2011 and February 2012. PTC was preoperatively confirmed by fine-needle aspiration biopsy (FNAB) and diagnosed according to the National Cancer Institute classification system. 8 Preoperative PET–CT was recommended for each patient. Patients who showed ambiguous 18F-FDG uptake for primary lesions and diffuse high uptake for the surrounding thyroid tissue were excluded from the study because diffuse uptake by thyroiditis may mask uptake by primary thyroid cancer. Clinical and demographic data for the study cohort were determined by retrospective review of medical records. All patients underwent prophylactic central neck dissection. Additional selective lateral neck dissection was performed in those patients with clinically confirmed lateral neck lymph node metastases.
The study protocol was reviewed and approved by the Institutional Review Board of the Chung-Ang University Medical Centre, Seoul, Republic of Korea, and the requirement for informed consent was waived.
PET–CT Scanning
A PET–CT scanner (Gemini TF, Philips, Hamburg, Germany) was used to perform 18F-FDG PET–CT ≥7 days after FNAB. After fasting for ≥8 h, each patient received 370 MBq 18F-FDG intravenous injection; PET–CT scanning was performed 60 min later. Patients were scanned from the middle of the skull to the upper thigh. PET images were reconstructed onto a 128 × 128 matrix using the ordered-subsets expectation maximization algorithm (four interactions and eight subsets) with attenuation correction. CT images were reconstructed onto a 512 × 512 matrix and converted using 511 keV equivalent attenuation factors for attenuation correction. PET, PET–CT and CT images were reviewed using a dedicated workstation. Extended Brilliance Workspace software (version 3.5.2.2260; Philips Medical Systems, Best, Netherlands) was used to construct three-dimensional displays (transaxial, coronal and sagittal) using CT, PET and PET–CT images, and the maximum intensity projection displays of PET data.
A single nuclear medicine specialist quantified 18F-FDG uptake using standardized uptake values (SUV) calculated as follows: SUV = (decay corrected activity [kBq]/tissue volume [ml])/(injected 18F-FDG activity [kBq]/body mass [g]). The SUV of a lesion was obtained by placing regions of interest (ROIs) manually around the lesion. The maximum SUV (SUVmax) within a ROI was used to minimize partial volume effects. Any visually discernible 18F-FDG uptake within thyroid nodules was classified as PET positive. If there was no visibly higher 18F-FDG uptake than in the surrounding thyroid tissue, the tumour was classified as PET negative. For PET-negative results, the SUVmax of PTC lesions were obtained by reference to ultrasonographic and CT images. The SUVmax of PET-negative results was identical to the 18F-FDG value of surrounding thyroid tissue.
