Abstract
Obstructive sleep apnea (OSA) is a sleep-breathing disorder with repetitive total or partial airway occlusion events. Its importance lies in the high prevalence and presence in all age groups and on cardiovascular (CV), metabolism, neurocognitive function, mental health, and overall quality of life. Furthermore, the coexistence of multiple comorbidities in patients suffering from OSA plays an essential role in these entities’ development, perpetuation, and clinical control. In the past years, efforts have been made to understand the pathophysiology of each event, the spectrum of the disease, and the different phenotypes, as well as to refine the diagnostic process and, thus, direct treatment toward a more comprehensive and personalized approach. Many uncertainties remain regarding which patients are at higher CV risk and who derive the greatest benefit from treatment. In this promising scenario, new tools have been developed, including advanced diagnostic technologies, molecular biomarkers, and predictive statistic models tailored for precision medicine. However, to effectively understand and manage this heterogeneous and complex pathology, well-designed studies are essential to validate precision medicine models and ensure their efficient integration into routine clinical practice in a simple, accessible, and practical manner. In this article, we present a comprehensive review of the most relevant scientific evidence concerning the diagnosis and treatment of OSA. We examine significant cluster studies available to date and aim to provide the reader with personalized recommendations for diagnosing and treating OSA. Additionally, we propose new pathways for advancing precision medicine in the field of sleep-disordered breathing.
Introduction
Sleep disorders are a common pathology, and among them, obstructive sleep apnea (OSA) is the most prevalent. It is difficult to know precisely the worldwide prevalence of OSA due to the heterogeneity and complexity of its clinical expression, which leads to underdiagnosis, especially in groups without risk factors and symptoms highly suggestive of OSA. However, some studies have attempted to collect these data.1,2 These studies show that sleep-disordered breathing (SDB) affects all age groups, with a prevalence of at least 10% of the general population. OSA is characterized by total or partial occlusions of the upper airway during sleep. Concerning respiratory events, the primary underlying issue is the inability of the muscles that dilate the upper airway to neutralize the negative pressures created in the airway during inhalation. Several factors contribute to the narrowing of the oropharynx, including abnormal craniofacial anatomy, excess soft tissue in the neck, and the upward movement of fluid in the body when lying down. These factors increase the collapsing forces within the airway. Additionally, the effectiveness of the upper airway dilating muscles can be negatively impacted by the onset of sleep, reduced responsiveness of the genioglossus, and ventilatory instability, especially after arousal, as well as by the loop gain. 3 These respiratory events have important implications: OSA leads to brain arousal, changes in intrathoracic pressure, and intermittent episodes of hypoxemia and re-oxygenation. These events activate several pathways, including oxidative stress, sympathetic activation, inflammation, hypercoagulability, endothelial dysfunction, and metabolic dysregulation. 4 Although the exact mechanisms linking OSA to cardiovascular (CV) disease are not yet fully understood, it is suggested that these abnormal fluctuations in CV function may contribute to an increase in CV morbidity and mortality. 5 OSA is a significant independent risk factor for various CV outcomes, including hypertension, arrhythmia, coronary heart disease, heart failure, stroke, and sudden death. Also, these mechanisms lead to a disruption of homeostatic metabolic processes, which are further exacerbated by the presence of obesity or high-fat diets. 6 However, the clinical significance of these disorders remains unclear.
In addition to the complex pathophysiology, where each patient’s characteristics play a crucial role, the clinical expression and long-term complications also vary depending on individual susceptibility and adaptation capacity. In this framework of complexity and heterogeneity, with consequences at the level of CV and metabolic disease, deterioration in the quality of life related to sleep, decrease in work performance, and increase in traffic accidents, OSA is currently a global health problem underdiagnosed and undertreated.
In recent years, precision medicine has emerged, referring to the tailoring of clinical management to each patient’s unique characteristics. 7 Precision medicine offers significant potential for creating more accurate diagnoses, tailoring specific therapies to patients most likely to benefit, and avoiding treatments that could cause adverse effects or be ineffective for particular individuals. 8 It has been progressively incorporated into chronic diseases, such as OSA.
New approaches are needed that take into account the various clinical phenotypes and endotypes, as well as the high prevalence of SDB. We require new diagnostic methods that utilize innovative technologies to provide alternatives for sleep staging and respiratory evaluation beyond the apnea–hypopnea index (AHI) and to explore the role of biomarkers and bio-signals in disease classification. 9 In addition, personalized patient management could be enhanced by linking treatment selection to clinical and pathophysiological phenotypes and endotypes, while also assessing treatment outcomes beyond AHI measurements. Some tools have been developed, including control panels, multidimensional algorithms, and fingerprint tools, to integrate the relevant pathophysiological mechanisms better and assess their impact on each patient based on their susceptibility and adaptive capacity.10–12 These tools could help guide clinical decisions for diagnosis, treatment, and follow-up. The new clinical management guidelines for OSA aim to incorporate this multidisciplinary approach; however, there is still much field to explore, and these tools require further validation. 13
This article reviews key scientific evidence on diagnosing and treating OSA. We aim to provide clear recommendations based on personalized medicine and suggest ways to enhance precision medicine for sleep-related breathing issues.
Literature search methodology
An initial MEDLINE search was performed using the keywords “obstructive sleep apnea,” “cardiovascular risk,” and “precision medicine” for the publication years 1990 to 2025. A second MEDLINE search used additional keywords linked to each paragraph and subparagraph of this review, including “endotypes,” “phenotypes,” “clinical features,” “cluster analysis,” “diagnosis,” “guidelines,” “expert opinion,” “panel,” “treatment,” “polysomnography,” “risk stratification,” “biomarkers, “biosignals,” “prevention,” “inflammation,” “endothelial dysfunction,” “hypertension,” “ “autonomic dysfunction,” “risk factors,” “upper airway,” “comorbidity,” “treatable treats,” “obesity,” “mortality,” “cardiovascular disease,” “hypertension,” “positive airway pressure,” “mandibular advancement devices,” “diet,” “positional therapy,” “machine,” and “biomarkes”. Data from systematic reviews were highlighted.
Endotypes, phenotypes, and cluster analysis in obstructive sleep apnea
Obstructive sleep apnea is a highly complex disorder, and its heterogeneity demands a precise characterization of endotypes and phenotypes to optimize therapeutic decision-making.
