Abstract
Background:
Pulmonary segmentectomy is increasingly recognized as a viable alternative to lobectomy for early stage non-small-cell lung cancer (NSCLC), offering comparable oncological outcomes with potentially reduced morbidity. Identifying reliable predictors for postoperative complications and prolonged air leak (PAL) is crucial for optimizing patient selection. While multifactorial scoring systems exist, their complexity limits clinical utility and the predictive value of single factors, such as forced expiratory volume in 1s (FEV1) and diffusing capacity for carbon monoxide (DL CO ), remains underexplored.
Objectives:
This study aimed to evaluate the ability of preoperative FEV1 and DL CO to predict complications (Clavien-Dindo ⩾ 3a) and PAL in patients undergoing pulmonary segmentectomy.
Design:
A retrospective, single-center study compared outcomes between patients undergoing segmentectomy (n = 33) and lobectomy (n = 126) for NSCLC.
Methods:
Patient characteristics, complication rates, and PAL incidence were analyzed. Logistic regression and ROC curve analyses assessed the predictive accuracy of FEV1 and DL CO for complications and PAL.
Results:
Baseline characteristics, including FEV1 and DL CO , were comparable between the segmentectomy and lobectomy groups (p > 0.05). FEV1 was identified as a significant predictor of complications, with lower values associated with increased risk. DL CO exhibited an even stronger predictive value for complications in the segmentectomy cohort, with an AUC of 0.924, indicating excellent predictive accuracy. In contrast, neither FEV1 nor DL CO demonstrated significant predictive value for PAL, which occurred in 30% of segmentectomy and 20% of lobectomy patients (p > 0.05).
Conclusion:
Preoperative FEV1 and DL CO are valuable predictors of complications (Clavien-Dindo ⩾ 3a) in pulmonary segmentectomy, with DL CO showing high predictive accuracy. However, their inability to reliably predict PAL highlights the need for multifactorial models to enhance risk assessment. Despite the limited sample size, our findings align with larger studies and reinforce the clinical utility of FEV1 and DL CO for preoperative risk stratification in segmentectomy patients.
Plain language summary
Lung segment removal, also known as pulmonary Segmentectomy, is a surgery increasingly performed to treat early-stage lung cancer. It offers the potential for preserving lung function compared to larger procedures like lobectomy, which removes an entire lung lobe. However, predicting complications after segmental resection remains a challenge, as this surgery has unique risks due to its technical complexity. While lung function tests, such as FEV1 (a measure of how much air a person can forcefully exhale in one second) and DLCO (a test that assesses how well oxygen passes from the lungs into the blood), are well-established predictors of risk for larger surgeries like lobectomy, their role in segmental resection is less understood. We studied patients undergoing segmental resection or lobectomy and compared their complication rates, focusing on serious complications and prolonged air leaks (PAL), a common post-surgical issue. We analyzed whether FEV1 and DLCO could reliably predict these complications in patients undergoing segmental resection. Using statistical tools, we assessed how accurately these tests could estimate a patient’s surgical risk. Our study found that FEV1 is a useful predictor of serious complications, while DLCO demonstrated an even higher accuracy. However, neither test was effective at predicting prolonged air leaks. Interestingly, patients undergoing segmental resection had complication rates similar to those undergoing lobectomy, suggesting this procedure can be performed safely with proper risk assessment. These results highlight the value of simple, preoperative lung function tests for assessing risk before surgery. While FEV1 and DLCO are strong tools for predicting general complications, additional factors must be considered to predict prolonged air leaks. As segmental resection becomes more common, further research is needed to refine risk prediction and improve patient outcomes.
Introduction
Pulmonary segmentectomy has become an increasingly important surgical option in the treatment of early stage non-small-cell lung cancer (NSCLC). Historically, lobectomy has been regarded as the gold standard for resectable NSCLC.1,2 The paradigm has shifted with accumulating evidence supporting the safety and efficacy of pulmonary segmentectomy. Notably, the JCOG0802/WJOG4607L randomized trial demonstrated that segmentectomy not only achieved non-inferior survival compared to lobectomy but also conferred superior overall survival in patients with peripheral NSCLC ⩽ 2 cm. 3 These findings are further supported by the CALGB140503 trial, which showed similar disease-free and overall survival rates between sublobar resection and lobectomy, highlighting segmentectomy’s role in preserving lung function without compromising oncological outcomes. 4 Other studies have also supported the feasibility and effectiveness of segmentectomy as a standard surgical approach for selected patients with small, early stage NSCLC.5 –9
Despite its benefits, segmentectomy is associated with unique challenges in the postoperative period. Prolonged air leak (PAL), a common complication after anatomical lung resections, 10 remains a significant clinical concern due to its association with extended hospital stays, increased healthcare costs, and higher rates of additional complications.11,12 PAL might be a particularly distinct problem in segmentectomy as a result of the segmental plains.
General complications following lung surgery can also impact recovery and long-term outcomes. Identifying patients at increased risk for such complications is, therefore, crucial for optimizing perioperative management and surgical decision-making. 13
Preoperative pulmonary function tests, particularly forced expiratory volume in 1 second (FEV1) and diffusing capacity of the lung for carbon monoxide (DL CO ) predict very accurately postoperative outcomes in thoracic surgery. While these parameters are well-established in the context of lobectomy, 14 their specific predictive value for complications and PAL in patients undergoing segmentectomy remains unclear. Given the increasing adoption of segmentectomy as a preferred surgical strategy, understanding the role of FEV1 and DL CO in this context is crucial.
