Abstract
Background:
The presence of anti-melanoma differentiation-associated gene 5 (MDA5) antibodies in dermatomyositis (DM) is associated with an increased risk of developing rapidly progressive interstitial lung disease (RP-ILD) and a poor prognosis.
Objectives:
We aimed to explore whether tofacitinib could improve the prognosis of Anti-MDA5 antibody positive DM-interstitial lung disease (ILD).
Design:
Systematic review and meta-analysis.
Data Sources and Methods:
Studies were included if they compared mortality rate and infection events in patients with anti-MDA5 antibody positive DM-associated ILD who were treated with or without tofacitinib.
Results:
The systematic review and meta-analysis included a total of 148 patients from four cohort studies. Fifty-eight patients with anti-MDA5 antibody positive DM-ILD who received combined treatment-containing tofacitinib were enrolled in the experimental group. Additionally, 90 DM-ILD patients who did not receive tofacitinib-based therapy were included in the control group. The pooled risk ratio (RR) for all-cause mortality was 0.61 (95% CI, 0.41–0.91, p = 0.02) with I2 = 0 indicating no heterogeneity among the included studies. For virus infection risk, the pooled RR was 1.92 (95% CI, 0.90–4.10, p = 0.09), while bacterial and fungal infection-associated RRs were found to be 1.29 (95% CI, 0.65–2.55, p = 0.47) and 1.15 (95% CI, 0.46–2.89, p = 0.77), respectively. There was no statistically significant difference in infection risk between the two groups, and no heterogeneity was observed.
Conclusion:
Our findings suggest that tofacitinib may reduce the risk of all-cause mortality in patients with anti-MDA5 antibody-positive DM-ILD without an increased risk of additional infections.
Trial registration:
PROSPERO: CRD42023445427; https://www.crd.york.ac.uk/prospero/
Plain language summary
Keywords
Introduction
The presence of anti-melanoma differentiation-associated gene 5 (MDA5) antibodies in dermatomyositis (DM) indicates an important subtype of DM, 1 which is prone to rapidly developing progressive interstitial lung disease (RP-ILD). 2 Despite the application of conventional treatments involving high-dose glucocorticoids (GCs) combined with cyclophosphamide (CYC), or calcineurin inhibitors such as tacrolimus (TAC) and cyclosporine A (CsA), the 6-month mortality rate remains alarmingly high at 50%. 3 Therefore, it is imperative for us to explore novel therapeutic approaches aiming at controlling the rapid progression of this disease.
Tofacitinib, a Janus kinase (JAK) inhibitor targeting JAK1 and JAK3, exerts its function by inhibiting the phosphorylation and activation of JAKs. This leads to the prevention of signal transducer and activator of transcription (STAT) phosphorylation and subsequent gene transcription, ultimately resulting in decreased cytokine production and modulation of the immune response. 4 The effectiveness and safety of tofacitinib have been validated in the treatment of various conditions, including ulcerative colitis (UC), 5 rheumatoid arthritis (RA), 6 and psoriatic arthritis. 7
The pathogenesis of DM is not fully understood, type I IFN signaling plays a crucial role in the development of this disease, which is activated in the muscle and skin of patients with DM. 8 Inhibiting the JAK-STAT pathway has been found to reduce IFN signaling, supporting the mechanism of action of tofacitinib. Additionally, in vitro research has shown that tofacitinib can inhibit the pro-inflammatory and profibrotic effects by targeting T cells derived from amyopathic dermatomyositis (ADM)-ILD patients. 9 Clinically, several published studies with small sample sizes have demonstrated the usefulness of tofacitinib in treating anti-MDA5 antibody-positive DM-associated ILD.10,11
In this systematic review and meta-analysis, we aimed to explore whether tofacitinib could improve the prognosis of anti-MDA5 antibody-positive DM-ILD.
Methods
The search flow diagram, which depicts the systematic review and meta-analysis, is presented in Figure 1. The protocol was conducted in accordance with the PRISMA statement. 12

Study selection algorithm.
Search strategy and study selection
The MEDLINE and Embase databases were systematically searched from their inception until July 8, 2023. The search strategy used the term “Dermatomyositis” OR “DM” OR “Idiopathic Inflammatory Myopathy” AND “Tofacitinib” (Supplemental Document 1).
Inclusion criteria
We included studies with patients diagnosed with DM according to the modified Sontheimer’s definition 13 or Bohan–Peter’s classification criteria 14 or 2017 European League Against Rheumatism/American College of Rheumatology (EULAR/ACR) classification criteria. 15 Additionally, patients had to be anti-MDA5 antibody-positive and had interstitial lesions confirmed by pulmonary computed tomography. The experimental group received treatment with Tofacitinib. Language was not a limiting factor.
