Abstract
Background:
Cystic fibrosis (CF) is characterized by chronic neutrophilic inflammation in the airways. Elexacaftor/tezacaftor/ivacaftor (ETI) therapy has demonstrably improved clinical outcomes and quality of life in people with CF (pwCF), but its effects on systemic inflammatory parameters remain unclear.
Objective:
To evaluate the impact of ETI on systemic inflammation in children and adolescents with CF.
Design:
Retrospective, dual-center observational, propensity score-matching study of pediatric pwCF on ETI.
Methods:
PwCF aged ⩽ 18 years treated with ETI at two Italian reference centers were included in this study. Data on immunoglobulins (Ig) (A, G, and M), γ-globulin, leukocyte levels, percent predicted forced expiratory volume in the first second (ppFEV1), sweat chloride (SC) concentration, and sputum cultures were collected at baseline, 12, and 24 months of treatment. Laboratory data of a control group (pwCF, not in ETI therapy, same demographic characteristics as the study group) were also collected.
Results:
Sixty-six patients (30 males, median age: 12 years, F508del homozygous: 23) were included. Mean IgG levels (SD) significantly decreased (p = 0.001) from 1168.20 mg/dl (344.41) at baseline to 1093.05 mg/dl (258.73; 12 months) and 1092.87 mg/dl (232.42; 24 months). Similar reductions were observed for IgA and γ-globulin; IgM reduction was not statistically significant. Leukocyte levels also decreased significantly from 8.04 × 103/µl (3.23 × 103) at baseline to 6.61 × 103/µl (1.74 × 103) (12 months) and 6.45 × 103/µl (1.70 × 103; 24 months). As for the control group, no significant changes in the levels of Ig, leukocytes, and γ-globulin were detected throughout the study period (p > 0.05).
The mean (SD) ppFEV1 and the overall mean (SD) SC concentration significantly decreased during the follow-up. Regarding cultures, 18 (27%) of the 27 patients positive (41%) for Staphylococcus aureus at baseline became negative during treatment. Three patients (4%) with persistently positive cultures for Pseudomonas aeruginosa during the first 12 months, became negative after 24 months. One patient (1.5%), with a baseline positive culture for Pseudomonas Aeruginosa, showed negative cultures after 12 months.
Conclusion:
ETI treatment improved respiratory outcomes and significantly reduced values of IgG, IgA, γ-globulin, and leukocytes, suggesting an effect on the systemic inflammatory response. Further research is warranted to elucidate the role of inflammatory parameters in monitoring response to therapy.
Plain language summary
• Inflammation plays a pivotal role in Cystic Fibrosis (CF) progression
• The new combination therapy Elexacaftor/Tezacaftor/Ivacaftor (ETI) improves lung function, nutritional status, and quality of life in eligible CF patients. Limited data exists on the impact of ETI on inflammation in CF.
• This retrospective study demonstrates that ETI appears to positively impact inflammation markers in children and adolescents with CF.
• Further research is needed to better understand how inflammation parameters can help monitor the response to therapy
Introduction
Cystic fibrosis (CF) arises from variants in the cystic fibrosis transmembrane conductance regulator (CFTR) gene, leading to epithelial chloride channel dysfunction and impairing the water–salt balance across various organ systems. 1 This dysfunction results in reduced or absent flow of chloride/bicarbonate ions and the formation of thickened secretions, causing symptoms such as recurrent lung infections, pancreatic insufficiency, liver cirrhosis, intestinal obstruction, and male infertility. 2 The interplay of obstruction, infection, and inflammation is pivotal in CF lung disease pathogenesis and progression. 3 Studies suggest that individuals with CF exhibit increased susceptibility to inflammation, possibly due to dysregulated CFTR protein leading to overproduction of proinflammatory cytokines by respiratory epithelial cells.4 –6
The correlation between serum immunoglobulin (Ig) levels, particularly IgG, and pulmonary involvement is well established, with higher IgG levels indicative of chronic inflammation and poorer respiratory outcomes.7 –9
The emergence of highly effective modulator therapies (HEMT) targeting CFTR protein has revolutionized CF management.10–11 Elexacaftor/tezacaftor/ivacaftor (ETI) is the first triple combination modulator approved for patients with responsive CFTR variants. 12 In Italy, ETI is approved for use in people with CF (pwCF) aged 6 and above with at least one F508del variant 13 and has demonstrably improved lung function, nutritional status, and quality of life in this population.14 –16 These improvements are likely attributed to enhanced mucociliary clearance and reduced inflammation. 17
Against this backdrop, we conducted a retrospective study to evaluate the impact of ETI therapy on inflammation parameters in pediatric CF patients with a focus on respiratory outcomes.
