Abstract
Background:
The role of the gut-lung axis in respiratory disease is increasingly recognised. Much emphasis has been placed on gastro-oesophageal reflux disease; however, oesophageal dysmotility may also play a significant role. Azithromycin, a known prokinetic, has been shown to be of major benefit in a number of respiratory diseases, but the relationship between oesophageal function and the lung has not been examined.
Objectives:
We assessed the feasibility of performing continuous cough monitoring and repeated high-resolution oesophageal manometry (HROM) in patients with chronic respiratory disease.
Design:
We conducted an open-label, single-arm, feasibility trial.
Methods:
Azithromycin 250 mg once daily was given to patients with chronic respiratory disease who reported a chronic cough. All participants were monitored continually for at least 1 week prior to and 4 weeks after azithromycin with the Hyfe Cough Tracker. Participants also had HROM performed at two time-points, immediately before and 4 weeks after initiation of azithromycin. Feasibility outcomes pertaining to recruitment, data quality, and acceptability of trial processes were assessed. Exploratory outcome data for metrics of oesophageal function were also analysed.
Results:
A total of 30 participants (57% female, mean age 65.2 (SD = 11.3)) were recruited over a 10-month period, giving a recruitment rate of three patients per month in a single centre. A total of 87% (n = 26) of participants completed all three study visits. All pre-specified data quality outcomes met their ‘green’ traffic light stop-go criteria. HROM demonstrated that the majority (52%) of participants had abnormal oesophageal function, as defined by the Chicago Classification, at baseline. Changes in oesophageal function were not significantly associated with changes in objective or subjective cough measures, except for a weakly negative correlation with the Hull Airway Reflux Questionnaire score.
Conclusion:
A large-scale trial examining the effect of azithromycin on the relationship between oesophageal function and cough in respiratory disease is feasible and acceptable to patients.
Trial registration:
This trial was prospectively registered ClinicalTrials.gov ID: NCT05469555.
Plain language summary
This study explores the connection between gullet function and cough, particularly in patients with chronic respiratory diseases. It focuses on azithromycin, a medication known to improve many different respiratory conditions. The researchers conducted a trial involving 30 patients who experienced chronic cough. They monitored patients’ coughs continuously and assessed their oesophageal function using a specialized test both before and after starting azithromycin. As oesophageal testing is known to be an invasive procedure, it was vital that the acceptability to patients of this procedure was tested. The trial showed that it is feasible to conduct this type of research, as most participants completed the study and reported that they were not put off by the investigation. The results of this study suggest that a larger clinical trial is required.
Introduction
The importance of the gut-lung axis in the development and progression of respiratory disease has been highlighted in numerous studies in recent years.1 –3 There is debate as to what aspect of gastrointestinal pathology is most pertinent in respiratory disease, with many believing that acidic gastro-oesophageal reflux disease (GORD) may be chiefly responsible. However, there have been negative trials of proton-pump inhibitors, the cardinal therapeutic for GORD, for respiratory diseases such as chronic cough and asthma.4,5
Alternatively, aspiration of non-acidic refluxate may be responsible. This mechanism has been termed ‘Airway Reflux’ 6 since it may afflict any supra-oesophageal airway. In the lung, chronic ‘silent’ aspiration of gastrointestinal content from the oesophagus causes damage. The epithelium responds by releasing alarmins such as Adenosine Triphosphate (ATP). ATP then acts on P2X3 receptors which depolarise vagal sensory afferents. How this leads to the chronic hypersensitivity in cough hypersensitivity syndrome 7 is unknown. We have previously shown that up to two-thirds of patients with diverse chronic respiratory diseases have oesophageal dysfunction, when measured by high-resolution oesophageal manometry (HROM). 8
The macrolide antibiotic azithromycin mimics a panacea in respiratory medicine. Multiple trials showing its efficacy in reducing exacerbation frequency in asthma, chronic obstructive pulmonary disease (COPD), and bronchiectasis.9 –11 Despite its exceptional clinical effects, no current hypothesis explains such diverse activity; however, immunomodulatory and antibacterial mechanisms have been suggested.12,13 Azithromycin is an agonist of the hormone motilin which regulates Gastrointestinal (GI) motility14 –16 but its effects on oesophageal motility have been scarcely studied. It may be the case that improved GI motility, engendered by azithromycin, may reduce exacerbations and improve symptoms of such respiratory disease by improving airway reflux.