Exclusion criteria
Two investigators (Ruyi Zou and Jie Wei) independently reviewed the titles and abstracts of all included studies. Studies were excluded if (1) a full article was unavailable for review, (2) juvenile dermatomyositis, (3) involved animal, or in vitro research, (4) case report or review.
After assessing 13 studies, a total of 9 were excluded: 1 study was excluded due to insufficient survival data, and 8 studies were excluded because they lacked control group. Finally, four eligible studies were included in this study.
Data extraction
Two investigators (Yanhong Wang and Junjie Wang) independently extracted data from each study, including author names, geographical distribution, year of publication, study enrolment period, participant basic characteristics (number, age, sex), infection events, and the number of deaths in both groups. Disagreements between two investigators were settled by arbitration of the principal investigator (Minjie Lin).
Quality assessment
All the included studies are observational researches, and the assessment of bias was conducted using the Newcastle–Ottawa scale. 16 The risk of bias for the included studies was independently assessed by two investigators (Yanhong Wang and Cheng Tang), with any discrepancies resolved through arbitration of the principal investigator (Minjie Lin). A score ⩾ 5 was defined as “low risk of bias,” while studies with scores <5 were regarded as “high risk of bias.”
Outcomes
The primary outcome of this study was to compare risk of all-cause mortality and infection events between anti-MDA5 antibody-positive DM patients with ILD treated with or without tofacitinib. The effect size was estimated as risk ratio (RR) with 95% confident interval (CI).
Statistics
We extracted the survival rate and number of infection events in two groups from eligible studies, and pooled the data using a random-effect model.
The statistical heterogeneity of effect size across the included studies was assessed using both the Cochrane Q test and I2 statistic. p-Value of <0.1 on the Q test was considered statistically significant. Heterogeneity levels were categorized as acceptable (0%–50%) or high (51%–100%). 17 Funnel plot, Egger’s test, and Begg’s test were utilized to evaluate publication bias. All statistical analyses were conducted using RevMan5.4 and Stata 11 SE.
Results
Out of the 444 articles or abstracts initially identified in our search, 431 were excluded after a thorough review of their titles and abstracts. Subsequently, we conducted a full-text review of 13 articles and ultimately included four in our final meta-analysis (Figure 1).3,18–20
Characteristics of the included studies
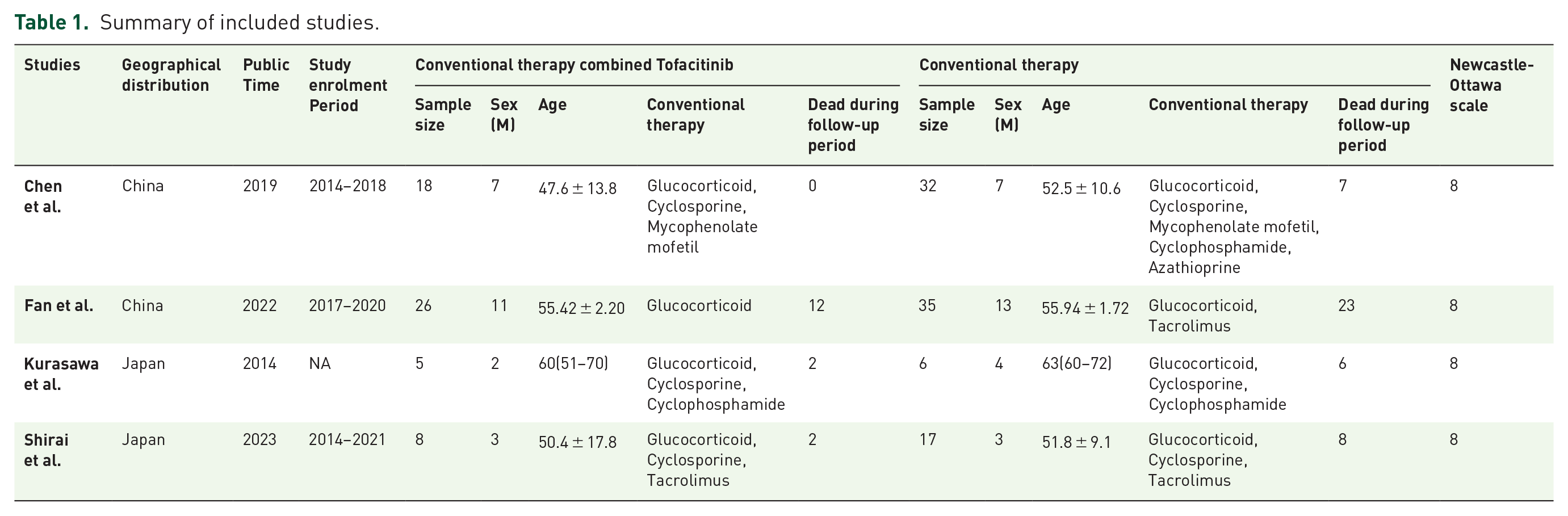
The included studies consisted solely of cohort studies. A total of 58 patients with anti-MDA5 antibody-positive dermatomyositis-associated interstitial lung disease (DM-ILD) who received combined treatment containing tofacitinib were enrolled in the meta-analysis. Additionally, 90 DM-ILD patients who did not receive tofacitinib-based therapy were included in the control group. All studies were evaluated using the Newcastle–Ottawa scale, and a score of 8 was achieved (Table 1), indicating that the included studies are of high quality.