Histological Classification
An experienced pathologist (H.S.R.) reviewed tumour pathology after thyroidectomy and tumours were classified according World Health Organization guidelines for classification of PTC. 9 Hashimoto’s thyroiditis was described according to histology, regardless of clinical symptoms or radiological findings. 10 Multicentricity was defined as more than two tumour foci in ipsilateral or contralateral lobes. Extrathyroidal extension was diagnosed when carcinoma extended into perithyroidal soft tissue, including adipose tissue and skeletal muscle, as well as around sizeable vascular structures and nerves. Lymph node metastasis included central and lateral neck metastasis.
Clinical and Demographic Definitions
Age was classified as younger (aged <45 years) or older (aged ≥45 years), as suggested by the American Joint Committee on Cancer staging manual. 11 Tumour size was classified as smaller (≤1 cm) or larger (>1 cm).
Statistical Analyses
Data were presented as
Results
The study included 194 patients with FNAB-confirmed PTC (40 males/154 females; mean age 48.6 ± 13.4 years, age range 19–90 years). PET–CT revealed 18F-FDG uptake for primary thyroid cancer in 138/194 of these patients (diagnostic sensitivity 71.7%). Postoperative histological findings revealed central lymph node metastasis in 70/194 patients (36.1%) and lateral neck lymph node metastasis in 24/194 (12.4%) patients. Preoperative PET–CT findings suggested central lymph node metastasis in three of 70 patients (sensitivity 4.3%). Preoperative PET–CT suggested the presence of lateral lymph node metastasis in 17 patients, two of whom were found to have no such metastasis, during surgery. The sensitivity of PET–CT for lateral neck lymph node metastasis was therefore 62.5% (15/24 patients), with a false-positive rate of 1.1% (two of 179 patients).
Demographic, clinical and pathological characteristics of patients with fine-needle aspiration biopsy-confirmed primary papillary thyroid cancer (PTC), included in a study to evaluate the clinical usefulness of preoperative positron emission tomography–computed tomography (PET–CT), stratified according to PET-positivity
a
(
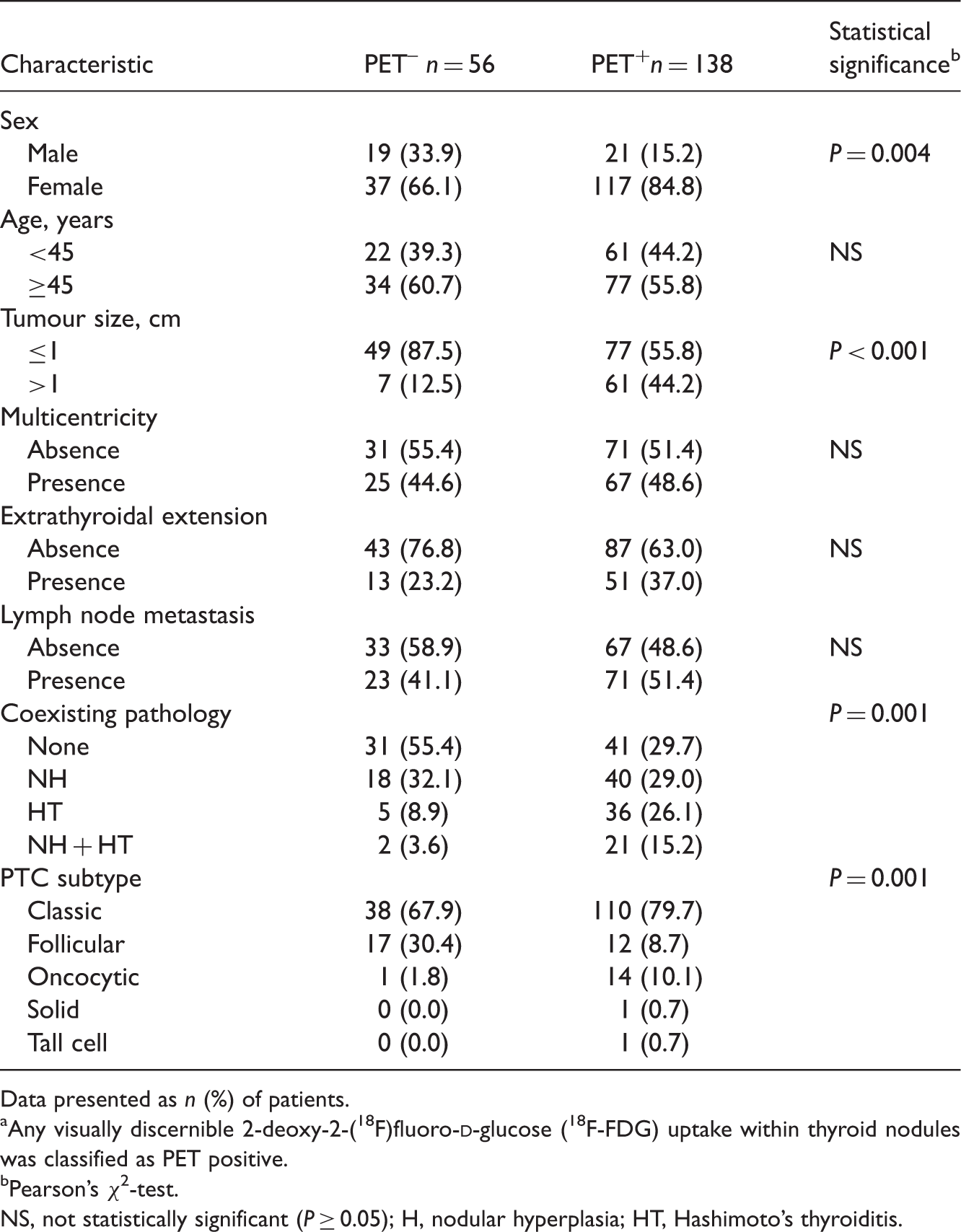
Data presented as
Any visually discernible 2-deoxy-2-(18F)fluoro-
Pearson’s χ2-test.
NS, not statistically significant (
Logistic regression analysis of demographic, clinical and pathological characteristics associated with preoperative positron emission tomography (PET) positivity
a
in patients with fine-needle aspiration biopsy-confirmed primary papillary thyroid cancer (PTC) (