Endotypes
The underlying endotypes include anatomical compromise, functional impairment of the pharyngeal dilator muscles, unstable ventilatory control (characterized by elevated loop gain), and a low arousal threshold. 14
Some anatomical factors contribute to the development of respiratory pauses. Some of them are shared between obese and nonobese patients, including macroglossia (enlargement of the tongue), retrognathia (recessed chin), alterations in soft palate tissue, and inflammation and edema of the larynx, which may be linked to smoking, alcohol use, or gastroesophageal reflux. 15 The volume of the tongue and lateral walls has been shown to increase the risk of OSA 16 independently. Nonobese patients with OSA exhibit excessive fat deposition, particularly in the anterolateral region of the upper airway, a narrower facial anterior–posterior distance, and a narrower bony pharynx.17,18 Regarding obese patients, the global concern over the “obesity epidemic” is rapidly growing, with projections indicating that both the prevalence of obesity and the average body mass index (BMI) will continue to rise. 19 One notable consequence of this trend is the increasing prevalence of obesity-related respiratory diseases, particularly OSA and obesity hypoventilation syndrome. Obesity is a significant risk factor for OSA: a 10% increase in weight is linked to a sixfold rise in OSA risk, 20 and it is suggested that human fatness is under substantial genetic control. 21 Patients with OSA caused by anatomical issues may benefit from surgical treatments such as increasing the size of the affected airway. Therapies such as continuous positive airway pressure (CPAP) or mandibular advancement devices (MAD) can also help manage. For those who are overweight or obese, these treatments should be accompanied by proper dietary management and physical activity to reach the ideal weight. The development of new antidiabetic drugs such as Glucagon-Like Peptide 1 Receptor Agonists GLP-1RA or tirzepatide could change the treatment paradigm for patients with moderate-to-severe OSA with obesity. These drugs have demonstrated a significant reduction in weight, OSA severity, and other related variables such as blood pressure, hypoxic load, inflammatory biomarkers, and sleep quality.22,23
Another important aspect is the role of the dilator muscles of the pharynx, particularly the genioglossus, which is the primary muscle involved in keeping the upper airway open during sleep. However, several factors—such as fatigue, neurological damage, myopathy, or dysfunction of the pharyngeal sensors—can impair the function of the genioglossus and lead to airway obstruction.24,25 Moreover, some authors have observed that awake patients with OSA exhibit increased electrical activity in the dilator muscle compared to healthy controls. 26 This issue may arise during the sleep phase when the reflex disappears, particularly in patients with an anatomical predisposition. In this context, hypoglossal nerve stimulation could serve as an effective and innovative alternative therapy for moderate-to-severe OSA in patients who cannot tolerate CPAP therapy. 27
Combining drugs with noradrenergic and antimuscarinic effects, like atomoxetine and oxybutynin, improves genioglossus muscle activity and upper airway patency during sleep. 28 These effects open new possibilities for the pharmacologic treatment of OSA.29,30
Respiratory center instability, also known as loop gain, refers to the sensitivity of the respiratory system in adjusting breathing in response to changes in carbon dioxide (CO2) partial pressure levels. Patients with high loop gain, such as those with OSA, exhibit an exaggerated response to these changes. 31 When the stimulation of the respiratory center decreases, the activity of the pharyngeal dilator muscles also decreases, increasing airway resistance and facilitating airway collapse. As a result, the clinical response to surgery may not meet expectations. 32 Alternative treatments, such as acetazolamide 33 or supplemental oxygen, have been suggested. 34 However, the complexity of the underlying pathophysiology makes it difficult to evaluate the results thoroughly.
Also, a low arousal threshold has been observed in some patients with OSA. It means that patients require minimal stimulation to respond to a respiratory event, making it challenging to gather respiratory stimulus signals needed to activate the dilator muscles of the pharynx, thus perpetuating the events. After arousal, most people experience a brief period of hyperventilation. If this hyperventilation is significant, it can lower the blood CO2 below the threshold required for normal breathing, leading to central apnea. Because the muscles that maintain the upper airway open rely on respiratory input, hypocapnia can reduce their activity, increasing the risk of airway collapse. Arousals interrupting breathing events in OSA are typically accompanied by pronounced hyperventilation, due to a significant increase in respiratory drive, leading to hypocapnia in some patients. Individuals with low arousal thresholds may wake before the dilator muscles can fully reopen the airway. Delaying arousal with sedatives could help treat the condition, provided the upper airway muscles can still respond adequately to respiratory stimuli to stabilize the airway before awakening. 35 However, caution is needed, as sedatives may prolong respiratory events in patients who cannot reopen the airway without arousal. 36
Additional factors contributing to disease development include end-expiratory lung volume, arousal intensity, and fluid redistribution.37,38
Phenotypes and cluster analysis
We can explore various approaches to the phenotypes described in OSA, focusing on anatomical and pathophysiological changes, sleep studies data, clinical evaluations, CV risk, and responses to treatment.
In recent years, there has been growing interest in the nonanatomical pathophysiological factors contributing to the development of OSA and how identifying these factors can help predict treatment response and the development of CV disease. 11 In this context, new pathophysiological phenotypes have been suggested, considering the various factors involved sharing some characteristics with the described endotypes.
Eckert et al. carefully define the proportions of key anatomical and nonanatomical contributors in a large cohort of patients with OSA compared to control subjects. 39 They propose a scale incorporating these four factors: passive critical closing pressure of the upper airway (an anatomic feature), overall loop gain, arousal thresholds to hypoxia and hypercapnia, and the reflex responses of upper airway dilator muscles to changes in negative interstitial pressure (nonanatomic features). Their results suggest that nonanatomic features play an important role in 56% of patients with OSA, emphasizing the need to look beyond purely anatomical characteristics.
