Abstract
Background:
A six-minute walk test (6MWT) is a reproducible, easily performed test, and is widely used to determine functional exercise capacity in patients with idiopathic pulmonary fibrosis (IPF). However, there is currently a paucity of data on the clinical significance of baseline and serial 6-minute walk tests in patients with IPF, especially in Asian patients.
Objectives:
We aimed to investigate the clinical significance of serial 6MWT in patients with IPF, especially in Asian patients.
Design:
This is a single-center retrospective cohort study.
Methods:
Clinical data of patients diagnosed with IPF at a tertiary center in Korea were retrospectively analyzed. IPF diagnosis was defined according to the clinical guidelines of the American Thoracic Society (ATS)/European Respiratory Society (ERS)/Japanese Respiratory Society/Latin American Thoracic Association.
Results:
There were 216 patients diagnosed with IPF from December 2012 to January 2022, of whom 198 had a baseline of 6MWT data. The mean age of the cohort was 66.9 ± 8.6, and 89% were male. The non-survivors showed significantly lower six-minute walk distance (6MWD), minimum saturation of peripheral oxygen (SpO2) during 6MWT, forced vital capacity, and diffusing capacity of the lung for carbon monoxide than survivors at baseline. A multivariate Cox analysis demonstrated that lower minimum SpO2 was independently associated with increased mortality rates (Hazard ratio (HR): 1.081, 95% confidence interval (CI): 1.024–1.142, p = 0.005). Higher mortality rates were also associated with echocardiographic-determined pulmonary hypertension (HR: 2.466, 95% CI: 1.149–5.296, p = 0.021) at diagnosis. Among 144 patients with 6MWT results at 12 months, patients with a decline of 50 m or more in the 6MWD showed poorer overall survival than others (median survival: 45.0 months vs 58.0 months, p < 0.001).
Conclusions:
Baseline lower minimum SpO2 during 6MWT was an independent prognostic factor in patients with IPF, and a decline in 6MWD in serial follow-up was also associated with a poorer prognosis. These findings suggest that both baseline 6MWT and follow-up data are important in the prognostication of patients with IPF.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, and ultimately fatal fibrosing interstitial lung disease with a median survival of 3–5 years. 1 Although antifibrotics have been shown to improve overall survival and slow the decline in lung function,2–5 the prognosis remains poor. Several prognostic factors for IPF have been identified, including low forced vital capacity (FVC), low diffusing capacity of the lung for carbon monoxide (DLco), and the composite GAP index which is composed of gender, age, and physiology (% predicted FVC and DLco).6–8
The six-minute walk test (6MWT) is a reproducible, easily performed, and widely used test to determine functional capacity in patients with various cardiopulmonary diseases including heart failure, chronic obstructive pulmonary disease, pulmonary hypertension (PH), and IPF. 9 Since it is affected by many factors other than the severity of lung disease, including frailty, deconditioning, and musculoskeletal disorders, it provides important information about patients’ overall functional ability. 10 Several studies have demonstrated the significant utility of the 6MWT in assessing disease status and in playing a prognostic role in patients with IPF.11–14 However, there are currently few large-scale studies regarding the clinical significance of serial 6MWT and its predictive ability in determining prognosis in patients with IPF, especially in Asian patients. Therefore, we aimed to evaluate the clinical implications of baseline and serial 6MWT in patients with IPF.
Material and methods
Study population
There were 216 patients diagnosed with IPF who were initially screened between December 2012 and January 2022. Eighteen patients without available baseline 6MWT results were excluded. Finally, 198 patients who had baseline 6MWT data comprised the final cohort.
IPF diagnosis was performed by using the diagnostic criteria set by the American Thoracic Society (ATS)/European Respiratory Society (ERS)/Japanese Respiratory Society/ Latin American Thoracic Association in 2018. 15 Patients with definite connective tissue disease or an exposure history related to the possible causes of interstitial lung diseases were excluded. For patients diagnosed before 2018, the diagnosis of IPF was confirmed through discussions on whether IPF diagnosis was appropriate according to the recent international guideline. 15 In cases where classification was ambiguous or surgical biopsy was performed, a multidisciplinary approach was employed.
Data collection
Clinical and survival data for all patients were collected from the medical records of the Asan Medical Center, at the time of their initial IPF diagnosis. FVC, and the single breath diffusing capacity for carbon monoxide (DLco) were measured according to the ATS/European Respiratory Society recommendations16–18 and expressed as percentages of the normal predicted values. The 6MWT was performed according to the ERS/ATS recommendations. 19 Echocardiographic PH (intermediate to high probability) was defined as a ⩾2.9 m/s maximal tricuspid regurgitation velocity (TR Vmax) according to the 2022 European Society of Cardiology/European Respiratory Society guidelines. 20 Lung transplantation was considered equivalent to death in terms of outcomes. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guideline. 21 The study protocol was approved by the Institutional Review Board of Asan Medical Center (IRB No.: 2023-1105). The requirement for informed consent was waived due to the retrospective nature of the study.
