Abstract
Cases of pulmonary squamous cell carcinoma (SCC) metastases to the nasal region are highly uncommon and frequently manifest with poor prognosis. However, the use of anti-programmed cell death protein 1 (PD-1) inhibitors in pulmonary SCC with nasal metastasis has not been documented. In this study, a case of pulmonary SCC metastasizing to the nose was discussed. Nasal nodules appeared in the patient; initially, these were considered benign lesions. It was determined following a pathological biopsy that the patient had nasal metastases of lung SCC. The lung tumor had decreased after two cycles of standard chemotherapy (paclitaxel + carboplatin); however, the nasal tumor continued to grow. Consider that nasal metastases are not responsive to chemotherapy. A multidisciplinary consultation believed that immunotherapy had potential benefits for metastatic tumors and decided to use a chemotherapy + immunotherapy regimen. The nasal metastatic tumor gradually returned to normal as a result of the combination of standard chemotherapy and immunotherapy (Tislelizumab) from the third cycle onward. After six cycles of combined treatment, the patient began maintenance monotherapy. During treatment, the patient’s condition remained stable without progression or distant metastasis. This case highlights the potential of combining anti-PD-1 therapy with standard chemotherapy as an effective salvage strategy for chemotherapy-refractory nasal metastases of pulmonary squamous cell carcinoma, thereby contributing to improved patient survival.
Introduction
Globally, lung cancer is the most prevalent malignancy in terms of both incidence and mortality.1,2 The liver, bones, and brain are the most common sites of metastasis from lung cancer, and metastasis to the tip of the nose is very rare. 3 When patients first present with nasal symptoms, the absence of systemic symptoms often leads to misdiagnosis of nasal masses as other more common illnesses, thus delaying cancer treatment.4,5 Further, skin metastases from internal cancers generally have a poor prognosis, and the median survival of patients with nasal metastases from lung cancer is only 3–6 months.6,7
Previously, the therapeutic options for pulmonary squamous cell carcinoma (SCC) with nasal metastasis have been restricted. Recently, the emergence of anti-programmed cell death protein 1 (PD-1) inhibitors has led to a substantial improvement in treatment, significantly increasing the survival rates of lung cancer patients. 8 Blocking the interaction between programmed death-ligand 1 (PD-L1) and its receptor programmed death 1 (PD-1) restores the antitumor activity of T cells, thereby increasing the immune response of the body against the tumor cells. 9 The efficacy of PD-1 inhibitors is not only influenced by PD-L1 expression but also by a range of tumor-intrinsic molecular/genetic factors and the tumor immune microenvironment (e.g., tumor mutational burden (TMB) and IFN-γ signaling status). 10 However, anti-PD-1 inhibitors have not been reported to be used in the treatment of pulmonary SCC with nasal metastasis. Therefore, we report the first case of pulmonary SCC with nasal tip metastasis that was successfully treated with Tislelizumab, an anti-PD-1 inhibitor, along with standard chemotherapy. At the same time, related research and potential mechanisms are reviewed to provide a reference for the treatment of lung SCC with rare metastatic sites. This case report was prepared in accordance with the CARE reporting checklist (Supplemental Material). 11
Case presentation
It was a case of a 59-year-old male patient who had a 20-year history of smoking. He experienced a nodule formation in April 2023, which was slightly raised above the nasal cavity surface and located approximately 0.5 cm from the right nostril. After scratching caused ulceration and bleeding, the surface of the right nasal tip became red and swollen, with the affected area gradually enlarging (Figure 1(a)). A systemic antibiotic and local remedies were administered at an external hospital, but no improvement was observed (Figure 4). During May 2023, the patient experienced a primarily dry cough, sometimes accompanied by sputum and occasionally blood-streaked. He visited our respiratory department on July 10, 2023, with a primary complaint of a 2-month history of a persistent cough. His physical examination revealed a 1.7 × 1.5 cm round erythematous nodule with central elevation on the nasal tip (Figure 1(b) and (c)). An irregular mass located in the middle and lower lobes of the right lung, adjacent to the right hilum, was identified on chest-enhanced CT. The mass, which measured approximately 11 × 8.9 × 9.4 cm, was unclearly defined and associated with obstructive atelectasis and pneumonia. This was considered to be a malignant neoplastic lesion, likely central lung cancer with mediastinal, right hilar, and hepato-gastric space lymph node metastases (Figure 2(a) and (b)). A nasal tissue histopathological biopsy reveals SCC. The carcinoma is contained inside the dermis and subcutaneous tissue; it has not spread to the surface squamous epithelium or skin appendages. There is no dysplasia observed in the skin’s surface squamous epithelium or appendages. This is consistent with the characteristics of metastatic cancers (Figure 3(a) and (b)). Bronchoscopy showed obstruction of the right intermediate bronchus with mucosal infiltration changes. SCC was detected through an endoscopic biopsy at the opening of the right intermediate bronchus. The pathological examination of SCC reveals that the tumor cells have significant pleomorphic, pathological mitotic patterns, intercellular bridges, and single-cell keratinization are visible (Figure 3(c)). Whole-body bone imaging revealed abnormally increased radioactivity in the left acromion. Antinuclear antibody (ANA): 1:1000 cytoplasmic granular type and anti-RO-52(+). Genetic testing for pulmonary SCC showed no mutations, with PD-L1 expression in the solid tumor at 15% (Figure 3(d)). The characteristics of the nasal lesion were consistent with metastatic presentation. Combined with the histopathological and imaging results of the lung and nasal lesions, the diagnosis was pulmonary SCC, stage T4N2M1b IVA, with metastases to the left acromion and nasal region (Figure 4).