A narrative review examines the phenotype of nonobese patients with OSA. 15 Several clinical features define this subgroup: patients tend to be women, snorers, nonsmokers, nondrinkers, younger and have smaller neck and waist circumferences. Additionally, they typically present with snoring but do not experience significant EDS, which is their primary reason for seeking consultation. From a polysomnography (PSG) perspective, nonobese patients with OSA exhibit distinct patterns, including lower AHI, shorter durations of oxygen desaturation, fewer desaturations per hour, and higher average oxyhemoglobin saturation compared to other groups. Pathophysiologically, these patients show reduced muscle responsiveness in the upper airway during sleep, a low respiratory arousal threshold, and a higher loop gain. Early recognition of this condition in these patients is crucial, as they often do not fit the “classic” OSA phenotype. Additionally, because they tend to be younger, they usually have poorer adherence to CPAP therapy, complicating their treatment. 40
Regarding the phenotype of patients responding to MAD by evaluating clinical, craniofacial, PSG, physiological, and treatment characteristics, its identification may open new clinical management opportunities for this group of patients. 41 This opportunity could simplify the approach in patients with moderate OSA without EDS, without other sleep disorders and without a high CV load in whom treatment with MAD could be considered as initial, even without having to perform a drug-induced sleep endoscopy. Effectively, studies have shown that MAD treatment can lead to clinical improvement and a significant reduction in AHI. 42
One of the first attempts to explore the heterogeneity of OSA clinical presentations using a data-driven approach was conducted by Lichuan Ye et al. 43 This novel approach, based on cluster analysis, examines relationships among patient groups to identify those who are similar to each other but significantly different from others. This approach enables a refined diagnosis by identifying symptom patterns and clinical phenotypes in 822 patients with newly diagnosed moderate to severe OSA. They established three clusters: Cluster 1 (32.7% of the cohort): “Disturbed Sleep Group”—This group is more likely to experience symptoms related to insomnia; Cluster 2 (24.7% of the sample): “Minimally Symptomatic Group”—Individuals in this cluster generally exhibit milder symptoms compared to the other two groups, but they have the highest probability of comorbidities such as high blood pressure, diabetes, or CV disease; Cluster 3 (42.6% of the cohort): “Excessive Daytime Sleepiness (EDS) Group”—This group shows greater EDS, as measured by the Epworth Sleepiness Scale, and a high likelihood of issues related to insufficient nighttime rest. These findings suggest that identifying clinical presentations based on combinations of symptoms and comorbidities can provide a more comprehensive understanding of OSA than relying solely on indices such as the AHI or the Oxygen Desaturation Index (ODI). Cluster analysis, based on symptoms, physical examination, risk factors, and comorbidities conducted in the French registry (Highlight a category within the topic) of patients with OSA, reveals even greater heterogeneity, with six OSA clusters identified. 44 Cluster 1: “The young symptomatic” (1,823 patients, 10%) is represented by young, overweight, symptomatic OSA patients with few or no co-morbidities compared to the other clusters; Cluster 2: “The older obese” (4200 patients, 23%) includes those who are elderly minimally symptomatic obese OSA with few co-morbidities; Cluster 3: “The multi-disease old obese” (3363 patients, 18.4%) is composed of the oldest patients, minimally symptomatic with multimorbidity OSA; Cluster 4: “The young snorers” (2715 patients, 14.9%) that includes young, overweight, minimally symptomatic OSA without co-morbidities; Cluster 5: “The drowsy obese” (3511 patients, 19.2%) agglutinates middle age, with few symptoms of OSA and few co-morbidities patients but experiencing daytime drowsiness; and Cluster 6: “The multi-disease obese symptomatic” (2642 patients, 14.5%) includes middle age, symptomatic, multimorbidity OSA patients with particularly poor lifestyle habits. In this line, Romero Peralta et al. conducted a cluster analysis involving 2525 patients from the Apnea-HUGU cohort. 45 They built the clusters using general data, comorbidities, sleep symptoms, anthropometric measurements, physical examinations, and sleep study results. Four distinct clusters were described: Cluster 1 (32.7% of the cohort): “Young obese male without comorbidity with moderate OSA and ORL malformations”—In them, the symptoms have a significant impact on their daily activities and driving, but they have low comorbidity and less oximetric impact compared to the other clusters. Cluster 2 (10% of the cohort): “Middle-aged obese male with very severe OSA symptomatic with comorbidity without developed CV disease”—this group has the highest frequency of EDS and witnessed apneas with significant oximetric impact. Cluster 3 (22.6% of the cohort): “Non-obese woman with mood disorders.” This group is characterized by a higher prevalence of antidepressants and anxiolytics use. The presence of snoring and the clinical pretest probability of suffering OSA are the lowest, and the apnea is moderate without oximetric impact. Cluster 4 (16.4% of the cohort): “Symptomatic obese male with established CV disease and severe OSA”—The EDS is present but with little impact on their daily life. Despite severe OSA, the oximetric impact is less pronounced than in cluster 2. These findings support the previous conclusions discussed. The search for clusters based on anatomical or pathophysiological alterations has been a key focus of other studies. Su-Jung Kim et al. aimed to identify and characterize phenotypes of OSA using a multiperspective clustering approach that incorporates craniofacial risks, obesity, apnea severity, arousability, symptoms, and comorbidities. 46 This study involved 421 Korean patients with OSA. Three phenotypic clusters were identified and characterized. Cluster 1 (39% of the cohort): “Non-craniofacial phenotype”—This group consisted of non-obese patients with moderate-to-severe OSA, exhibiting no skeletal discrepancies. Members of this cluster have a low arousal threshold (ArTh), minimal sleepiness, and low comorbidity. Cluster 2 (33% of the cohort): “Craniofacial skeletal phenotype”—This cluster included nonobese patients with moderate OSA who exhibited noticeable skeletal discrepancies. They also have a low ArTh, mild EDS, and low comorbidity. Cluster 3 (28% of the cohort): “Complicated phenotype, 28%”—Patients are obese with severe OSA and displayed skeletal discrepancies. This group have a high ArTh, EDS, and a high incidence of hypertension. This multiperspective clustering approach may provide a foundation for more precise therapeutic decision-making, including craniofacial skeletal interventions, beyond the usual characterization of OSA subgroups. Different studies have shown that OSA is a common disorder, with varying incidence rates between genders. Recent population-based studies indicate that the prevalence of OSA is approximately 22.5% in women (ranging from 3.7% to 63.7%) and around 27.3% in men (ranging from 9% to 86%). 47 Despite this significant prevalence among women, OSA has traditionally been viewed as a condition predominantly affecting men. In a recent cluster analysis exclusively within a female cohort of 1886 women diagnosed at a tertiary hospital Sleep Unit by cardiorespiratory polygraphy and based on general data, clinical variables, comorbidities and sleep study results, Fontanilles et al. revealed a heterogeneous presentation of OSA in women. 48 This study identified four phenotypic subtypes. Cluster 1 (27% of the cohort): “Middle-aged paucisymptomatic women without CV risk factors”—This group presents moderate OSA with mild oximetric impact. Cluster 2 (47% of the cohort): “Older paucisymptomatic women with established CV disease and severe OSA”—Older patients with greater CV disease burden and obesity, showing a significant oximetric impact from severe OSA. Cluster 3 (47% of the cohort): “Middle-aged women with “classic” symptoms and CV risk factors.” This group includes younger patients with moderate OSA and mild oximetric impact. Cluster 4 (14% of the cohort): “Middle-aged women with mood disorders, nonrestorative sleep and CV risk factors.” These patients suffer from moderate OSA with mild oximetric repercussions. Another interesting perspective is the cluster analysis based on PSG variables conducted by V Zinchuk et al. from a sample of 1247 participants. 49 The analysis examined four key pathophysiological domains of OSA linked to CV disease: sleep architecture disturbance, autonomic dysregulation, breathing disturbance, and hypoxia. Seven phenotypes were identified: “Mild,” “Periodic limb movements of sleep,” “Non rapid eye movement (NREM) and arousal,” “Rapid eye movement (REM) and hypoxia,” “Hypopnea and hypoxia,” “Arousal and poor sleep,” and “Combined severe.” CV risk was assessed using the Framingham risk score, and comorbidity burden was measured by the Charlson comorbidity index. The clusters could also be stratified by OSA severity. NONE/MILD OSA GROUP: “Mild” was the largest cluster (n = 533), whose patients were younger, had high sleep efficiency, normal REM sleep, and low comorbidity. The “Periodic limb movements of sleep” cluster (n = 119) had a median AHI of 10, higher Periodic Limb Movement Index, and elevated comorbidity rates compared to the ‘mild’ cluster; MODERATE OSA GROUP: the ‘NREM and poor sleep’ (n = 186) and ‘REM and hypoxia’ (n = 168) clusters had equivalent median AHIs. Poor sleep characterized ‘NREM and poor sleep’, while ‘REM and hypoxia’ showed the highest frequency of REM-related apneas and lowest oxygen saturation (72.8%). Both clusters were more obese and had higher rates of CPAP or home oxygen use; SEVERE OSA GROUP: the ‘Hypopnea and hypoxia’ cluster (n = 75, median AHI of 44) showed hypopneas with desaturations and preserved sleep architecture, while the ‘Combined severe’ cluster (n = 126, median AHI of 84) experienced severe hypoxic burden and high arousal indices. The ‘Arousal and poor sleep’ cluster included older patients with less CPAP use and showed a trend toward higher prevalence of CV issues compared to the ‘mild’ cluster. Adjusted analyses revealed increased risk for the ‘Periodic limb movements of sleep’, ‘Hypopnea and hypoxia’, and ‘Combined severe’ clusters compared to ‘Mild’. A follow-up analysis using conventional AHI severity categories found that moderate and severe OSA were not associated with increased risk compared to the mild/none category.