This study aims to evaluate the predictive value of FEV1 and DL CO for general complications and PAL in patients undergoing pulmonary segmentectomy, with a comparison to lobectomy patients. By focusing on these routinely assessed pulmonary function parameters, this study seeks to provide clinically relevant insights to enhance risk stratification and improve perioperative management in thoracic surgery.
Material and methods
Study design and population
This study is a retrospective analysis of patients who underwent either segmentectomy or lobectomy for NSCLC at the Department for Thoracic Surgery at the Lung Center Coswig, Saxony, Germany, from January 2023 to July 2024. Inclusion criteria encompassed all patients receiving anatomic resection (segmentectomy or lobectomy) for NSCLC. Exclusion criteria included patients who had extended resections (such as additional wedge resection beyond the primary anatomical resection), combined chest wall resections, or incomplete records. Out of 236 patients, we identified 159 patients meeting inclusion and exclusion criteria (33 segmentectomies and 126 lobectomies). The inclusion and exclusion criteria for the study population are detailed in Table 1. Data were extracted and analyzed from a prospective database recording all complications during the clinical trials. Clinical parameters were extracted from the clinical information system.
Inclusion and exclusion criteria of the study population.

All anatomical resections performed at the Department for Thoracic Surgery, Lung Center Coswig, between January 2023 and July 2024 were initially included. Extended resections (e.g., additional wedge resections), combined chest wall resections, and incomplete patient records were excluded. The final cohort consisted of 159 patients.
NSCLC, Non-small cell lung cancer.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 15
Prolonged air leak
PAL was defined as the presence of an air leak persisting beyond postoperative day 5.
Surgical treatment
The indication for anatomical resection was established in all cases following a multidisciplinary tumor board decision. All patients were functionally evaluated according to the current guidelines of ERS/ESTS. 16
Segmentectomy procedure
Segmentectomy was indicated for NSCLC smaller than 2 cm, located peripherally within a well-defined segment or segment group. It involves the complete dissection and division of the segmental arteries, bronchi, and veins. A systematic lymph node dissection was performed in all cases, including nodes at the base of the segmental hilum.
To define the intersegmental planes, intravenous indocyanine green (ICG) was used to highlight the anatomical boundaries of each segment. In our cohort, ICG was exclusively used in video-assisted thoracoscopic surgery (VATS) segmentectomies to visualize the intersegmental plane. In open procedures, traditional inflation-deflation techniques were applied instead. For VATS cases, a uniportal approach with a 4–6 cm axillary incision in the fourth intercostal space was used. Mechanical staplers were used to divide the intersegmental planes. Simple segmentectomy was defined as a resection of a segment with only one intersegmental plane, whereas complex segmentectomy includes segments with more than one intersegmental plane.
In cases performed with open thoracotomy, the planes were divided either with staplers or by using electrocautery combined with a continuous suture to unite the parenchymal surfaces.
Air leaks were assessed at the end of the procedure using a submersion technique and ventilator measurement. Significant air leaks were addressed with sutures as needed or with sealants applied selectively at the surgeon’s discretion. Chest tube was inserted perioperatively, connected to a digital drainage system, and removed when air leaks were below 10 ml/min and fluid drainage was less than 400 mL in the last 24 h.
Lobectomy procedure
Lobectomy was chosen when the tumor size or location required resection of the entire lobe for optimal oncologic outcomes. Lobectomy involves systematic dissection and division of the lobar arteries, bronchi, and veins. Complete lymph node dissection was performed at both hilar and mediastinal stations.
For VATS lobectomies, a uniportal approach with a 4–6 cm axillary incision in the fourth intercostal space was used. Mechanical staplers were employed to divide the vascular and bronchial structures as well as the fissures. In open thoracotomy cases, the structures were divided using staplers or surgical ties, the fissure was completed with staplers or electrocautery as required.
Similar to segmentectomy, air leaks were evaluated at the end of the operation using a submersion technique and ventilator assessment. Significant leaks were closed with sutures if possible. Sealants were used selectively. No additional measures, such as buttressed sutures or pleurodesis were applied. One or two chest tubes were placed and connected to a digital drainage system, with removal criteria based on air flow stability below 10 mL/min and fluid output of less than 400 mL in 24 h.
Data collection
Data were retrospectively collected from patient records, focusing on key preoperative and postoperative variables relevant to predicting postoperative complications. The primary markers of interest were preoperative pulmonary function tests, specifically FEV1 % (forced expiratory volume in 1 second) and DL CO % (diffusing capacity of the lung for carbon monoxide).
Additional variables included patient demographics (age, sex), smoking status (active smoker, pack-years), and comorbidities. Patient-specific risk stratification parameters included the ASA score, ECOG performance status, and SUVmax (maximum standardized uptake value from preoperative PET/CT imaging). Surgical details encompassed the type of surgical approach (VATS or thoracotomy), the necessity of conversion (from VATS to thoracotomy), the type of resection (segmentectomy or lobectomy), and the specific segment or lobe resected. In addition, the method used for parenchymal closure—either stapler line or classical suture—was recorded. Key postoperative outcomes included the incidence of PAL, duration of chest tube drainage (in days), complications classified by Clavien-Dindo grade, and 30-day mortality.