Diagram illustrating the evolution of the patient’s nasal condition. (a) 1.0 × 0.9 cm smooth, unevenly bordered red nodule on the nose surface. (b) Round, erythematous, and enlarging in the center, the nodule grows to a size of around 1.7 × 1.5 cm. (c) Following a local pathological biopsy, the patient’s condition shows a minor increase in edema in comparison to the previous state. (d) Following the initial chemotherapy treatment, the tumor experienced a substantial increase in size, measuring 2.5 × 3.0 cm, and projecting visibly above the skin surface due to significant dilatation of capillaries. (e–h) Following the second round of chemotherapy, the tumor measured 3.0 by 3.5 cm and was almost straight throughout the nose. It has a rough surface with blisters, mild scaling, and apparent dilatation of the capillaries. (i–l) Following the initial immunotherapy, the nasal mass deepened to a deep red but remained smaller than previously, with no noticeable elevations or depressions. Capillary dilatation was observed at the interface with the normal skin. (m–p) Following the second immunotherapy treatment, the mass size decreased, and the surface nearly reverted to the level of normal skin. The center showed a deep red color, accompanied by capillary dilation at the periphery, while the middle displayed normal skin. (q–t) On January 10, 2024, the nasal mass was no longer present, suggesting that the nose had entirely regained its previous shape.

Imaging results during patient treatment. Initial diagnosis: Enhanced computed tomography of the lung window (a) and mediastinal window (b) revealed an irregular tumor extending from the right hilar region to the right middle and lower lobes. Following two rounds of chemotherapy, a chest computed tomography revealed a reduction in the size of the lesions observed in the lung window (c) and mediastinal window (d). Chest computed tomography revealed that the lesions in the lung window (e) and mediastinal window (f) had further reduced in comparison to before after three cycles of immunotherapy and five rounds of chemotherapy.

Patient’s histopathological examination. The tissue samples were stained using the Hematoxylin and Eosin (HE) staining technique. The samples were observed with a magnification of 10 × 10 (a) and 2 × 10. (b): The carcinoma does not spread to the surface squamous epithelium or skin appendages; instead, it is found in the dermis and subcutaneous tissue. The surface squamous epithelium and appendages of the skin are free of atypia. (c)Tumor cells show considerable atypia at 40 × 10 magnification, with abnormal mitotic structures visible as well as intercellular bridges and single-cell keratinization. (d) The tumor percentage score for programmed death-ligand 1 (PD-L1) is 15%.