Summary of included studies.
Efficacy and infection events of tofacitinib
The pooled RR for all-cause mortality was 0.61 (95% CI, 0.41–0.91, p = 0.02), with an I2 value of 0. In tofacitinib group, 16 out of 57 patients died during follow-up period, resulting in a mortality rate of 28.07%, while in the control group, there were 44 deaths out of 90 patients, resulting in a mortality rate of 48.89% (Figure 2). The pooled RR for virus infection risk was 1.92 (95% CI, 0.90–4.10, p = 0.09; Figure 3), and bacterial infection-associated RR was 1.29 (95% CI, 0.65–2.55, p = 0.47; Figure 4), and for fungal infection, the associated RR was calculated as1.15 (95% CI, 0.46–2.89, p = 0.77; Figure 5). In the tofacitinib group, 10/57(17.54%) patients suffered from virus infection, 6/31 (19.35%) suffered from bacterial infection, and 7/31 (22.58%) suffered from fungal infection when the control group were 7/41 (17.07%), 5/41 (12.20%), and 8/41 (19.51%) respectively, with no heterogeneity found (I2 = 0; Figure 3–5). Only two studies were included in Figures 3–5, because the other studies did not contain the needed information about infection.

Forest plot for pooled risk ratio for all-cause mortality.

Forest plot for pooled risk ratio for virus infection risk.

Forest plot for pooled risk ratio for bacterial infection risk.

Forest plot for pooled risk ratio for fungal infection risk.
Evaluation of publication bias
The funnel plot appeared to indicate potential publication bias across the four studies using RevMan 5.4 (Supplemental Figure 1). However, after reanalysis with Egger’s and Begg’s methods using Stata 11 SE (Supplemental Figures 2 and 3, Supplement Table 2), there was no statistical significance. Therefore, we concluded that studies included in the analysis did not have publication bias.
Discussion
This systematic review and meta-analysis included 148 patients from 4 studies (58 anti-MDA5 antibody positive DM- ILD treated with tofacitinib and 90 patients without tofacitinib). The findings suggest that tofacitinib may reduce the risk of all-cause mortality. Regarding infection risk, there was no statistical significance in the occurrence of viral, bacterial, or fungal infections between the two groups.
Patients with anti-MDA5 antibody-positive DM are prone to developing ILD, particularly RP-ILD. 21 Although the specific pathogenic mechanisms of anti-MDA5 antibody-positive DM-ILD are unknown, potential environmental triggers, especially viral infections and genetic susceptibility have been indicated to play a role in this disease. 22 MDA5 protein is encoded by the interferon-induced helicase C domain 1 (IFIH1) gene. It functions as a sensor for detecting the presence of viruses and gets activated by viral RNA. Its activation is necessary for viral immunity but can also lead to interferon-driven auto-inflammatory diseases. 22 Certain human leukocyte antigen (HLA) regions, such as HLA-DRB1*0101, *0405, *1201, *0901, *0401, and *1202 have been found to be associated with susceptibility to anti-MDA5 antibody positive DM.23–25 During this process, several components of the type I IFN signaling pathways play crucial roles including IFN-induced protein with tetratricopeptide repeats 2(IFIT2), IFN-induced protein with tetratricopeptide repeats 3(IFIT3), myxovirus resistance 1 (MX1), interferon regulatory factor 7 (IRF7), C-C motif ligand 2 (CCL2), and clusterin (GLU). 26
Due to the high mortality rate of anti-MDA5 antibody-positive DM, especially combined with ILD, there is an urgent need for novel therapies. Currently, pirfenidone, plasma exchange, rituximab, intravenous immunoglobulin, and tofacitinib are being applied (1). Tofacitinib, a JAK inhibitor widely used in rheumatoid arthritis (RA), has shown efficacy as a monotherapy in reducing signs and symptoms of RA and improving physical function. 27 Up to 8.5 years’ follow-up results have demonstrated a low incidence of adverse events including opportunistic infections, tuberculosis, tumors, cardiovascular events, and gastrointestinal perforation. 28 Additionally, a survey conducted in the United States using email questionnaires found that 56.4% of patients were more inclined to choose oral medication such as tofacitinib over biologics that required injection. 29 Based on its efficacy, safety, and convenience, tofacitinib has been applied in the treatment of anti-MDA5 antibody-positive DM. Chen et al.’s 3 study demonstrates that compared to a historical control group (25 out of 32, 78%), a combination regimen with tofacitinib can improve the survival rate of anti-MDA5 antibody-positive DM-ILD within 6 months after ILD onset (18 out of 18, 100%). In a prospective open-label pilot study on refractory DM, patients treated with tofacitinib showed an improvement in median total improvement score (TIS) with five out of ten patients (50%) experiencing moderate improvement and the other half experiencing minimal improvement on TIS. Additionally, the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) significantly decreased from 28 ± 15.4 to 9.5 ± 8.5 (p = 0.0005). 