Statistics
Statistical analysis was conducted using MedCalc® Statistical Software version 20.104 (MedCalc Software Ltd., Ostend, Belgium). Comparisons between the segmentectomy and lobectomy groups were conducted using t-tests for continuous variables. To assess group comparability beyond functional parameters, additional clinical and oncologic variables were analyzed, including ASA classification, ECOG performance status, SUVmax, history of ischemic heart disease, peripheral arterial vascular disease, previous malignancies, COPD, emphysema, smoking status and cumulative smoking exposure (pack-years). T-tests or chi-squared tests were applied as appropriate, depending on the nature of the variable. These analyses were performed to validate the comparability of the segmentectomy and lobectomy cohorts with respect to relevant baseline characteristics.
The incidence of PAL and general complications (Clavien-Dindo ⩾ 3a) was compared between the segmentectomy and lobectomy groups using both chi-squared tests and Fisher’s exact tests. The relationship between PAL and general complications and preoperative predictors—specifically FEV1 and DL CO (both treated as continuous variables)—was evaluated.
Logistic regression models were employed to assess the relationships between FEV1, DL CO , and postoperative outcomes in both the segmentectomy cohort and the combined cohort of lobectomy and segmentectomy patients. In the segmentectomy cohort, the associations of FEV1 and DL CO with postoperative complications and PAL were analyzed. For the combined cohort, the association between FEV1 and complications was also evaluated. In an additional exploratory analysis, logistic regression was performed to assess whether PAL and clinically relevant complications (Clavien-Dindo ⩾ 3a) could be independently predicted by preoperative and patient-specific factors or surgical techniques. The variables included in the model were ASA score, ECOG performance status, history of ischemic heart disease, and peripheral arterial disease; surgical access (VATS vs thoracotomy), conversion from VATS to thoracotomy, and for segmentectomy cases, the complexity of the procedure (simple vs complex segmentectomy). These were selected based on their known clinical relevance and availability in the dataset. Odds ratios (OR) with 95% confidence intervals (CI) were calculated to quantify the strength of associations, and statistical significance was defined as p < 0.05.
Results
A total of 159 patients undergoing anatomical lung resection for NSCLC were included in the analysis, comprising 33 segmentectomies (21%) and 126 lobectomies (79%). The primary focus of this study was the segmentectomy group, while the lobectomy group served as a comparison cohort. Basic patient characteristics are summarized in Table 2.
Baseline characteristics of patients undergoing lobectomy and segmentectomy, stratified by occurrence of PAL.

Continuous data are expressed as median (range), and categorical data as absolute numbers (percentage). p-Values reflect comparisons between the lobectomy and segmentectomy groups. Bold values indicate statistical significance (p < 0.05).
COPD, chronic obstructive pulmonary disease; DLCO, diffusing capacity of the lung for carbon monoxide (% predicted); FEV1, forced expiratory volume in 1 second (% predicted); PAL, prolonged air leak; TT, open thoracotomy; VATS, video-assisted thoracic surgery.
The median age was similar across both groups: 69 years (range 52–84) in the segmentectomy group and 68 years (range 34–83) in the lobectomy group. Male gender was predominant in both groups, with 64% in the segmentectomy cohort and 66% in the lobectomy cohort. In addition to age and gender, further baseline characteristics were compared to assess group comparability. The ASA classification (mean 3.04 in segmentectomy vs 2.97 in lobectomy, p = 0.23), ECOG performance status (0.4 vs 0.35, p = 0.65), and prevalence of cardiac comorbidities (14.3% vs 17.1%, p = 0.80) did not differ significantly between groups. Similarly, there were no significant differences in the rates of peripheral arterial vascular disease (5.7% vs 8.6%, p = 0.74), previous malignancies (31.4% vs 20.7%, p = 0.26), or smoking exposure as measured by mean pack-years (33.9 vs 28.9, p = 0.27).
However, the mean SUVmax of the primary tumor was significantly lower in the segmentectomy group (10.92) compared to the lobectomy group (14.78, p = 0.04), suggesting a potential difference in tumor biology and metabolic activity.
The 33 segmentectomies were divided between 13 right-sided and 20 left-sided resections. The most frequently performed resections included S6 on the right side (5 cases) and the upper division (S1–3) on the left side (11 cases). Detailed distribution of segmentectomies is provided in Table 3.
Overview of pulmonary segmentectomies performed, classified by anatomical complexity and occurrence of PAL.

Complex, complex segmentectomy involving multiple intersegmental planes; PAL, prolonged air leak; simple, simple segmentectomy involving a single intersegmental plane.
Preoperative pulmonary function was assessed using FEV1 and DL CO . The median FEV1 was 81% in both the segmentectomy and lobectomy groups (p > 0.05, Figure 1). The median DL CO was comparable between the segmentectomy group (66%) and the lobectomy group (70%), with no statistically significant difference (p > 0.05, Figure 2).

Shows the distribution of predicted FEV1 (%) in the lobectomy and segmentectomy groups. The median FEV1 was identical between the two groups at 81%, with a similar interquartile range and overall variability. No statistically significant difference was observed between the two groups (p > 0.05). A single outlier is noted in the segmentectomy group.

Illustrates the distribution of predicted DLCO (%) in the lobectomy and segmentectomy groups. The median DLCO was slightly lower in the segmentectomy group (66%) compared to the lobectomy group (70%). However, this difference was not statistically significant (p > 0.05). The interquartile range and the overall spread of DLCO values were comparable between the two groups.