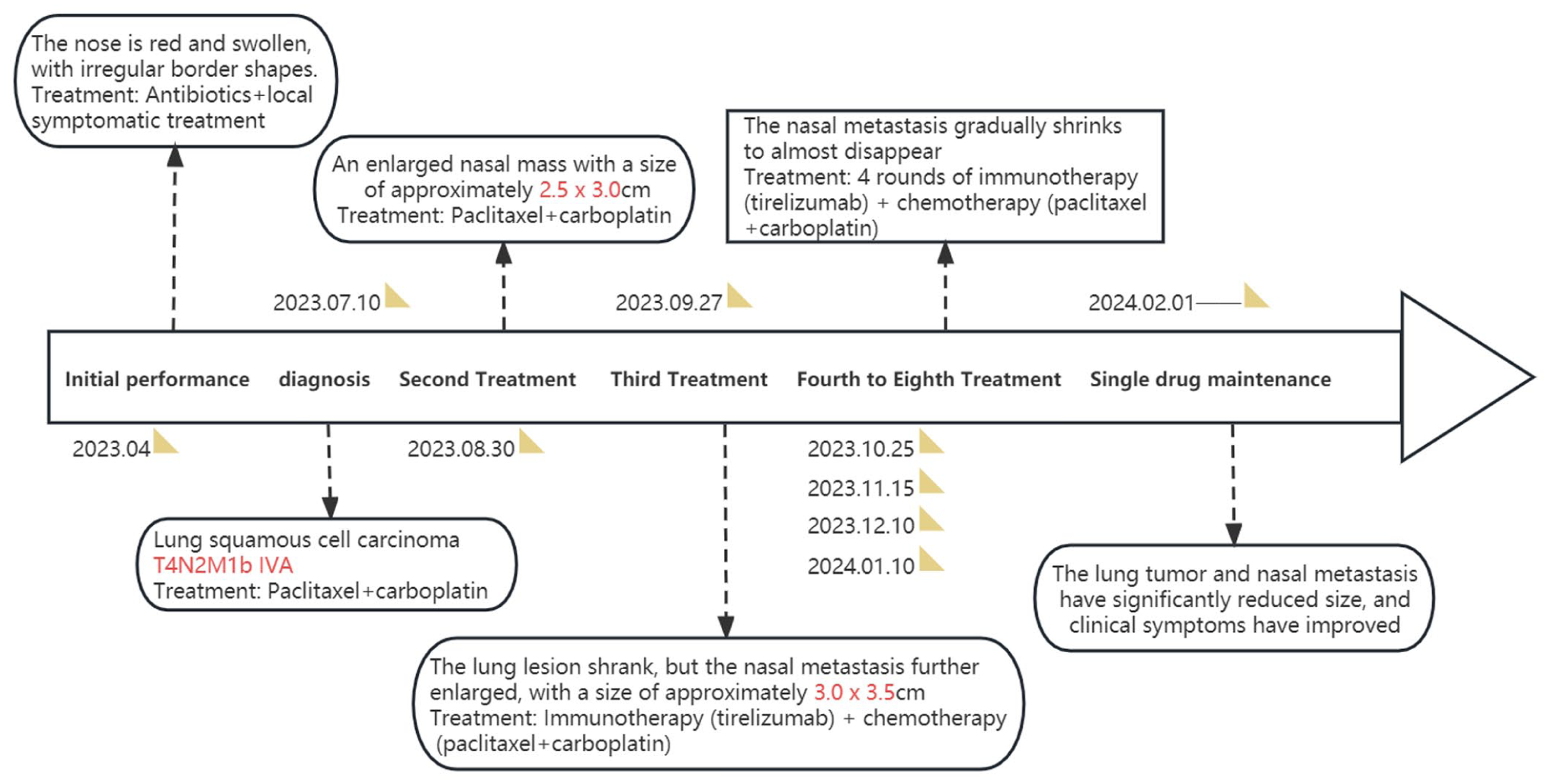
Treatment timeline.
The patient did not receive tumor immunotherapy as a result of aberrant immunological markers from the screening. The first systemic chemotherapy (paclitaxel 210 mg + carboplatin 500 mg) was administered on August 9, 2023. A second chemotherapy session was scheduled for the patient 3 weeks later, and they visited on August 30, 2023. Physical examination showed an enlarged nasal mass with substantial capillary extension on the surface of the nasal metastasis, approximately 2.5 × 3.0 cm (Figure 1(d)). Paclitaxel 210 mg + carboplatin 500 mg were administered as the second systemic chemotherapy on August 31, 2023 (Figure 4). The patient returned to the respiratory department 3 weeks later. Physical examination at this time revealed that the nasal metastasis had continued to expand, causing an uneven surface and extensive capillary expansion. It measured approximately 3.0 × 3.5 cm and was disseminated throughout the nasal area (Figure 1(e)–(h)). The repeat chest CT revealed an irregular mass in the middle and lower lobes of the right lung, adjacent to the right hilum, with unclear boundaries. The mass was slightly smaller than the lesion identified on July 5, 2023 (Figure 2(c) and (d)). Repeat antinuclear antibody (ANA) test showed 1:1000 cytoplasmic granular type and anti-RO-52 (+).