Table 1 summarizes the cluster analysis studies. These findings reveal how a combination of anatomical, clinical and PSG variables, influenced partly by varying individual polygenic contributions, can be grouped into clusters to facilitate understanding. OSA could also be categorized in other important ways for management. Some potential groups might include elderly patients, individuals exposed to environmental risk factors or those with CV comorbidities presenting as their first clinical symptom.
Cluster analysis in obstructive sleep apnea.
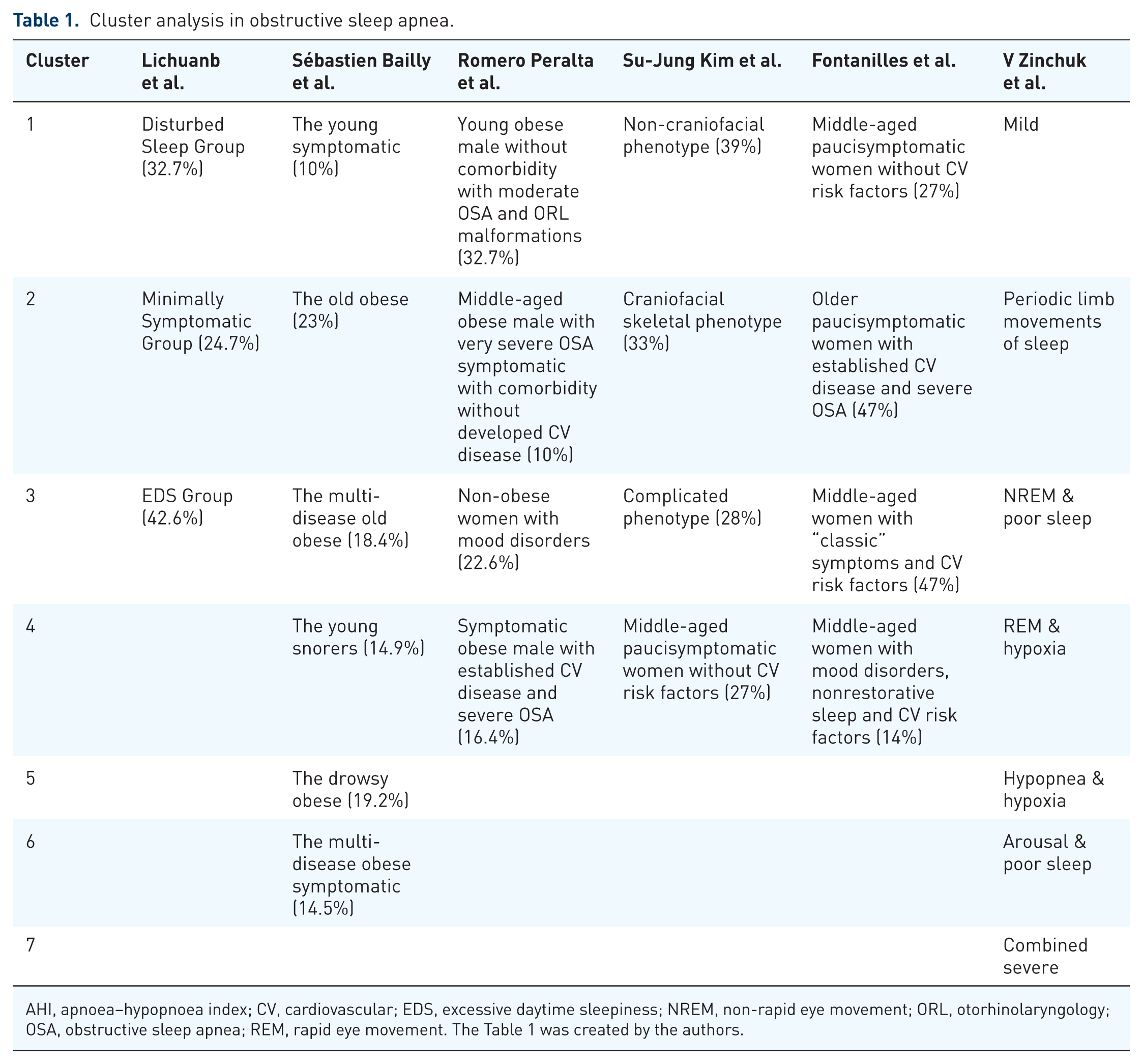
AHI, apnoea–hypopnoea index; CV, cardiovascular; EDS, excessive daytime sleepiness; NREM, non-rapid eye movement; ORL, otorhinolaryngology; OSA, obstructive sleep apnea; REM, rapid eye movement. The Table 1 was created by the authors.