Segmentectomies were predominantly performed via VATS (30 of 33 cases, 91%), with 8 cases (24%) requiring conversion to thoracotomy. Thoracotomy was the primary approach in three cases (9%). PAL occurred in 10 segmentectomy patients (30%). Among these, four cases (40%) required conversion to thoracotomy and one case (10%) was performed as a primary thoracotomy. The chest tube duration was significantly longer in patients with PAL (median: 11 days, range 8–20 days; 95% CI = 9.95, 16.05) compared to those without PAL (median: 4 days, range 2–16 days; 95% CI = 3.57, 5.99); p < 0.0001, unpaired t-test), indicating a highly significant association between PAL and prolonged chest drainage.
Complications greater than Clavien-Dindo grade 2 were observed in seven segmentectomy patients (21%). Among patients without PAL, complications occurred in two cases (6%), including lingula torsion requiring re-intervention and chest wall hematoma. In the PAL group, five patients (50%) experienced severe complications, including transfusion-requiring bleeding, myocardial infarction, pneumonia, severe subcutaneous emphysema, upper gastrointestinal bleeding. Minor complications (Clavien-Dindo grade 1), such as small pneumothoraces, occurred in five patients overall but were not associated with PAL. No 30-day mortality was recorded in the segmentectomy group. The distribution of severe complications (Clavien-Dindo grades 3a and higher) is detailed in Table 4. The categorized postoperative complications in the segmentectomy group are summarized in Table 5.
Postoperative complications and mortality stratified by procedure type and occurrence of PAL, categorized by Clavien-Dindo classification.
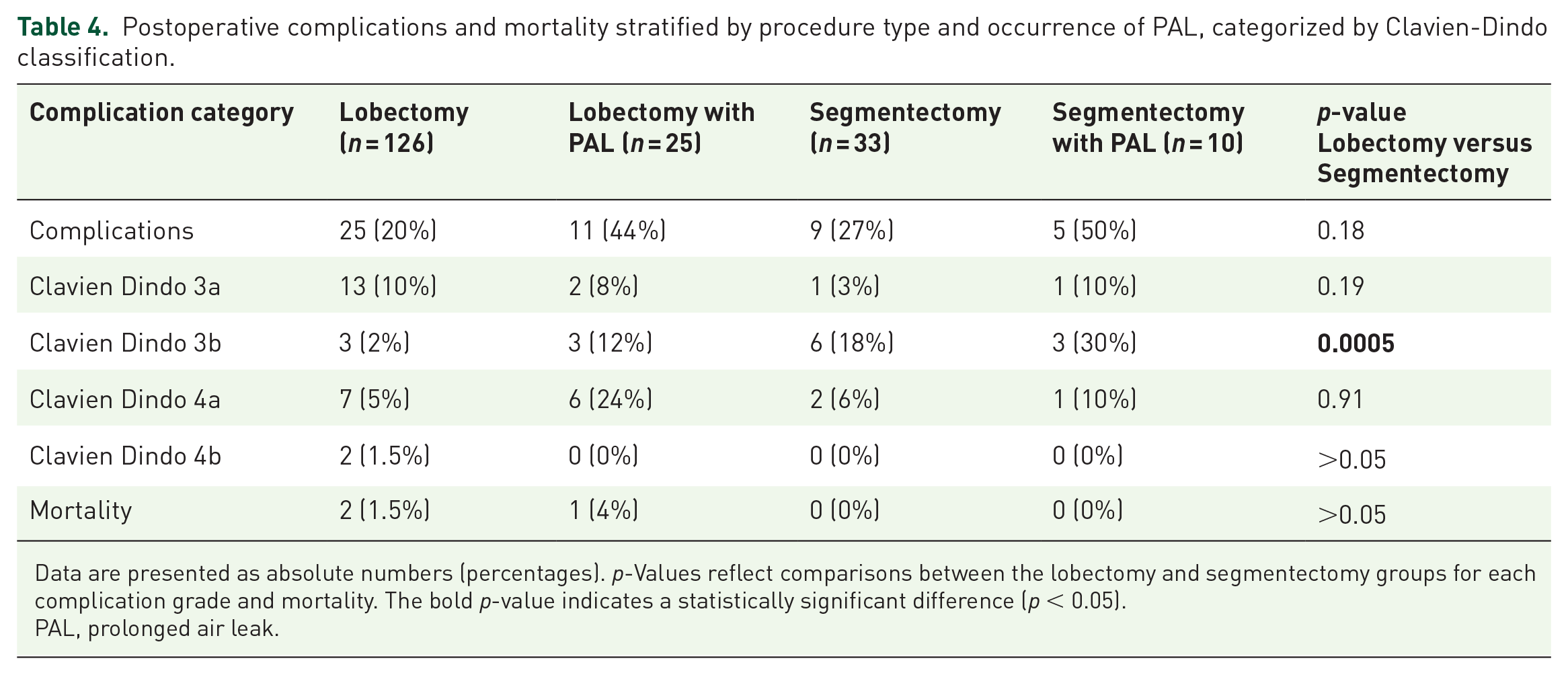
Data are presented as absolute numbers (percentages). p-Values reflect comparisons between the lobectomy and segmentectomy groups for each complication grade and mortality. The bold p-value indicates a statistically significant difference (p < 0.05).
PAL, prolonged air leak.
Classification of postoperative complications in the segmentectomy group by category, clinical examples, number of affected patients, and Clavien-Dindo severity range.

