Abstract
Background:
Comorbid rheumatoid arthritis (RA) is known to be associated with excess mortality in patients with bronchiectasis. However, whether excess mortality is affected by RA seropositivity and is altered by using disease-modifying anti-rheumatic drugs (DMARDs) remains unknown.
Objectives:
To assess the association between comorbid RA and mortality in participants with bronchiectasis, plus the impacts of seropositivity and DMARDs on this association.
Design:
A retrospective cohort study.
Methods:
Mortality rates were compared between participants with bronchiectasis-RA overlap syndrome (BROS) (n = 3355; 2632 seropositive RA (SPRA) and 723 seronegative RA (SNRA)) and 1:5 age- and sex-matched participants with bronchiectasis only (n = 16,240) who were enrolled between 2010 and 2017 in the Korean National Health Insurance Service database. The participants were followed up from 1 year after RA diagnosis or the corresponding index date to the date of death, censored date, or 31 December 2020.
Results:
During a median follow-up of 5.8 years (interquartile range, 4.2–7.8 years), participants with BROS revealed a 2.09-fold higher mortality risk compared with participants with bronchiectasis only, even after adjusting for potential confounders (95% confidence interval (CI), 1.88–2.33). In an analysis of RA serologic status using a fully adjusted model, participants with SPRA and those with SNRA showed 2.34-fold (95% CI, 2.09–2.62) and 1.29-fold (95% CI, 1.01–1.65) increased risks, respectively, than participants with bronchiectasis only. DMARDs use was related to increased mortality.
Conclusion:
The presence of RA doubles the mortality risk in patients with bronchiectasis. Increased mortality risk was more evident in patients with SPRA and those who use DMARDs. Causality cannot be ascertained, but these data suggest that rheumatic inflammation may affect disease progression and excess mortality in patients with BROS.
Introduction
Bronchiectasis has been recognised as an extra-articular manifestation of rheumatoid arthritis (RA),1,2 with a cumulative incidence of approximately 7% during a 9-year follow-up, 3 and international bronchiectasis registries have also reported RA as an aetiology of bronchiectasis.4–8 Furthermore, the risk of developing bronchiectasis was significantly higher in patients with seropositive RA (SPRA) than in those with seronegative RA (SNRA). 3 Considering that SPRA usually denotes a higher inflammatory state than SNRA, 9 the effect of RA seropositivity on bronchiectasis development is consistent with our current concept that inflammation plays a pivotal role in bronchiectasis pathogenesis.10,11
Bronchiectasis-RA overlap syndrome (BROS) is clinically relevant because its coexistence is related to excess mortality.12–14 If rheumatic inflammation is associated with the development of bronchiectasis, 3 we can hypothesise that inflammation also affects excess mortality in BROS. Furthermore, excess mortality in BROS could plausibly be alleviated by disease-modifying anti-rheumatic drugs (DMARDs) that control rheumatic inflammation. Alternatively, DMARDs use may indicate more severe RA, and these drugs carry a risk of increasing infection, thus potentially worsening outcomes. However, such hypotheses, particularly regarding the impact of DMARDs on mortality, are difficult to evaluate in clinical trials or observational studies without long-term follow-up data.
Therefore, this study aimed to assess the association between comorbid RA and mortality in participants with bronchiectasis, as well as the impact of seropositivity on this association. Furthermore, we aimed to examine the association between DMARDs and mortality in BROS and identify the factors affecting the association between BROS and mortality.
Methods
Study data
We used a dataset from the Korean National Health Insurance Service (NHIS), which covers 97% of the Korean population as a mandatory universal healthcare programme. The NHIS database includes demographics, the International Classification of Diseases 10th revision (ICD-10) diagnostic codes, prescriptions and healthcare utilisation. In addition, the NHIS offers a biannual health screening programme for all individuals aged ⩾40 years; the health screening examination includes weight, height, a self-administered questionnaire on health behaviours and laboratory tests.3,15,16
Study population
The study population comprised patients with bronchiectasis. A bronchiectasis diagnosis is defined by at least one claim under ICD-10 diagnosis code J47 and exclusion of those with cystic fibrosis (ICD-10 code E84). The operational definition has been widely used in prior bronchiectasis studies, with reliable validity.3,17–24 Participants with bronchiectasis were categorised into the RA cohort (those with BROS) and matched controls (those with bronchiectasis only).