To understand the effect of azithromycin on oesophageal function and its impact on clinical outcomes in respiratory disease, a prospective clinical trial is necessary. As HROM is an invasive procedure, it was unclear whether participants would tolerate multiple procedures to better understand their disease. We therefore set out to perform a study to assess the feasibility and acceptability of performing repeated HROM studies to measure oesophageal functioning in participants with a broad range of chronic respiratory diseases, particularly those who reported cough as a prominent symptom.
Methods
To inform a potential randomised controlled trial of the effect of azithromycin on oesophageal dysmotility and how this relates to respiratory symptoms, we undertook an open-label single-arm feasibility trial. Patients were recruited from Hull University Teaching Hospitals NHS Trust between July 2023 and May 2024 and all provided written informed consent to participate. We report the findings of this study using the Consolidated Standards of Reporting Trials (CONSORT) checklist extension to pilot and feasibility trials. 17
Study protocol
Patients with chronic respiratory disease who were being considered for long-term azithromycin therapy by their treating clinician were eligible to be recruited to this study. Eligibility was assessed by a Hull Airway Reflux Questionnaire score above the upper limit of normal (>14). 18 A target sample size of 30 patients was set, as is common in UK feasibility studies. 19
The trial comprised three study visits, one screening visit, one pre-azithromycin initiation visit, and one follow-up visit. The schedule of events for the study, including all investigations that participants undertook at each visit, can be seen in Figure 1. At screening the main exclusion criteria were a contraindication to macrolide antibiotics, either due to allergy, sensitivity, or QTc prolongation (>450 ms for males, >470 ms for females) on electrocardiogram (ECG). Informed written consent was provided.

Schedule of events for the clinical trial.
The nature of the participants’ respiratory disease, with particular regard to their cough, was assessed through validated questionnaires at screening. Participants were then provided with a continuous cough monitor to record cough counts for the duration of the study. Participants also completed an invitation experience questionnaire in which they were asked to describe their experience of being invited to participate in the trial.
Participants re-attended for their pre-azithromycin initiation visit (Visit 2) approximately 1 week after screening, at which point they undertook their baseline HROM investigation. Following this test, they commenced azithromycin 250 mg once daily. Participants were advised to contact the trial team if they experienced any common adverse effects of azithromycin, including gastrointestinal discomfort, nausea, diarrhoea, or vomiting. If the participants reported any of these symptoms, the study team advised the patient to reduce their dose to 250 mg three times per week in the first instance. The patient was advised to discontinue altogether if the adverse effects were intolerable or if the patient did not wish to continue.
Participants re-attended for their final follow-up visit (Visit 3) approximately 1 month after their pre-azithromycin visit. During this visit, they had a further HROM study performed, and all other clinical assessments were repeated. Participants then completed a study experience survey to assess the acceptability of the trial.
High-resolution oesophageal manometry protocol and metrics
All participants were investigated using a standardised HROM technique. Participants were advised to fast 4 h prior to the investigation and were investigated with a 36-channel combined manometric and impedance monitoring catheter. This catheter was inserted trans-nasally to a length of 60 cm from the nostril, and suitably adjusted to record pressure along the length of the patient’s oesophagus and within the oesophago-gastric junction.
A standardised protocol of oesophageal function tests was followed, with further tests utilised if felt appropriate by the clinical scientist performing the test. The standardised protocol included:
Ten individual supine wet (5 mL) swallows, with an interval of 20 s between each swallow
A baseline oesophago-gastric junction resting measurement during quiet respiration for a minimum of three respiratory cycles
Five individual supine wet (2 mL) swallows, with an interval of 2 s between each swallow (multiple rapid swallow sequence)
Ten individual upright solid swallows (bread)
Each investigation was analysed manually by an expert clinical scientist and a full analysis report was produced. A list of the measured metrics of oesophageal functioning, along with a brief explanation of their significance, is given in Table 1. Oesophageal motility was assessed based on Version 4.0 of the Chicago Classification 20 and included the distal contractile integral (DCI), integrated relaxation pressure (IRP), and lower oesophageal sphincter resting pressure (LOSP).
HROM metrics with brief explanations regarding their measurement and significance.

HROM, high-resolution oesophageal manometry.
Feasibility outcomes
To assess the feasibility of a larger randomised controlled trial, the following feasibility outcomes were used:
Recruitment: eligibility to consent ratio, recruitment rate (participants recruited to the trial per month), and participant retention to follow-up.