On the basis of a comprehensive multidisciplinary team (MDT) evaluation, combined with the potential benefits of immunotherapy in metastatic disease, the resources of our hospital, and the patient’s choice, we ultimately decided to use chemotherapy combined with immunotherapy (tislelizumab) as the treatment strategy for this case. The first immunotherapy (Tislelizumab 200 mg) was administered on September 27, 2023(Figure 4), in combination with the third systemic chemotherapy (paclitaxel 210 mg + carboplatin 500 mg). The patient was re-examined 3 weeks later, on October 24, 2023. The physical examination revealed that the nasal metastasis was dark red with fewer capillaries on the surface, without any significant protrusions or depressions, and the mass had substantially reduced in size in contrast to the previous visit (Figure 1(i)–(l)). The fourth systemic chemotherapy (paclitaxel 210 mg + carboplatin 500 mg) was administered in combination with the second immunotherapy (Tislelizumab 200 mg) on October 25, 2023 (Figure 4). The patient’s nose returned to its normal size 3 weeks later, and the mass was dark red in the center, with some capillary expansion at the margins and normal skin visible in between (Figure 1(m)–(p)). Repeat chest CT indicated that the lesion had reduced in size compared to previous scans (Figure 2(e) and (f)). A combination of the fifth systemic chemotherapy (Paclitaxel 210 mg + Carboplatin 500 mg) and the third immunotherapy (Tislelizumab 200 mg) was administered on November 15, 2023 (Figure 4). Three weeks later, on December 10, 2023, the sixth systemic chemotherapy was administered in combination with the fourth immunotherapy (Figure 4). The nasal skin had largely returned to its normal state upon examination on January 10, 2024 (Figure 1(q)–(t)). On January 10, 2024, the fifth immunotherapy was administered in combination with the seventh systemic chemotherapy (Figure 4). The patient resumed routine monotherapy maintenance treatment (paclitaxel + tislelizumab) after completing seven cycles of systemic chemotherapy and five cycles of Tislelizumab immunotherapy (Figure 4). No immune-related adverse events (irAEs), such as immune-related pneumonitis, colitis, or hepatitis, were observed during the immunotherapy period. The lung tumor and nasal metastases showed significant regression, clinical symptoms were markedly improved, progression-free survival (PFS) has exceeded 12 months, and the patient remains under regular follow-up. The patient’s diagnosis and treatment timeline is illustrated in Figure 4.
Discussion
Lung cancer typically metastasizes to the liver, bones, and central nervous system, with skin metastasis being highly uncommon. 12 Lung cancer skin metastases are most frequently observed in the chest, abdomen, scalp, and face. However, nasal metastases are highly rare. 3 To the best of our knowledge, there have been only 15 reported cases of primary lung cancer metastasizing to the nasal region. The demographic data, prognosis, treatment, and disease details of these patients have been documented (Table 1).4,5,13 –20 Among them, the majority of lung cancer metastasis to the nose cases were male patients. Histopathological examination revealed 10 cases of SCC, 1 case of adenosquamous carcinoma, 1 case of oat cell carcinoma, 1 case of anaplastic large cell carcinoma, 1 case of large cell undifferentiated carcinoma, and 1 case of unspecified lung cancer. Further, in most of the cases, the patients were found to have advanced-stage lung cancer and unfortunately passed away within 1–9 months. In the absence of systemic signs and symptoms, cases that present with nasal symptoms as their first manifestation frequently results in the nasal mass being misdiagnosed as other more prevalent conditions, such as non-melanoma skin cancer, nasal tumors, inflammatory tumors, or infectious diseases, particularly when there is no evidence of a history of internal malignancies.4,5 Initially, the nasal lesion (Case 15) manifested as a small, localized redness and swelling that was slightly elevated above the epidermis and had a smooth surface nodule. It progressively developed into a mass that covered the majority of the nasal skin, with an irregular surface and capillary growth, similar to a tumor. However, the pathological morphological diagnosis of SCC metastasizing to the skin from the lungs is difficult due to the rarity of such metastasis in comparison to primary skin SCC and the absence of organ-specific immunohistochemical markers in SCC. Regardless of the primary site, SCC expresses the same squamous cell differentiation antigens, such as p63 or CK5/6. 13 To date, advancements in morphology, immunohistochemical markers, and molecular genetics have not fully characterized SCC. 21 Typically, a combination of clinical case data, clinical histopathological characteristics, tissue morphology examination, and molecular genetics is necessary to determine whether it is a primary tumor or SCC metastasis to the skin.