To establish the RA cohort, of the 92,336 individuals aged ⩾40 years and diagnosed with RA between 2010 and 2017, we initially included 54,910 who had undergone a national health screening examination within 2 years prior to their RA diagnosis. Among the remaining 52,985 participants with RA, after excluding 1925 with missing data, we finally enrolled 3355 participants with BROS who were eligible for age- and sex-matching (ratio 1:5) (Figure 1).

Flow chart of the study population.
To establish matched controls, we initially evaluated a control pool consisting of 1,255,036 individuals who were approximately matched 1:10 to 92,336 individuals with RA, based on age and sex. Of the 1,255,036 individuals, 641,341 underwent health screening in the same year when the matched participants with RA received health screening. Among the remaining 641,341 controls, after excluding 24,343 individuals with missing data, 16,240 matched participants with bronchiectasis only were eligible for 1:5 age- and sex-matching with the RA cohort (Figure 1).
Main exposures
RA was the primary exposure of this study. It was identified according to the following criteria: ICD-10 codes for RA (M05 for SPRA and M06, except M06.1 and M06.4, for SNRA), prescription records of any DMARDs for ⩾180 days (both SPRA and SNRA), and enrolment in the rare intractable diseases (RID) programme for SPRA. The RID programme offers a copayment reduction of up to 10% for rare and intractable diseases. Patients with SPRA are eligible only for the RID programme when the following criteria are met: a diagnosis certification submitted by physicians (mostly rheumatologists) and a positive test for rheumatoid factor or anti-cyclic citrullinated peptide antibodies.3,25,26 The registration date for the RID programme or the date of RA diagnosis was the index date in this study.
In addition to RA diagnosis and seropositivity, DMARD use was assessed as a study exposure. In this study, DMARDs included biologic (adalimumab, etanercept, infliximab, golimumab, rituximab, abatacept and tocilizumab) and targeted synthetic DMARDs (tofacitinib), but not conventional synthetic DMARDs such as methotrexate and hydroxychloroquine. That was because major rheumatology organisations recommend that all patients diagnosed with RA be started on DMARD therapy as soon as possible,27–29 and we defined RA based on the criteria of DMARDs prescription records for at least 180 days. Therefore, all study participants may have been taking conventional systemic DMARDs at baseline, and this study only considered biologic and targeted synthetic DMARDs in analyses.
Study outcome
The primary outcome was to compare mortality risk between participants with BROS and those with bronchiectasis only. The study participants were followed up from 1 year after their RA diagnosis or the corresponding index date to the date of death, censored date, or 31 December 2020 (the end date of the study).
Covariates
The low-income group included participants comprising a quarter of the lowest-income and Medicaid groups. Smoking status was classified as never, former, or current. In former and current smokers, cumulative smoking exposure was measured by multiplying the average daily cigarette consumption in packs by the smoking period in years.3,30 Regular physical activity was defined as ⩾ 30 min of moderate physical activity ⩾ 5 times per week or ⩾ 20 min of vigorous physical activity ⩾ 3 times per week.20,31 Body mass index (BMI) was calculated as weight in kilograms divided by the square of the height in metrers, and BMI ⩾ 25.0 kg/m2 was defined as obesity based on Asian recommendations.23,32–34 Comorbidities were also determined based on ICD-10 codes, prescriptions and health screening results as previously described.15,17,35–37
Statistical analysis
Characteristics of participants according to the presence and serologic status of RA were compared by calculating the standardised differences; an absolute standardised difference of >0.20 was interpreted as a meaningful difference. 38 The incident rate was calculated by dividing the number of deaths by the total follow-up duration (1000 person-years). A cumulative incidence plot was used to compare mortality rates between the BROS and bronchiectasis-only cohorts, as well as among the SPRA, SNRA and bronchiectasis-only cohorts. A log-rank test was employed to identify significant differences between the groups. Mortality hazards were estimated using Cox proportional hazards regression analysis and presented as a hazard ratio (HR) with a 95% confidence interval (CI). Multivariable analysis was adjusted for demographic characteristics (age and sex), socioeconomic status (low income) and lifestyle habits potentially affecting deaths (smoking status, alcohol consumption, physical activity and obesity) in Model 1. The variables included in Model 1, plus comorbidities (diabetes mellitus, hypertension, dyslipidaemia and chronic kidney disease), were adjusted in Model 2. To evaluate the factors affecting the association between RA and mortality, we performed stratified analyses according to the variables listed in Table 1. Stratified analysis used only a fully adjusted model (Model 2). All statistical analyses were performed using Statistical Analysis System version 9.4 (SAS Institute Inc., Cary, NC, USA), and p-values < 0.05 were considered significant.