Data quality: completion of clinical outcomes at each visit and patterns of missing data for the trial measures. Completion of participant symptom questionnaires throughout the study. Completion of trial processes, principally HROM.
Acceptability of assessment: this was primarily the willingness of participants to undertake both HROM tests. We also assessed this using the invitation and study experience questionnaires.
A ‘traffic light’ stop-go criteria was used to assess the feasibility of a larger study, based on these findings. This will inform the larger trial protocol and potential recruitment target.
Exploratory clinical outcomes
The following physiological and clinical outcomes were assessed for a signal of change to inform primary and secondary endpoint selection for a larger randomised controlled trial:
Oesophageal function: we evaluated the effect of azithromycin on the measures of oesophageal contraction and swallow coordination, as described in Table 1. This was done by comparing the HROM studies for individual participants before and 1 month after initiation of azithromycin treatment.
Subjective symptom burden: Symptoms were assessed before and 1 month after initiation of azithromycin. These were measured using validated questionnaires to assess the severity of symptoms as well as health-specific quality of life. The specific results of these will be reported and discussed elsewhere.
Relationship between oesophageal motility and symptoms: The change in oesophageal functioning and both objective and subjective measures of respiratory symptoms was examined in each patient.
Objective cough frequency: Effect of azithromycin therapy on continuous objective cough frequency as measured by the Hyfe Cough Tracker©, Wilmington, Delaware. As above, findings from these assessments will be reported elsewhere.
Statistical analysis
Participant characteristics are summarised using descriptive statistics. Medians (range) are reported for non-parametric data, mean (standard deviation) for continuous, normally distributed data, and raw count (number (%)) are reported for nominal data. The primary feasibility outcomes are presented using raw numbers and percentages. Comparison of baseline and follow-up HROM and questionnaire data was performed using paired t-tests for normally distributed data and Mann–Whitney U testing for skewed data. Correlations between symptoms and HROM metrics were performed using Pearson’s correlation coefficient for normally distributed data and Spearman’s rank correlation for non-normal data. All data were analysed using IBM SPSS Statistics 29 (IBM Corp., Armonk, NY, USA).
Results
Study recruitment
Recruitment took place between July 2023 and May 2024, and 30 potential participants were assessed for eligibility, with all participants who were screened being deemed eligible for inclusion, giving an eligibility rate of 100%. Of these 30 participants, all 30 provided informed consent to participate in the trial and completed the initial study visit, giving a consent rate of 100%.
Patient characteristics
Of the 30 participants recruited to this study 60% (n = 18) had a diagnosis of chronic cough, 23% had a diagnosis of interstitial lung disease (n = 7), and 17% had a diagnosis of obstructive airways disease (n = 5). The cohort had a mean age of 65.2 (SD = 11.3), and 57% (n = 17) were female. All baseline characteristics are displayed in Table 2.
Baseline characteristics of participants included in the study.

COPD, chronic obstructive pulmonary disease; FEV1, Forced expiratory volume in 1 second; FVC, Forced Vital Capacity; HARQ, Hull airway reflux questionnaire; VAS, visual analogue scale.
Feasibility outcomes
The eligibility:consent ratio for the study was 1:1, as all participants who were deemed eligible for the study agreed to participate. The recruitment rate for the study was calculated by the total number of participants recruited per month for the duration of the study; this worked out as three participants per month (30 participants recruited over a 10-month period). Participant retention was 93% (n = 28) at visit 2 and 87% (n = 26) of all participants who were recruited were able to complete the study. The study therefore met all of its primary recruitment and retention feasibility outcomes, indicating that a larger randomised controlled trial is feasible. All feasibility outcomes and their respective ‘traffic light’ stop-go criteria are presented in Table 3.
Results of the feasibility outcomes with their associated stop-go criteria to be successfully deemed feasible in a large-scale randomised clinical trial.