Complications are grouped by clinical category with representative examples. Numbers indicate affected patients within each category. Severity is expressed as the observed Clavien-Dindo classification range.
Clavien-Dindo, Clavien-Dindo classification of surgical complications.
A detailed comparison of postoperative complications revealed significantly higher complication rates (Clavien-Dindo ⩾ 3a) in patients with PAL (50%) compared to those without PAL (18%; χ2 = 11.84, p = 0.00058). Furthermore, complications in PAL patients were significantly more severe, as demonstrated by higher median Clavien-Dindo scores (median 2.5) compared to non-PAL patients (median 1.0; Mann–Whitney U = 1802, p < 0.00001). A detailed breakdown of individual complication types is presented in Table 6.
Frequency of specific postoperative complications in patients with and without PAL.

Complication rates are expressed as percentages of patients within each group. P-values were calculated using chi-square or Fisher’s exact test as appropriate, comparing the PAL and non-PAL groups. Bold values indicate statistically significant differences.
MI, Myocardial Infarction; PAL, prolonged air leak.
Logistic regression analysis focused on the segmentectomy cohort identified DL CO as a significant predictor of complications (OR 0.97, p < 0.001, Area under the ROC curve (AUC): 0.924), with lower values associated with an increased risk. FEV1 showed a weaker but statistically significant association (OR 0.98, p = 0.0425, Area under the ROC curve (AUC) 0.764). Neither DL CO nor FEV1 significantly predicted the occurrence of PAL in this cohort. An additional multivariate logistic regression was performed to evaluate whether PAL or clinically relevant complications (Clavien-Dindo ⩾ 3a) could be independently predicted by preoperative and surgical factors. The model included ASA score, ECOG performance status, history of ischemic heart disease, peripheral arterial disease, surgical access (VATS vs thoracotomy), conversion to thoracotomy, and segmentectomy complexity (simple vs complex). None of the variables demonstrated statistically significant associations with PAL or complication occurrence (p > 0.05). However, a nonsignificant trend toward higher complication risk was observed for reduced ECOG performance (OR 3.6, p = 0.11) and COPD (OR 3.4, p = 0.24). In segmentectomy patients, complex procedures showed a tendency toward increased PAL risk (OR 3.8, p = 0.17).
PAL occurred in 25 lobectomy patients (20%), with a median chest drainage duration of 14 days (range 6–35 days; 95%CI = 11.74, 19.08) compared to 5 days (range 2–13 days; 95% CI = 4.76, 5.51) in non-PAL patients, p < 0.00001 (unpaired t-test). Severe complications (Clavien-Dindo ⩾ 3a) were observed in 25 lobectomy patients (20%), and two 30-day mortalities occurred in this group (one due to stroke and one due to pulmonary embolism). While FEV1 was also a significant predictor of complications in the lobectomy cohort, neither FEV1 nor DL CO predicted PAL occurrence. Table 7 provides an overview of categorized postoperative complications in the lobectomy group.
Classification of postoperative complications in the lobectomy group by category, clinical examples, number of affected patients, and Clavien-Dindo severity range.

Complications are grouped by clinical category with representative examples. Numbers indicate affected patients within each category. Severity is expressed as the observed Clavien-Dindo classification range.
Clavien-Dindo, Clavien-Dindo classification of surgical complications.
The incidence of PAL was higher in the segmentectomy group (30%) compared to the lobectomy group (20%). However, statistical analysis revealed that this difference was not significant, as demonstrated by both the chi-squared test (χ2 = 1.11, p = 0.291) and the Fisher’s exact test (p = 0.238).
Similarly, the rate of general complications (Clavien-Dindo ⩾ 3a) was 27% in the segmentectomy group and 20% in the lobectomy group. Statistical tests again showed no significant difference between the two groups, with a chi-squared value of χ2 = 1.8 (p = 0.18) and a Fisher’s exact test p = 0.350.
Discussion
Segmentectomy can be technically demanding and the resected intersegmental planes might cause a different complication profile compared to lobectomy. Since two prospective trials proved a better clinical outcome and comparable oncological results, segmentectomy will be increasingly performed in the future. In addition, newly implanted lung cancer screening programs in many countries will lead to more detected small lesions and will lead to the revival of this technique. Therefore, it is important to investigate the risk profile of segmentectomy and identify strong prognosticators for segmentectomy. In lobectomy, there was an intensive investigation over several decades, and FEV1 is clearly the strongest prognosticator.
Our study demonstrated that the segmentectomy and lobectomy groups did not significantly differ in terms of baseline characteristics, including age, gender, preoperative FEV1, and DL CO . A more detailed group comparison further confirmed the overall comparability: ASA classification, ECOG performance status, cardiac comorbidities, peripheral arterial disease, prior malignancies, and cumulative tobacco exposure (pack-years) were similar between both cohorts, with all comparisons yielding nonsignificant p-values.
The only statistically significant difference was observed in the metabolic activity of the tumors, as measured by SUVmax, which was lower in the segmentectomy group (mean 10.9) compared to the lobectomy group (mean 14.8, p = 0.04). This may reflect a tendency toward selecting patients with lower tumor aggressiveness for segmentectomy, in line with its established indication for small, peripheral tumors ⩽2 cm.
Importantly, there were no significant differences in the incidence of PAL or major postoperative complications (Clavien-Dindo ⩾3a) between the two groups, despite the difference in resection extent. These findings suggest that, within a real-world population, segmentectomy can be performed safely without an increased risk of severe complications, even when accounting for a broad range of clinical variables.