Baseline characteristics of study participants with bronchiectasis.

Data are presented as mean ± SD or number (percentage).
ASMD, absolute standardised mean difference; BMI, body mass index; CKD, chronic kidney disease; PY, pack-years; RA, rheumatoid arthritis; SNRA, seronegative rheumatoid arthritis; SPRA, seropositive rheumatoid arthritis.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 39
Results
Baseline characteristics
The mean age of the study participants was 63.6 years (standard deviation, 9.6), and 71.3% were female. The characteristics were well-balanced between the BROS and bronchiectasis-only cohorts (absolute standardised mean difference for all variables < 0.2). In the RA cohort (n = 3355), 2632 (78.5%) had SPRA and 723 (21.5%) had SNRA. Clinical characteristics were not significantly different between participants with SPRA and those with SNRA (Table 1).
Mortality risk in participants with bronchiectasis according to the presence of RA and RA serologic status
A total of 1654 participants died (1176 in participants with bronchiectasis only and 478 in those with BROS; 7% and 14%, respectively) during the follow-up period of 100,056 person-years, corresponding to a median of 5.8 years (interquartile range, 4.2–7.8 years). Participants with BROS showed a 2.07-fold higher mortality risk compared with those with bronchiectasis only in an unadjusted model (95% CI, 1.86–2.30); this association was consistent in the two adjusted models (Model 1: adjusted HR, 2.09 (95% CI, 1.88–2.33); Model 2: adjusted HR, 2.09 (95% CI, 1.88–2.33)) (Table 2). A significant difference was also evident in the cumulative incidence probability of mortality between the BROS and bronchiectasis-only cohorts (Figure 2(a)).
Mortality risk according to RA status and serologic status of RA in participants with bronchiectasis.

Control group denotes participants with bronchiectasis only. Model 1 was adjusted for age, sex, low-income status, smoking, alcohol consumption, physical activity and obesity. Model 2 was further adjusted for diabetes mellitus, hypertension, dyslipidaemia and chronic kidney disease in addition to the variables adjusted for Model 1.
CI, confidence interval; HR, hazard ratio; PY, person-years; RA, rheumatoid arthritis; SNRA, seronegative rheumatoid arthritis; SPRA, seropositive rheumatoid arthritis.
