Abstract
Background:
Benralizumab is a monoclonal antibody treatment for severe eosinophilic asthma (SEA). Few studies investigated its role in airway inflammation and its correlation with lung function.
Objectives:
The aim of the present study is to assess its effect after 1 year of treatment, focusing on airway inflammation.
Design:
This is a retrospective observational study, in an Italian tertiary reference centre specialised in diagnosis and management of severe asthma patients.
Methods:
We conducted a monocentric retrospective study including SEA patients treated with benralizumab for 1 year. Clinical, functional and inflammatory data were collected at baseline, 6 (T6) and 12 (T12) months.
Results:
Twenty-two SEA patients on benralizumab were included. We observed a reduction in exacerbations rate and systemic steroid treatment (p < 0.0001) as well as an improvement in asthma control (p < 0.0001), health-related quality of life (p = 0.017) and lung function pre-BD FEV1 (L) (p = 0.02) and percentage (p = 0.004) and post-BD FEV1 (L) (p = 0.01) and percentage (p = 0.003) from baseline to T6 and T12. A reduction in sputum eosinophil percentage was observed at T6 and T12 (p < 0.005). We found a positive correlation between the variation of sputum eosinophils percentage and FEV1 (L) at T12 (rho = −0.79, p = 0.04). Moreover, the improvement of FEF25%–75% from baseline to 6 (rho = −0.53, p = 0.03) and 12 (rho = −0.62, p = 0.01) months negatively correlated with the duration of asthma disease.
In our cohort 12/22 patients were super-responders at T6 and 15/22 at T12. Furthermore, clinical remission was reached by 12/22, and all of them obtained blood and sputum eosinophils counts normalisation.
Conclusion:
Our data confirm that it is a rapid and effective treatment for SEA acting on clinical, functional, systemic and airway inflammatory outcomes. Our results highlight the role of induced sputum as a promising non-invasive technique to investigate pathophysiologic mechanisms in severe asthma treated with biologics. Finally, a negative correlation between small airway improvement and the duration of asthma may suggest that a prompt referral to asthma centres may delay lung function worsening. Additional studies are needed to investigate more in-depth the role of induced sputum in the management of asthma, response to treatment and remission.
Plain language summary
Background: Asthma is estimated to affect almost 5%-8% of the European adult population, and 5%–10% of these patients suffer from a severe asthma form. Severe asthma is characterised by chronic airway inflammation despite traditional inhaled treatment and patients may experience acute flare ups requiring courses of steroids and eventually hospitalization leading to a poor quality of life. New biological drugs have been introduced to treat severe asthma characterized by inflammation in the blood and in the lungs. Benralizumab is a well known biological option for severe asthmatic patients. Methods: We provide data on 22 patients followed up in our tertiary severe asthma centre in Tradate, Italy, affected by severe eosinophilic asthma treated with benralizumab for at least one year. Results: Our data confirmed a rapid effect of benralizumab on respiratory symptoms, exacerbations and quality of life. In addition, we documented a significant improvement in lung function along with a normalization of inflammation in the blood and in the lungs, assessed with a non-invasive tool: the induced sputum. Conclusions: Interestingly, our data highlight the importance of induced sputum as a promising non-invasive technique to investigate pathophysiologic mechanisms in severe asthma treated with biologics. Additional studies are needed to implement its role in the management of asthma also in terms of response to treatment towards a personalized approach.
