Abstract
Background and aims:
Severe asthma may require the prescription of one of the biologic drugs currently available, using surrogate markers of airway inflammation (serum IgE levels and allergic sensitization for anti-IgE, or blood eosinophils for anti-IL5/IL5R). Our objective: to assess upper and lower airway inflammation in severe asthmatics divided according to the eligibility criteria for one of the target biologic treatments.
Methods:
We selected 91 severe asthmatics, uncontrolled despite high-dose ICS-LABA, and followed for >6 months with optimization of asthma treatment. Patients underwent clinical, functional and biological assessment, including induced sputum and nasal cytology. They were then clustered according to the eligibility criteria for omalizumab or mepolizumab/benralizumab.
Results:
Four clusters were selected: A (eligible for omalizumab, n = 23), AB (both omalizumab and mepolizumab, n = 26), B (mepolizumab, n = 22) and C (non-eligible for both omalizumab and mepolizumab, n = 20). There was no difference among clusters for asthma control (Asthma Control Test and Asthma Control Questionnaire 7), pre-bronchodilator forced expiratory volume in 1 s, serum IgE and fractional exhaled nitric oxide levels. Sputum eosinophils were numerically higher in clusters AB and B, in agreement with the higher levels of blood eosinophils. Allergic rhinitis was more frequent in clusters A and AB, while chronic rhinosinusitis with nasal polyps prevalence increased progressively from A to C. Eosinophils in nasal cytology were higher in clusters AB, B and C.
Conclusion:
Eosinophilic upper and lower airway inflammation is present in the large majority of severe asthmatics, independently from the prescription criteria for the currently available biologics, and might suggest the use of anti-IL5/IL5R or anti IL4/13 also in patients without blood eosinophilia.
The reviews of this paper are available via the supplemental material section.
Introduction
For several years, physicians interested in respiratory disease have been strongly limited in treating severe asthmatic patients because of the absence of a shared definition of severe asthma, the poor knowledge of the pathogenic mechanism drawing symptoms and clinical manifestations, and the absence of a really effective treatment other than oral corticosteroid. Nowadays, the full understanding of the specific interleukin and chemokine pattern involved in the inflammatory Type 2 cascade responsible for eosinophilic severe asthma allows to target these patients and treat them according to disease endotype.1,2
Many biomarkers have been considered to target Type 2 eosinophilic asthma. Blood eosinophilia, fractional exhaled nitric oxide (FeNO) and total IgE are the easier biomarkers to measure, able to provide biologically relevant information to aid clinical decision-making in eosinophilic and allergic asthma. 3 Moreover, sputum eosinophilia is a constant feature over time in patients with severe eosinophilic asthma 4 and eosinophilia in cytological analysis of nasal smear is considered a biomarker well related to sputum eosinophilia in patients with chronic eosinophilic airway inflammation.5,6 Currently, we have available target therapy against IgE (omalizumab), IL5 (mepolizumab), IL5 receptor (benralizumab) and anti-IL4/13 (dupilumab). Although these treatments are directed against different points of Type 2 inflammatory cascade, they may be proposed in the same patient phenotype with eosinophilic and/or allergic asthma 7 as well as in patients with nasal polyps, with or without asthma. 8
Therefore, the main challenge for the physician will be to identify the right patient for the right treatment, in order to avoid treatment failure and optimize healthcare costs.
Our study is a single centre, cross sectional, observational study in which we have enrolled patients with severe asthma who attended our clinic from 2012 to 2018 (AUOP in Pisa, Tuscany, Italy). Each patient received a complete clinical, functional and biological evaluation to identify the phenotype and the endotype of the upper and lower airway disease. At the end of the enrolment, each patient was assigned to a specific group according to the eligibility criteria needed to prescribe one of the target biologic treatments available at that time, and we compared these clusters to identify potential differences in the main clinical, biological and functional features.
We hypothesized that some clinical and functional findings may help in selecting severe asthmatics to treat with biologics, also without the evidence of the eligibility criteria for the biologic drugs, in according with the need of a multidisciplinary approach in the management and treatment of the severe uncontrolled asthma.
Furthermore, the upper and lower airway inflammation, assessed by using induced sputum and nasal cytology examination, may provide significant information to suggest a biologic treatment in a patient with severe uncontrolled asthma, supposedly not eligible for biologic treatment.
