Abstract
Background:
Chronic obstructive pulmonary disease (COPD) is a progressive lung disease marked by airway inflammation and obstruction. Ensifentrine is a novel inhaled PDE3 and PDE4 inhibitor with both bronchodilator and anti-inflammatory effects.
Objectives:
Comprehensively review the available evidence on ensifentrine and its potential role in COPD management.
Design:
Systematic review and meta-analysis with trial sequential analysis of randomized clinical trials.
Data sources and methods:
We systematically searched PubMed, Scopus, ScienceDirect, Cochrane Library, and Medline for clinical trials published between 2018 and August 2024 that evaluated the safety and efficacy of ensifentrine in patients with COPD. We assessed study quality using the RoB 2 tool and conducted the meta-analysis with the “meta” package in R (version 4.3.2), using the mean difference with a 95% confidence interval to evaluate changes in outcomes.
Results:
Five studies met the predefined inclusion criteria with 2519 participants. At week 12, the pooled analysis indicated that forced expiratory volume in 1 s (FEV1) and trough FEV1 were significantly increased in the ensifentrine group (mean difference (MD): 91.32; 95% CI: 69.63 to 113.01) and (MD: 40.90; 95% CI: 19.65 to 62.15), respectively. At week 24, the pooled analysis indicated that the evaluating respiratory symptoms total score was significantly decreased in the ensifentrine group (MD: −0.81; 95% CI: −1.36 to −0.27), transition dyspnea index score was significantly increased in the ensifentrine group (MD: 0.96; 95% CI: 0.62 to 1.29), no significant difference was observed in rescue medication use (MD: −0.30; 95% CI: −0.60 to 0.00), and no significant difference was observed in St. George’s Respiratory Questionnaire total score (MD: −1.46; 95% CI: −3.22 to 0.30). Based on subgroup analysis, higher doses were associated with more favorable results.
Conclusion:
In conclusion, owing to its dual effects, ensifentrine has a significant impact on improving pulmonary function and quality of life with minimal side effects. Promising results are expected if implied by synergizing with other drugs, however, more studies are needed to study the long-term effect on disease progression.
Trial registration:
The study protocol was published via PROSPERO: International Prospective Register of Systematic Reviews (#CRD42024570799).
Plain language summary
Ensifentrine for COPD: A New Treatment That Improves Lung Function and Symptoms Background: Chronic Obstructive Pulmonary Disease (COPD) is a lung condition that makes breathing difficult. Current treatments help, but many patients still struggle with symptoms. Ensifentrine is a new inhaled medication that works by targeting two enzymes (PDE3 and PDE4) to reduce inflammation and open airways. Study Goal: This review analyzed five clinical trials (2,519 patients) to see if ensifentrine improves lung function and quality of life in COPD patients. Key Findings: Lung Function: Ensifentrine significantly improved FEV1 (a measure of lung capacity) by 91 mL at 12 weeks and morning trough FEV1 by 41 mL. Symptoms: It reduced respiratory symptoms (E-RS score) by 0.81 points and improved breathlessness (TDI score) by 0.96 points at 24 weeks. Quality of Life: Higher doses (1.5–3 mg) improved St. George’s Respiratory Questionnaire (SGRQ) scores early (4 weeks), but effects were smaller by 24 weeks. Safety: No major side effects were reported compared to placebo. Limitations: It did not reduce the need for rescue inhalers, suggesting it’s better for long-term management than quick relief. Conclusion: Ensifentrine is a promising add-on treatment for COPD, improving lung function and symptoms with minimal side effects. Higher doses and longer use showed better results. Future studies should explore its long-term benefits and role in COPD care guidelines.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a chronic respiratory condition characterized by airway inflammation and obstruction, leading to significant morbidity and mortality worldwide.1 –3 While these diseases share some common features, they exhibit distinct clinical presentations. COPD, which includes emphysema, is a progressive disease often exacerbated by environmental factors. 4 It imposes a substantial burden on patients and healthcare systems, necessitating ongoing research and the development of effective therapeutic strategies.
Phosphodiesterases (PDEs) are enzymes that control many bodily functions by regulating the amount of cyclic nucleotide signaling molecules inside cells. The levels of cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate in airway smooth muscles are controlled by PDE3. Inhibiting PDE3 causes airway smooth muscle to relax.5 –7 PDE4 controls cAMP levels and plays an important role in inflammatory cell activation. Therefore, PDE4 inhibition has anti-inflammatory effects.8 –10 These data indicate that the simultaneous inhibition of PDE3 and PDE4 may have additional or combined benefits in terms of reducing inflammation and expanding the airways. This is because of the presence of both PDE isoforms in inflammatory cells and airway smooth muscles. 11
Ensifentrine is a novel inhaled medication that inhibits both PDE3 and PDE4. Previous studies have shown that it effectively dilates the airways and reduces inflammation in both healthy individuals and patients with COPD.12 –15 Recently approved by the FDA, ensifentrine offers a potentially new approach for managing these respiratory conditions. Prior to this study, patients with COPD who received medication for a maximum of 6 days provided lung function and safety data. However, no symptom assessments were performed during this period. 15 The goal of ensifentrine inhalation is to reduce the systemic side effects associated with oral PDE inhibitor administration. 16
This systematic review aimed to comprehensively evaluate the available evidence on ensifentrine, focusing on its pharmacological properties, clinical outcomes, and potential role in the management of these chronic respiratory diseases. We synthesized data from multiple studies. We aim to provide a comprehensive understanding of the therapeutic potential of ensifentrine and its role in the evolving landscape of COPD treatment.
Methods
Search strategy and eligibility criteria
This systematic review adhered to PRISMA guidelines. 17 All stages were performed following the Cochrane Handbook for Systematic Reviews of Interventions, version 6.3. 18 The protocol of this systematic review and meta-analysis was registered with the International Prospective Register of Systematic Reviews (PROSPERO) in August 2024 with registration number CRD42024570799.