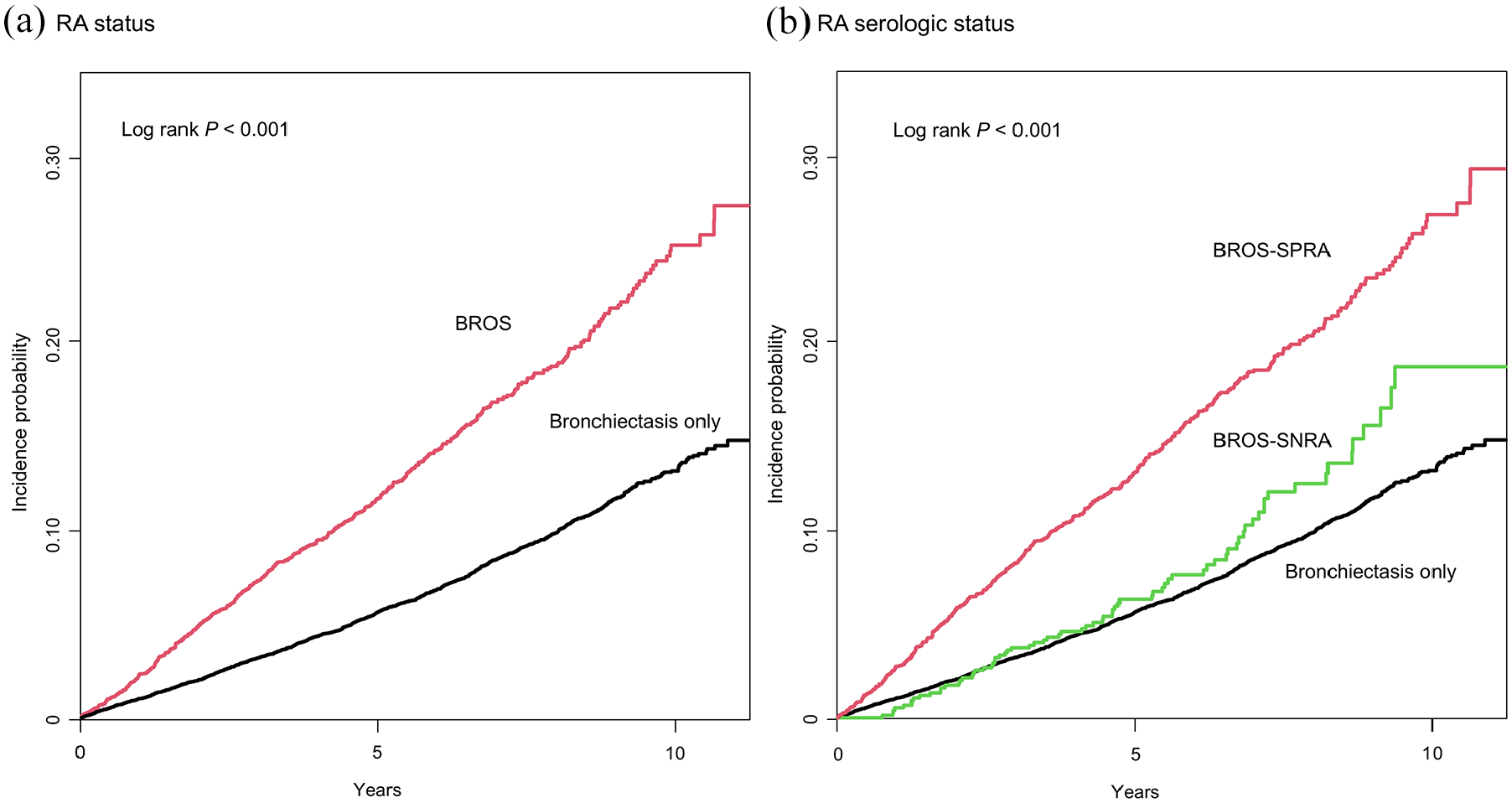
Cumulative incidence probability of mortality in participants with bronchiectasis according to (a) rheumatoid arthritis status and (b) serologic status of rheumatoid arthritis.
An analysis according to RA serologic status revealed that participants with SPRA and those with SNRA showed 2.34-fold (95% CI, 2.09–2.62) and 1.29-fold (95% CI, 1.01–1.65) increased mortality risks, respectively, compared with those with bronchiectasis-only in the fully adjusted Model 2. Compared with the SNRA group, the SPRA group had a higher mortality risk (Model 2: adjusted HR 1.77 (95% CI, 1.37–2.29)) (Table 2). The analysis of the cumulative incidence probability of mortality showed similar results (Figure 2(b)).