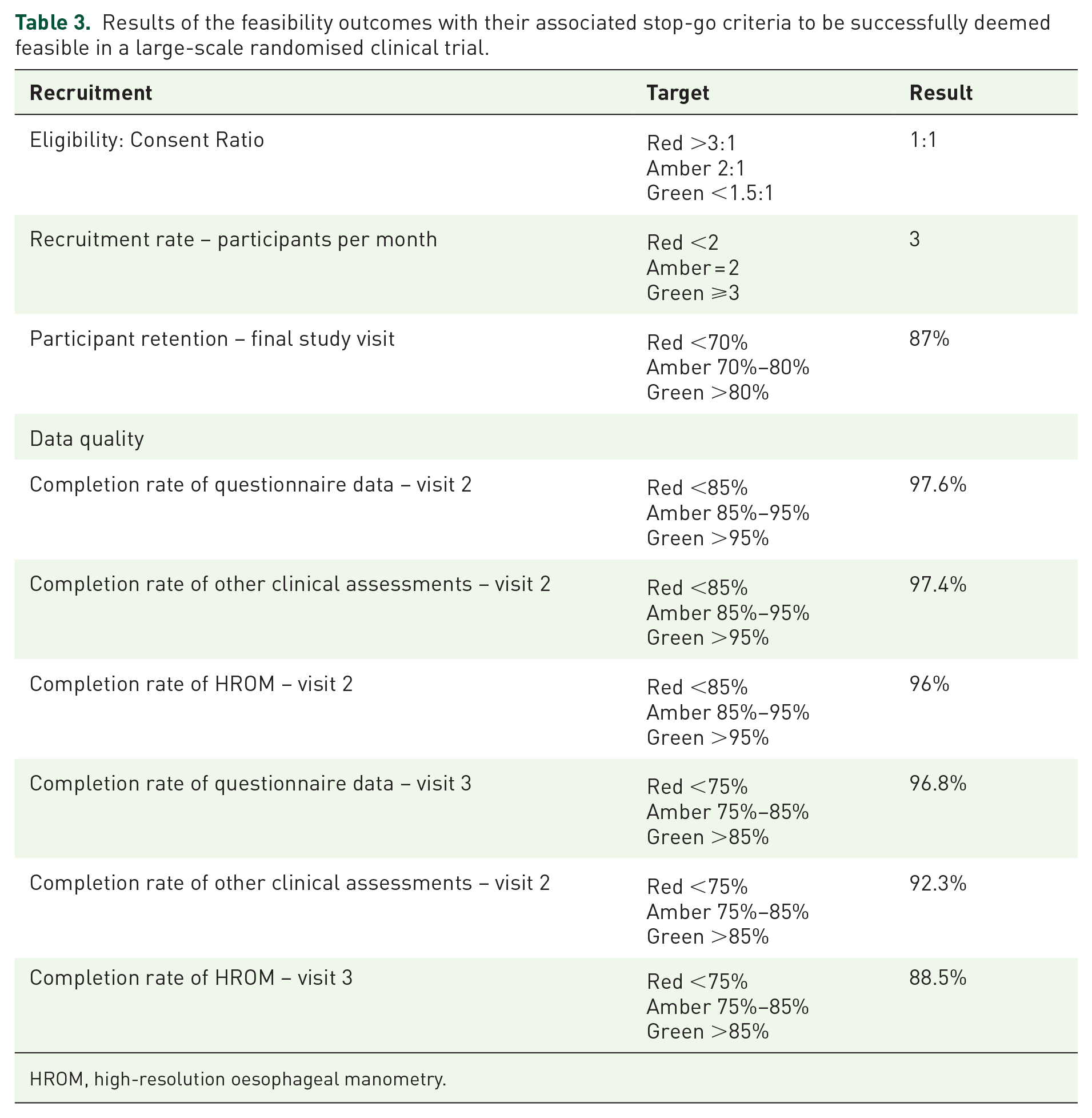
HROM, high-resolution oesophageal manometry.
Data quality
For the initial visit, all clinical assessments were carried out, with no missing data. The flow of participants throughout the clinical trial can be seen in Figure 2.

CONSORT diagram of patient flow in the trial.
Pre-azithromycin initiation visit (visit 2)
For the 28 participants who attended visit 2, 97.6% of questionnaire data was collected, 96% (n = 27) of participants had their initial HROM performed, and 97.4% of other clinical assessments such as spirometry, ECG, and physical examination were completed.
Follow-up visit (visit 3)
Of the 26 participants who remained in the trial until the final study visit, 96.8% of questionnaire data were able to be collected. 88.5% (n = 23) participants had both their initial and follow-up HROM performed as two participants declined a second study and one participant was unable to have the procedure performed due to an anatomical issue at visit 2. A total of 92.3% of the other clinical asssessements were completed for participants at visit 3.