We conducted a literature search for relevant studies published between 2018 and 2023 in PubMed, Scopus, ScienceDirect, and Cochrane Library databases using a combination of search keywords and relevant medical subject headings (MeSH terms) related to COPD and nebulized ensifentrine as (Ensifentrine OR RPL554 OR LS193855) AND (COPD OR “chronic obstructive pulmonary disease” OR emphysema OR “chronic bronchitis”). In addition, we reviewed the reference lists of the selected articles to ensure a comprehensive search. Two independent reviewers conducted a literature search and selected studies based on the following inclusion criteria: (1) Patients aged ⩾18 years diagnosed with COPD, emphysema, or chronic bronchitis; (2) Studies evaluating multiple doses of Ensifentrine administered once or twice daily in the target population, compared to traditional treatments. The exclusion criteria were as follows: (1) History of asthma or other pulmonary disease. (2) Patients who had recently received COPD therapy. (3) Reviews. (4) Case reports. (5) Conference abstracts.
Study selection and data extraction
Two authors independently evaluated the titles and abstracts of all the identified articles against the predetermined inclusion criteria. Full-text reviews of potentially relevant studies were independently conducted to confirm their eligibility. The final selection of studies was reached through consensus among all authors. The same reviewers systematically extracted the data using a standardized form. Extracted information included baseline characteristics (mean forced expiratory volume in 1 s (FEV1) in mL and St. George’s Respiratory Questionnaire (SGRQ) total score), efficacy outcomes (spirometry at weeks 1, 2, 3, and 4), and safety endpoints (adverse events). For continuous outcomes with missing data, a multiple imputation approach was employed, under the assumption that the data were missing at random.
Quality of studies and risk of bias assessment
Two authors independently assessed the quality of the included studies. We assessed the risk of bias in the randomized clinical trials included in our study using the RoB2 tool.18,19 This tool evaluates five key domains of bias: randomization process, deviations from intended interventions, missing outcome data, outcome measurements, and selection of reported results. Each domain was answered, and an overall judgment was provided for the trial. The risk of bias was categorized as low, concerning, or high.
Statistical analysis
The meta-analysis was conducted using the “meta” package on R version 4.3.2. 20 The mean difference with 95% confidence intervals was used to assess the difference in the mean change between the ensifentrine and placebo groups. A fixed-effects model utilizing the Mantel-Haenszel method was used when heterogeneity was absent (I2 < 50%, p > 0.10). However, if heterogeneity did arise (I2 ⩾ 50%, p < 0.10), a random-effects model based on the DerSimonian and Laird method was employed. Sensitivity and publication bias were assessed using leave-one-out plots and Egger’s test, respectively. Given that the studies employed different samples for different doses, we treated each dose as an independent study with subsequent subgrouping based on dose and weeks until follow-up to allow a more accurate interpretation of the results. We further strengthened our conclusions by conducting trial sequential analysis (TSA) using Copenhagen Trial Unit TSA analysis software. 21 For TSA, the alpha error rate was set at 5%, while the power of the study was set at 80%. Furthermore, the required information size was calculated using two different methods, the empirical method and the low risk of bias method, both of which utilized the pooled results from our meta-analysis. The statistical significance for all meta-analytic results was set at p < 0.05.
Results
Search results
Our search strategy identified 249 studies from the searched databases. After removing duplicates, 175 studies remained, which were then screened by title and abstract. This screening led to the exclusion of 158 studies. The remaining 17 articles were further evaluated by the same authors, resulting in 5 studies that met our inclusion eligibility criteria12,13,22 –24 shown in Figure 1.

The search and selection process in PRISMA flowchart.
Characteristics of included studies
Five studies were ultimately included in this review, comprising a total of 2519 participants, with 1780 in the intervention group and 739 in the control subjects. The publication dates ranged from 2013 to 2023. Three studies reported a mean participant age of over 40 years. One study was conducted in the United States, one in the United Kingdom, one in Bulgaria and the Czech Republic, and two were international collaborations. The follow-up duration varied across studies, ranging from 12 h to 48 weeks. The baseline characteristics of all included studies are summarized in Table 1. Additional outcomes of the included studies are shown in Table S1.
Characteristics of included studies.
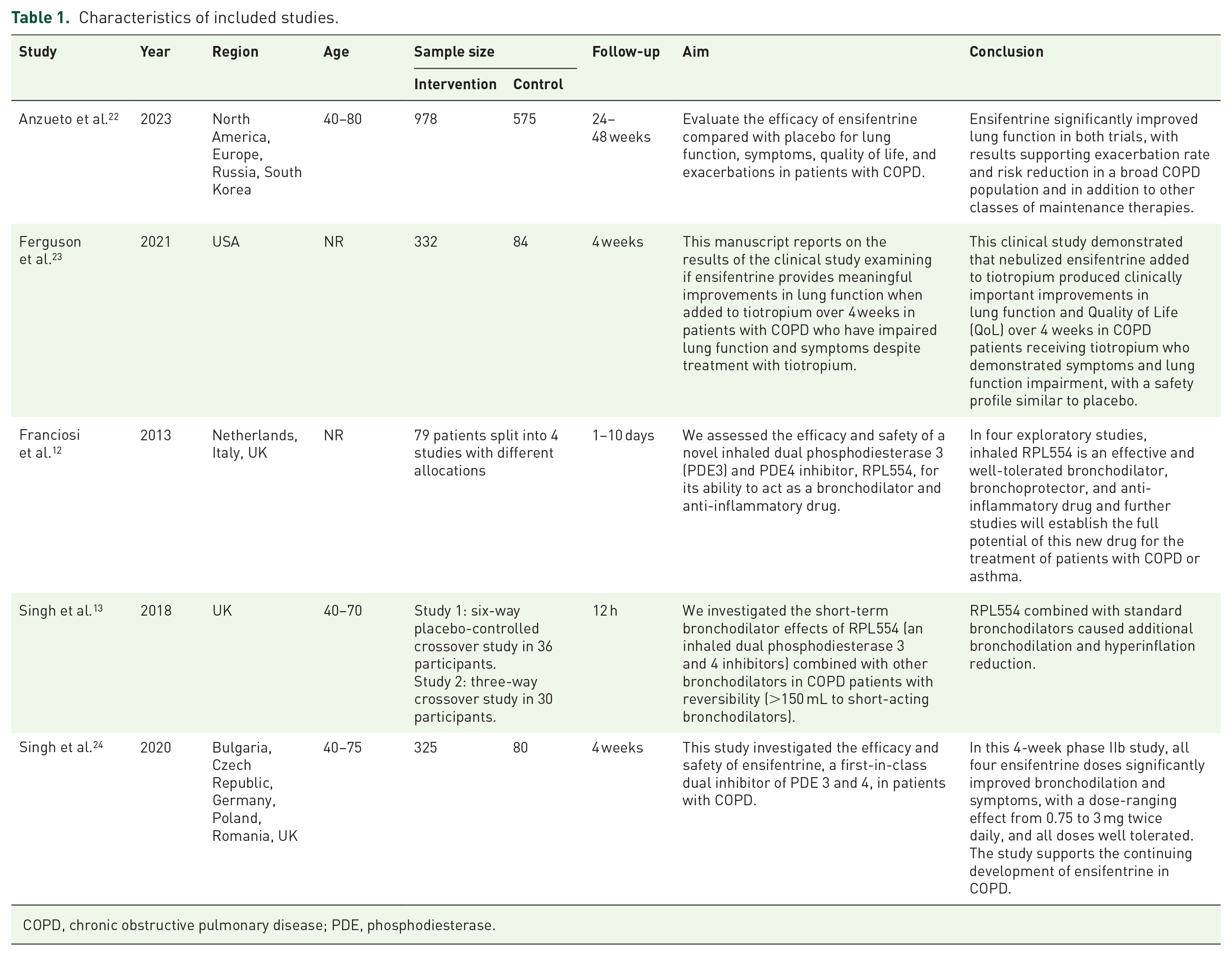
COPD, chronic obstructive pulmonary disease; PDE, phosphodiesterase.