In our segmentectomy cohort, patients with PAL experienced a significantly prolonged chest tube duration compared to those without PAL (median 11 days, range 8–20 days vs 4 days, range 2–16 days, p < 0.00001), highlighting PAL as a clinically relevant complication with direct impact on recovery and resource use. Notably, the maximum drainage duration of 16 days in the non-PAL group was due to a postoperative chylothorax in one patient, which was managed conservatively and partially in an outpatient setting.
The numerical imbalance between the segmentectomy (n = 33) and lobectomy (n = 126) cohorts reflects the underlying clinical indications that guided treatment decisions within our patient population. However, this imbalance accurately reflects clinical reality, as segmentectomy is indicated only for a highly selected subgroup of NSCLC patients—those with peripheral tumors ⩽2 cm and no clinical suspicion of lymph node metastasis—whereas lobectomy remains the standard of care for a significantly broader patient population. Thus, our study did not intend to directly compare two matched surgical methods. Rather, it aimed to assess clinical characteristics and risk factors within a carefully selected segmentectomy cohort in comparison with the broader lobectomy cohort. Consequently, the observed imbalance should not be interpreted as a methodological weakness but rather as a representation of clinical practice.
Regarding the association between specific segments and the occurrence of PAL (see Table 3), no statistically robust conclusions could be drawn due to the small number of cases per segment. Although anatomical and technical differences between segments could theoretically complicate the identification and delineation of intersegmental boundaries, our data did not demonstrate a clear relationship between the complexity of segmental resection and PAL occurrence. The incidence of PAL was similar in simple (6 out of 20; 30%) and complex segmentectomies (4 out of 13; 31%). Future studies with larger sample sizes are needed to further clarify segment-specific risks.
The comparison of postoperative complications between patients with and without PAL revealed a significantly higher rate of clinically relevant complications (Clavien-Dindo ⩾ 3a) in the PAL group. Furthermore, the severity of complications was significantly greater in this group, as indicated by a higher median Clavien-Dindo score. These findings emphasize that PAL is associated not only with a higher frequency of complications but also with increased clinical burden and severity.
One likely explanation is that certain complications—such as pneumothorax and subcutaneous emphysema—are directly related to the presence of PAL. These events typically require invasive interventions (e.g., re-drainage or surgical revision) and are thus classified as at least Clavien-Dindo grade 3a, which contributes to the overall complication severity in this group.
Another possible interpretation is that prolonged drainage duration in PAL patients leads to a cascade of downstream effects, such as immobilization, delayed recovery, or increased susceptibility to secondary complications. However, while such mechanisms are plausible, our dataset does not confirm a higher burden of baseline comorbidities or worse pulmonary function in the PAL group. Therefore, these factors remain speculative and cannot be substantiated within the scope of this study.
In summary, the observed association between PAL and both increased complication rates and severity highlights the clinical relevance of PAL and the need for strategies aimed at its prevention and early management.
Regarding predictive factors, FEV1 was identified as a statistically significant predictor for general complications. Lower FEV1 values were associated with a significant increase in the risk of complications. However, FEV1 alone was not a reliable predictor for PAL in this study. Similarly, DL CO emerged as an even stronger predictor for general complications in the segmentectomy group, with lower DL CO values significantly increasing the complication risk. However, DL CO also showed no significant predictive value for PAL.
While FEV1 is classically used for preoperative risk stratification in lobectomy, our analysis was designed to evaluate predictive factors within the segmentectomy cohort. In this context, DL CO emerged as a strong and statistically significant predictor of postoperative complications. This may reflect the particular physiological burden of segmentectomy, which—despite preserving more lung volume than lobectomy—involves extensive parenchymal dissection and division of intersegmental planes, typically using stapling or suturing techniques. These interventions may lead to compression or trauma in adjacent lung tissue, potentially impairing gas exchange and thereby making DL CO as a measure of diffusion capacity, a more sensitive indicator of postoperative risk.
DL CO was analyzed as a continuous variable in our study and showed a significant inverse correlation with the rate of general postoperative complications, while its association with PAL remained statistically nonsignificant. This underscores the role of reduced diffusing capacity as a marker for overall physiological vulnerability, rather than a specific predictor of air leak. We did not apply predefined DL CO thresholds in our statistical model. However, when interpreting our findings in light of previous research, the study by Aguinagalde et al. 17 provides valuable reference points. Their analysis of over 2,500 patients undergoing anatomical resection revealed inflection points in postoperative risk—particularly a sharp increase in morbidity and mortality below ppoDL CO values of 60% in thoracotomy and 45% in VATS procedures. Although our sample size precluded stratified subgroup analysis by DL CO thresholds, the observed trend in our cohort supports these broader findings. Future studies should explore whether similar functional cutoffs can be validated for segmentectomy, potentially allowing for refined, threshold-based risk stratification in clinical decision-making. However, the excellent predictive accuracy of DL CO for general complications in our analysis (AUC of 0.924) underscores its substantial clinical value even without clearly established thresholds. Future research involving larger patient cohorts may explore the utility of threshold-based risk models and assess whether combining selected markers could indeed lead to clinically relevant improvements in predictive performance.