Methods
Patients and study protocol
All patients with a diagnosis of asthma according to GINA guidelines 9 were evaluated. All of them have been followed in our clinic for 1 year at least, during which the adherence to anti-asthmatic treatment was assessed and corrected, comorbidities were checked and appropriately managed and the pharmacologic treatment optimized. At the end of this screening period, patients were considered affected by severe asthma according to the ERS/ATS definition of severe asthma 10 and were enrolled in the present study. Patients already under treatment with biologic drugs were excluded; therefore, a sample size of 91 severe asthmatic patients was considered for this study.
All patients underwent a clinical, functional and biological characterization of upper and lower airway disease; then, we assigned each patient to a specific group according to the eligibility criteria to be treated with anti-IgE (omalizumab) or with anti-IL5/IL5R (mepolizumab and benralizumab, the only biologic dugs currently licensed in Italy for the treatment of severe asthma at that time). We obtained four clusters of severe asthmatic patients: cluster A (patients eligible for anti-IgE); cluster B (patients eligible for anti-IL5/5R); cluster AB (patients eligible for both anti-IgE and anti-IL5/5R); cluster C patients with no evidence of the eligibility criteria for omalizumab and mepolizumab or benralizumab.
The study had been approved by the local Ethic Committee (Comitato Etico Area Vasta Nord Ovest, CEAVNO) as a part of a large observational registry on Severe Asthma In Italy (SANI project) (no. 1245/2016) and all patients signed an informed consent for the use of personal data.
Methods
All patients enrolled in the study attended our Unit on two different days, after withdrawal of pharmacological therapy in the last 24 h (apart from inhaled salbutamol as rescue medication), to undergo the followed exams:
On the first day (Visit 1): blood analysis for eosinophils count and serum total IgE; pre and post bronchodilator spirometry, made according to European Respiratory Society reference value and using the same equipment in each patient (Elite Series plethysmography Medical Graphics, St Paul, Minnesota, USA), measurement of FeNO at a flow rate of 50 ml/s, using a chemiluminescent analyser (HypAir FeNO, Medisoft, Belgium) and collection of induced sputum for the cytological examination, according to European Respiratory Society Task Force recommendations 11 and as previously described; 12
On the second day (Visit 2), visits no more than 4 weeks apart, patients performed ENT visit with fibre-optic rhinoscopy to characterize the upper airways involvement and to define characteristic of nasal disease and inflammation. Particularly, all patients underwent nasal cytological analysis of nasal smear. The possible nasal disease diagnoses were: (a) normal (no sinonasal endoscopic alterations); (b) allergic rhinitis if at least one skin test result was positive; if not, rhinitis was defined as “non-allergic” and further specified with nasal cytology according to the predominant cellular population; (c) chronic rhinosinusitis with (CRSwNP) and without (CRSsNP) nasal polyps. CRSwNP and CRSsNP patients were further subclassified according to predominant nasal cellular population by cytological analysis.
During the first visit, for each patient were also collected: demographic and anthropometric data (age, gender, weight, height, body mass index), smoking habit, familial history of asthma, age of asthma onset and nasal disease onset, allergic sensitization and aspirin intolerance. The number of asthma exacerbations in the past 12 months and the level of oral corticosteroids treatment assumed in the past year were also recorded.
Asthma control was evaluated by Asthma Control Test (ACT) and Asthma Control Questionnaire (ACQ7). The disease-specific quality of life instruments were also adopted, such as the SinoNasal Outcome Test (SNOT) and Asthma Quality of Life Questionnaire.
Statistical analysis
Data are reported as mean ± SD or median and range for continuous normally or non-normally distributed variables, and as absolute frequencies and percentages for nominal variables. Categorical variables (gender, atopy, early onset asthma, control of asthma, exacerbations, sputum eosinophilia) were compared by Chi-Square analysis. Continuous data were compared using unpaired t-test or ANOVA test for age and pulmonary function, and Mann–Whitney or Kruskal–Wallis test for non-parametric data.
Statistical analysis was carried out using SPSS 20 (SPSS Inc., Chicago, IL, USA).
Results
We enrolled 91 patients with diagnosis of severe asthma. The asthma control, as evaluated by ACT and ACQ tests, showed a partial/poor asthma control in the majority of patients (mean values on the whole population: ACT 18 ± 4.5, ACQ 1.61 ± 1), despite a high level of pharmacologic treatment (high-dose inhaled corticosteroids/long-acting beta2 agonist (ICS/LABA) combinations in all patients, with the addition of tiotropium (in 41 out of 91) and montelukast (in 52 out of 91). In the previous year, the number of asthma exacerbations was 1.5 per patient and the average days of oral corticosteroids (OCS) use was 115 days per person. A definite nasal disease was confirmed in almost all patients, with prevalence on CRSwNP (43%) and allergic rhinitis (23%).