Materials and methods
Study design, subjects, and data
This was a retrospective, dual-center observational, propensity score-matching study. Inclusion criteria were as follows: having a diagnosis of CF according to Farrell et al., 18 and with at least one F508del CFTR variant, undergoing ETI treatment, and being ⩽ 18 years before starting the drug. Data were retrieved from the hospital’s electronic database system at baseline and after 12 and 24 months of therapy. Key exclusion criteria included the following: liver cirrhosis or history of solid organ or hematologic transplantation and presence of CFTR variant other than F508del, not eligible for ETI prescription, according to Italian legislative guidelines.
The dosing regimen for ETI was tailored to the patient’s weight and followed the protocol outlined below 19 :
For patients weighing <30 kg:
ELX: 100 mg once daily
TEZ: 50 mg once daily
IVA: 75 mg every 12 h
For patients weighing >30 kg:
ELX: 200 mg once daily
TEZ: 100 mg once daily
IVA: 150 mg every 12 h
Primary outcome measures included Ig (classes A, G, and M), leukocyte, and γ-globulin levels over the initial 24 months of ETI treatment. The same data of a control group of people with CF not eligible for ETI therapy and intentionally with the same demographic characteristics as the study group was also collected.
Secondary outcomes comprised percent predicted forced expiratory volume in the first second (ppFEV1) adjusted for age and sex, percentage of positive sputum cultures, change in sweat chloride (SC) concentration from baseline, and nutritional status assessed via body mass index (BMI) z-score. Normal ranges for IgG, IgA, and IgM were determined following Bayram et al. 20
Ig levels and assessment of lung function were conducted during the period without pulmonary exacerbations (PEx) 21 or in the next quarterly visit an episode of PEx. Microbiological status was assessed in line with standard care protocols. 22 Chronic Pseudomonas aeruginosa (PsA) infection was defined according to the modified Leeds criteria. 23
The study was conducted following STROBE guidelines for observational cohort studies (Supplemental File). 24
Statistical analysis
Statistical analysis was performed using SPSS. Results are expressed as means and standard deviations (SD), means and 95% confidence interval (CI), or medians and interquartile range (IQR), as appropriate. The Wilcoxon signed rank test and the paired t-test, as appropriate, were used to compare data, and p-values <0.05 were considered significant.
Results
Characteristics of the study population
Sixty-six patients (30 males), aged 6–8 years (median age: 12 years; IQR: 11–14), were included. Baseline demographics and patient characteristics of the study group are summarized in Table 1.
Clinical characteristics of the study population at initiation of ETI.

Immunological and inflammatory parameters
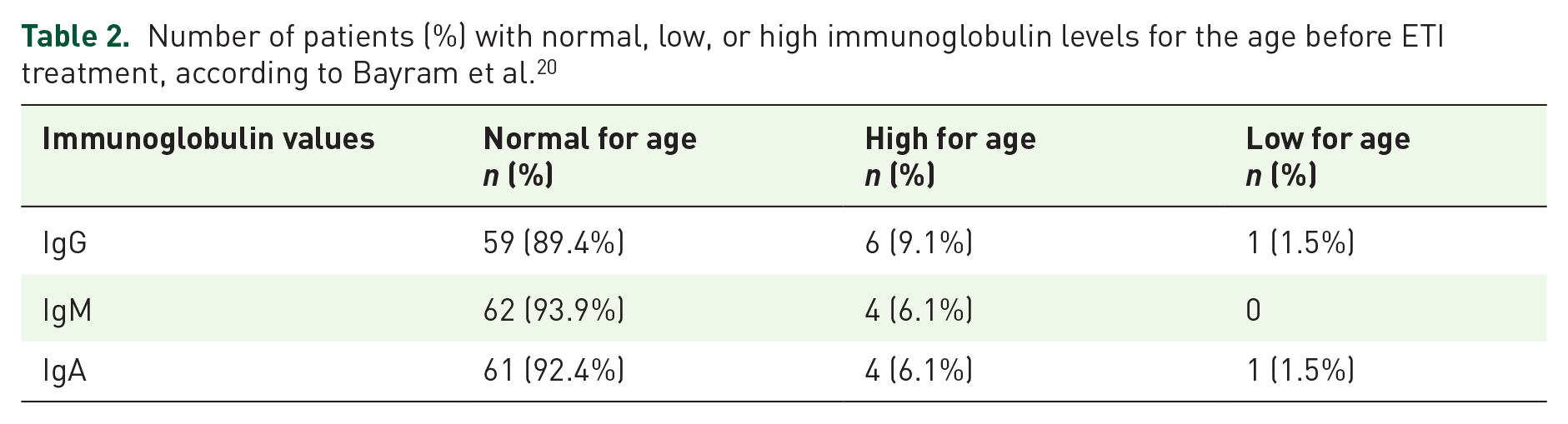
Immunoglobulin levels for age before ETI treatment are summarized in Table 2.