Introduction
Asthma is a chronic airway disease characterised by recurrent respiratory symptoms, including wheezing, coughing and chest tightness, caused by usually reversible airflow limitation due to inflammation and bronchial remodelling. 1
This condition affected an estimated 330 million people in 2022 and in Europe asthma is estimated to affect between 5.1% and 8.2% of the European adult population. 2 The 5%–10% of these patients suffer from severe asthma form, characterised by poor symptom control, despite medium or high dose of inhaled corticosteroids (ICS) plus a second controller and/or systemic corticosteroids (OCS), which are often responsible for adverse events. 3
Severe asthma patients experience frequent exacerbations which lead to a reduction in pulmonary function and need for hospital admission, causing impaired quality of life for patients and relevant costs for the health system economic impact of OCS-related adverse events in severe asthma patients. 3
Severe asthma is a heterogeneous inflammatory disease with two different phenotypes according to the presence of at least one biomarker (Type 2 high) among blood eosinophilic count ⩾0.150 g/L), exhaled fraction of nitric oxide (FeNO ⩾ 20 ppb), sputum eosinophilia (⩾2%) and atopy (perennial aeroallergens positive skin tests and/or presence of specific Immunoglobulin E) or none of them (Type 2 low). 1
Eosinophilic inflammation is present in more than 50% of patients with asthma 4 and is triggered by T2 cellular responses involving T helper 2 (Th2) lymphocytes and group 2 innate lymphoid cells (ILC2). 5
The secretory pattern of these cells includes several cytokines, and among them, interleukin-5 (IL-5) plays a central role in the differentiation, growth, activation, survival and recruitment of eosinophils from blood to the airways. 6
Increased sputum and blood eosinophils count are associated with poor asthma control 7 and are one of the main risk factors for asthma exacerbations and decline in lung function,8,9 taking into account that eosinophilic airway inflammation contributes to persistent airflow limitation in severe asthma. 10
Identifying patients with T2 high phenotype is important to guide decisions on biological treatment options.
Current recommendations suggest that biological therapy for the treatment of patients with severe, uncontrolled eosinophilic asthma, including anti-interleukin-5 receptor. 1
Benralizumab is an anti-interleukin 5 receptor (IL5R) monoclonal antibody (mAb) that induces a direct, rapid and almost complete depletion of eosinophils by enhancing antibody-dependent cell-mediated cytotoxicity, an apoptotic process of eosinophil elimination involving natural killer cells. 11
Randomised clinical trials (RCT) and real-life studies have demonstrated the safety and efficacy of benralizumab in severe eosinophilic asthma (SEA) in reducing annual exacerbation rates, the burden of oral corticosteroids dependence while improving lung function and asthma control.5,12 –20
However, few real-life studies have investigated the role of benralizumab on airway inflammation and its association with lung function improvement. 21
In particular, the effects of benralizumab on airway inflammation have been poorly investigated with a non-invasive approach like induced sputum.
The aim of this real-life observational cohort study is to assess the effect of benralizumab on clinical, inflammatory and functional characteristics, focusing on airways inflammation in SEA patients investigated with induced sputum and followed up for 12 months.
Materials and methods
A retrospective cohort study was carried out between May 2019 and October 2023, in an Italian asthma reference centre (Istituti Clinici Scientifici (ICS) Maugeri, IRCCS, Tradate, Italy) specialised in diagnosis and management of severe asthma patients.
Demographic, clinical and biological data were collected in a de-identified database.
The study was performed according to the Declaration of Helsinki and approved by the Institutional Review Board of ICS Maugeri, Tradate (Identifier: p3/16). All participants gave written informed consent for their inclusion in the research. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 22
Patients and study design
All SEA outpatients treated with anti-IL5Rα (benralizumab) therapy for at least 12 months were consecutively enrolled. Data were recorded anonymously in an electronic case report form. Patients were assessed at baseline and then after 6 and 12 months of treatment with benralizumab. None of them received any biological therapy before.
Diagnosis of severe asthma was made according to GINA guidelines. 1 Benralizumab was started at the physician’s discretion in accordance to Italian Medicines Agency prescription criteria for severe asthma. It was administered subcutaneously at a dose of 30 mg every 4 weeks for the first three doses and every 8 weeks thereafter.
We reviewed patients’ electronic medical records to collect the following data: baseline characteristics (age, sex, body mass index (BMI) smoking history), asthma onset (childhood-onset or adult-onset), atopy, symptoms control, episodes of acute exacerbations, pharmacological treatment and comorbidities.