Displays confirmed cases of cutaneous metastases of the nose originating from lung cancer.
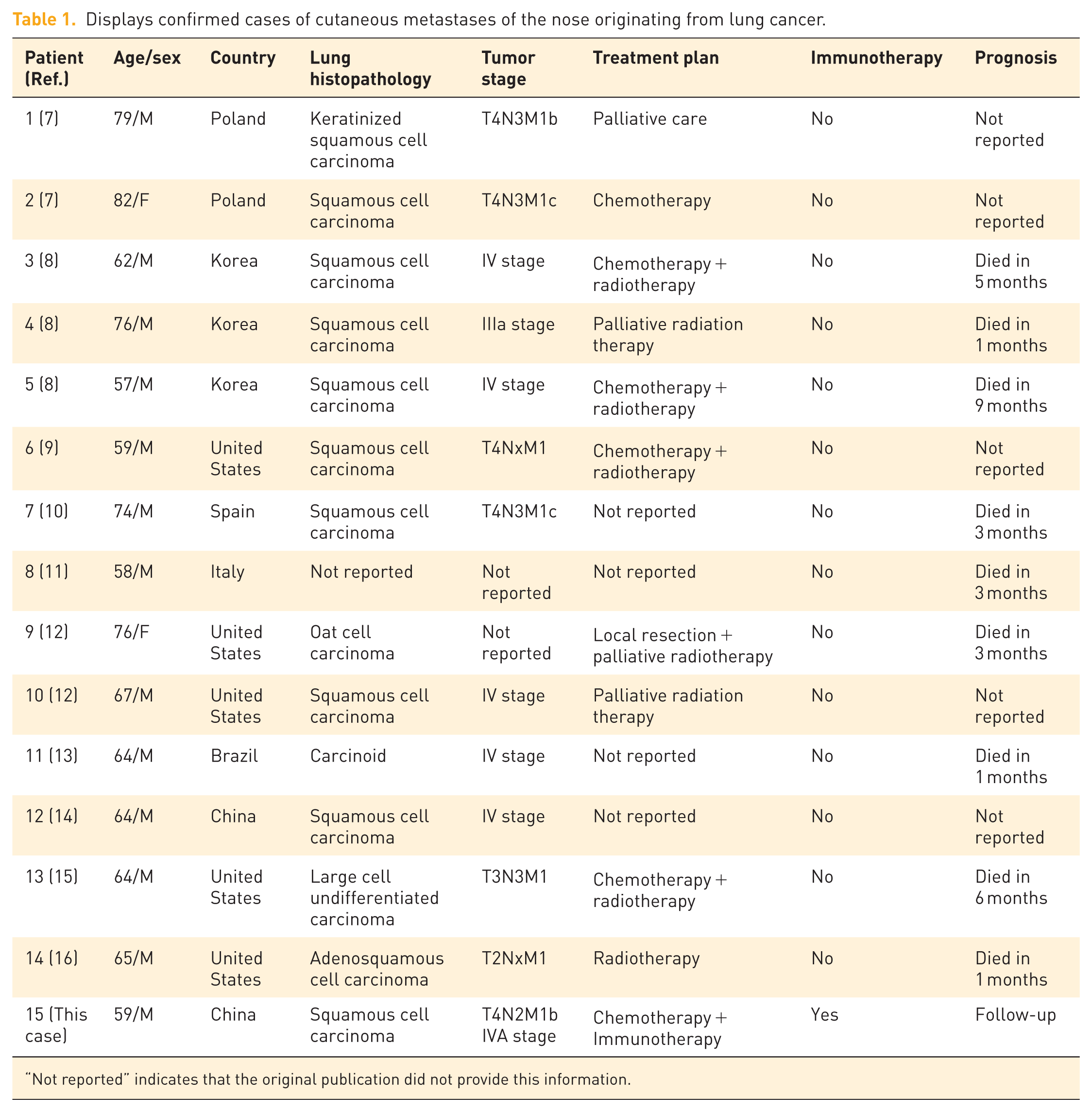
“Not reported” indicates that the original publication did not provide this information.