Fifty-three per cent of patients had a Type 2 inflammation according to blood eosinophils cut off of 300 cell/µl, and blood eosinophilia was significantly related to sputum eosinophilia, although sputum analysis was more sensitive than blood analysis in detecting the presence of eosinophilic inflammation (Figure 1); in effect, 74% of patients with blood eosinophils <300 cell/µl had sputum eosinophilia (sputum eosinophils ⩾3%) in comparison with 19% of patients with blood eosinophils ⩾300 cell/µl and no sputum eosinophilia.

Scattergram plot of sputum eosinophil percentages versus blood eosinophil levels in the whole sample of severe asthmatics.
Considering these patients according to the eligibility criteria for anti-IgE and anti-IL5/IL5R treatment, we obtained four clusters of patients: A (eligible for omalizumab), AB (eligible for both omalizumab and mepolizumab or benralizumab), B (eligible for mepolizumab or benralizumab) and C (non-eligible for both omalizumab and mepolizumab or benralizumab), composed of 23, 26, 22 and 20 patients respectively. Table 1 shows the main demographic and clinical data of these different clusters. The prevalent gender was female. There was no difference among groups in terms of mean age, asthma control (ACT and ACQ), pre-bronchodilator forced expiratory volume in 1 s and median value of serum IgE. Aspirin intolerance was more frequent in group B patients. As expected from the inclusion criteria, blood eosinophils were significantly higher in groups AB and B in comparison with groups A and C.
Demographic and clinical data of the patients divided into four clusters according to the eligibility for biologic treatment.

ACT, Asthma Control Test; AQLQ, Asthma Quality of Life Questionnaire; ASA, acetylsalicylic acid; CRS, chronic rhinosinusitis; M/F, male/female; Pre-BD FEV1, pre-bronchodilator forced expiratory volume in 1 s; SNOT, SinoNasal Outcome Test; SPT, skin prick test.
In Table 2 are reported the characteristics of upper and lower airway inflammation as well as the diagnosis of upper airway disease. Sputum eosinophils were numerically higher in groups AB and B (Figure 2), in agreement with the higher levels of blood eosinophils in these two groups in comparison with groups A and C, while FeNO levels were similar among the different groups with a tendency to have higher values in group B patients (Figure 3). The diagnosis of upper airway disease differed significantly among the groups: allergic rhinitis was more frequent in patients of groups A and AB, while CRSwNP increased progressively from group A to group C, with the highest prevalence in group C. Presence of eosinophils in nasal cytology was higher in groups AB, B and C, in comparison with A group (although this difference was not statistically significant).
Sputum inflammatory cell percentages and diagnosis of upper airway diseases of the patients divided into four clusters according to the eligibility for biologic treatment.

CRSsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; FeNO, fractional exhaled nitric oxide; Pts, patients.

Sputum eosinophil percentages in the different groups according to the eligibility for the currently available biologics.