Continuous assessment of cough
Participants were provided with a physical paper cough diary which comprised 30 days of consectuive cough visual analogue scale (VAS) scoring, of the 26 participants who completed the study 73% (n = 19) were able to return a completed diary. Of the participants who completed the study, 92% (n = 24) had satisfactory levels (>12 h per day for 90% of days monitored) of continuous cough recordings using the Hyfe Cough Tracker©.
Invitation and study experience surveys
Of the participants who attended the screening visit, 77% (n = 23) completed the invitation experience survey. One of the core components of this survey was an eight item Likert scale questionnaire, exploring the patient’s understanding and thoughts around participating in the trial. The results from this questionnaire can be seen in Figure 3. When asked whether participants may have been discouraged from participating in the trial by having an invasive investigation (i.e., HROM) 74% responded either ‘disagree’ or ‘strongly disagree’. When asked what the participants’ primary motivation for participating in the trial was 56% (n = 13) stated it was to ‘improve their cough’, 48% (n = 11) reported it was to help other people who may have a similar condition, 17% (n = 4) stated they wanted to better understand why they cough, and 17% (n = 4) wanted to help to find a new treatment for chronic cough (each participant could give more than one reason).
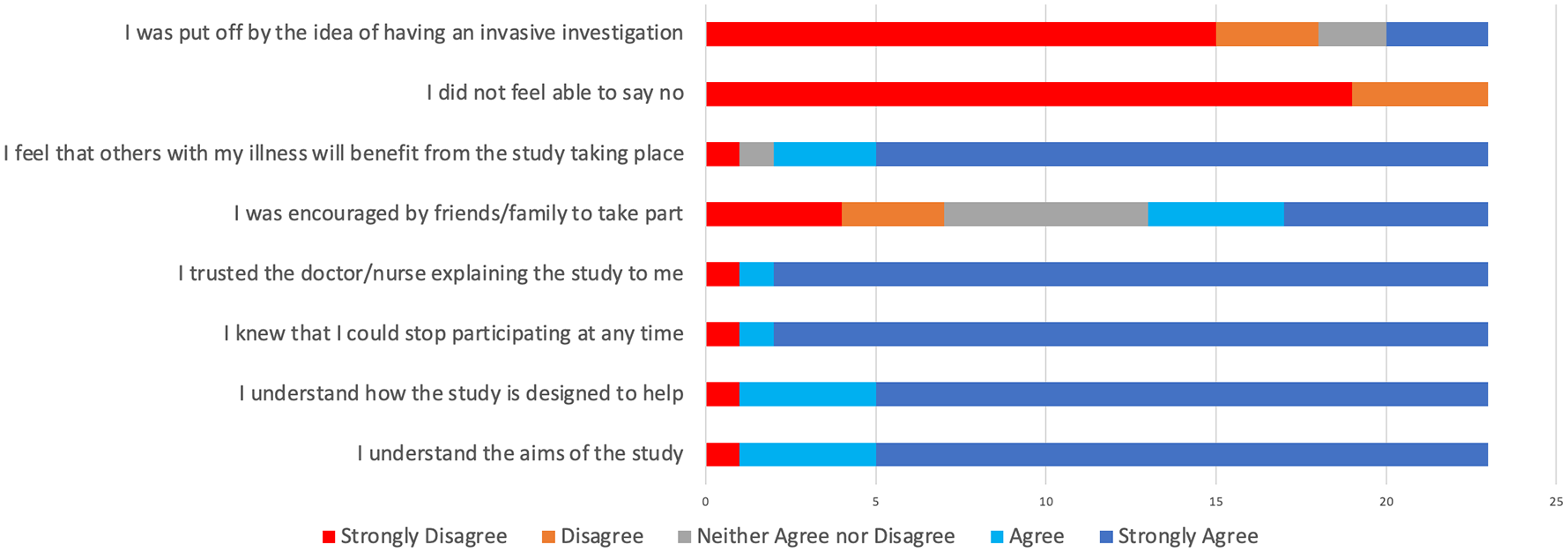
Responses given by study participants to the trial invitation experience survey.
The proportion of participants who attended visit 3 and completed the study experience survey was 50% (n = 13). When asked their overall experience of the trial 92% (n = 12) stated it was ‘good’ and 8% (n = 1) reported it to be ‘fairly good’.