Number of patients (%) with normal, low, or high immunoglobulin levels for the age before ETI treatment, according to Bayram et al. 20
Overall mean (SD) IgG values significantly decreased (p = 0.001) from 1168.20 (344.41) mg/dl at baseline (95% CI (1085.11, 1251.29) to 1093.05 (258.73) g/dl after 12 months (95% CI: 1030.63–1155.46) and 1092.87 (232.42) after 24 months of ETI treatment (95% CI: 1011.05–1174.69; Table 3).
Laboratory parameters. Values are expressed as mean (95% CI).

Indicates statistically significant values.
IgA levels decreased from 173.08 (98.39) (95% CI: 149.35–196.82) to 156.17 (80.15) mg/dl (p < 0.05; 95% CI: 136.83–175.51) after 12 and 160.52 (69.31) mg/dl after 24 months of treatment (95% CI: 136.12–184.92). IgM values also decreased from 133.92 (56.94) mg/dl at baseline (95% CI: 120.19–147.66) to 130.60 (61.78) mg/dl at 12 months (95% CI: 115.69–145.50) and 137.54 (77.36) at 24 months (95% CI: 110.31–164.78), but this variation was not statistically significant.
Concurrently, mean (SD) γ-globulin levels significantly reduced from 15.32 (3.92; 95% CI: 14.16–16.47) to 14.13 (2.75) g/l after 12 months of treatment (p = 0.001) (95% CI: 13.32–14.95) and 14.87 (2.48) g/l (p < 0.05) after 24 months (95% CI: 13.75–15.98).
Mean (SD) leukocyte values decreased from 8.04 × 103/µl (23 × 103) (95% CI: 7.26 × 103–8.82 × 103) to 6.61 × 103/µl (1.74 × 103) during the first 12 months of ETI treatment (p = 0.00009) (95% CI: 6.19 × 103–7.03 × 103) and to 6.45 × 103/µl (1.70 × 103/µl) after 24 months (p < 0.05) (95% CI: 5.86 × 103–7.04 × 103).
No significant changes in the levels of Ig, leukocytes, and γ-globulin were detected over time (p > 0.05) in the control group (N.55, 38 males; median age at initiation of study: 14.4 years; Supplemental Table (A–C)).
Lung function
At baseline, the mean ppFEV1 (SD) was 85.7 (17.1). This increased to 100.7 (16.3) after 12 months of ETI treatment, representing a mean absolute ppFEV1 improvement of 14.8% (95% CI: 12.3–17.3; Figure 1). After 24 months of treatment, the mean (SD) ppFEV1 had mildly declined to 95.5 (16.9).

Absolute changes from baseline in ppFEV1.
A significant difference was observed in the number of respiratory exacerbations requiring oral antibiotics between the 2 years before and after starting ETI (p < 0.05). However, no significant difference was found in the number of respiratory exacerbations requiring parenteral therapy (p > 0.05).
Bacterial pathogens
Eighteen (27%) of 27 patients who were positive (41%) for Staphylococcus aureus at baseline showed negative cultures during treatment. Three patients (4%) had persistent positive cultures for PsA during the first 12 months of treatment, but these cultures became negative after 24 months of treatment. One patient (1.5%), with a baseline culture positive for PsA, showed negative cultures after 1 year of treatment.
Nutritional status
The mean (SD) BMI z-score at baseline was −0.34 (1.01) and progressively increased to −0.04 (0.95) after 12 months of ETI treatment (p = 0.0001) remaining stable after 24 months (−0.05 (0.70)).
The mean (95% CI) absolute increase in BMI z-score was 0.31 (CI 95%: 0.14–0.47; p < 0.0001) after 12 months of ETI treatment.
Sweat chloride
The overall mean (SD) SC concentration was 109.2 (38.8) mmol/l at baseline, significantly decreased to 50.2 (25.2) mmol/l during the first 12 months of ETI treatment and further to 47.0 (22.2) mmol/l after 24 months (p < 0.0001).
The mean absolute change from baseline was −56.9 mmol/L (95% CI: −64.3 to –49.4) after 12 months of treatment.
Discussion
Inflammation plays a pivotal role in CF progression. Despite showing significant quality-of-life improvements with ETI treatment, further research is warranted to assess the effects of CFTR modulators on inflammatory response and immune processes.
In this study, we report the trend of inflammatory parameters in children and adolescents with CF during ETI treatment.
