Abstract

Dear Editor,
We enjoyed reading Obeidat and colleague’s retrospective cohort study on sodium-glucose cotransporter-2 inhibitor (SGLT2i) use following pulmonary hypertension diagnosis and its association with clinical outcomes. 1 Given that TriNetX is a relatively new tool to conduct retrospective studies, 2 research design errors that introduce bias can be easily made. We have identified two issues with Obeidat and colleague’s studies, which may account for the improved clinical outcomes seen in the SGLT2i cohort.
Firstly, the authors selected the index event as the date of the first post-pulmonary hypertension diagnosis-SLGT2i use in the SLGT2i cohort. However, the index event for the control cohort is simply the date of pulmonary hypertension diagnosis. Therefore, the observation period of 365 days likely represents a period of enhanced clinical monitoring for the control cohort, as they are likely symptomatic at the time of receiving a pulmonary hypertension diagnosis. In contrast, the SGLT2i cohort’s observation period could occur after a patient’s symptomatology has already been relatively stabilised. For example, an SGLTi2 cohort patient could present with pulmonary hypertension in 2012, experiencing hospitalisation, intubation and acute right heart failure in the year following this diagnosis. Then, in 2019, long after the patient’s pulmonary hypertension is successfully stabilised, they are prescribed an SGLT2i for an unrelated indication. It is therefore possible that the poorer clinical outcomes in the control group are simply representative of the likely morbidity and mortality that occur immediately following pulmonary hypertension diagnosis.
Secondly, we believe that the authors have not successfully matched laboratory values between cohorts. We re-calculated standard differences comparing the mean laboratory outcomes matched in Obeidat and colleague’s group 1 pulmonary hypertension analysis and found that body-mass index (BMI) remained unbalanced post-matching (Table 1). It is likely that only the proportion of patients receiving each test was balanced, rather than the results of the laboratory tests themselves. This oversight is common in TriNetX-based studies3,4 and can be remedied by balancing the proportion of patients falling within pre-determined range values for each laboratory outcome. For example, to balance BMI, the proportions of patients that fall within healthy (18–25 kg/m2), overweight (25–30 kg/m2) and obese (>30 kg/m2) could be balanced by setting these ranges at the input stage.
Re-calculations of standard differences for post-matching laboratory values in the group 1 pulmonary hypertension analysis.
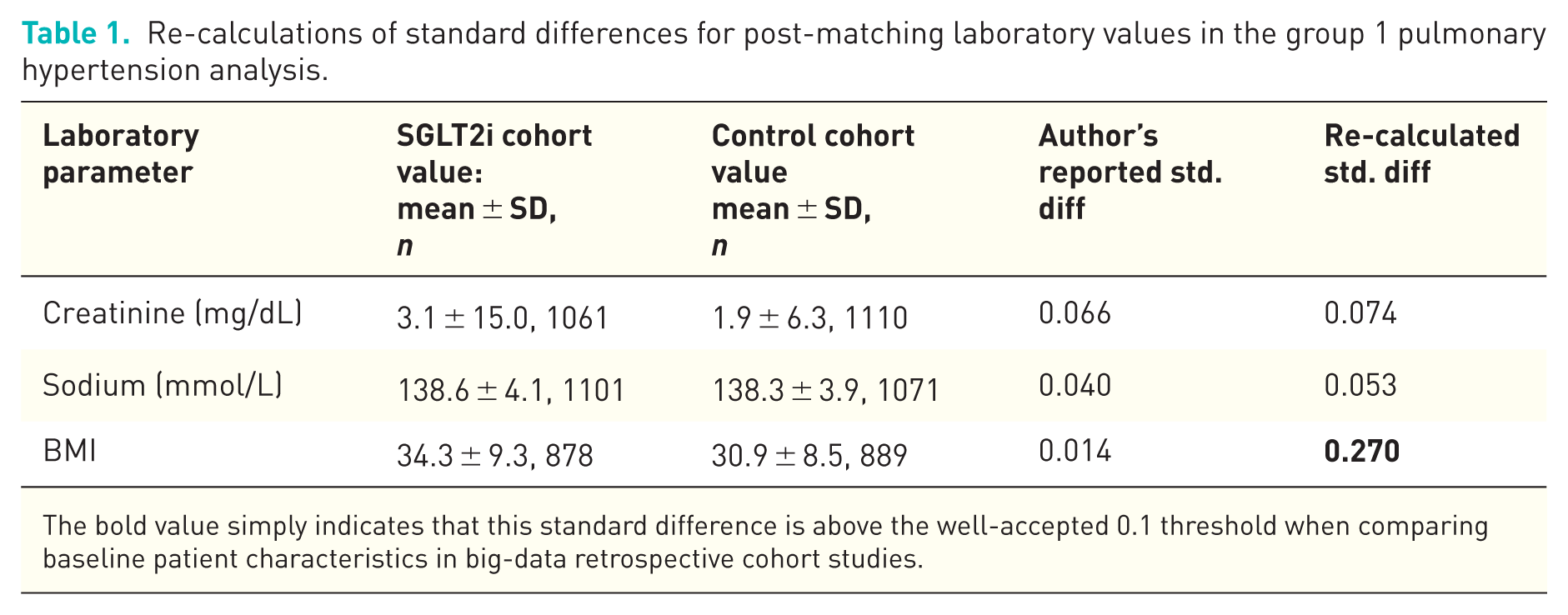
The bold value simply indicates that this standard difference is above the well-accepted 0.1 threshold when comparing baseline patient characteristics in big-data retrospective cohort studies.
To address these limitations, the study could be repeated by defining SGLTi use within a specified timeframe post-pulmonary hypertension diagnosis (e.g. within 1 year of diagnosis). Then, the index events for both cohorts can be defined as the date of pulmonary hypertension diagnosis, with an outcome period of 1–2 years post-pulmonary hypertension diagnosis. This design, in combination with properly balanced laboratory confounders, would produce more robust clinical outcomes data. Unfortunately, we were unable to perform this replication directly as the clinical codes used were not reported in the original publication. We would, therefore, encourage Obeidat and colleagues to perform this re-analysis to determine whether these limitations influenced the outcomes of their study.