Any visually discernible 2-deoxy-2-(18F)fluoro-
Maximum standardized uptake value (SUVmax) of preoperative positron emission tomography (PET) in patients with fine-needle aspiration biopsy-confirmed primary papillary thyroid cancer (PTC), stratified according to demographic, clinical and pathological characteristics (

Data presented as median (interquartile range).
Mann–Whitney
NS, not statistically significant (
Demographic, clinical and pathological characteristics of patients with fine-needle aspiration biopsy-confirmed primary papillary thyroid cancer (PTC), stratified according to absence or presence of extrathyroidal extension (

Data presented as
Pearson’s χ2-test; bFisher’s exact test.
NS, not statistically significant (
Demographic, clinical and pathological characteristics of patients with fine-needle aspiration biopsy-confirmed primary papillary thyroid cancer (PTC), stratified according to absence or presence of lymph node metastasis (

Data presented as
Pearson’s χ2-test.
NS, not statistically significant (
Demographic, clinical and pathological characteristics of patients with fine-needle aspiration biopsy-confirmed primary papillary thyroid cancer (PTC), stratified according to histological subtype (nonfollicular or follicular;

Data presented as
Pearson’s χ2-test.
NS, not statistically significant (
Logistic regression analysis of clinical and pathological characteristics associated with follicular histological subtypes of primary papillary thyroid cancer (

SUVmax, maximum standardized uptake value.
Discussion
The sensitivity of preoperative 18F-FDG PET–CT for thyroid cancer has been reported to be 60% in unclassified tumour types. 12 The present study found a PET-positive rate of 71.7%, and significant associations between positivity and clinicopathological parameters including female sex, larger tumour size, nonfollicular tumour type and presence of coexisting pathology (nodular hyperplasia or Hashimoto’s thyroiditis).
The presence of a positive PET signal indicates increased glucose and 18F-FDG uptake in a cancer cell. Tumours in female patients were more frequently PET positive and had higher quantitative 18F-FDG values than those in male patients, in the present study. Studies have indicated a poorer prognosis of well-differentiated thyroid cancer in men than in women.13,14 In addition, it has been suggested that 18F-FDG uptake in differentiated thyroid cancer is a sign of higher-grade malignancy,
15
and there may be a relationship between solute carrier family 2 (facilitated glucose transporter), member 1 (
The presence of coexisting pathologies (such as nodular hyperplasia or Hashimoto’s thyroiditis) significantly increased the likelihood of PET positivity in the present study. No previous report has shown a relationship between PET positivity and coexisting benign thyroid disease. The phosphatidylinositol 3-kinase(PI3K)/protein kinase B (Akt) pathway has been found to be involved in both Hashimoto’s thyroiditis and well-differentiated thyroid cancer. 18 It is possible, therefore, that other molecules (such as those related to 18F-FDG uptake) may be coexpressed in thyroid cancer with thyroiditis. Evidence suggests that nodular hyperplasia itself may be PET positive, 19 leading to possible confusion between the PET signals of these two conditions. The absence of any significant differences in median SUVmax between PTC (with or without coexisting pathologies) in the present study may have been due to the relatively small study cohort.
Follicular variant PTC was significantly less likely to be PET positive and had significantly lower SUVmax than other subtypes, in the present study. The clinical implications of follicular variant PTC have not yet been clearly elucidated. Some studies have reported that the follicular variant had preferable clinicopathological features to the classic type;20,21 others showed no between-group differences.22,23 It has been suggested that central node dissection may be unnecessary in follicular variant PTC because of the absence of central lymph node metastasis. 24 Data from the present study suggest that PET negativity may be helpful for preoperative determination of follicular variant PTC.
It has been suggested that PET positivity in papillary thyroid microcarcinomas (i.e. those that are ≤1 cm) implies lymph node metastasis and extracapsular tumour extension. 25 In contrast, others have shown that PET positivity is unrelated to extrathyroidal extension. 26 Extrathyroidal extension was associated with larger tumour size in the present study, and tumour size was related in turn to PET positivity and high SUVmax. When considering the relationships between independent variables such as tumour size, subtype, lymph node metastasis and SUVmax, PET positivity was not an independent factor for extrathyroidal extension. Other studies did not consider the PTC subtype,25,26 which may act as a confounding factor. The rate of extrathyroidal extension is thought to differ between study centres due to subjectivity in the criteria for defining minimum extrathyroidal extension. 27 In a similar manner, the presence of lymph node metastasis could differ according to the bounds of central neck dissection. These differences could affect the relationships between 18F-FDG uptake and extrathyroidal extension or lymph node metastasis. Prospective, multicentre studies with common pathological criteria defining extrathyroidal extension and PTC subtype, in addition to common principles for central neck dissection, would be needed to overcome this interobserver variability.
In conclusion, the value of preoperative PET–CT in PTC remains unclear. PET-positive results and high SUVmax were not related to significant clinical factors such as extrathyroidal extension or lymph node metastasis, in the present study. PET positivity was, however, related to larger tumour size and indicated possible coexisting pathology. In addition, PET negativity or low SUVmax suggested possible follicular variant PTC.
Footnotes
Acknowledgement
The authors thank Dr. JW Seok (Department of Nuclear Medicine, Chung-Ang University Hospital, Seoul, Republic of Korea) for his assistance in reviewing PET–CT images.
Declaration of Conflicting Interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