Quality assessment
We conducted a quality assessment of five clinical trials. Using the ROB-2 Quality Assessment Tool as a reference, four studies were classified as good quality, while one study was rated as moderate. The latter received this rating due to a lack of justification for the randomization process, missing data, and some bias in the measurement of the outcome. Further details of the quality assessment are provided in Figure 2 and Figure S1.

Risk of bias assessment traffic light plot.
Outcomes
Evaluating respiratory symptoms total score
Pooled data from the two studies were analyzed to assess the effects of varying ensifentrine doses. A significant difference was found between patients receiving 3 mg of ensifentrine and the control subject, with a mean difference (MD) of −0.81 (95% CI: −1.31 to −0.31), forest plot shown in Figure 3. A funnel plot is shown in Figure S2.
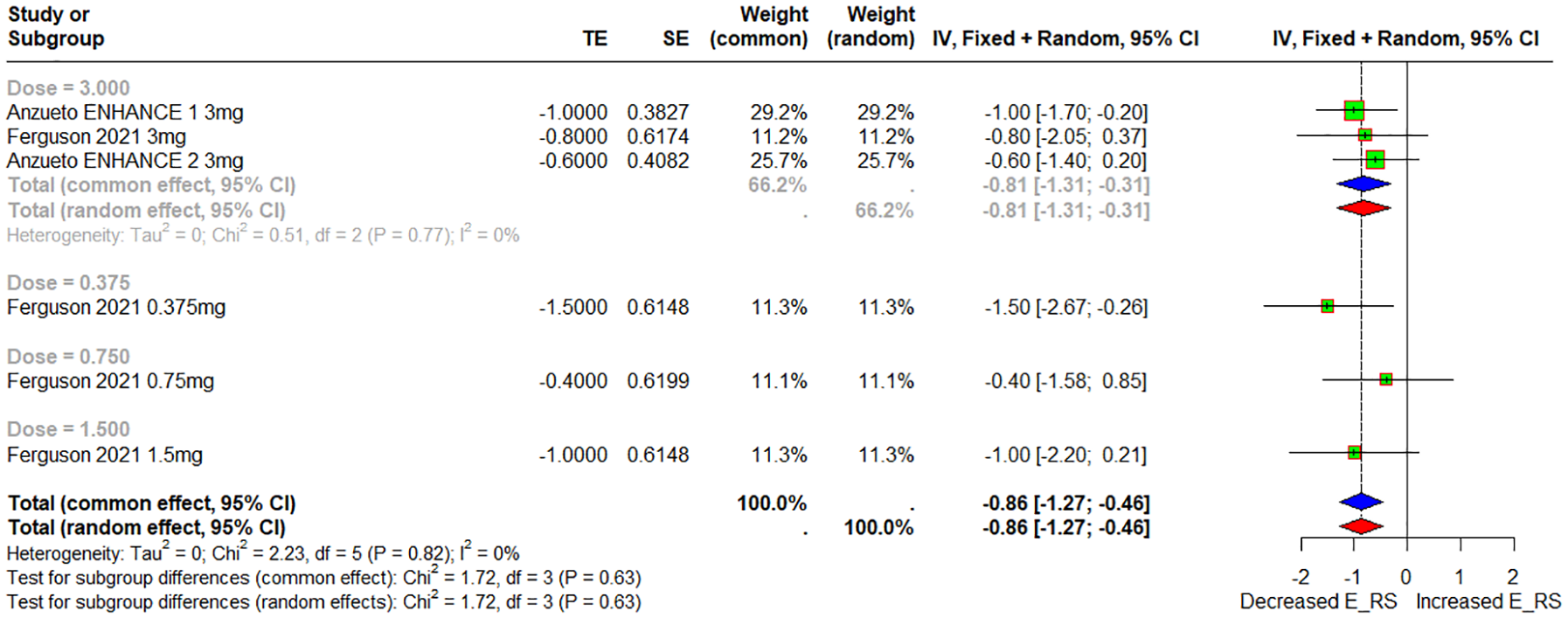
Fores plot of E-RS total score with dose subgroups.
When evaluating outcomes over time, a significant difference was noted after 24 weeks in patients receiving 3 mg of ensifentrine compared to control subjects, with an MD of −0.81 (95% CI: −1.36 to −0.27). After 4 weeks, a similar analysis of different doses of ensifentrine showed a significant MD of −0.93 (95% CI: −1.53 to −0.32) when compared to control subjects, forest plot shown in Figure S3.