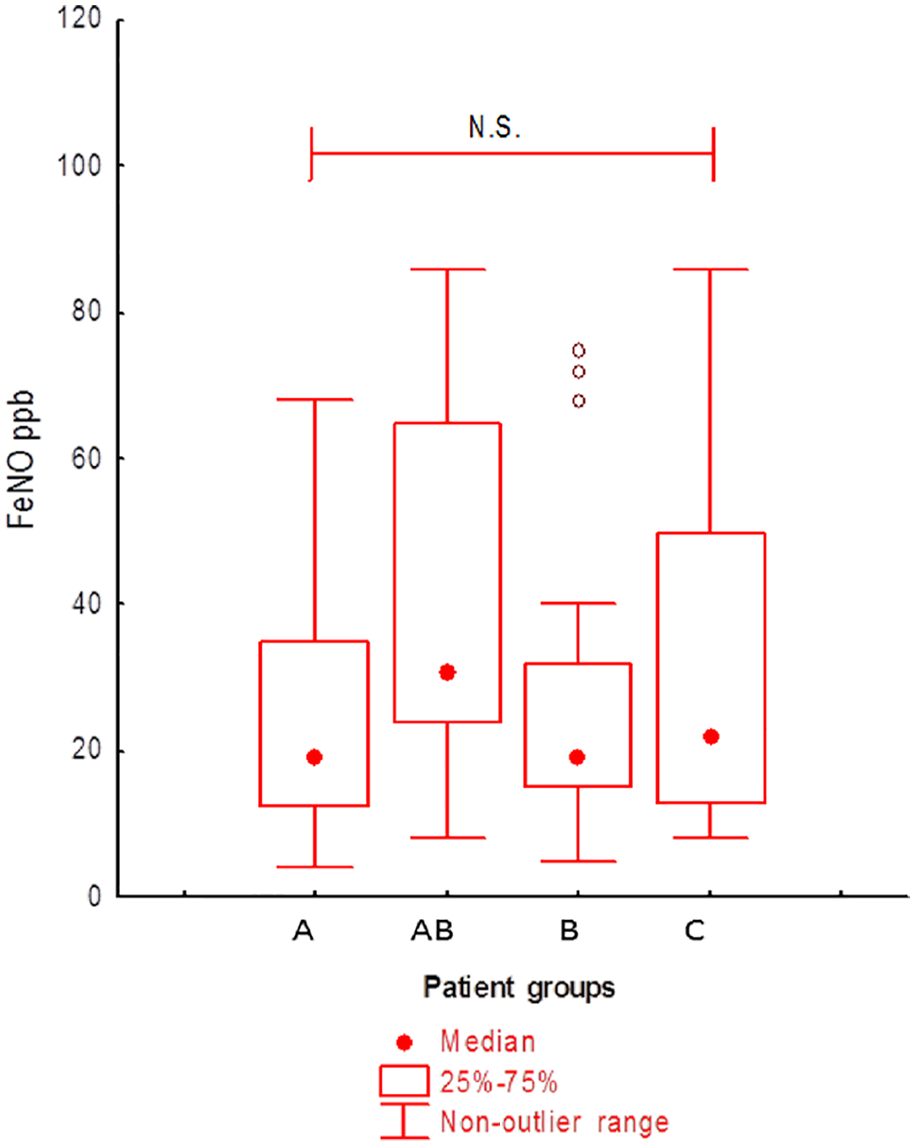
Exhaled nitric oxide levels (FeNO) in the different groups according to the eligibility for the currently available biologics.
Considering the prescription criteria of the anti/IL 4–13 (not yet available in Italy), we found a substantial possibility to use dupilumab in B and AB groups (blood eosinophils more than 150/mm3 and/or FeNO more than 25 ppb), expecting a similar improvement in asthma control to other biologics targeting Type 2 inflammation. More exciting, in the C group we have found that 50% of patients had a level of FeNO higher than 25 ppb; these patients, without evidence of blood eosinophilia and atopy, had sputum eosinophilia (median value 17%) and nasal polyps in 57% of the cases, with recurrence after surgery in 75% of patients. In addition, in this subgroup, eosinophils in nasal smear were detected in 43% of patients. In the remaining 50% of the C group patients, FeNO was lower than 25 ppb, but sputum eosinophils were high (median value 16%), nasal polyps were demonstrated in the 37% of patients, with 66% of disease recurrence after surgery.
Discussion
Eosinophilic asthma is the most frequent asthma phenotype, involving both atopic and not-atopic patients. Peripheral blood biomarkers such as blood eosinophils and total IgE are simple to detect, but not sufficient to characterize the specific endotypes of asthma. In the present study, we confirmed that upper and lower airways eosinophilic inflammation is more represented in severe asthmatics eligible for anti-IL5/IL5R, but that it is also present in patients not eligible for the currently available biologics.
The clinical impact of nasal disease was very relevant in all patients: the presence of nasal comorbidity expressed as the presence of rhinitis (allergic/non-allergic) or chronic rhinosinusitis (with/without nasal polyps), was elevated in all groups, and the mean SNOT-22 was abnormal (>9) in all groups, indicating a moderate (21–49) or severe nasal disease (⩾50). In contrast with data from the literature from real life in a very large sample of severe asthmatics, 13 in our single centre experience the nasal polyposis did not affect the control and severity of asthma more than the other forms of nasal comorbidities.