Effect of azithromycin on oesophageal function – exploratory outcomes
The majority of participants included in this study had abnormal oesophageal function present on their baseline HROM study (52%, n = 14), over half were also found to have a hiatus hernia (55%, n = 15). The Chicago classification for all participants at baseline was either normal (48%, n = 13), ineffective oesophageal motility (41%, n = 11), or absent contractility (11%, n = 3). The proportions of participants with these three classifications were not significantly different on repeat HROM testing at follow-up, as there were no instances of change of the Chicago classification in any participant.
There was no significant difference between the DCI at baseline and follow-up (459 vs 493, p = 0.362), although the median change in DCI in all participants was an increase of 12.8% (range = −78.3% to 293.1%). The proportion of participants who had a >30% improvement in their DCI was 42% (n = 10). Individual participant data for changes in DCI is displayed in Figure 4.

Individual patient data for DCI measurements at baseline and follow-up for all participants.
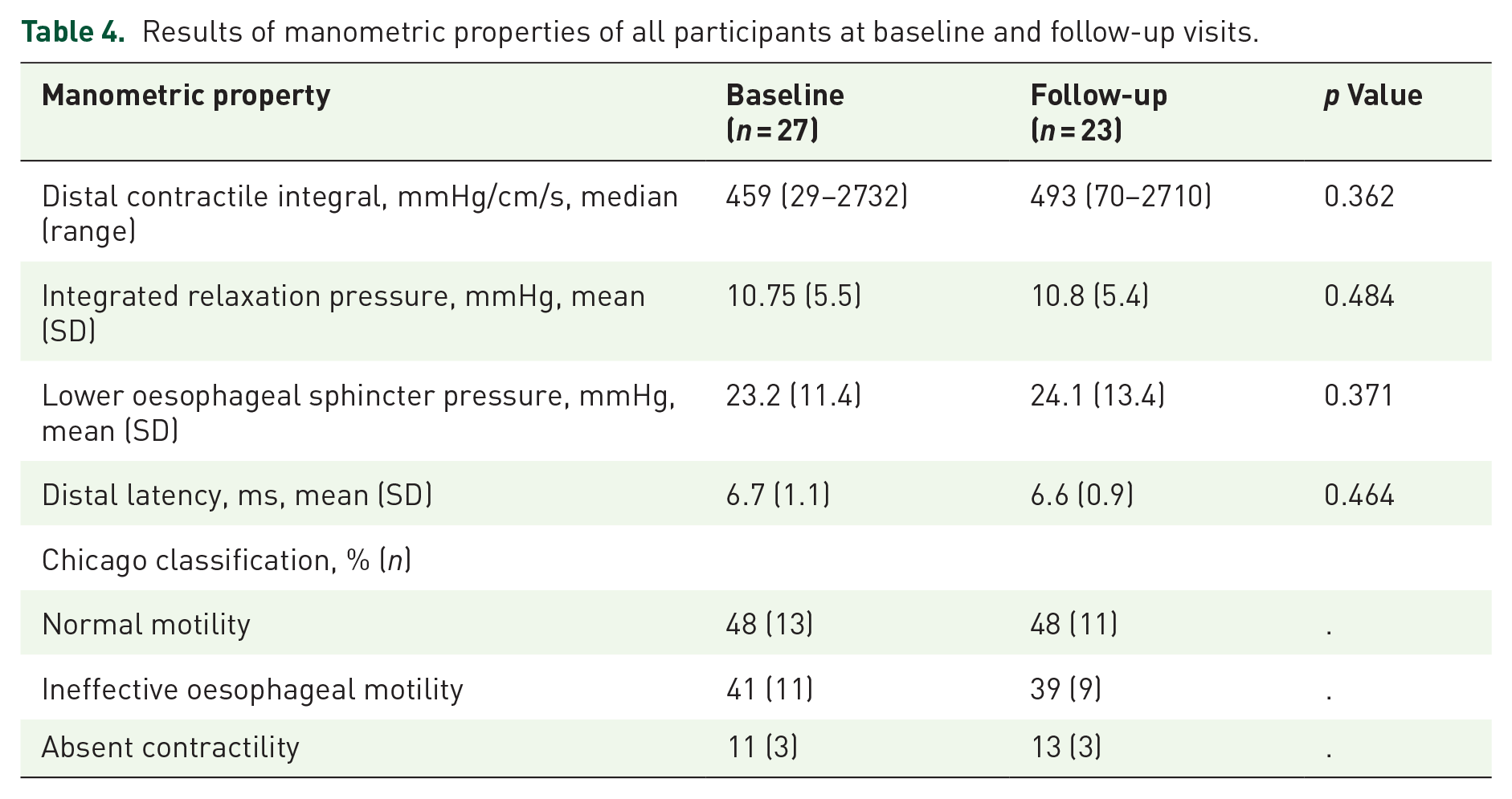
There were no significant differences between baseline and follow-up measurements of IRP (10.75 (SD = 5.5) vs 10.8 (SD = 5.4), p = 0.484) or LOSP (23.2 (SD = 11.4) vs 24.1 (SD = 13.4), p = 0.371). All data for manometric findings can be found in Table 4.