Precision medicine: Diagnosis, treatment, and follow-up of patients with obstructive sleep apnea
Diagnosis and risk stratification
The introduction of precision medicine in OSA is essential to implementing cost-effective diagnostic, treatment, and follow-up tools. The rising prevalence of OSA in recent decades has coincided with the development of new, simplified devices that facilitate the rapid and economical diagnosis of patients with an intermediate to high probability of OSA. The effectiveness of cardiorespiratory polygraphy has been compared to PSG in terms of its diagnostic capability and role in guiding therapeutic decisions. 50 Some researchers have even gone further by incorporating simpler devices, such as nocturnal pulse oximetry, for high-risk patients preselected using a screening questionnaire like the modified Berlin questionnaire, since a primary care unit. 51 These strategies will help us reach a larger population without the need for evaluation by a specialized sleep unit. This diagnostic approach has been included in some of the recent guidelines. 13 The diagnostic criteria for OSA have evolved and become more complex since their initial definition in 1990. 52 The most recent criteria, established by the American Academy of Sleep Society in 2014, 53 and reviewed in 2023, 54 reflect advancements in scientific understanding. Recognizing the limitations of a reductionist approach based solely on the AHI, which can sometimes fail to align with the severity of a patient’s symptoms, the Society now advocates for a more holistic perspective. This approach includes considering mental health, metabolic issues, and CV comorbidities as essential components of OSA. In line with this concept, some authors have proposed models or frameworks that integrate diagnostic and therapeutic approaches, considering a multidimensional perspective of the disease, its pathophysiology, clinical impact, and response to treatment. Their goal is to develop precision medicine strategies to address the needs of complex patients who do not fit the general recommendations outlined in clinical management guidelines.
In 2018, a group of experts within the European Respiratory Society (ERS) reviewed changes and perspectives on OSA. 11 They proposed a multicomponent grading system to assess the severity of this condition, which takes several factors into account, starting with an AHI of 15 or higher. This system combines symptomatology derived from patients’ medical histories, the Epworth Sleepiness Scale, instances of daytime drowsiness, and results from objective vigilance tests. Additionally, it considers the impact of OSA on the CV system and metabolism along with any accompanying comorbidities. Four groups are thus established based on the severity of symptoms and the impact on end organs. Patients with minor end-organ impact can be categorized into Group A if they exhibit mild symptoms, or Group B if they present with severe symptoms. Conversely, individuals with a major impact on their end organs and mild symptoms are placed in Group C. In contrast, those with severe symptoms are assigned to Group D. This classification helps in understanding the varying degrees of severity and potential complications associated with their condition. This grading system could help guide clinical decisions regarding the management of OSA.
Martínez-García et al. considered various parameters related to the severity of the disease, biological activity, some that analyze the impact on the patient, and pathophysiological traits. 12 The analysis and combination of these parameters aim to unify various aspects of precision medicine, including exposome-genome interactions, endotype-phenotype relationships, biomarkers available in routine clinical practice, and treatable traits that can provide insights into underlying pathophysiology and inform treatment. Their innovative approach relies on simple, economical, and easily accessible measures. For assessing severity, they consider factors such as the AHI, the percentage of sleep time with oxygen saturation below 90% (T90), high blood pressure, BMI, and comorbidities. To evaluate the impact on patients, they use validated quality of life scales, measures of EDS, symptoms of depression, and attention or vigilance scales. At the biological level, they suggest using biomarkers that provide insights into the endotype associated with intermittent hypoxia, sleep fragmentation and related consequences or comorbidities, as well as sympathetic activation, such as ambulatory blood pressure monitoring. Additionally, biomarkers linked to CV stress, like hypoxic burden, metabolic control (e.g., glycosylated hemoglobin), and inflammation (e.g., C-Reactive Protein), could also be included. Regarding treatable pathophysiological features, they identify four main domains: three related to non-anatomical dysfunction (high loop gain, low arousal threshold, and upper airway muscle dysfunction) and one related to anatomical muscle dysfunction, as previously mentioned.
Sánchez-de-la-Torre et al. propose an approach divided into two main processes. 10 The first process involves risk stratification, which groups clinical features and comorbidities to determine whether a diagnostic test is necessary and, if so, to assess its complexity. The second process focuses on individualized or combined interventions based on the patient’s specific phenotype, with a personalized follow-up plan.
More recently, Yu Li et al. proposed classifying OSA patients based on clusters of symptoms, PSG phenotypes, physiological endotypes, and circulating biomarkers to establish subtypes of OSA. 55 This approach is complex because it involves grouping patients according to their symptoms and requires conducting PSG to identify variables such as hypoxic burden, heart rate response to respiratory events, delta wave activity, and the respiratory events load. Regarding phenotypes, they consider factors such as collapsible upper airway, low arousal threshold, increased loop gain, and reduced pharyngeal muscle responsiveness. For biomarkers, the focus is on those that provide significant information about CV risk, such as C-Reactive Protein and adhesion molecules related to inflammation and oxidative stress.
Some authors have proposed a three-dimensional model to evaluate the severity of OSA based on the number of respiratory events and their acute and chronic systemic effects. 11 Due to individual susceptibility and ability to adapt to these changes, this model offers a spectrum of disease that varies from low exposure and high impact to high exposure and low impact. The effect of therapy on this bidirectional relationship varies depending on the presence of comorbidities.
Management: Effective available therapeutic alternatives and response to treatment
Initial treatment is usually directed at the underlying causes of respiratory events. Since obesity is the primary risk factor associated with OSA, weight loss should be considered a first-line intervention to achieve an ideal weight through a healthy, balanced diet and regular physical activity. In those patients with an indication, GLP-1RA, approved for obesity and diabetes mellitus, shows promising therapeutic prospects in OSA.22,23 Additionally, positional therapy with specific devices may be an alternative in patients with positional OSA since it has proven to be an effective therapy with good tolerance and compliance. Some subgroups of patients with anatomical or dynamic alterations in the upper airway may benefit from surgery at that level or hypoglossal nerve stimulation.