30 In newly onset adults with anti-MDA5 antibody-positive DM, tofacitinib also demonstrated efficacy. According to Wang et al.s’ 10 study, responses were observed in 10 patients (71.4%) after 6 months of treatment with GCs combined with tofacitinib, with two patients achieving major improvement, seven achieving moderate improvement, and one achieving minimal improvement. Furthermore, studies about anti-MDA5 antibody-positive DM suggested that the rate of adverse events of tofacitinib was very low.3,18 Tofacitinib has two dosages, an 11 mg once-daily extended-release formulation and a 5 mg twice-daily immediate-release formulation. In our four included studies, three studies used the 5 mg immediate-release formulation, while one study did not describe the drug dosage and formulation. Observations by Wang et al. 31 suggest that the extended-release tofacitinib formulations have therapeutic efficacy in controlling disease activity of refractory Behçet’s disease. Considering the convenience of the extended-release formulation, we plan to use it in the future in our center for anti-MDA5 antibody-positive DM-ILD.
Advantages and limitations
To the best of our knowledge, this is the first systematic review and meta-analysis that focuses on the efficacy and safety of tofacitinib in anti-MDA5 positive DM-ILD. Furthermore, all results demonstrate homogeneity, which enhances the robustness of our conclusions. However, our study has several limitations. Notably, the number of patients included in our study is small. Second, all four studies included are cohort studies and prospective randomized controlled trials are needed. Additionally, there were variations in the severity of anti-MDA5 antibody-positive DM-ILD among patients, and severe cases were more likely to receive tofacitinib, potentially leading to confounding and selection bias. Finally, all four studies involved patients from Japan and China, and different human races may exhibit distinct clinical characteristics and responses to tofacitinib.
Conclusion
Our findings suggest that tofacitinib could decrease the risk of all-cause mortality in patients with anti-MDA5 antibody-positive DM-ILD without increasing infection risk. We look forward to more randomized controlled trials being carried out in the future to validate this conclusion.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241294000 – Supplemental material for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tar-10.1177_17534666241294000 for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis by Yanhong Wang, Ruyi Zou, Jie Wei, Cheng Tang, Junjie Wang and Minjie Lin in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666241294000 – Supplemental material for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tar-10.1177_17534666241294000 for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis by Yanhong Wang, Ruyi Zou, Jie Wei, Cheng Tang, Junjie Wang and Minjie Lin in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-3-tar-10.1177_17534666241294000 – Supplemental material for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tar-10.1177_17534666241294000 for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis by Yanhong Wang, Ruyi Zou, Jie Wei, Cheng Tang, Junjie Wang and Minjie Lin in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-png-4-tar-10.1177_17534666241294000 – Supplemental material for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis
Supplemental material, sj-png-4-tar-10.1177_17534666241294000 for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis by Yanhong Wang, Ruyi Zou, Jie Wei, Cheng Tang, Junjie Wang and Minjie Lin in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-png-5-tar-10.1177_17534666241294000 – Supplemental material for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis
Supplemental material, sj-png-5-tar-10.1177_17534666241294000 for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis by Yanhong Wang, Ruyi Zou, Jie Wei, Cheng Tang, Junjie Wang and Minjie Lin in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-png-6-tar-10.1177_17534666241294000 – Supplemental material for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis
Supplemental material, sj-png-6-tar-10.1177_17534666241294000 for The efficacy and safety of tofacitinib in anti-melanoma differentiation-associated gene 5 antibody positive dermatomyositis associated interstitial lung disease: a systematic review and meta-analysis by Yanhong Wang, Ruyi Zou, Jie Wei, Cheng Tang, Junjie Wang and Minjie Lin in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