Results of manometric properties of all participants at baseline and follow-up visits.
Relationship between HROM and symptoms – exploratory outcomes
As previously stated, the effect of azithromycin on respiratory symptoms will be detailed elsewhere; however, we performed an exploratory analysis of the relationship between the change in objective and subjective measures of cough with the HROM findings.
There was no significant relationship between changes in DCI and changes in cough index (coughs per hour) in participants who had adequate cough monitoring performed, Pearson’s correlation = 0.226 (p = 0.325). There was also no significant correlation between the change in cough severity VAS scores or Leicester cough questionnaire (LCQ) scores and change in DCI, Pearson’s correlation = 0.199 (p = 0.362) and 0.174 (p = 0.450), respectively. There was, however, a significant but weak association between the change in Hull airway reflux questionnaire (HARQ) score and the change in DCI measurement, with a Pearson’s correlation of −0.448 (p = 0.032). The scatter plots examining these relationships can be seen in Figures 5–7.

Scatter plot examining the relationship between change in cough index (number of coughs per hour) and DCI between baseline and follow-up.

Scatter plot examining the relationship between change in HARQ scores and DCI between baseline and follow-up.

Scatter plot examining the relationship between change in LCQ scores and DCI between baseline and follow-up.
Discussion
The results of this trial show that a definitive large-scale randomised controlled trial to assess the effectiveness of azithromycin on oesophageal function and symptom burden in participants with chronic respiratory disease is feasible and acceptable to participants. The findings of numerical improvements of DCI measurements in this group of participants after 1 month of azithromycin treatment suggests that investigating this as a primary endpoint in a large-scale trial is warranted. This is further evidenced by the fact that almost half of participants had an improvement of their DCI measurement ⩾30% at follow-up. We believe that this signifies a clinically important rise as previous data has shown an approximate 30% intra-individual variability between two separate HROM recordings. 21
In this study, all the pre-specified stop-go criteria for feasibility outcomes were achieved, including recruitment rate and participant retention. In addition, all pertinent data quality targets were met. The only exception to this was the completion rate of the paper cough VAS diary by participants, which we believe is likely attributable to the cumbersome nature of the document. In a large-scale trial, we believe that a digital cough VAS diary would be much more user-friendly and would lead to a lower rate of data attrition.
In the conception phase of the study, one of the chief concerns pertaining to the protocol was the willingness of participants to undergo two HROM investigations. Although it is not a painful procedure, it has risks such as bleeding and discomfort 22 and it is plausible that participants may not wish to undergo an investigation twice as part of a research study. However, the results from our invitation experience survey showed that 78% (n = 18) of participants selected ‘strongly disagree’ or ‘disagree’ responses when asked whether the thought of having an invasive investigated discouraged them from taking part in the study. Furthermore, only two participants (that did not withdraw from the study) who were able to have their initial HROM study declined to have a further test at follow-up. This indicates that the study protocol for measuring oesophageal function was acceptable to participants and would be feasible in a larger study.