Asthma onset was defined as adult-onset when symptoms developed in adult age (⩾18 years) and childhood-onset when developed before age 18 years.
Atopy, according to the World Allergy Organisation definition, was assessed by prick tests for the most common inhalant allergens and/or through the concentration of total and specific IgE in serum (dust mites: Dermatophagoides pteronissinus and farinae, pollens: Ambrosia elatior, Betula verrucosa, Corylus avellana, Parietaria officinalis, Graminaceae mix, Olea europaea, Alms incana; moulds: Altentaria alternata and Aspergillus fumigatus; cat and dog epithelia).
The status of disease control was scored with the asthma control questionnaire (ACQ-6) 23 and Asthma Control Test (ACT). 24
The impact on overall health, daily life and perceived well-being was measured with St. George’s Respiratory Questionnaire (SGRQ). 25
Asthma exacerbations were recorded during each visit and defined as a change in symptoms and lung function from the patient’s usual status. 26
Super-responders were considered patients free of both exacerbations and oral corticosteroid therapy for asthma as defined by Kavanagh et al. 27
Response to treatment was also investigated in terms of remission on treatment according to Menzies-Gow. 28 Clinical remission was achieved after at least 12 months of treatment in case all the three following criteria were met: (1) no relevant symptoms or asthma exacerbations; (2) no systemic corticosteroid treatments; (3) lung function improvement and stabilisation. Clinical remission and the absence of inflammation or bronchial hyperresponsiveness would then define a complete on-treatment remission. 28
Functional evaluation included: Lung function tests (Pony FX Spirometer, Cosmed, Chicago, IL, USA) such as forced expiratory volume in 1 s (FEV1) (Litres, L and percentage, %, of predicted value), forced vital capacity (FVC) (L and % of predicted value), and FEV1/FVC) and forced expiratory flow rate between 25% and 75% of forced vital capacity (FEF25%–75%), were performed according to the American Thoracic Society/European Respiratory Society guidelines. 29
To assess inflammatory patterns, blood and induced sputum samples were collected during each visit in patients free of exacerbations over the previous month. Sputum was induced with hypertonic inhaled saline solution (4.5%) and processed with dithiothreitol (DTT) according to European Respiratory Society International Guidelines. 30
Inflammatory patterns were defined as follows: eosinophilic when eosinophils ⩾3% and neutrophils <61%, neutrophilic when neutrophils ⩾61% and eosinophils <3%, mixed granulocytic when eosinophils ⩾3% and neutrophils ⩾61% and paucigranulocytic when eosinophils <3% and neutrophils <61%.31,32
Statistical analysis
Qualitative variables were described with absolute and relative (percentage) frequencies, whereas quantitative ones were summarised using medians and interquartile ranges (IQR). Spearman correlation coefficients were computed to examine the relationships between non-parametric study variables. Variations across three time points (i.e. T0, T6 and T12) were assessed using repeated measures analysis of variance (Friedman test), followed by posthoc analysis adjusted for multiple comparisons using Bonferroni correction. A p-value< 0.05 was considered statistically significant. Statistical computations were performed using STATA17 software.
Results
Twenty-two SEA outpatients treated with benralizumab therapy for at least 12 months were consecutively studied taking advantage of induced sputum to evaluate inflammation. Clinical, demographic, functional and inflammatory characteristics of all patients are listed in Table 1.
Demographic, functional and inflammatory characteristics at baseline of patients treated with benralizumab.

Data are presented as Mean (±standard deviation) or Median (interquartile range).
Four patients without IS pattern.
ACT, asthma control test; ACQ6, asthma control questionnaire 6; ACQ7, asthma control questionnaire 7; BMI, body mass index; FeNO, fractional exhaled nitric oxide; FEV1, forced expiratory volume in the 1st second; FVC, forced vital capacity; FEF 25–75, forced expiratory flow rate between 25% and 75% of forced vital capacity; HFA, hydrofluoroalkane; IgE, immunoglobulin E; IS, Induced Sputum; OCS, systemic corticosteroids; Pre-BD, pre-bronchodilator spirometry; Post-BD post-bronchodilator spirometry.