Leave-on-out-Analysis (LOOA) plot showed a consistent pattern of mean difference, and the exclusion of an individual study did not notably alter the pooled mean difference as all studies produced a similar effect (MD: −0.86, 95% −1.27 to −0.46, p-value < 0.01, I2 = 0). The LOOA plot is shown in Figure S4.
Following TSA, the RIS for the evaluating respiratory symptoms (E-RS) total score was 2127 patients. The Z-curve crossed the upper monitoring boundary, indicating a statistically significant favorable effect of ensifentrine, although the required sample size was not reached. Given that the Z-curve crosses the monitoring boundary, we can conclude that the results of our meta-analysis are robust and conclusive. TSA Low-risk of bias (LRB) and empirical plots are shown in Figures S5 and S6.
FEV1 change
Pooled data from two studies were analyzed to assess the effects of varying ensifentrine doses on FEV1. In the comparison between patients receiving 3 mg of ensifentrine and control subjects, a significant improvement in FEV1 was observed, with an MD of 90.92 mL (95% CI: 70.27 to 111.57), forest plot shown in Figure 4. A funnel plot is shown in Figure S7.
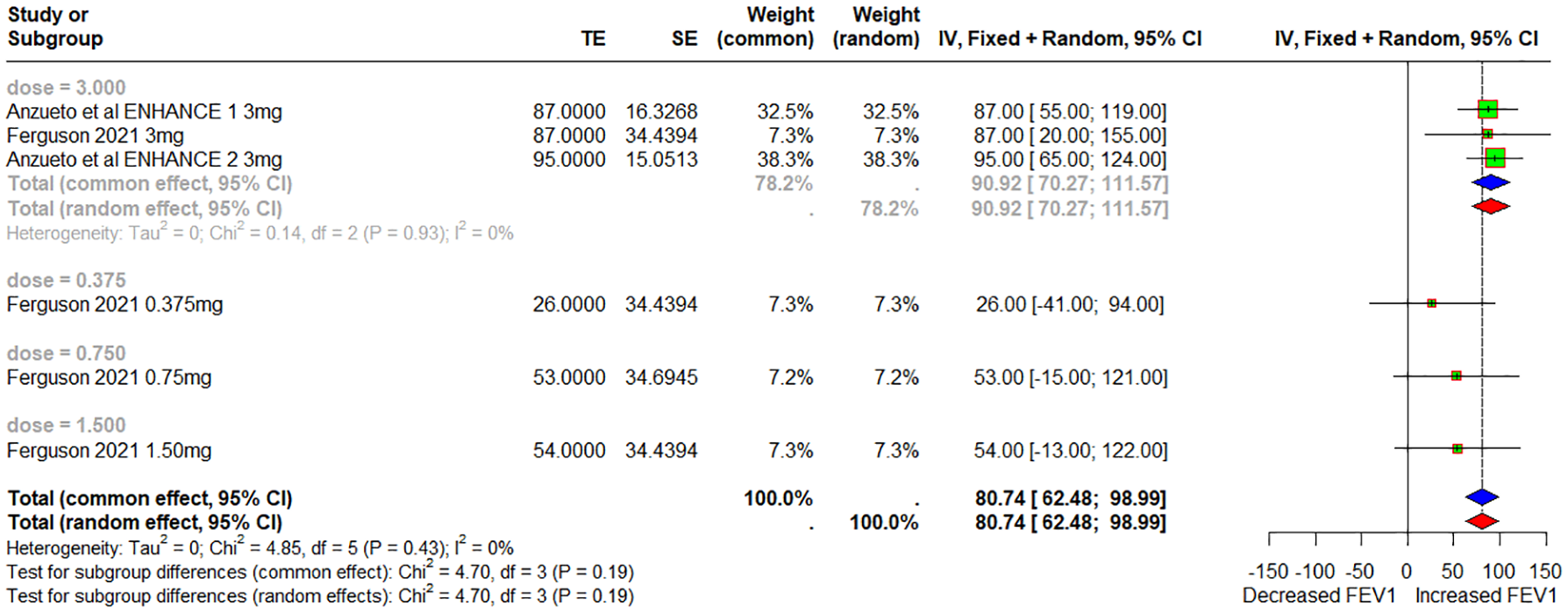
Forest plot of FEV1 change with dose subgroups.
At 12 weeks, patients receiving 3 mg ensifentrine also showed a significant increase in FEV1 compared to control subjects, with an MD of 91.32 mL (95% CI: 69.63 to 113.01). After 4 weeks, a comparison of different doses of ensifentrine revealed a significant FEV1 improvement, with an MD of 55.01 (95% CI: 21.2 to 88.82) when compared to control subjects, forest plot shown in Figure S8.
LOOA plot showed a consistent pattern of mean difference and the exclusion of an individual study didn’t notably alter the pooled mean difference as all studies produced a similar effect (MD: 80.74, 95% 62.48, 98.99, p-value < 0.01, I2 = 0). LOOA plot is shown in Figure S9.
TSA indicated that an RIS of 224 patients was necessary to ensure low bias and reliable results. This threshold was exceeded by our cohort, with the Z-curve crossing both the lower conventional and monitoring boundaries, confirming the robustness and conclusiveness of our meta-analysis without the need for further trials. TSA LRB and empirical plots are shown in Figures S10 and S11.
Morning trough FEV1
Pooled data from two studies were analyzed to assess the effects of varying ensifentrine doses on morning trough FEV1. Patients receiving 3 mg ensifentrine demonstrated a significant improvement in morning trough FEV1 compared to control subjects, with an MD of 40.90 mL (95% CI: 19.65 to 62.15), forest plot shown in Figure 5. Funnel plot is shown in Figure S12.
Forest plot of morning through FEV1 with dose subgroups.
At 12 weeks, patients receiving 3 mg ensifentrine also showed a significant increase in the morning through FEV1, with an MD of 42.65 mL (95% CI: 20.10 to 65.21) compared to control subjects. However, after 4 weeks, a comparison of different doses of ensifentrine and control subjects revealed no significant difference, with an MD of 6.07 (95% CI: −25.07 to 37.89), forest plot shown in Figure S13.