Mortality risk in participants with bronchiectasis according to RA and exposure to biologic or targeted synthetic DMARDs
In an analysis according to RA and exposure to biologic or targeted synthetic DMARDs in the fully adjusted Model 2, participants with BROS exposed to DMARDs and those unexposed to biologic or targeted synthetic DMARDs showed 2.96-fold (95% CI, 2.25–3.89) and 2.03-fold (95% CI, 1.83–2.25) increased mortality risks, respectively, than participants with bronchiectasis-only (Table 3).
Mortality risk according to RA status and exposure to biologic or targeted synthetic disease-modifying anti-rheumatic drugs in participants with bronchiectasis.

Control group denotes participants with bronchiectasis only.
Model 1 was adjusted for age, sex, low-income status, smoking, alcohol consumption, physical activity and obesity. Model 2 was further adjusted for diabetes mellitus, hypertension, dyslipidaemia and chronic kidney disease in addition to the variables adjusted for Model 1.
BROS, bronchiectasis-rheumatoid arthritis overlap syndrome; b/ts, biologic or targeted synthetic; CI, confidence interval; DMARD, disease-modifying anti-rheumatic drug; HR, hazard ratio; PYs, person-years; RA, rheumatoid arthritis.
Stratified analyses
Demographics, income level and comorbidities had no significant effect on the association between RA or serologic status of RA and mortality risk (p for interaction > 0.05). However, the association between RA and mortality risk was more robust in obese participants (adjusted HR, 2.52 (95% CI, 2.07–3.06)) compared with non-obese participants (adjusted HR, 1.97 (95% CI, 1.76–2.21)) (p for interaction = 0.035); similar results were shown in analyses classified by serologic status (p for interaction = 0.030). In addition, in a stratified analysis based on smoking history, the association was only significant in never-smokers (adjusted HR, 2.27 (95% CI, 2.02–2.56)), ex-smokers (⩾20 pack-years) (adjusted HR, 2.34 (95% CI, 1.74–3.15)) and current smokers (⩾20 pack-years) (adjusted HR, 1.91 (95% CI, 1.36–2.70)) (p for interaction = 0.009); however, this trend was not observed in analyses classified by serologic status (Table 4).
Stratified analysis of mortality risk according to RA status and serologic status of RA in participants with bronchiectasis.

Data are presented as hazard ratios (95% confidence intervals). Hazard ratios were adjusted for age, sex, low-income status, smoking, alcohol consumption, physical activity, obesity, diabetes mellitus, hypertension, dyslipidaemia and chronic kidney disease.
CKD, chronic kidney disease; NA, not applicable; PA, pack-year; RA, rheumatoid arthritis; SNRA, seronegative rheumatoid arthritis; SPRA, seropositive rheumatoid arthritis.
Discussion
The present study showed that comorbid RA approximately doubled the mortality risk in patients with bronchiectasis, even while adjusting for potential confounders. The excess mortality was more apparent in participants with SPRA than in those with SNRA compared with those with bronchiectasis only.