SEA patients were prevalently females (68.2%), with a mean age of 56.2 years and a BMI of 25.6, uncontrolled asthma, mean exacerbation rate of 2.1 per year and poor health-related quality of life (mean SGRQ total score 26.9). Chronic rhinosinusitis with Nasal Polyposis was the most common comorbidity affecting 68.2% of patients.
They had impaired lung function with pre-bronchodilator (BD) FEV1/FVC of 69.3% and post-BD 72.7%. The majority of patients (81.8%) were on oral corticosteroids maintenance treatment with a median dose of 5 mg/day of prednisone equivalent and had received up to three courses of systemic steroids within the last year.
None of them received biological therapy before benralizumab. Their airway inflammatory patterns were as follows: 66.7% have eosinophilic asthma, 22.2% mixed granulocytic, 5.6% neutrophilic and paucigranulocytic asthma.
The patients were followed up after 6 and 12 months with the same tests performed at baseline (Table 2).
Outcomes after 6 and 12 months of benralizumab treatment.

Post-hoc analysis adjusted for multiple comparisons using Bonferroni correction; data are presented as median (interquartile range).
ACT: asthma control test; ACQ6: asthma control questionnaire 6; ACQ7: asthma control questionnaire 7; IS, Induced Sputum; FeNO: fractional exhaled nitric oxide; FEV1: forced expiratory volume in the 1st second; FVC: forced vital capacity; FEF25%–75%: forced expiratory flow rate between 25 and 75% of forced vital capacity; Pre-BD pre-bronchodilator spirometry, Post-BD post-bronchodilator spirometry
T0 versus T6 and versus T12 p < 0.0001
T0 versus T6 and versus T12 p < 0.0001
T0 versus T6 and versus T12 p < 0.0001
T0 versus T6 and versus T12 p < 0.0001
T0 versus T6 and versus T12 p < 0.0001
T0 versus T6 p = 0.003; T0 versus T12 p = 0.005
T0 versus T6 p = 0.001; T0 versus T12 p = 0.004
T0 versus T6 p = 0.007; T0 versus T12 p = 0.001
T0 versu T12 p ⩽ 0.0001; T6 versus T12 p = 0.005
T0 versus T6 p = 0.0002; T0 versus T12 p < 0.0001
T0 versus T6 p = 0.0001; T0 versus T12 p < 0.0001
T0 versus T6 p = 0.002; T0 versus T12 p = 0.022
T0 versus T6 p = 0.007
T0 versus T6 p = 0.01; T0 versus T12 p = 0.005
T0 versus T6 p = 0.002; T0 versus T12 p = 0.01
T0 versus T6 p = 0.0006; T0 versus T12 p = 0.006
T0 versus T6 p = 0.0002
T0 versus T6 p = 0.002; T0 versus T12 p = 0.001
T0 versus T6 p = 0.001; T0 versus T12 p = 0.0009
T0 versus T6 p = 0.0001; T0 versus T12 p = 0.0008.
In our cohort, we confirmed a significant reduction in the number of exacerbations rate after 6 months and maintained over time (p < 0.0001).
We observed a significant decrease in OCS dose after 6 months from 5 (4.4–6.3) to 0 (0.0–5.0) mg of prednisone equivalent (p < 0.0001), which was maintained at 12 months (p < 0.0001) with 59% and 94% of patients free of OCS therapy at 6 and 12 months respectively. No change in ICS dose was reported after 6 and 12 months.
In our cohort 54.5% (12/22) of cases were super-responders after 6 months and 68% (15/22) after 12 months. Furthermore 54.5% (12/22) were in clinical remission, and considering only this group a normalisation of blood count and sputum eosinophils percentage was observed in 100% of patients (12/12 and 6/6, respectively;).