A previous study analyzing the effects of CFTR modulators on inflammatory markers in pwCF, like Jarosz-Griffiths et al., 25 suggests that CFTR modulators have anti-inflammatory properties; however, inflammation persists, albeit reduced, during ETI treatment.26,27
Our study revealed a significant decrease in overall mean IgG, γ-globulin, and leukocyte levels after 24 months of treatment. These findings align with Schnell et al.’s 28 prospective monocentric study involving CF people aged at least 12 years treated with ETI, which observed reductions in Ig A, G, M, and leukocyte levels. Lepissier et al., in a study on an adult cohort of patients, noted reductions in blood and sputum inflammatory biomarkers within a month of ETI treatment, particularly for C-reactive protein, leukocytes, and polymorphonuclear neutrophil counts. 29
Other studies 30 reported normalized neutrophil counts, decreased proinflammatory cytokine levels31,32 and chemokines, 33 and enhancements in monocyte and macrophage function34 –36 in pwCF undergoing ETI treatment.
As it is known, IgA is a protective barrier defending mucosal surfaces against microorganisms and IgG is involved in the humoral immune response. 37
Elevated IgG and IgA levels are associated with poorer clinical outcomes and reduction in lung function in pwCF.38–39 Wheeler et al. found that children with hypogammaglobulinemia G exhibited better lung function, nutritional status, fewer PEx, and less colonization with PsA, with a slower decline in lung function. 7 Gur et al. described a positive correlation between IgG levels and some markers of disease severity such as, among others, lung clearance index and C-reactive protein. 8 In our study, only 1 out of 66 patients (1.5%) had low total IgG levels for age before ETI treatment; this too small subgroup making it difficult to compare adequately with the remaining study population.
Some authors speculated that IgA and IgG levels should be considered as a routine strategy to indirectly assess lung function impairment because their elevation is inversely related to the ppFEV135 –37 or, more generally, their levels could be a useful outcome marker in the follow-up of pwCF. 9
Regarding sputum cultures, previous studies have demonstrated that ETI treatment led to a reduction in CF pathogens40,41; however, most individuals had the same bacteria before CFTR modulator treatment. 41 In this research, it was registered a lower percentage shift to negative PsA cultures after 12 months of treatment (25%) than reported in previous studies.28,42
A notable improvement in lung function was also observed, consistent with data from the study by Middleton et al. and by Wainwright et al. 43
In our study cohort, it is interesting to note that the mean ppFEV1 at 24 months (95.50 (16.99)), although remaining higher than baseline (85.7 (17.1)), was lower than that the value reached at 12 months (100.7 (16.3)). This decline might be explained by reduced adherence or discontinuation of multiple therapies, as reported elsewhere. 44 This finding warrants further investigation in larger studies with longer follow-ups.
As previously reported in other studies,14 –16,43 SC concentrations decreased significantly, halving their baseline value after 12 months of treatment and, additionally, BMI increased.
Despite these findings, our study has limitations, including its retrospective design, small sample size recruited from only two Italian CF Centers, a small number of evaluated immune endpoints, and lack of data on concomitant medications or discontinuation of therapies after starting ETI that might influence inflammatory parameters or clinical outcomes. In addition, we did not perform a sample size calculation nor an analysis stratified by age, sex, previous treatment with other CFTR modulators, or genotype. Furthermore, the limited number of samples may have affected the statistical significance of your results. While we did not evaluate cytokine levels, these are not routinely measured in the clinical practice for CF treatment.
Conclusion
ETI treatment could reduce serum levels of IgG, IgA, and leukocytes, suggesting its potential anti-inflammatory effect and significantly improving respiratory function (as measured by ppFEV1), and nutritional status of children and adolescents with CF.
Larger-scale studies are warranted to elucidate the role of this inflammatory parameter in monitoring response to therapy.
Supplemental Material
sj-doc-1-tar-10.1177_17534666251314706 – Supplemental material for Elexacaftor/tezacaftor/ivacaftor and inflammation in children and adolescents with cystic fibrosis: a retrospective dual-center cohort study
Supplemental material, sj-doc-1-tar-10.1177_17534666251314706 for Elexacaftor/tezacaftor/ivacaftor and inflammation in children and adolescents with cystic fibrosis: a retrospective dual-center cohort study by Angela Pepe, Cristina Fevola, Daniela Dolce, Silvia Campana, Novella Ravenni, Giovanni Taccetti, Donatello Salvatore and Vito Terlizzi in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251314706 – Supplemental material for Elexacaftor/tezacaftor/ivacaftor and inflammation in children and adolescents with cystic fibrosis: a retrospective dual-center cohort study
Supplemental material, sj-docx-2-tar-10.1177_17534666251314706 for Elexacaftor/tezacaftor/ivacaftor and inflammation in children and adolescents with cystic fibrosis: a retrospective dual-center cohort study by Angela Pepe, Cristina Fevola, Daniela Dolce, Silvia Campana, Novella Ravenni, Giovanni Taccetti, Donatello Salvatore and Vito Terlizzi in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