Brownstein et al. described 117 cases of lung cancer with skin metastases, in which the most common histopathological type was adenocarcinoma, followed by SCC, and undifferentiated carcinoma. 22 However, poorly differentiated squamous cell carcinoma was the predominant type of nasal metastasis in the 15 cases that we examined. This may be associated with the nasal anatomy and tumor metastasis mechanisms, which have not yet been completely elucidated. Existing studies classify this distant metastasis into three pathways: arterial embolism, retrograde spread via the vertebral venous plexus, and lymphatic dissemination. 23 Lung cancer is more likely to disseminate through arterial embolism. Emboli are formed by tumor cells in the lungs, which enter the pulmonary veins, migrate through the left atrium into the systemic circulation, and enter into the nasal supplying vessels. Another potential mechanism for nasal metastasis is that when intrathoracic pressure is substantially raised, emboli containing tumor cells migrate upward via the venous plexus to the cranial venous sinuses. The pterygoid plexus and cavernous sinus can interact with the vertebral system and have the potential to move tumor cells to the nasal cavity. 24 Further, the connection between regional lymphatics and the thoracic duct provides another route to pulmonary vascular circulation. 23 To date, no systematic molecular studies have been conducted on the mechanisms of nasal metastasis in lung squamous cell carcinoma (LUSC). However, recent advances in understanding the molecular basis of distant metastasis in LUSC may offer valuable insights for investigating this rare metastatic pattern. Multiple signaling pathways and regulatory molecules have been identified as being associated with LUSC dissemination. For instance, the FGF19–GLI2 axis promotes tumor progression and distant metastasis by activating components of the Hedgehog pathway, while the CXCR4/SDF-1 signaling pathway plays a critical role in stem cell–mediated migration and pulmonary colonization. 25 In lymphatic spread, hub genes such as CDK1, PLK1, and CLDN3 are linked to cell cycle regulation, chromosomal stability, and epithelial barrier integrity, respectively. 26 In particular, the loss of CLDN3 disrupts tight junctions and enhances tumor invasiveness. 27 Furthermore, there is growing evidence that circulating tumor cells (CTCs) may acquire metastatic traits at early stages of tumor progression. Experimental models have identified β-actin mRNA–positive CTCs as markers of early hematogenous dissemination and distant organ colonization. 28 Future studies are needed to explore whether similar molecular mechanisms may underlie this rare pattern of nasal metastasis.
Lung cancer with nasal metastasis as the first symptom is usually accompanied by metastasis to other parts of the body, with a poor prognosis and a median survival of 3–6 months. 6 The majority of the patients who were diagnosed with lung cancer with nasal metastasis passed away within 3–9 months. Previously, the treatment for advanced lung cancer primarily involved chemotherapy, radiotherapy, and targeted therapy, all of which had limitations and poor therapeutic outcomes. Recently, the treatment model and prognosis for non-small cell lung cancer (NSCLC) patients have been substantially altered by the extensive use of immune checkpoint inhibitors (ICIs) that target the PD-1/PD-L1 axis. The PD-1/PD-L1 immune checkpoint plays a crucial role in the mechanism of immune evasion by malignant tumors. PD-1 on the surface of T cells binds with PD-L1 on tumor cells, inhibiting T-cell-associated kinases. This inhibits T cells from responding to malignancies, making it impossible for them to identify and eliminate tumor cells, which eventually results in immune evasion.29,30 Furthermore, the binding of PD-1 with its ligands PD-L1 and PD-L2 can impede the proliferation of T lymphocytes and the production of cytokines such as IL-2 and IFN-γ, block the proliferation and differentiation of B lymphocytes, and inhibit the secretion of immunoglobulins, thus compromising the immune response of effector cells. 30 These ICIs can inhibit the binding of PD-1 on T cells with PD-L1 ligands on tumor cells, reactivating T cells to kill tumor cells. To date, there have been many clinical studies that have demonstrated that the combination of ICIs with chemotherapy significantly extends overall survival (OS) and PFS in patients with PD-L1 expression levels ⩾1% compared to traditional chemotherapy alone. This combination also provides durable responses and good risk-benefit profiles.31 –33 Clinically, ICIs combined with chemotherapy have been established as first-line treatment for advanced NSCLC.