Statistical analysis
All data are expressed as mean ± standard deviation or median (interquartile ranges) for continuous variables and percentages for categorical variables. The student’s t-test was used for continuous data, and Pearson’s chi-square test or Fisher’s exact test was used for categorical data. In this study, we employed a SpO2 cutoff value of 88% during 6MWT and a decline in 6MWD of 50 m to analyze clinical outcomes based on its importance established in several studies on the prognosis of patients with interstitial lung diseases.9,22–25 The Kaplan–Meier estimates and the log-rank test were used for survival analysis. The follow-up time was calculated from the date of IPF diagnosis to the date of death or time of censoring (July 2023). Risk factors for all-cause mortality were analyzed using the Cox proportional hazards analysis. Variables with p < 0.1 in the unadjusted analysis along with those considered clinically significant were entered into multivariable models. When two variables demonstrated a strong correlation (Pearson’s correlation coefficient >0.6), only one was selected for inclusion in the multivariate analysis to avoid overfitting. Additionally, variables with excessive missing data were excluded from the multivariate analysis logistic regression analysis was used for the determination of risk factors for PHT. p-values <0.05 were considered statistically significant. Statistical analysis was performed by R software version 4.2.1 (the R Foundation, Vienna, Austria).
Results
Baseline characteristics
The baseline characteristics of the 198 patients who formed the final cohort are shown in Table 1. Of these, there were 64 (32.3%) with biopsy-proven IPF. The mean age of the cohort was 66.9 years, and 89% were male. The median follow-up period was 28.5 months (interquartile range: 20–37 months). During the follow-up period, 78 patients were transplanted (n = 41) or died. The non-survivors were younger (mean age: 64.6 vs 68.4, p = 0.002), had lower FVCs (mean values: 69.9 vs 75.5, p = 0.008), lower DLco values (mean values: 48.5 vs 59.5, p < 0.001), shorter 6MWD (mean values: 400.1 vs 437.4, p = 0.012), lower minimum SPO2 values during the 6MWT (mean values: 88.9 vs 92.1, p < 0.001), and had more frequent echocardiographic PHs at IPF diagnosis (16.4% vs 3.2%, p = 0.003), compared to the survivors. During the follow-up, 74 patients (37.3%) experienced an acute exacerbation (AE), and non-survivors experienced AE more often than survivors (62.8% vs 20.8%, p < 0.001). Most patients (94.4%) received pirfenidone, and 9% received nintedanib of which six patients (3.0%) had a history of taking both drugs. There was no significant difference in antifibrotics use between non-survivors and survivors (Table 1).
Comparison of baseline characteristics between the non-survivors and survivors among patients with IPF.

Data are presented as mean ± standard deviation, or number (%).
BMI, body mass index; CT, computed tomography; DLco, diffusing capacity of the lung for carbon monoxide; FVC, forced vital capacity; IPF, idiopathic pulmonary fibrosis; KL-6, Krebs von den Lungen-6; PFT, pulmonary function test; 6MWD: six-minute walk distance; PH, pulmonary hypertension; SpO2: saturation of peripheral oxygen.
Prognostic factor for mortality and survival analysis
During follow-up, the median survival of all patients was 50.0 months (95% confidence interval (CI): 45.0–58.0 months). In the unadjusted Cox analysis, lower FVC% and DLCO%, shorter 6MWD, lower minimum SpO2, and PH at diagnosis were significantly associated with mortality in patients with IPF (Table 2). In the multivariable Cox analysis, only lower minimum SpO2 values were independently associated with higher mortality (hazard ratio (HR), 1.081; 95% CI: 1.024–1.142, p = 0.005) along with echocardiographic PH at the time of IPF diagnosis (HR, 2.466; 95% CI 1.149–5.296, p = 0.021).
Prognostic factors for mortality in patients with IPF.

Data are presented as mean ± standard deviation, or number (%).
Excluded in the multivariate analysis because of significant correlation with Minimum SpO2, 6MWT (r = 0.649, p < 0.001).
Excluded in the multivariate analysis due to a large number of missing data.
Continuous variables.
BMI, body mass index, forced vital capacity; CT, computed tomography; DLco, diffusing capacity of the lung for carbon monoxide; IPF, idiopathic pulmonary fibrosis; KL-6, Krebs von den Lungen-6; 6MWD, six-minute walk distance; SpO2, saturation of peripheral oxygen.