LOOA plot showed a consistent pattern of mean difference and the exclusion of an individual study didn’t notably alter the pooled mean difference as all studies produced a similar effect (MD: 30.42, 95% 12.02 to 48.82, p-value < 0.01, I2 = 0). The LOOA plot is shown in Figure S14.
TSA indicated that a total of 3490 patients were required to draw conclusive results regarding morning trough FEV1. The Z-curve surpassed the lower conventional boundary and approached the lower monitoring boundary, suggesting a potentially beneficial effect of ensifentrine on morning trough FEV1. TSA LRB and empirical plots are shown in Figures S15 and S16.
TDI score
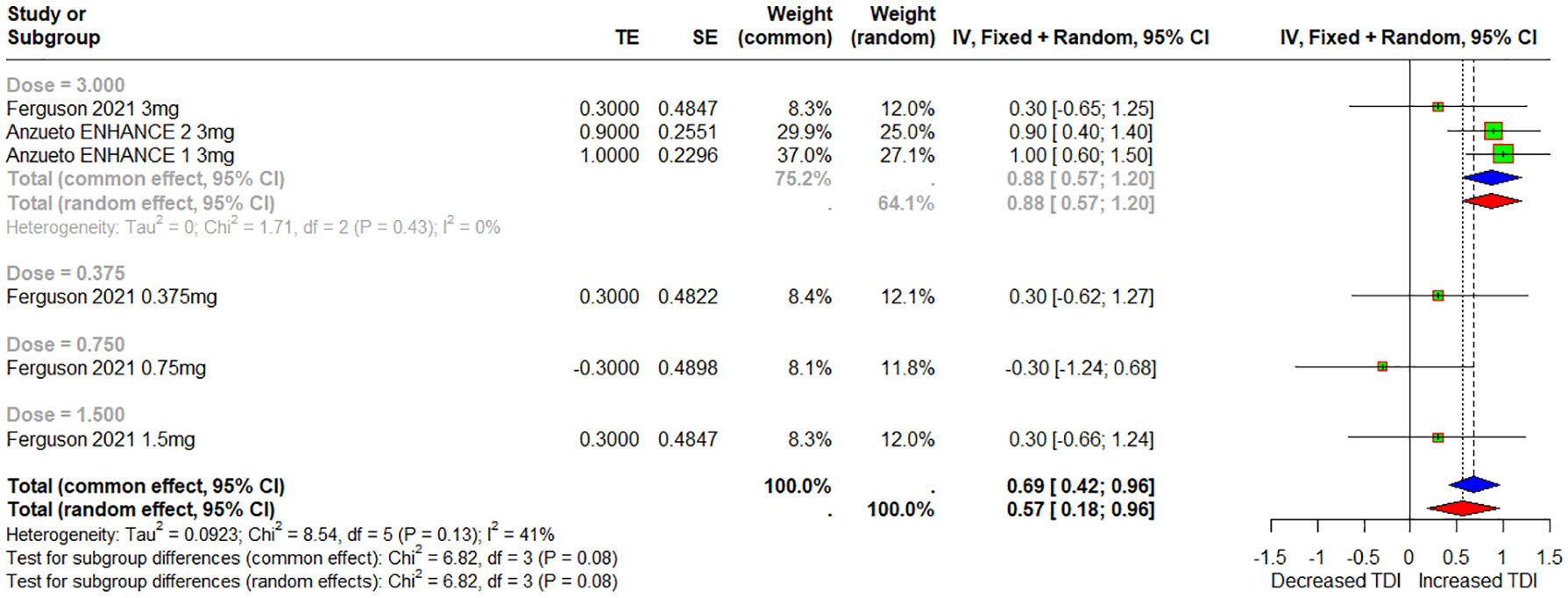
Pooled data from the two studies were analyzed to assess the effects of varying ensifentrine doses on transition dyspnea index (TDI) scores. Patients receiving 3 mg of ensifentrine showed a significant improvement in TDI score compared to control subjects, with an MD of 0.88 (95% CI: 0.57 to 1.20), forest plot shown in Figure 6.

Forest plot of TDI score with dose subgroups.
At 24 weeks, a significant increase in TDI score was observed in patients receiving 3 mg of ensifentrine compared to control subjects, with an MD of 0.96 (95% CI: 0.62 to 1.29). However, after 4 weeks, comparisons of different doses of ensifentrine and control subjects revealed no significant difference, with an MD of 0.15 (95% CI: −0.32 to 0.63), forest plot shown in Figure S17.
LOOA plot showed a consistent pattern of mean difference and the exclusion of an individual study didn’t notably alter the pooled mean difference as all studies produced a similar effect (MD: 0.69, 95% 0.42, 0.96, p-value < 0.01, I2 = 42). LOOA plot is shown in Figure S18.
TSA indicated that 4189 patients were required to ensure robust evidence. Our cohort included 1794 patients, with the Z-curve crossing the conventional line of significance and approaching the lower monitoring boundary, suggesting a beneficial effect of ensifentrine on the TDI score. TSA LRB and empirical plots are shown in Figures S19 and S20.
Rescue medication use
Pooled data from three studies were analyzed to assess the effects of varying the ensifentrine dose. No significant differences were observed across different doses when compared to control subjects, with an MD of −0.25 (95% CI: −0.52 to 0.02) for 3 mg, MD of −0.01 (95% CI: −0.39 to 0.36) for 0.75 mg, and MD of −0.46 (95% CI: −0.93 to 0.01) for 1.5 mg, forest plot shown in Figure 7. The funnel plot is shown in Figure S21.

Forest plot of rescue medication use with dose subgroups.
At 24 weeks, no significant difference was observed between patients receiving 3 mg of ensifentrine and control subjects, with an MD of −0.30 (95% CI: −0.60 to 0.00). Similarly, after 4 weeks, no significant difference was found when comparing different doses of ensifentrine to control subjects, with an MD of −0.17 (95% CI: −0.47 to 0.12), forest plot shown in Figure S22.
LOOA plot showed a consistent pattern of mean difference and the exclusion of an individual study didn’t notably alter the pooled mean difference as all studies produced a similar effect (MD: −0.19, 95% CI −0.30 to −0.07, p-value < 0.01, I2 = 73). The LOOA plot is shown in Figure S23.