Our findings align with and contribute to the growing body of evidence supporting the clinical role of segmentectomy in early stage NSCLC. The JCOG0802/WJOG4607L trial demonstrated the non-inferiority and even superiority of segmentectomy over lobectomy in terms of overall survival, while showing comparable rates of postoperative complications, including PAL. 3 Similarly, other studies, such as the CALGB140503 trial, found no significant differences in perioperative morbidity or mortality between lobectomy and sublobar resections, highlighting the safety and efficacy of segmentectomy in carefully selected patients. 18 These findings reinforce the clinical validity of segmentectomy as a treatment option for small, peripheral NSCLC.
The perioperative complication rates observed in our study (20% for lobectomy and 27% for segmentectomy) closely mirror the 26% and 27% rates reported in the JCOG trial for lobectomy and segmentectomy, respectively. 3 Regarding PAL, our rates (30% in the segmentectomy group and 20% in the lobectomy group) are higher than those reported in the CALGB post hoc analysis (7% and 9%). However, it is important to note that the CALGB trial includes wedge resections within the sublobar resection cohort, which may not be directly comparable to our segmentectomy cohort. Moreover, other prospective studies, such as Stamatis et al., have confirmed comparable morbidity rates between segmentectomy and lobectomy, further supporting the generalizability of our results. 19
Our findings can be contextualized by comparison with the recent analysis by Gooseman et al., who evaluated PAL in 191 segmentectomy patients and identified low DL CO , low BMI, and complex segmentectomies as significant predictors of PAL. In contrast, our analysis did not find DL CO , FEV1, or the complexity of the segmental resection to be significantly predictive of PAL, which occurred in 6 of 20 simple and 4 of 13 complex segmentectomies. While DL CO was a robust predictor of general complications in our cohort, it failed to predict PAL reliably. This discrepancy may in part be explained by the distribution of segment types within our study population. Specifically, more than half of the segmentectomies in our cohort consisted of left upper division (S1–3) or superior segment resections (S6), which are commonly classified as “simple segmentectomies” due to involving only a single intersegmental plane. In total, only 13 out of 33 procedures (39%) met the criteria for complex segmentectomy. As such, the relatively small number of complex segmentectomies may have limited the statistical power to detect a potential association with PAL. In addition, differences in surgical techniques, case selection, and perioperative management may contribute to the divergence in findings. Nevertheless, our observed PAL rate of 30% aligns reasonably well with existing literature, including the 19% incidence reported by Gooseman et al., further supporting the notion that PAL is a multifactorial complication influenced by both patient- and technique-related factors. From a technical standpoint, achieving precise anatomical dissection and reliable parenchymal sealing remains the primary challenge in segmentectomy. Stapler technology significantly facilitates secure and reproducible sealing of the parenchyma, particularly in minimally invasive procedures. Laser technology, widely adopted in pulmonary metastasectomy for parenchymal dissection, theoretically provides superior precision and reduced trauma; however, its utility in anatomical segmentectomy remains limited by practical constraints, restricting its benefit predominantly to open thoracotomy procedures. Additional technical advancements, such as intraoperative tissue sealants, surgical patches (e.g., polyglycolic acid or collagen sponges) in cases of detected air leaks, reinforced staple lines when high risk for PAL is anticipated, digital chest drainage systems and consistent postoperative drainage management protocols, also have the potential to further reduce PAL incidence. Although these approaches were not systematically investigated in our study, their selective clinical application could represent valuable strategies for the future management of PAL in pulmonary segmentectomy patients.
Interestingly, Yamauchi et al. identified a high FEV1 as a predictor of PAL, which contrasts with traditional assumptions that low pulmonary function is associated with increased PAL risk. This discrepancy may stem from the study’s specific focus on patients with residual air leaks on postoperative day 1, as highlighted by the authors. They suggest that better lung function (FEV1 > 80%) might contribute to higher airflow at the leakage site, potentially impeding spontaneous healing. These findings underscore the multifactorial nature of PAL and the need for individualized risk assessments that consider context-specific variables.
In addition, Kawatani et al. pointed to technical factors such as cranial side free space and electrocautery as risk factors for late-onset pulmonary fistula, emphasizing the significance of surgical technique in preventing postoperative complications. 20
Our findings highlight the utility of preoperative pulmonary function tests, particularly DL CO , in assessing the risk of general complications. However, the absence of significant predictive value for PAL suggests the need for multifactorial models to improve risk stratification in patients undergoing anatomical lung resection. Either FEV1 or DL CO has been studied and incorporated into multifactorial scoring systems, such as the ESTS score, IPAL and others.21 –26 While these systems achieve moderate predictive accuracy, their complexity and the inclusion of variables such as pleural adhesions or the extent of resection21,24—which are often unknown preoperatively—make them impractical for assessing surgical risk prior to the operation.
Our study was specifically designed to evaluate readily available single-parameter predictors (FEV1 and DL CO ), emphasizing their practical applicability in routine clinical settings. Although combining multiple clinical markers into predictive models has previously been extensively studied, such multifactorial analyses typically do not substantially outperform simpler, clinically feasible parameters. In this context, we deliberately focused on individual parameters rather than combinations, while acknowledging that future refinements—such as the incorporation of defined DL CO thresholds in combination with selected clinical risk factors—may further enhance predictive accuracy.
DL CO and FEV1 have shown individual predictive value for general complications, yet their utility for reliably predicting PAL remains limited. Reported paradoxical associations—such as higher FEV1 values linked to increased PAL risk 27 —underscore the multifactorial and context-dependent nature of this complication. Our findings support the interpretation that neither parameter alone is sufficient for accurate PAL prediction. This reinforces the need for clinically practical risk stratification tools that rely on robust, preoperatively available data.