We found a significant improvement in ACT, ACQ6 and SGRQ scores as compared to baseline (Figure 1, Panel (a)–(c)).
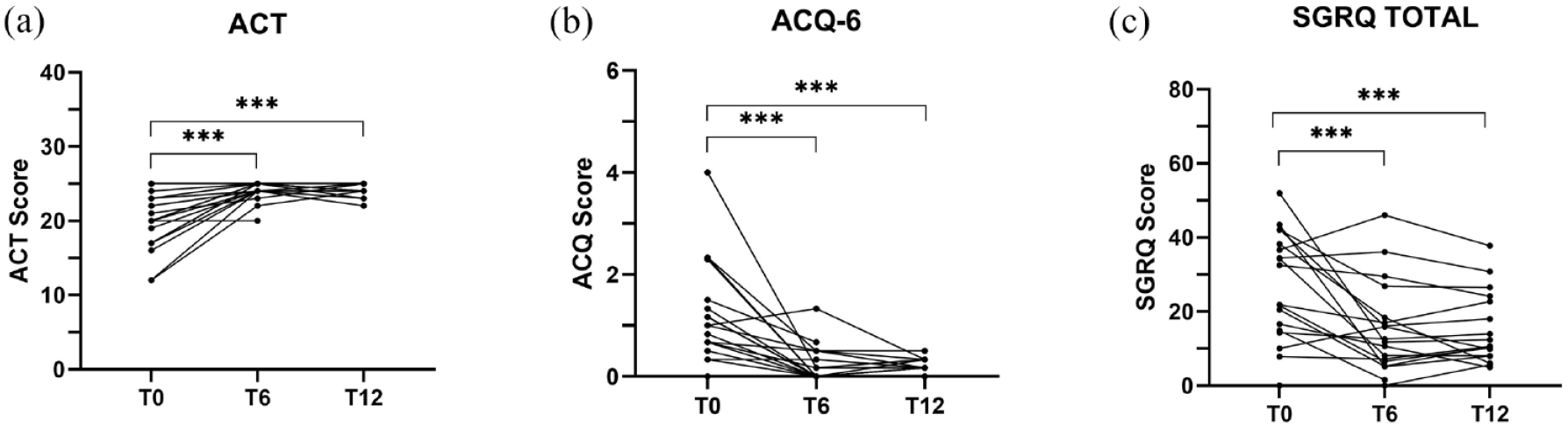
Evolution of disease control (panels (a) and (b)) and health status (panel (c)) after 6 and 12 months of treatment in SEA with benralizumab: (a) ACT, (b) ACQ-6, (c) SGRQ.
In particular, a 4 point-increase in the ACT score after 6 months (p < 0.0001) was observed; the increase was maintained up to 12 months (p < 0.0001) with almost 45% patients improving their ACT score by at least 3 points at 6 months.
Furthermore, a significant improvement in ACQ6 (p < 0.0001) (with a reduction of 0.9 points at 6 months, maintained over 12 months) was found and about 64% of treated patients improved their ACQ6 score (by at least 0.5 points).
Our data also showed a significant improvement in asthma health-related quality of life with a median change in SGRQ total score of 15 points at 6 months (p = 0.003) and 16.6 point after 12 months (p = 0.005): the minimum clinically significant difference of 4 points was reached by (11/18) 61% of the population, with (9/18) 50% of patients showing a reduction of at least 12 points.
Change in inflammatory biomarkers during treatment as compared to baseline was also analysed: a significant percent reduction in sputum eosinophils at 6 months (p = 0.001) was observed in all patients and maintained after 12 months (p = 0.004; Figure 2, Panel (a)). Eighteen patients out of twenty-two were able to cough up sputum at baseline, interestingly, the proportion of patients decreased to 39% (11/18) after 6 months and 50% (9/18) at 12 months compared to baseline.