Table 3 presents the risk factors for mortality in patients with IPF (Table 3). In the unadjusted logistic analysis, younger age, lower lung function (FVC and DLCO), lower minimum SpO2 during 6MWT, and emphysema on CT were associated with an increased risk of mortality. In the multivariate logistic analysis, lower minimum SpO2 was independently associated with higher mortality along with younger age (Table 3).
Risk factors for mortality in patients with IPF.

Excluded in the multivariate analysis because of significant correlation with Minimum SpO2, 6MWT (r = 0.649, p < 0.001).
Excluded in the multivariate analysis due to a large number of missing data.
Continuous variables.
BMI, body mass index, forced vital capacity; CT, computed tomography; DLco, diffusing capacity of the lung for carbon monoxide; KL-6, Krebs von den Lungen-6; IPF, idiopathic pulmonary fibrosis;6MWD, six-minute walk distance; SpO2, saturation of peripheral oxygen.
Patients with minimum SpO2 <88% during 6MWT had a worse survival than those with minimum SpO2 ⩾ 88% (median survival: 29.0 months vs 50.0 months, p < 0.001; Figure 1). Also, Patients with baseline PH showed poorer survival outcomes compared to those without (median survival: 21.0 months vs 50.0 months, p < 0.001; Figure 2(a)). Those with PH at the time of IPF diagnosis along with those who developed PH during follow-up also showed poorer overall survival than those who didn’t develop PH during the follow-up (median survival: 36.0 months vs 58.0 months, p = 0.001; Figure 2(b)).
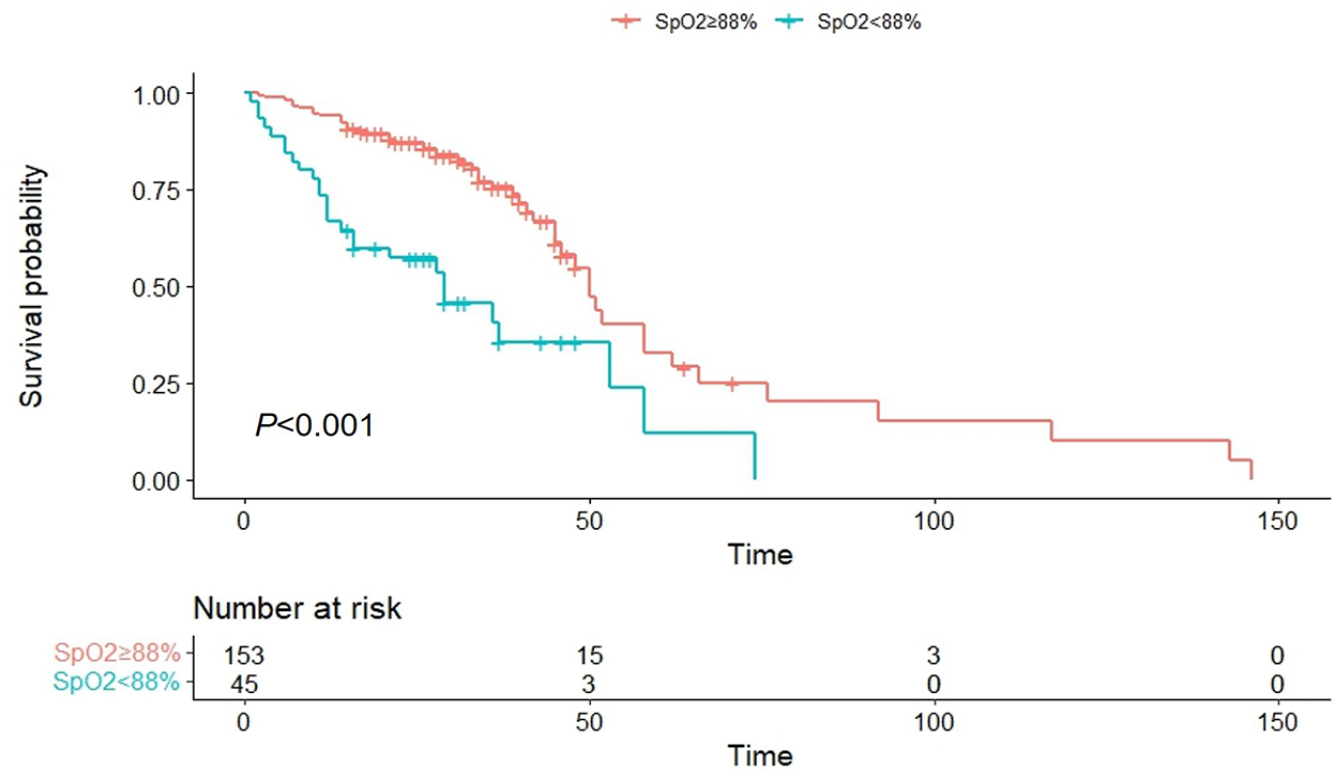
Comparison of overall survival curve between IPF patients with minimum SpO2 at 88% or higher and patients with minimum SpO2 below 88% during 6MWT.