TSA indicated that 2500 patients were required to validate the pooled results. The Z-curve did not cross either the conventional or monitoring boundaries but intersected the lower futility boundary, indicating a lack of effect of ensifentrine on rescue medication usage. This aligns with our findings and suggests that further clinical trials are unnecessary. The TSA empirical plot is shown in Figure S24.
SGRQ total score
Pooled data from three studies were analyzed to assess the effects of various ensifentrine doses on the SGRQ total score. Patients receiving 3 and 1.5 mg of ensifentrine demonstrated significant improvements in SGRQ total scores, with MD of −2.00 (95% CI: −3.37 to −0.63) and −3.53 (95% CI: −6.14 to −0.93), respectively. In contrast, no significant difference was observed at the 0.75 mg dose, with an MD of −2.49 (95% CI: −5.09 to 0.10), forest plot shown in Figure 8. A funnel plot is shown in Figure S25.

Forest plot of SGRQ total score with dose subgroups.
At the 24-week mark, the comparison between patients receiving 3 mg of ensifentrine and control subjects revealed no significant difference, with an MD of −1.46 (95% CI: −3.22 to 0.30). However, after 4 weeks, a significant decrease in the SGRQ total score was observed when comparing different doses of ensifentrine to control subjects, yielding an MD of −3.18 (95% CI: −4.48 to −1.89), forest plot is shown in Figure S26.
LOOA plot showed a consistent pattern of mean difference and the exclusion of an individual study didn’t notably alter the pooled mean difference as all studies produced a similar effect (MD: −2.45, 95% CI: −3.42 to −1.47, p-value < 0.01, I2 = 0). The LOOA plot is shown in Figure S27.
TSA indicated that a sample size of 2200 was necessary to ensure the validity of our meta-analysis results. Our cohort exceeded this requirement, with the Z-curve intersecting both the conventional and monitoring boundaries, confirming the effect of ensifentrine on the SGRQ scores. Thus, our analysis is definitive, and no further trials are necessary to confirm this association. TSA LRB and empirical plots are shown in Figures S28 and S29.
Adverse events
Pooled data from four studies were analyzed to assess the overall incidence of treatment-emergent adverse events (TEAEs) across different doses of ensifentrine. Patients receiving 0.375 and 0.75 mg showed no statistically significant difference between the two groups, with a risk ratio (RR) of 0.85 (95% CI: 0.51 to 1.42). Similarly, the 1.5 mg dose yielded a nonsignificant RR of 0.95 (95% CI: 0.64 to 1.39), and the 3 mg dose had an RR of 1.06 (95% CI: 0.89 to 1.27).
The 5 and 6 mg doses were each reported in a single study, with RRs of 1.04 (95% CI: 0.57 to 1.89) and 0.79 (95% CI: 0.33 to 1.90), respectively. The overall pooled incidence across all doses resulted in an RR of 1.00 (95% CI: 0.87 to 1.15), with no heterogeneity (I2 = 0%). The corresponding forest plot is shown in Figure 9. The funnel plot showed a symmetrical distribution of studies suggesting no publication bias.

Forest plot of any treatment-emergent adverse events in ensifentrine and placebo groups.
In addition, pooled analyses of adverse event risk ratios for the 3 mg dose of ensifentrine showed no significant associations with hypertension, diarrhea, or serious adverse events (SAEs). The risk ratios were 2.22 (95% CI: 0.90 to 5.49, I2 = 0%, p = 0.7944) for hypertension, 1.53 (95% CI: 0.49 to 4.73, I2 = 0%, p = 0.5041) for diarrhea, and 1.01 (95% CI: 0.68 to 1.49, I2 = 0%, p = 0.7719) for SAEs. The forest plots for diarrhea, hypertension, and SAEs are shown in Figures S30–S32. Funnel plots for diarrhea, hypertension, and SAEs are shown in Figures S33–S35.
Discussion
Relying on available pharmacotherapies for COPD without evaluating their impact on patients’ quality of life, may contribute to increased mortality. 25 An analytical and evidence-assisted measurement of the novel therapeutic intervention ensifentrine to determine its effect on symptomatology, quality of life, frequency, and severity of exacerbation is desirable. This commenced by utilizing the available data to date to reach the primary outcomes through the current meta-analysis. According to our findings, ensifentrine showed time-dependent improvement in FEV1, morning trough FEV1, E-RS, and TDI scores at the 3 mg dose without reductions in rescue medications and even showed an increase in doses compared to the placebo. However, the morning trough FEV1 and TDI did not improve at 4 weeks, even at different doses. Quality of life improved at higher doses after 4 weeks, with an insignificant reduction (SGQR) at 24 weeks.
Unlike synergistic β2 agonists and steroids, improved bronchiolar structural remodeling and harnessing inflammation with isolated PDE 3 and 4 inhibition may be cumbersome without holding a superior position that emphasizes PD3/4 blockade following a single drug. 26 With inflammatory consideration, lowering the lipopolysaccharide-induced cytokine concentration with this synergistic inhibition was sustained even under oxidative stress, in contrast to steroids. 27 In addition, the additive effect fostered a significant anti-inflammatory role on CD4+ and CD8+ T lymphocytes, IL-1β, VCAM-1, and eosinophil adhesion, which are key regulators involved in the exacerbation of the inflammatory cascade. 28 Apart from this, relaxation of inherent tone and halting allergen or LTC4-induced contraction may provide a bronchodilatory benefit with such synergism, which could be potentiated with other bronchodilators. 11 Recent advancements have suggested its involvement in the practical reversal of paralyzed mucociliary escalators, further amplifying its efficacy in quality care among patients with obstructive patterns. 29
A randomized controlled double-blind crossover study yielded an improved effect of ensifentrine on FEV1 among patients with asthma, which is consistent with our study results, 14 which ultimately reduced residual volume and bronchiolar resistance. The population under study was asthmatic, and the maximum outcome was evaluated for only 12 h, in contrast to our study, where the effect of ensifentrine was calibrated for 4 weeks. Another aspect noted in that study was the reduced effect of ensifentrine compared to salbutamol at the same dose, except for the 24 mg dose of ensifentrine. Moreover, this study must be extended to weeks or months to achieve comparable outcomes in patients with asthma. In addition to the beneficial effects of ensifentrine monotherapy, its combined effect with salbutamol (p = 0.004) and ipratropium (p = 0.005) further improved FEV1, as demonstrated in another analytical study. An improvement of 384 and 385 mL in FEV1 was calculated from a baseline of 302 mL after combined treatment with salbutamol and ipratropium, respectively. 30
We found improved morning trough FEV1 at 3 mg with statistical significance (MD 40.90, 95% CI: 19.65 to 62.15) without any improvement at lower doses. 0.3, 0.7, and 1.5 mg, which is dose-dependent. Improvement in morning trough FEV1 for ensifentrine versus placebo was also found to be dose-dependent, increasing from 80 mL (p = 0.01) to 110 (0.006) mL when the dose went from 1000 to 3000 µg. 31 Statistically insignificant outcomes were found after 4 weeks, even at a higher dose, which might be due to the lack of plasma levels during this period. However, the duration was fixed for 1 week in that study, making a direct comparison of dose with duration impossible with our findings.