Evolution of airway eosinophilic inflammation (panel (a)) and lung function (panels (b) and (c)) after 6 and 12 months of treatment in SEA with benralizumab.
We observed a significant improvement in lung function tests pre- and post-BD after 6 months and maintained at 12 months: pre-BD FEV1 (L) and percentage (p = 0.02 and p = 0.004 respectively); post-BD FEV1 (L) and percentage (p = 0.01 and p = 0.003 respectively; Table 2).
Patients under benralizumab showed a progressive increase in post-BD FEV1 (L) which reached 300 mL after 6 months with 68.2% of the patients improving their FEV1 (L) value and maintaining it after 12 months of treatment (Figure 2, Panel (b)).
We found a significant positive correlation between the variation of both sputum eosinophils percentage and FEV1 (L) after 12 months of benralizumab treatment (rho = −0.79, p = 0.04), whilst no correlation was found for blood eosinophil counts and FEV1 (L).
An improvement in small airways function was displayed in FEF25%–75% after 6 months (p = 0.001) of benralizumab, and maintained after 12 months (p = 0.0009) (Figure 2, Panel (c)). The improvement of FEF25%–75% from baseline to 6 (rho = −0.53, p = 0.03) and 12 (rho = −0.62, p = 0.01) months of treatment negatively correlated with the duration of asthma disease.
Discussion
In this real-life cohort study all SEA patients achieved significant improvement in clinical, functional and inflammation outcomes (evaluated through induced sputum and blood eosinophils) after a 12-month period of treatment with benralizumab in line with previous findings.
We found for the first time an inverse correlation between the decrease of induced sputum eosinophils % and the improvement in FEV1 (L) after 12 months of treatment, underlining the role of eosinophilic inflammation in lung function impairment.
Three RCTs have demonstrated a reduction in exacerbation rates, improvement in quality of life and lung function12,13,33; two RCTs15,17 have highlighted a glucocorticoid-sparing effect and recent real-life studies have also confirmed these findings.5,18,34,35
We observed a significant improvement in the first six-month period in symptom control (45% of cases), in exacerbation rate (100% of cases), in health-related quality of life (61% of cases) along with a reduction of OCS maintenance (94% of cases). This achievement has been maintained at 12 months follow-up, in line with RCTs and real-life studies.5,18,34,35
Although reduction in exacerbation rates and in OCS use has been considered the gold standard for biological therapy response, additional criteria have been recently introduced to identify patients more prone to respond to biological treatment, defined as super-responders.
In our cohort 54.5% of cases were super-responders after 6 months in line with the results of the PROMISE 34 study and 68% after 12 months similar to that reported by Miralles-Lopez et al. 36
Clinical remission was reached by 54.5% of patients and all of them obtained a resolution of asthma-related airway inflammation assessed with IS.
Our data confirm benralizumab to be a rapid and effective treatment for SEA.
The majority of patients (13/22, 59%) showed airflow limitation (post-BD FEV1 or FEV1/FVC < 75%) 10 at baseline, in the presence of blood and airway eosinophilic inflammation.
Our results demonstrated that there is a statistically significant improvement in lung function, a reduction in sputum eosinophils percentage after 6 months and a normalisation (<3% eosinophils in induced sputum) at 6 and 12 months in 91% and 100% of patients respectively. In addition, our data show an inverse correlation between the variation of both FEV1 and sputum eosinophils percentage from baseline to 6 and 12 months of benralizumab treatment in SEA being statistically significant after 12 months (p = 0.04).