Considering the overall population of our series of severe asthmatic patients, 23 patients (A group) were clearly candidates for treatment with anti-IgE and 22 (B group) for treatment with anti-IL5 or anti-IL5 receptor. Indeed, group A patients presented allergic sensitization to perennial allergens, with high levels of IgE, no evidence of blood eosinophilia and lower local eosinophilic inflammation (median value of sputum eosinophils was 4%, FeNO was low and eosinophils were present in the nasal smear in only 13% of these patients). In this group, allergic rhinitis was the prevalent diagnosis of upper airway disease. Eighty-seven per cent of the patients of this group were poly-sensitized allergic patients.
On the other hand, hypereosinophilia was the main feature of patients of the B group. Indeed, they had both airway (high level of eosinophils in sputum, nasal mucosa and high level of FeNO) and peripheral eosinophilic inflammation. The 36% of patients of this group presented atopy but only for seasonal allergens and the median value of IgE was similar to the other groups. CRSwNP was the prevalent nasal disease diagnosis (in 50% of patients) and the Sampter’s triad affected 23% of patients. Therefore, a target treatment against IL5 or its receptor is strongly indicated in this group of patients.
Patients of AB group could be proposed for both anti IgE and anti IL5 or IL5 receptor treatment. In the literature it is well described that the prevalence of atopy increases with level of eosinophilia 14 and we observe that this group of patients presented the same median value of IgE as the other groups. According to the literature, also in our patients the level of IgE, similar in all groups, cannot be considered predictive of response to target treatment and cannot help us to choose the best treatment. 15 On the other hand, the majority of these patients presented a very high level of blood eosinophilia, but recent data of the literature, from real life observation, shows that blood eosinophilia is not always predictive of the clinical response to anti IL5 treatment. 16 On the other hand, our results underline that the combination of blood eosinophilia with local eosinophilic inflammation (eosinophils in sputum and nasal smear, high FeNO levels) might support the choice of a treatment with anti IL5 or IL5R. Additional clinical features, such as the presence of nasal polyps, should be also considered to orient the choice of treatment because of the poor response to the anti-IgE therapy described in the literature in patients with comorbidities such as nasal polyps17,18 and the initial data of the efficacy of anti-IL5 on nasal polyps treatment. 19
Finally, group C patients resulted not eligible for the target treatments currently available in our country, due to the absence of atopy for perennial allergen and to the low level of blood eosinophils. Our study provides for this group of patients some evidence of a dissociation between the level of peripheral inflammation, which was low, and local eosinophilic inflammation, which was often increased, with evidence of eosinophilic inflammation similar to the B group. We can also observe that a significant number of these patients had nasal polyposis. Considered together, these features strongly suggest the prescription of anti-IL4-13 in these patients, regardless of blood eosinophilia or FeNO level. In these patients, the optimal therapeutic choice should be based on patients’ characteristics and positive patients’ feedback towards the possible therapeutic schemes of biologics proposed. However, more studies are needed to identify specific mediators involved in the activation of upper airway-Type 2 inflammation before proposing biologic treatment in patients with such biologic features.
This observation confirms that there is a subgroup of severe asthmatic patients with activation of Type 2 inflammatory cascade who may have low level of peripheral eosinophilic inflammation in front of a significant eosinophilic recruitment into upper and lower airway tissues. In this group of patients, allergens do not seem to be the main triggers for this sustained eosinophilic inflammation. Therefore, we can assume an overexpression of cytokines involved in eosinophils recruitment in the airway, such as eotaxin 3 (CCL26).
Nowadays, target treatment, such as anti IL-4/13, capable of blocking eosinophils cascade activation and the cytokines involved in the tissue recruitment of eosinophils with minimal peripheral eosinophilia, are close to being available on the market. 20 This new type of treatment might be proposed in patients with a dissociation between airway and blood eosinophilic inflammation, as demonstrated by the subgroup analysis of a recent randomized clinical trial with dupilumab. 21
The discrepancy between local and peripheral eosinophilic inflammation is well known: in some studies, a consistent proportion (ranging from 30% to 70%) of severe asthmatics had sputum eosinophilia with normal values of blood eosinophils.22–24 We can suppose that this partial correlation between local and peripheral eosinophilic inflammation may be due to “single spot” assessment of these patients, and that repeated measurements of blood eosinophils may catch in these patients an occasional blood eosinophilia which may make these patients candidates for an anti IL5 or IL5R treatment. On the other hand, we have demonstrated in a group of severe asthmatics who performed repeated sputum measurements over years, that sputum eosinophilia was a constant feature in more than 80% of these patients. 4 The degree of concordance between blood and sputum eosinophilia should be confirmed with repeated measurements of both blood and sputum eosinophils in the same severe asthmatics; at the present time, this information is still lacking. Longitudinal observations on large groups of severe asthmatics (as obtained with the use of national registries) might clarify this point. 25
Some papers have been recently published with the aim to characterize the proportion of severe asthmatics who are candidates for the current biologic treatments, according to the prescriptive criteria in the different countries.26–28 These studies, some of them with a sample size comparable to that of our study, showed that a majority of severe asthmatics were eligible for at least one biologic drug, with a large overlap between anti-IgE and anti-IL5/5R. Differently from these papers, our study investigated the presence of eosinophilic inflammation in upper and lower airways, showing that, in patients without prescriptive criteria for anti-IgE or anti-IL5/5R, eosinophilic inflammation in induced sputum and/or nasal cytology might indicate the use of anti-eosinophilic biologic drugs, including also anti-IL4/13, therefore increasing consistently the possibility to manage appropriately this subgroup of severe asthmatics.