To our knowledge, this case is the first to use Tislelizumab combined with standard chemotherapy for pulmonary SCC with nasal metastasis. In this case, the patient’s lung lesions were reduced after two cycles of chemotherapy, but the nasal metastasis continued to progress. The nasal metastasis progressively decreased until it almost disappeared after the implementation of immunotherapy in the third cycle. The efficacy of anti-PD-1 inhibitors for metastatic tumors in rare sites is further confirmed by the high efficacy of immunotherapy in treating the nasal metastasis of pulmonary SCC. Several mechanisms have been proposed to explain this effectiveness. (1) Enhancement of Tumor Antigen-Specific Immune Responses: Anti-PD-1 inhibitors block the action of immune checkpoints, such as PD-1 and CTLA-4, thus lifting the inhibition of T-cell antitumor functions. This T-cell activation enables the cells to effectively recognize and attack tumor cells. Given that metastatic lesions often share surface antigens with the primary tumor, the enhanced systemic immune response can target multiple lesions simultaneously. 34 (2) Modulation of the Tumor Microenvironment: These inhibitors can modulate the tumor microenvironment to be more conducive to immune cell infiltration and activation. For instance, they can reduce the proportions of tumor-associated immunosuppressive cells (e.g., regulatory T cells) and increase those of effector T cells, creating an environment hostile to tumor survival, which can also inhibit the growth of distant metastatic lesions. 35 (3) Promotion of Immunological Memory: Anti-PD-1 inhibitors not only trigger immediate immune responses but also promote long-term immunological memory. This memory allows the body to respond rapidly to tumor recurrence or the development of new metastases, limiting further progression. 36 (4) Overcoming Immune Evasion Mechanisms: Metastatic tumors often evade immune surveillance by upregulating the levels of PD-L1 to inhibit T-cell activity. Anti-PD-1 inhibitors block these pathways, enabling the immune system to recognize and attack metastatic tumor cells that previously escaped detection, which is crucial for the elimination of distant metastases. 37 In summary, the effectiveness of anti-PD-1 inhibitors against distant metastatic lesions can be attributed to their ability to enhance systemic immune responses, modulate the tumor microenvironment, promote immunological memory, and overcome immune evasion. These mechanisms collectively contribute to the unique advantages of anti-PD-1 inhibitors in treating metastatic cancers. However, the efficacy of immunotherapy on nasal metastases of lung SCC could not be evaluated in previous studies due to the rarity of nasal metastasis. Therefore, this gap is addressed by the present research.
This study has several limitations. First, as a single case report, the findings may not be generalizable to all patients with pulmonary SCC and nasal metastases. Second, although a literature review was included, the number of reported cases involving nasal metastasis remains extremely limited, and the available data are heterogeneous in terms of treatment approaches and clinical outcomes. Therefore, further case accumulation and larger studies are needed to validate our observations. Third, this case lacked immunohistochemical or molecular characterization of the nasal metastatic lesion, such as PD-L1 expression, immune cell infiltration (e.g., CD8+ T cells and Treg cells), or mutational profiling. This limits our ability to investigate the biological mechanisms underlying the response to immunotherapy in rare-site metastases. It is recommended that future research focus on comprehensive immunological and molecular characterization of both primary and metastatic sites, as this may provide insights into tumor microenvironment heterogeneity, inform individualized treatment strategies, and enhance prognostic accuracy.
Conclusion
Nasal metastasis from pulmonary SCC is rare, and the prognosis of previously reported cases has been poor. This is the first case to report the combination of standard chemotherapy and the anti-PD-1 inhibitor Tislelizumab for pulmonary SCC with nasal tip metastasis. The treatment demonstrated significant efficacy in chemotherapy-refractory nasal metastases of pulmonary SCC. In advanced pulmonary SCC, this study offers a treatment strategy for rare-site metastasis.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666251384018 – Supplemental material for Tislelizumab in conjunction with chemotherapy for the treatment of nasal metastasis from lung squamous cell carcinoma: a case report and literature review
Supplemental material, sj-pdf-1-tar-10.1177_17534666251384018 for Tislelizumab in conjunction with chemotherapy for the treatment of nasal metastasis from lung squamous cell carcinoma: a case report and literature review by Kang Luo, Liwan Dai, Kexin Lin, Hengchang Zhang, Kaili Pi, Na Yang, Yuan Gao, Jing Yu, Jinxing Wu and Qian Xiao in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