MAD has evolved over the years and now represents the second most common therapeutic option, after CPAP therapy, showing satisfactory results. This device is designed to mechanically advance the mandible, enlarging the upper airway and reducing its likelihood of collapse. Various clinical trials and meta-analyses have assessed the effectiveness of MAD compared to CPAP therapy.56–58 Both treatments are effective in reducing excessive daytime sleepiness with similar outcomes. One advantage of MAD is its potential to enhance CPAP tolerance and improve overall quality of life for patients. Current research indicates that while MADs generally have a lesser effect on reducing AHI than CPAP, they tend to have higher patient adherence. 59 However, the effectiveness of MAD is diminished when patients sleep in a supine position or during the REM sleep phase. Consequently, ERS recommends MAD for patients with mild to moderate OSA, minimal daytime symptoms, and no significant CV comorbidities. 60 MADs are also suitable for patients with severe OSA who cannot tolerate CPAP therapy and have unacceptably low adherence to CPAP treatment. 59 However, in terms of CV risk, limited studies have shown no significant differences between the two therapies.57,61
CPAP therapy is the most common and widely used treatment for mild to severe OSA. Early initiation of CPAP and high adherence to its use are linked to optimal treatment outcomes. However, adherence to this therapy varies widely, and optimal utilization and outcomes are achieved only in a minority of patients. Information on the impact of CPAP and MAD on reducing CV risk is scarce. Most studies have focused on the effects of these interventions on OSA symptoms, particularly excessive daytime sleepiness, with positive results. Numerous studies have demonstrated the advantages of consistent CPAP treatment in the primary prevention of CV issues among patients who also experience excessive daytime sleepiness. However, a contentious question remains regarding the necessity of treating asymptomatic OSA and whether such treatment can prevent primary CV or metabolic problems. To date, the homogeneous beneficial effects of CPAP treatment, based on an intention-to-treat analysis, on CV morbidity in asymptomatic patients have not been established. In non-sleepy patients with OSA, effective use of CPAP for more than 4 h per night may be associated with a reduced recurrence risk of major adverse cardiac or cerebrovascular events. 62 However, the effectiveness of this treatment in secondary CV prevention remains unproven63–65 and only adequate adherence to CPAP could confer a protective effect. Given the significant variability in how patients with OSA respond to CPAP treatment and the problem of low adherence rates among many individuals, it is essential to prioritize the ability to predict treatment responses individually. Tailoring treatment approaches to meet each patient’s needs can significantly enhance effectiveness. Furthermore, there is an urgent need to develop innovative strategies and algorithms that integrate the various treatment options available for OSA, ensuring that every patient receives personalized care. Consequently, it is essential to conduct well-designed studies that analyze each treatment, identify the specific phenotype and endotype of patients who would benefit from individual or combined therapies, and evaluate the reliability of incorporating these models into routine clinical practice.
Regarding dual therapies, the “MIMOSA” clinical trial, a parallel, randomized, controlled, superiority clinical trial, evaluated 187 adult participants, overweight men and women, diagnosed with moderate-to-severe OSA. 66 Participants were randomly assigned to one of three treatment groups: standard care, a Mediterranean diet, or a Mediterranean lifestyle intervention (which included dietary changes, advice on physical activity, and sleep hygiene), all alongside CPAP treatment. The results showed that adding a dietary or lifestyle intervention to standard care led to more significant improvements in the severity of OSA and its symptoms compared to standard care alone. These benefits were observed regardless of CPAP use and weight loss. A recent Spanish study examined an interdisciplinary weight loss and lifestyle intervention involving Spanish men with moderate–to-severe OSA, who were also overweight or obese and receiving CPAP therapy. 67 The intervention led to significant and lasting improvements in the severity of OSA, associated health issues, and overall health-related quality of life. Thus, this approach is a key strategy for addressing the substantial impact of this increasingly prevalent (SDB). In two phase 3, double-blind, randomized, controlled trials involving adults with moderate-to-severe OSA and obesity, participants who were not receiving CPAP therapy at baseline were enrolled in trial 1, while those already receiving CPAP therapy were enrolled in trial 2. 22 The participants were assigned in a 1:1 ratio to receive either the maximum tolerated dose of tirzepatide or a placebo for 52 weeks. The results showed that among individuals with moderate-to-severe OSA and obesity, tirzepatide reduced the AHI, body weight, hypoxic burden, C-Reactive Protein concentration, and systolic blood pressure and improved sleep-related patient-reported outcomes. These findings open a window of opportunity to consider these drugs as part of the first-line treatment for this subgroup of patients.
As we mentioned, OSA is a heterogeneous disease, making it challenging to determine which patients are at higher CV risk and which would benefit from CPAP treatment. This complexity has driven the development of personalized medicine, incorporating various biological, sleep-related, and anatomical parameters to better identify patients at CV risk and those who may respond to different therapies. One key question is which patients might benefit from CPAP treatment as a preventive measure against CV disease, particularly since the AHI alone is not a definitive indicator. Current evidence suggests that patients with OSA and refractory hypertension, particularly those with nocturnal hypertension and a non-dipper blood pressure pattern, could benefit the most.68–73 However, the variability in treatment response, particularly in terms of blood pressure control, raises questions about the underlying factors influencing this variability.
A special group of interest is “mild OSA,” a common condition that can lead to potential neurocognitive and CV complications. Research suggests that those patients with mild OSA who experience subjective sleepiness may benefit from treatment. 74 Additionally, in the general population, mild OSA does not appear to be associated with an increased risk of CV mortality. However, evidence regarding the effects of mild OSA on neurocognition, quality of life, mood, elevated blood pressure, and CV outcomes is inconclusive. Therefore, further research is needed in this area to clarify these issues.
Beyond the objective clinical responses observed in clinical trials, recent years have seen increasing interest in the role of biomarkers and biosignals in CV risk stratification and prediction of treatment responses, particularly for primary CV prevention
At the biomolecular level, intermittent hypoxia associated with OSA triggers inflammation and oxidative stress, processes that are linked to the development of CV disease. Inflammatory markers such as C-Reactive Protein, fibrinogen, interleukin-6, and tumor necrosis factor-alpha are commonly elevated in OSA patients, but there is currently no evidence suggesting that these markers decrease with CPAP treatment.75–77 Thus, these markers may serve primarily as prognostic indicators. Conversely, other biomarkers that protect against oxidative stress, such as decreased nitric oxide and increased isoprostanes, may improve following CPAP therapy. These biomarkers could potentially help identify patients who would benefit from treatment to reduce CV risk.78–80 Endothelial dysfunction in OSA is associated with CV diseases, such as atherosclerosis, coronary heart disease, and hypertension, as evidenced by elevated adhesion molecules in the blood. Research indicates that CPAP treatment may lower these adhesion molecule levels, possibly serving as a predictor of which patients will benefit from the CPAP therapy in terms of reducing CV risk. 81 Additionally, OSA is often associated with metabolic dysfunction. Several studies suggest that CPAP treatment may improve these metabolic alterations, leading to better control of lipid profiles and insulin resistance, which in turn reduces the risk of metabolic syndrome and type 2 diabetes. High blood pressure is one of the most extensively studied bio parameters linking OSA to CV disease. Evidence indicates that CPAP improves blood pressure control. However, it is important to note that there is no evidence that CPAP improves the CV profile in patients with already established CV disease. In this context, the blood pressure response to CPAP treatment varies significantly in patients with OSA and resistant arterial hypertension. One recent study aimed to identified patient with resistant hypertension whose condition would improve with CPAP treatment. 82 The study evaluated the expression of CV system–related circulating miRNAs in plasma samples from patients with resistant arterial hypertension and OSA, both at baseline and after 3 months of consistent CPAP therapy, using an 84-miRNA array. Three specific miRNAs were identified that could predict favorable blood pressure responses to CPAP treatment. Therefore, a unique cluster of three plasma miRNAs measured before CPAP treatment can forecast how patients with resistant arterial hypertension and OSA will respond to CPAP in terms of blood pressure control. Furthermore, CPAP therapy is associated with changes in CV-related miRNAs, which may influence the risk of CV disease in patients suffering from OSA and resistant arterial hypertension.