Despite the complexity of segmentectomy and its anatomical variability, our analysis did not identify any other single patient-specific or surgical factor as a statistically significant independent predictor of PAL or major complications. Variables commonly presumed to influence postoperative risk—such as ASA classification, ECOG performance status, ischemic heart disease, peripheral vascular disease, or even surgical access (VATS vs thoracotomy)—failed to demonstrate robust predictive value in our multivariate models.
These findings do not negate the relevance of clinical judgment; rather, they highlight the multifactorial and perhaps elusive nature of postoperative complications. Although more fragile patients may present with lower DLCO values—potentially acting as a confounder in the association between DL CO and postoperative outcomes—we did not perform a statistical adjustment for this effect. Nonetheless, our broader comparability analysis, which showed no significant differences in comorbidities or functional parameters between groups, suggests that major confounding bias is unlikely.
Interestingly, neither the complexity of the segmentectomy (simple vs complex) nor conversion to thoracotomy emerged as significant contributors to PAL risk. It may well be that PAL, like many things in surgery, resists simple prediction—demanding instead a nuanced understanding of surgical technique, tissue behavior, and patient physiology. Until more refined models emerge, our data support the use of established parameters such as DL CO and FEV1 as solid indicators of general complication risk.
This study has several limitations. First, as a retrospective single-center study, it is inherently subject to selection bias. The decision to perform a segmentectomy was made by the surgical team involved in the treatment of each patient at the respective time. This approach may have introduced a bias influenced by individual preferences and experiences. The current German national guidelines recommend sublobar resections primarily for patients with compromised pulmonary function, while lobectomy remains the gold standard for lung cancer treatment, which may explain the disproportionate group sizes observed in this study. Furthermore, the relatively small sample size of the segmentectomy group limits the statistical power and the generalizability of the findings.
Another limitation is the lack of a standardized protocol for assessing intersegmental planes during surgery, which may have influenced outcomes such as PAL. In our analysis, surgeon-specific factors were not systematically evaluated. Given the technical complexity of pulmonary segmentectomy, individual surgical experience, case volume, operative technique, and preferences for parenchymal sealing methods likely influenced outcomes such as PAL. The absence of data regarding these surgeon-related variables represents a limitation, as they may have contributed significantly to variability in complication rates. Our study is limited by the relatively small sample size in the segmentectomy group (n = 33), which reduces the statistical power and generalizability of the findings. To quantify this limitation, a post hoc power analysis could be performed, enabling a clearer interpretation of nonsignificant results and providing guidance for required sample sizes in future studies. Finally, as a single-center study, the results may not fully reflect practices or outcomes at other institutions with different patient populations or surgical techniques. However, it is important to note that these data represent real-world clinical practice, and similar variations or challenges likely exist in many surgical teams. Despite these limitations, the study also has notable strengths. A key strength lies in its focus on two widely available and clinically established parameters, FEV1 and DL CO , which are routinely assessed preoperatively. These parameters are simple to obtain and interpret, making them highly relevant for clinical practice. Unlike complex scoring systems, which are rarely applied in everyday workflows, FEV1 and DL CO offer actionable insights for risk stratification and decision-making. Moreover, this study provides valuable insights into the predictive value of these parameters specifically in segmentectomy patients, a group for which robust data are still limited. By emphasizing single-factor predictors, this study highlights practical tools for clinicians while identifying the need for more comprehensive predictive models.
Conclusion
Our investigation emphasizes the predictive capabilities of FEV1 and DL CO in the context of pulmonary segmentectomy, a procedure increasingly recognized for its oncologic and functional benefits. In our segmentectomy cohort, DL CO emerged as a particularly robust predictor of general complications, achieving an exceptional Area Under the Curve (AUC) of 0.924. This level of discrimination underscores DL CO ’s clinical utility in identifying patients at heightened risk for postoperative complications. Such a result highlights its role as an actionable single-parameter tool in preoperative risk assessment, particularly relevant in the context of limited data focusing specifically on segmentectomy.
Despite these promising findings, the performance of FEV1 and DL CO as single predictors of PAL was limited. This outcome is consistent with the inherent complexity of PAL, a multifactorial complication influenced by a range of perioperative and patient-specific variables. Even established multifactorial scoring systems, such as the ESTS and IPAL, have demonstrated only moderate predictive accuracy for PAL and remain insufficiently validated for broad clinical application. Against this backdrop, the inability of DL CO and FEV1 to reliably predict PAL in our segmentectomy cohort is less surprising and highlights the need for innovative models that incorporate a broader spectrum of predictive parameters.
While our cohort is relatively small, it remains highly representative of real-world segmentectomy practice and the observed complication and PAL rates align with those reported in larger, multicenter studies. This comparability supports the validity of our findings, even as it underscores the need for larger-scale studies to confirm and refine our observations.
In summary, our study highlights the strengths and limitations of single-parameter tools such as DL CO and FEV1 in the nuanced field of thoracic surgery. While DL CO demonstrated excellent predictive accuracy for general complications in the segmentectomy cohort, both parameters fell short in addressing the multifaceted challenge of PAL prediction. This reinforces the need for comprehensive, validated models tailored to the complexities of thoracic surgical outcomes, particularly in procedures as intricate as segmentectomy.