This study revealed that comorbid RA was related to excess mortality in participants with bronchiectasis, which was consistent with the results of previous studies.12,13 Notably, we demonstrated that participants with SPRA and those with SNRA had approximately 2.3- and 1.3-fold higher mortality risks, respectively, than the matched controls, suggesting that bronchiectasis-RA mortality is increased by seropositivity, potentially associated with increased inflammation. Despite the consistent results regarding excess mortality, De Soyza et al. 12 reported that the coexistence of bronchiectasis and RA was not associated with higher rates of bronchiectasis exacerbation or bronchiectasis-related hospitalisations. Thus, we postulate that comorbid RA and its management may not increase the exacerbation risk and directly affect the clinical course of bronchiectasis, but the underlying rheumatic inflammation may lead to subclinical worsening. Rheumatic inflammation may worsen comorbidities contributing to mortality in bronchiectasis, such as cardiovascular and malignant diseases.21,26,40,41 This explanation is consistent with our current understanding of bronchiectasis as an inflammatory disease and raises the potential for controlling inflammation as a target in managing BROS.42,43
Therefore, we tested the hypothesis that DMARDs would reduce excess mortality by achieving better RA control. In addition, in a recently published study on RA-interstitial lung disease, biologic DMARDs tended to exhibit a survival benefit, although the difference was not statistically significant. 44 However, in the present study, previous biologic and targeted synthetic DMARDs were unexpectedly associated with increased mortality in patients with BROS. The first potential explanation for this phenomenon is that participants with severe BROS, who had already progressed to the disease state, may have received DMARDs compared to participants with less severe BROS, which was consequently related to increased mortality. Second, DMARDs may negatively affect bronchiectasis in patients with RA by increasing the risk of opportunistic infections and consequent recurrent infections.45,46 One Taiwanese study reported that biologic DMARDs were associated with an increased risk of tuberculosis and non-tuberculous mycobacterial infection in patients with BROS, but not with a higher mortality. 47 A limitation of our study is the absence of data on the causes of death. If such data were available, we could better explain the relationships between increased mortality and the use of previous biologic and targeted synthetic DMARDs; therefore, further studies are warranted to clarify this issue. Lastly, our analysis does not differentiate between DMARDs’ response – it is plausible that DMARDs, when given, may have been used with caution and not attained maximal RA control for fear of increased infection with DMARDs. Future studies, where possible, should include data on how well the DMARDs are titrated, for example, by reporting objective measures of RA control, such as DAS28-CRP.
Interestingly, in the stratified analyses in this study, obesity was related to increased mortality in participants with BROS, even while adjusting for potential confounders. The impact of obesity on RA mortality is controversial; some studies have reported increased mortality due to cardiovascular risk factors such as hypertension and metabolic syndrome, but other studies have shown that weight loss is a strong predictor of mortality in RA.48–50 The present study revealed that obesity robustly augmented the association between BROS and excess mortality. We interpreted the potential results of obesity-related various comorbidities, aligning with previous studies showing the negative impacts of comorbidities on long-term outcomes in bronchiectasis.21,40 To expand on this notion, we may need to develop management guidelines for BROS that incorporate unique features and clinical relevance.
A major strength is that this study is one of the largest studies on BROS, which reinforces the association between comorbid RA and excess mortality in bronchiectasis and further evaluates the impact of seropositivity and DMARDs on this association. Nonetheless, this study had some limitations. First, we used ICD-10 codes to define bronchiectasis and other comorbidities, which might be associated with over- or under-diagnosis. Second, the relatively small number of participants receiving DMARDs and the observational nature of the study may limit the analyses of the impact of DMARDs on mortality. Third, detailed information on bronchiectasis (e.g., exacerbation rates, hospitalisations and microbiological test results) and RA disease activity (e.g., joint erosive status and inflammatory markers) was unavailable due to the population-based study design. Fourth, RA-associated interstitial lung disease has been known to be more closely related to a poor prognosis in patients with RA than other pulmonary manifestations; however, we lack data on RA-associated interstitial lung disease that may have affected our study results. 51 Finally, as the aetiology and comorbidities of bronchiectasis vary by geographic region,5,8 our results should be applied to other countries with caution.
Conclusion
Participants with BROS had approximately twice the mortality risk of those with bronchiectasis only, even after adjusting for potential confounders. Increased mortality risk was most apparent in patients with SPRA, followed by those with SNRA and those with bronchiectasis only, implying that rheumatic inflammation may significantly affect disease progression and increased mortality in patients with BROS. Although DMARDs were associated with increased mortality, future studies are warranted to elucidate this issue and develop appropriate management strategies to ameliorate the disease burden of BROS.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251360071 – Supplemental material for Impact of rheumatoid arthritis, seropositivity and disease-modifying anti-rheumatic drugs on mortality risk in bronchiectasis
Supplemental material, sj-docx-1-tar-10.1177_17534666251360071 for Impact of rheumatoid arthritis, seropositivity and disease-modifying anti-rheumatic drugs on mortality risk in bronchiectasis by Hayoung Choi, Kyungdo Han, Jin Hyung Jung, Anthony De Soyza, Hyungjin Kim, Dong Wook Shin and Hyun Lee in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