Advances at the biomolecular level have been accompanied by technological advancements that have driven the creation of new devices capable of detecting beneficial biosignals. PSG remains the gold standard for diagnosing OSA. It includes recording nocturnal pulse oximetry to quantify hypoxia, along with other measurements such as AHI, ODI, and sleep architecture. However, some studies have questioned the utility of AHI as a marker of OSA severity and CV risk.11,83 The need to develop more precise markers to establish the clinical relevance of OSA and its association with CV risk is evident. Pulse wave analysis has been used to assess vascular properties. In a multicenter study, researchers incorporated pulse wave analysis into an algorithm that quantifies autonomic, vascular, and cardiac activity variables during sleep to stratify CV risk in patients with OSA. 84 While AHI, ODI, and cardiac risk index (based on pulse wave signals derived from pulse-oximetry,) were associated with increased CV risk, only cardiac risk index demonstrated an independent CV risk prediction effect. Other studies have also evaluated the response to CPAP treatment in high CV risk patients using the cardiac risk index and pulse wave measurement through photo-plethysmography before and after CPAP treatment. 85 These studies identified four clusters of patients with high CV risk who responded to CPAP therapy. Additionally, the results demonstrated that CPAP reduced cardiac index and that pulse wave monitoring using photo-plethysmography could help in identifying patients who would benefit from CPAP. Thus, nocturnal pulse wave analysis is considered a powerful tool for managing CV risk and assessing treatment benefits, adding a CV perspective to conventional OSA parameters. Abnormal cardiac autonomic modulation can provide valuable insights into understanding the diverse phenotypes of OSA. In this context, heart rate variability serves as a reliable neurocardiac tool to assess altered autonomic function and provides crucial CV information related to OSA. Heart rate variability estimates the variation and complexity of cardiac rhythm by measuring beat-to-beat intervals from an electrocardiogram. 86 Numerous studies indicate that HRV is an effective marker for screening and diagnosing OSA, and It correlates with disease severity and reflects the causal pathways involved in CV disease. 87 Short-term CPAP treatment partially improves cardiac autonomic activity in patients with moderate to severe OSA. Long-term CPAP treatment continues to positively affect both sympathetic and parasympathetic modulation, helping to reverse autonomic imbalance. This is indicated by an increase in high-frequency components, a decrease in low-frequency components, and a lower Low-Frequency/High-Frequency ratio. 88 Hypoxic burden is another measure proposed in recent years to overcome the limitations of the AHI in measuring CV risk. Measurement of T90 is a stronger predictor of CV disease and all-cause mortality than AHI. 89 However, it cannot capture intermittent hypoxia. It may be affected by other causes, such as Chronic obstructive pulmonary disease or nocturnal hypoventilation, unrelated to upper airway obstruction related to OSA. The HB was first defined by Azarbazin et al. in 2019. 90 This parameter can encapsulate the frequency, duration, and depth at which each respiratory event contributes to total hypoxemia, specifically by measuring the area under the curve of each desaturation following apnea or hypopnea events. A recent systematic review shows that HB has stronger relationship with major CV events and mortality than other respiratory parameters such as AHI or ODI. 91 Hypoxic burden also helps identify patients prone to arterial hypertension and can be used to monitor the treatment response in patients with moderate-to-severe OSA. In the post-hoc analysis of ISAAC study, 92 the authors analyzed its role as a predictor of treatment response and, observed that in patients with acute CV disease and a higher HB group, CPAP treatment significantly reduced the incidence of CV events. In contrast, CPAP-treated patients with a low hypoxic burden group, exhibited a trend toward a higher risk of CV outcomes than those receiving usual care (HR 1.33, 95% CI 0.79–2.25). The differential effect of the treatment depending on baseline hypoxic burden levels followed a dose-response relationship. Thus, HB is proposed as a predictive measure for morbidity and mortality associated with OSA. Regarding the respiratory arousal index, some authors have found, through multivariate analysis, that this index is the most critical risk factor for predicting non-dipper patterns, surpassing the clinical or classic PSG ones. 93
All this evidence serves as a foundation for future research that aims to provide valuable insights for medical practitioners.
Other considerations: Treatable treats and comorbidities in OSA
As we mentioned, the involvement of pathophysiological factors is also determined and influenced by comorbidities. It is known that OSA is often associated with various comorbidities, including metabolic, CV, renal, pulmonary, and neuropsychiatric disorders. 94 Growing evidence supports a bidirectional relationship between OSA and these comorbidities, particularly heart failure, metabolic syndrome, and stroke. The mechanisms underlying these bidirectional relationships differ across various comorbid conditions. An observational analysis using an extensive nationwide health claims database in the U.S. aims to clarify the impact of sex and age on the comorbidity burden in OSA patients. 95 The findings reveal that Type 2 diabetes and ischemic heart disease are more prevalent in men, while hypertension and depression are more common in women with OSA. In contrast, the differences between sexes regarding the prevalence of congestive heart failure, arrhythmias, and stroke are less pronounced. Additionally, the analysis shows that the prevalence of comorbidities increases with age, although the effect of age varies depending on the specific comorbidity. Notably, the divergence in comorbidity prevalence between OSA patients and controls becomes more pronounced after the sixth decade of life for most CV diseases, while depression follows an opposite trend. In a fully adjusted model, the odds of all comorbidities are significantly higher in patients with OSA. Martínez-García et al. 12 propose grouping these treatable traits into five spheres that can guide clinical management: upper airway conditions, pulmonary conditions, extrapulmonary comorbidities, sleep and lifestyle spheres. Daniil Lisik et al. 96 frame treatable traits into four categories: respiratory and sleep disturbances, daytime dysfunction, comorbidities, and adaptation and safety. Precision medicine should consider these factors to establish personalized management strategies.
Role of machine learning and artificial intelligence in the management of OSA
The role of machine learning (ML) techniques has been explored in various aspects of medicine, including diagnosis, risk stratification, treatment response, and personalized management. 97 Diagnosing OSA is time-consuming and resource-intensive. Additionally, the gold standard test—PSG—can yield inaccurate results because it is conducted in a hospital setting rather than the patient’s home. A meta-analysis of ML in this context shows that integrating ML into the diagnostic process is both feasible and effective, yielding good performance through various inputs, such as electrocardiograms, SpO2 levels, respiratory signals, electroencephalograms, sound, pressure sensors, and additional clinical information, including questionnaires or anthropometric data. 98 This approach holds promise in predicting CV mortality, assessing the severity and pathophysiology of respiratory events, and determining responses to surgical treatment or CPAP titration.