Strong evidence exists on the association of airflow limitation with sputum eosinophilia in SEA patients, 10 whilst the effect of benralizumab on airway remodelling in SEA patients has been poorly investigated and limited to invasive (i.e. bronchial biopsies)37,38 and/or radiological techniques (i.e. high-resolution computed tomography). 39
Data on bronchial biopsy specimens of asthmatic patients with persistent eosinophilic airway inflammation showed an increase in the thickness of the subbasement membrane associated with increased concentrations of transforming growth factor (TGF)-β and a reduction of the airway smooth muscle (ASM) mass after benralizumab treatment. 37 Eosinophils are the major TGF-β-expressing cell types in asthmatic patients and TGF-β is an important effector for ASM proliferation and migration, highlighting the key role of benralizumab on bronchial tissue and airway eosinophilic inflammation. 37
Airway remodelling is one of the factors that may lead to lung function decline and airflow obstruction in asthma. 40 In addition, airflow obstruction caused by chronic airway inflammation in severe asthma may be also due to the presence of mucus and mucus plugs in the airways, both strongly correlated with sputum eosinophilia. 41
Interestingly, 39% and 50% of our patients were not able to cough up a sputum after 6 and 12 months of treatment respectively. This could be linked to the massive reduction in eosinophils in the bronchial tissue, in line with data on biopsies.37,38
Although airway eosinophils are involved in mucus production additional studies are needed to implement the role of IS in monitoring SEA patients on biological treatment.
As mentioned before, the effect of benralizumab on lung function improvement through spirometry has been widely explored in RCT12 –14 and real-life studies;5,42 however, scarce and conflicting literature is available on the small airway disfunction (SAD).5,43
We remarkably found a swift improvement in small airways assessed with FEF25%-75% after 6 months of benralizumab, and maintained after 12 months as in previous real-life studies.6,43 Interestingly, our results also showed a statistically significant improvement of FEF25%–75% from baseline to 6 and 12 months of treatment, negatively correlated with the duration of asthma disease. These data support the idea that patients with severe asthma should be promptly guided to referral centres to start biological treatment if needed which may delay lung function worsening.
However, the reproducibility and comparability of FEF25%–75% are limited if not adjusted for lung volume, therefore, various physiological tests have been proposed as non-invasive measurements of SAD (e.g. impulse oscillometry) which may have better sensitivity than FEF25%–75% to detect SAD. 44 Additional studies are needed to address the lack of gold-standard determinants of the small airway in severe asthma, before and after biological treatment.
Limitations
Among the main study limitations, we report the retrospective design and the small number of patients (number of eligible patients in the clinical record), related to the single-centre involvement, may affect the statistical significances of results. However, our findings lay the basis for future studies based on induced sputum analysis.
Conclusion
In conclusion, our data confirm benralizumab to be a rapid and effective treatment for SEA acting on both systemic and airway inflammation.
Analysing the induced sputum, we were able to assess not only the decrease of the blood eosinophils count but also the eosinophils percentage in the airways.
To the best of our knowledge, even if in a small group of patients, we found for the first time an inverse correlation between the decrease of induced sputum eosinophils percentage and the improvement in FEV1 (L) after 12 months of treatment, underlining the role of eosinophilic inflammation in lung function impairment. The proportion of our patients able to cough up a sputum was significantly decreased after 6 months of treatment, suggesting the possible role of induced sputum as a non-invasive tool in the evaluation of hypersecretion. Future studies on the effect of biological treatment are needed to evaluate its effect on sputum production capability.
Furthermore, a negative correlation between small airway improvement and the duration of asthma may suggest that a prompt referral to asthma centres may delay lung function worsening.
These preliminary data stressed the key role of induced sputum in phenotyping asthmatic patients and lay the basis for further studies on the effects of biologics in severe asthma.
Interestingly, our data highlight the role of induced sputum as a promising non-invasive technique to investigate pathophysiologic mechanisms in severe asthma treated with biologics. Additional studies are needed to implement its role in the management of asthma also in terms of response to treatment and remission towards a personalised management.
Footnotes
Acknowledgements
None.
Author note
An Assocaite Editor of Therapeutic Advances in Respiraotry Disease is an author of this paper. Therefore, the peer review process was managed by alternative members of the Editorial Board and the submitting Editor had no involvement in the decision-making process.