Roflumilast, a PDE-4 inhibitor, was postulated to produce a significant improvement in FEV1 (MD: 51.53 mL, 95% CI: 43.17 to 59.90) and was found to be coherent with our primary endpoints (MD: 90.92 mL, 95% CI: 70.27 to 111.57). Existing with improved FEV1 (MD: 0.08 mL, 95% CI: 0.06 to 0.09, p < 0.00001), the homology of such adverse events exhibited with the addition of theophylline to inhaled bronchodilators portrays a positive influence of ensifentrine over other PDE inhibitors. 32 Ensifentrine can be recommended for cardiac patients as a safe cardiac profile without any arrhythmogenic risk, as reported in a recent study. Even with an increased dose of 9 mg, these findings characterize its future implications over other PDE inhibitors in patients with COPD and cardiac comorbidities. 33 Headache, cough, and dyspnea were found in up to 5%, 4.2%, and 1.8% of the ensifentrine group, respectively, compared with placebo reports of 2.5%, 1.2%, and 3.1%, suggesting similar effects without any fatal events, thereby providing validated data with a higher safety range. 34
One study explored its direct role in bronchodilation with a stepwise dose increase. Weighing its outcome (7.4 ± 1.9) at 20 µg/kg, the improved bronchodilation was noted at 55.2 ± 6.2, 65.1 ± 5.3 at 40 and 80 µg/kg ensifentrine, respectively. 35 Relative to this, the dose-dependent response from 0.75 mg (MD: −0.01, 95% CI: −0.39 to 0.36) to 3 mg (MD: −0.25, 95% CI −0.52 to 0.02) toward using rescue medication was statistically insignificant between ensifentrine and placebo in the current context, and this serves as an indirect measure of its anti-inflammatory and bronchodilatory effects on exacerbation.
The ensifentrine effect is assessed clinically based on E-RS scoring divided into breathlessness (>−1), cough/sputum (−0.7), and chest tightness (−0.7). 36 Concerning the forest plot (refer to the forest plots of ERS of weeks 4 and 24) evaluating variable drug doses’ effect on E-RS, an improved E-RS (>−1 to −2) with statistical significance was achieved after four weeks at 0.37 mg (MD: −1.50, 95% CI: −2.67 to −0.26) which increased (−1) for higher doses at 3 mg carrying statistical insignificance over this period. An in-depth understanding of this illustration strongly suggests the early bronchodilatory effect of ensifentrine, which could be inferred clinically as a more rapidly improved status of breathlessness among patients with COPD. 37 Although statistically insignificant, increased E-RS (−1 or <−1) at higher doses supports the delayed anti-inflammatory role of ensifentrine, which is clinically reflected in improved cough/sputum and chest tightness. In addition, when we extended therapy to 24 weeks with 3 mg ensifentrine, we found a sustained effect (−1) with statistical significance (MD: −1.0, 95% CI: −1.70 to −0.20) again supporting its delayed anti-inflammatory response presented as improved cough/sputum and chest tightness by the patients. In other words, a rapid bronchodilatory effect can be achieved within a shorter period, even at a low dose. However, persistent improvement in symptomatology was achieved only after prolonged therapy with a graded dose. In contrast to our study, Jones et al. suggested that improved breathlessness on the E-RS scale with long-acting anti-muscarinic aclidinium was achieved after 24 weeks, supporting the beneficial effect of ensifentrine and validating the dual role of early onset bronchodilatory and late anti-inflammatory effects. 38 As previously stated, ensifentrine was still found to have a more significant role than the PDE4 inhibitor roflumilast (roflumilast 0.805 vs placebo 0.927; rate ratio 0.868 (95% CI: 0.753–1.002), p = 0.0529), as postulated by Martinez et al. 39 Another study revealed insignificant improvements in COPD symptoms with roflumilast, arguing for the better efficacy of ensifentrine. 40
While we move toward the implications of COPD in patients’ lives, we aimed to assess its impact on daily activities and whole life using the TDI and SGRQ scales, respectively. 41 An improved TDI score was observed after reaching 3 mg ensifentrine statically (MD: 0.88, 95% CI: 0.57 to 1.20), as revealed in our analysis, with an insignificant impact at initial doses of 0.3, 0.7, and 1.5 mg. This could be explained by the achievement of a sustained response in the absence of all symptoms as scored by the E-RS, which reflects the effect model. When we plotted its role over time, a significant impact was observed after its use for a longer duration of 24 weeks (MD: 0.96, 95% CI: 0.62 to 1.29) in our case. This could be explained by the maintained anti-inflammatory response over a long period, with the ultimate marked reduction in exacerbations. 42 A persistent dyspnea-free period would improve daily activities, which could be translated as improved TDI. Furthermore, the early bronchodilatory effect does not have any role in patients’ daily performance owing to its narrow window of inflammation.43,44
In contrast to the TDI score, the SGRQ score has a broader impact on an individual’s life, and a longer duration is required to obtain accurate results. As viewed in various studies, the impact on the SGRQ was measured after a longer duration because a shorter duration may deviate from the actual results. Tashkin et al. forecasted the effects of ipratropium after a minimum of 6 months, which yielded a positive impact. 45 The improved quality of life at 4 weeks, but not at more extended periods (24 weeks) in our analysis, could be misleading, which might be due to its heightened perception resulting from better relief during the period of frequent exacerbations. 46 Reduced SGRQ at higher doses could be strengthened by inflammatory cessation, an ultimate long-term goal for qualitative life. 47 The wide confidence intervals in the statistically significant outcomes of our study must be interpreted with caution. However, the impact of breathlessness, cough, and sputum could not be distinguished for SGRQ, as in E-RS. 48
Our findings provide a guide for clinicians to consider the use of ensifentrine in symptomatic patients despite standard bronchodilator treatment. The statistically significant improvement in the E-RS score suggests the ability of the drug to reduce COPD-related symptoms by prescribing it to patients with persistent dyspnea despite treatment with Long-Acting Beta-Agonists (LABAs) and Long-Acting Muscarinic Antagonists (LAMAs). The observed increase in FEV1 indicates an enhancement in airflow and lung mechanics, which is beneficial for patients with frequent exacerbations or poor baseline lung function. However, its role as a first-line therapy should be considered in the current GOLD guidelines. 49