Regarding the pediatric population, ML algorithms have been employed to identify children at risk for OSA, to stratify OSA severity, and to predict persistent OSA despite initial surgical interventions.99–101 Given that children are a particularly vulnerable population with a high rate of underdiagnosis, ML could help address the limitations of traditional diagnostic methods. However, further prospective studies are necessary to establish the accuracy and generalizability of these algorithms. While the results so far are promising, some challenges must be addressed to successfully integrate these tools into OSA’s real-world diagnosis and management.
Expert opinion
It is essential to review and update the diagnostic criteria for OSA across its entire spectrum. Additionally, understanding and determining the most appropriate therapeutic strategies tailored to each patient profile is crucial. Given that OSA is a global health issue that affects all population groups and is frequently underdiagnosed, it is vital to identify the most effective interventions for each individual. The era of precision medicine has arrived in the field of OSA, opening a window of opportunity to incorporate aspects of personalized medicine into the clinical management of patients with this disease.
Understanding the interrelationship between individual vulnerability to developing the disease and its associated CV complications, as well as the adaptive capacity of each individual, allows for clinical management decisions focused on each patient. It is partly influenced by environmental exposure and other medical factors. This perspective has the potential to revolutionize routine clinical practice in the near future, allowing us to identify vulnerable, at-risk patients who will benefit from specific symptomatic or preventive treatment. In turn, avoid high resource use or even harm, prescribing therapies or clinical follow-up to patients who will not respond to a specific treatment or benefit from long-term CV disease prevention. For this proposal, it would be advisable to update the diagnostic approach to OSA. As previously mentioned, OSA is a heterogeneous, dynamic condition with clinical expressions that vary from one patient to another, resulting in diverse clinical and functional consequences. For this reason, we believe that cluster analysis for identifying phenotypes can help overcome the limitations of traditional symptom-based or “classic” anthropometric measures, aiming to capture the largest number of patients possible, especially considering the underdiagnosis and implications of the disease. Given the high healthcare burden and patient volume, clinical practice recommendations suggest that it is a priority to assign simplified sleep studies to patients with a high probability of OSA to avoid delays in diagnosis and treatment. Then, more complex methods such as cardiorespiratory polygraphy or PSG should be reserved for patients in a “gray” zone for refining the diagnosis. We consider the same approach for more complex cases where other comorbidities may condition the interpretation of the results in a simplified study. Furthermore, we consider that for patients in the “green” zone, with a low probability of OSA, economical and accessible diagnostic tools should be developed that could allow the rescue of those patients who, due to standard clinical practice, would otherwise be overlooked (Figure 1). In this sense, it has been suggested that the AHI cannot fully capture the spectrum of OSA and should not be considered the only variable for establishing the diagnosis of OSA. Therefore, we propose to incorporate multidimensional scales that consider other measures of nocturnal oxygenation, such as T90 or hypoxic burden, along with CV risk variables such as BMI and refractory high blood pressure or non-dipper pattern and CV comorbidities. If we can incorporate these variables into routine clinical practice questionnaires that assess sleep quality, excessive daytime sleepiness, and mood, while also collecting aspects related to the functional deterioration the disease causes for the patient and society, we could better identify patients for whom OSA has significant implications and requires treatment. The development and validation of these scales would be crucial for stratifying patients based on their risk of OSA and for guiding the diagnostic approach.

Approach to the diagnostic and therapeutic management of Obstructive Sleep Apnea based on precision medicine.
Generally, all patients should receive affordable lifestyle advice, including sleep hygiene and proper management of comorbidities and treatable traits. Regarding specific treatments for OSA, deciding which patient to treat is simple when they are symptomatic or have refractory high blood pressure or a non-dipper pattern. CPAP is the first-line treatment for most patients, as it has strong scientific support. In cases of craniofacial malformations, surgery or a MAD can be an effective alternative, especially in controlling symptoms. Positional therapy may also be considered for those whose OSA has a significant positional component. However, for patients who are intolerant to or poorly adapt to CPAP treatment or those who show an unexpected response to the prescribed treatment (e.g., CPAP, weight loss, surgery), the next step may not be immediately clear. Therefore, we recommend sequential therapy. It is crucial to reconsider the diagnosis and the necessity for treatment. Repeating a sleep study in conditions as close as possible to the patient’s usual sleeping environment, sometimes over several nights, can help clarify overdiagnosis or reassess severity. A holistic, multidisciplinary approach can identify the predisposing endotype at that moment and suggest trials with alternative therapies, such as weight loss drugs, diuretics or hypnotics. Furthermore, for highly symptomatic patients, such as those with residual excessive daytime sleepiness despite correct CPAP treatment, wakefulness-stimulating drugs like pitolisant or solriamfetol may serve as co-treatment.102,103
It is anticipated that in the near future, the results of biosignals from sleep studies, such as pulse wave or heart rate variability, along with biomarkers related to sympathetic activity, lipid profile or inflammation, and the genetic and molecular profile, could be incorporated into routine clinical practice to advance precision medicine. However, integrating these parameters into clinical practice remains challenging due to the current lack of well-designed clinical trials that provide robust scientific evidence, as well as their limited accessibility in sleep units. Nevertheless, incorporating these measures could significantly enhance the ability to assess treatment benefits, including both symptomatic and CV responses, while minimizing unnecessary exposure to potentially harmful therapies.
Conclusions and future perspective
There is a critical need for innovative approaches to syndrome definition that account for the distinct clinical phenotypes and the widespread prevalence of SBD. We must also consider specific comorbidities closely linked to OSA when assessing severity, particularly cardio-metabolic risks, worsening morbidity and mortality. By adopting new diagnostic strategies that leverage technological advances, we can enhance our understanding of sleep staging and respiratory evaluation beyond just the AHI, while also investigating the role of biomarkers and biometrics in classifying this disease. Moreover, precision medicine must be based on a careful initial selection, considering the clinical phenotype, primary endotype and treatment monitoring. This should go beyond symptoms and AHI, integrating more specific biosignals or biomarkers to guide adequate sequential treatment. The integration of ML in OSA is anticipated to progress quickly with significant potential. Advances in sensor technology, signal processing, and ML models will assist clinicians in diagnosing conditions, predicting therapeutic responses, assessing CV risk, optimizing treatment management, and classifying different types of OSA. However, these promising strategies require rigorous validation to ensure their effectiveness.
Footnotes
Acknowledgements
During the preparation of this paper the authors used Grammarly as assistive AI tools to improve manuscript comprehension. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