In conclusion, phenotyping severe asthmatic patients is essential to recognize specific endotypes of disease and to guide target treatment in a single severe asthmatic patient. Perennial atopy, in a patient with low airway and blood eosinophilic inflammation and allergic rhinitis, supports the choice for the anti-IgE treatment. The concordance between airway and blood eosinophilic inflammation, particularly in a patient with nasal polyps, should orient for anti-IL5/5 R target treatment. In atopic patients with evidence of blood eosinophilic inflammation, the level of blood eosinophilia and not the IgE level should guide the treatment choice; the level of airway eosinophilic inflammation and some clinical features such as nasal polyps or allergic rhinitis should be also considered. A severe asthmatic patient, non-atopic and non-eligible for anti IL5/5R treatment, should be evaluated for local eosinophilic inflammation (cytological examination of sputum and nasal smear, as suggested by Global Initiative for Asthma documents). 7 Indeed, this patient might have local airway inflammation caused by a different way of activation of eosinophilic inflammatory cascade, with more production of cytokines involved in the local recruitment of the eosinophils. This type of patient might benefit from a different type of target treatment such as anti-IL4/13.
A multidisciplinary approach, including at least pulmonology, allergist and ENT specialist, is needed to evaluate and treat a patient with severe uncontrolled asthma, particularly if he does not seem eligible for a biologic treatment. In these types of patients, frequently affected by multi-morbidities driven by Type 2 inflammation, the cytological examination of upper and lower airway disease should always be considered.
Supplemental Material
sj-pdf-1-tar-10.1177_1753466620965151 – Supplemental material for Upper and lower airway inflammation in severe asthmatics: a guide for a precision biologic treatment
Supplemental material, sj-pdf-1-tar-10.1177_1753466620965151 for Upper and lower airway inflammation in severe asthmatics: a guide for a precision biologic treatment by Manuela Latorre, Elena Bacci, Veronica Seccia, Maria Laura Bartoli, Cristina Cardini, Silvana Cianchetti, Ludovica Cristofani, Antonella Di Franco, Mario Miccoli, Ilaria Puxeddu, Alessandro Celi and Pierluigi Paggiaro in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_1753466620965151 – Supplemental material for Upper and lower airway inflammation in severe asthmatics: a guide for a precision biologic treatment
Supplemental material, sj-pdf-2-tar-10.1177_1753466620965151 for Upper and lower airway inflammation in severe asthmatics: a guide for a precision biologic treatment by Manuela Latorre, Elena Bacci, Veronica Seccia, Maria Laura Bartoli, Cristina Cardini, Silvana Cianchetti, Ludovica Cristofani, Antonella Di Franco, Mario Miccoli, Ilaria Puxeddu, Alessandro Celi and Pierluigi Paggiaro in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_1753466620965151 – Supplemental material for Upper and lower airway inflammation in severe asthmatics: a guide for a precision biologic treatment
Supplemental material, sj-pdf-3-tar-10.1177_1753466620965151 for Upper and lower airway inflammation in severe asthmatics: a guide for a precision biologic treatment by Manuela Latorre, Elena Bacci, Veronica Seccia, Maria Laura Bartoli, Cristina Cardini, Silvana Cianchetti, Ludovica Cristofani, Antonella Di Franco, Mario Miccoli, Ilaria Puxeddu, Alessandro Celi and Pierluigi Paggiaro in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The study was part of the participation of our centre in the national severe asthma registry (SANI).
Author contribution(s)
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