Although the current findings demonstrated a significant improvement in lung function and patient-reported outcomes with the use of ensifentrine, the impact of the drug on acute exacerbations of COPD (AECOPD) remains unclear because none of the included trials assessed exacerbation frequency or severity as a primary or secondary endpoint; thus, analysis of these outcomes was not possible. Knowing that exacerbations are a major driver of COPD morbidity, mortality, and healthcare costs, understanding the effects of ensifentrine and whether it can reduce AECOPD events is crucial for evaluating its full clinical utility. Future trials should prioritize long-term endpoints, including exacerbation rates, to determine the role of ensifentrine in preventing disease progression and hospitalization. Given the dual bronchodilator and anti-inflammatory mechanism of ensifentrine and its improved lung function and symptom control in stable COPD patients, evidence suggests its theoretical beneficial effects in AECOPD, and the drug may be considered as an add-on to inhaled corticosteroids or triple therapy. In addition, the significant improvement in the SGRQ total score indicated the possible benefit of the drug to those with COPD-related activity limitations, such as exercise intolerance. Despite improvements in lung function and symptom control, the drug did not reduce rescue medication use, suggesting that it had no effect on immediate symptoms. Clinicians should emphasize the use of short-acting bronchodilators for acute symptom management. Finally, ensifentrine demonstrated a favorable safety profile with no significant increase in TEAEs compared to placebo. The absence of a significant association with adverse events supports its use as an add-on therapy to standard care, with minimal risks.
Limitations
Of the limitations to be noted for future perspectives, generalizability must be addressed to obtain universal outcomes. Ensifentrine’s role in individual lives must be assessed from a broader perspective based on the SGRQ, for which the duration of studies must be extended to years. Insignificant results regarding rescue medication at different doses must be reconsidered by conducting studies among populations with larger sizes of moderate-to-severe COPD, accompanied by a higher equitable frequency of exacerbations. Adverse events with the specified doses mentioned in our study might not be appreciated, and these must be quantified at higher doses. In addition, the small number of included studies limits the strength of our findings, and further research is needed to evaluate the effect of ensifentrine on COPD. Finally, a key limitation in this review was the absence of data on AECOPD, as this outcome has not been reported in any of the included studies, and was unable to draw quantitative conclusions regarding the effect of ensifentrine on exacerbation frequency or severity, which is a critical part of the overall management of COPD.
Conclusion
In conclusion, owing to its dual effect, ensifentrine significantly improves lung function, symptom control, and quality of life in patients with COPD. The drug demonstrated dose- and time-dependent improvements in FEV1, morning through FEV1, E-RS, and TDI scores, showing both bronchodilator and anti- inflammatory effects. In contrast to the favorable safety profile of drugs, it did not decrease the need for rescue medication, indicating that it can be used as rescue medication and as an add-on maintenance therapy for current COPD treatment, rather than for acute symptom relief. Its role as an add-on therapy, particularly for patients with persistent dyspnea and exacerbation risk, should be further studied. Given its safety and efficacy, ensifentrine should be considered for integration into future COPD management guidelines, although long-term studies are required to confirm its effect on disease progression and whether ensifentrine can reduce the frequency and severity of AECOPD and improve clinical outcomes beyond pulmonary function.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251347775 – Supplemental material for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials
Supplemental material, sj-docx-1-tar-10.1177_17534666251347775 for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials by Bara M. Hammadeh, Osama M. Younis, Muaath I. Alsufi, Muhammad Idrees, Ayham Mohammad Hussein, Abdullah Yousef Aldalati, Fares A. Qtaishat, Banan Qatawneh, Al Bugazia and Raed A. Hamed in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251347775 – Supplemental material for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials
Supplemental material, sj-docx-2-tar-10.1177_17534666251347775 for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials by Bara M. Hammadeh, Osama M. Younis, Muaath I. Alsufi, Muhammad Idrees, Ayham Mohammad Hussein, Abdullah Yousef Aldalati, Fares A. Qtaishat, Banan Qatawneh, Al Bugazia and Raed A. Hamed in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-3-tar-10.1177_17534666251347775 – Supplemental material for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials
Supplemental material, sj-docx-3-tar-10.1177_17534666251347775 for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials by Bara M. Hammadeh, Osama M. Younis, Muaath I. Alsufi, Muhammad Idrees, Ayham Mohammad Hussein, Abdullah Yousef Aldalati, Fares A. Qtaishat, Banan Qatawneh, Al Bugazia and Raed A. Hamed in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-4-tar-10.1177_17534666251347775 – Supplemental material for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials
Supplemental material, sj-docx-4-tar-10.1177_17534666251347775 for Efficacy and safety of ensifentrine in treatment of COPD: a systematic review and meta-analysis of clinical trials by Bara M. Hammadeh, Osama M. Younis, Muaath I. Alsufi, Muhammad Idrees, Ayham Mohammad Hussein, Abdullah Yousef Aldalati, Fares A. Qtaishat, Banan Qatawneh, Al Bugazia and Raed A. Hamed in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