Of the candidate primary outcomes that were proposed, it appears that the change in DCI demonstrated the only numerical improvement in terms of oesophageal motility. The use of DCI is also concordant with the proposed mechanism by which azithromycin improves oesophageal motility. Moreover, there was a significant negative correlation between DCI and HARQ scores. This suggests that improved oesophageal contractility may be associated with reduced cough-related symptom burden, further suggesting that the relationship between DCI and objective and subjective measures of cough should be used as a potential primary endpoint in future trials. Azithromycin has been shown in vitro to be a potent motilin-receptor agonist 23 and such receptors are known to be expressed in the oesophagus. However, given that the primary effect of motilin agonism, specifically with macrolide antibiotics, in the oesophagus is a rise in lower oesophageal sphincter pressure24 –26 it may be surprising that our results did not show a signal of any change in this manometric parameter. This may, however, be explained by the fact that 50% of the participants in our study were found to have a hiatus hernia. The presence of such has been shown to reduce the LOSP significantly due to anatomical displacement, impairing the sphincter’s ability to maintain its normal pressure. 27 This may preclude the measurement of LOSP being a meaningful clinical endpoint in a definitive trial. Alternatively, pre-specified subgroup analysis stratifying patients by the presence of a hiatus hernia may reveal the true effect of azithromycin on these measures of oesophageal function.
Motilin agonism is not the only proposed mechanism for improvement in gastrointestinal motility with azithromycin therapy. There is evidence to suggest that deglycosylated azithromycin, a metabolite of azithromycin, may enhance intestinal smooth muscle function by binding to the structural protein transgelin. 28 This protein is predominantly distributed in smooth muscle tissue and exerts its effect via a c-terminal calponin-like molecule (CLIK23) and positively charged amino acids, which bind to actin filaments in the smooth muscle.29 –31 Transgelin promotes the aggregation of G-actin to F-actin. This then in turn increases the length and strength of fibre bundles in intestinal smooth muscle cells, increasing their contractility strength. 32 In a study performed in healthy volunteers, the presence of deglycosylated azithromycin in the faeces was significantly associated with increased intestinal peristalsis. 28 However, this study did note that there was no significant association between deglycosylated azithromycin levels and contractility in the stomach; furthermore, they reported that deglycosylated azithromycin lacked any motilin-receptor agonist activity. Taken together, this suggests that differences in azithromycin metabolism between participants may result in differential effects on gastrointestinal motility. Some participants may experience lower gastrointestinal effects such as abdominal cramps, flatulence, and diarrhoea, whereas others may have increased gastric peristalsis, which may be beneficial for participants with reflux-associated chronic cough. Unfortunately, this particular study did not examine the effect of transgelin activation by deglycosated azithromycin in the oesophagus, which may be different from that of both the gastric and intestinal cells. Further work is required on the potential mechanistic effects of azithromycin on both motilin agonism and transgelin binding in participants with chronic refractory cough. In a future definitive trial, measurement of clinical biomarkers, such as serum motilin may provide interesting mechanistic insights into the effect of azithromycin on gastrointestinal motility.
The primary limitation of this study is the lack of a control group. As all participants had received azithromycin, we are unable to demonstrate any signal of improvement in manometric parameters that may be attributable to the azithromycin therapy. This also does not allow us to perform a formal sample size calculation for a larger randomised controlled trial. However, as previously stated, it would be logical to use the cut-off of at least 30% improvement in DCI as the minimum clinically important difference (MCID) between groups. Moreover, the lack of patient, clinician, and HROM operator blinding to the treatment and the phase of treatment, that is, HROM before or after azithromycin treatment, leaves this study open to researcher bias. However, given that this was a feasibility study, where the primary endpoint was the acceptability of the trial processes to the participants and clinicians, the lack of blinding and randomisation is of lesser concern.
Conclusion
A definitive randomised controlled trial to assess the efficacy of azithromycin on oesophageal function, as measured by repeated HROM testing, and its relationship to cough in chronic respiratory disease is feasible and acceptable to participants. The primary outcome of this trial should be the relationship between oesophageal contractility, measured by DCI, and objective cough counts measured by continuous cough monitoring. Such future trials should utilise mixed methodologies to understand the effect of azithromycin from the patient’s perspective, as well as its in/ ex vivo effect on motilin agonism and transgelin activity.
Supplemental Material
sj-doc-1-tar-10.1177_17534666251360065 – Supplemental material for Azithromycin and oesophageal motility in chronic respiratory disease: a feasibility study
Supplemental material, sj-doc-1-tar-10.1177_17534666251360065 for Azithromycin and oesophageal motility in chronic respiratory disease: a feasibility study by Dominic L. Sykes, Kayleigh Brindle, Rohan Menon, Simon P. Hart, Jennifer Nielsen, Warren Jackson, John Gallagher, Elisabeth Kirton, Mengru Zhang, Alyn H. Morice and Michael G. Crooks in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