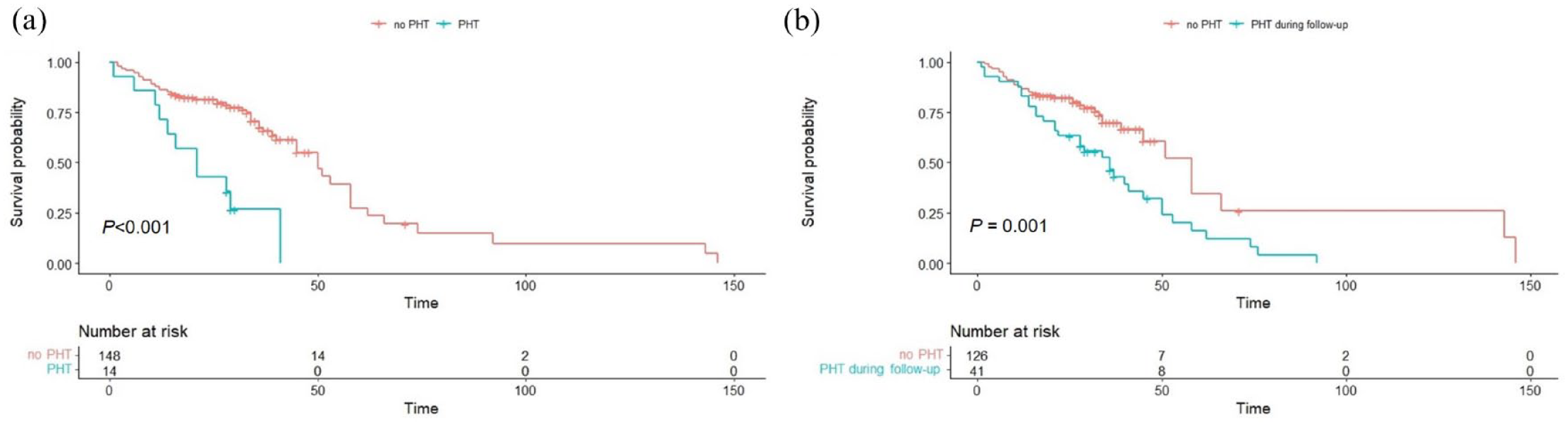
Overall survival curves in patients with IPF according to PH. (a) Comparison of survival curves according to baseline PH in patients with IPF. (b) Comparison of overall survival between patients with PH at the time of IPF diagnosis along with those who developed PH during follow-up and those without.
A comparison of patients stratified by baseline minimum SpO2 < 88% during the 6MWT compared to those with minimum SpO2 ⩾88% is presented in Supplemental e-Table 1. The group with excess desaturation were younger (mean age: 62.8 vs 68.1, p < 0.001), had lower FVC (mean values: 63.3 vs 76.1, p < 0.001), DLco (mean values: 38.1 vs 60.1, p < 0.001), shorter 6MWD (mean values: 368.2 vs 438.7, p = 0.001), and had more elevated Krebs von den Lungen-6 (KL-6) levels ⩾ 1000 U/mL (69.6% vs 15.9%, p < 0.001).
Echocardiographic pulmonary HTN
There were 162 patients with baseline echocardiography results, 14 of whom (8.6%) had echocardiographic evidence of PH at the time of IPF diagnosis, and 24 patients developed PH during the follow-up. The non-survivors were found to have PH more frequently than survivors (41.0% vs 4.2%, p < 0.001). Additionally, baseline PH was also an independent prognostic factor for mortality (Table 2). A comparison of baseline characteristics according to baseline echocardiographic PH is shown in Supplemental e-Table 2. Patients with PH had a lower body mass index (BMI), DLCO%, and a shorter 6MWD than those without.
In the unadjusted logistic regression analysis, risk factors for PH included older age, lower DLCO%, shorter 6MWD, and minimum SpO2 during the 6MWT. Due to the overfitting issue, only two variables were entered into the multivariate analysis, and DLCO% was excluded because of a significant correlation with minimum SpO2 during the 6MWT (r = 0.649, p < 0.001). The shorter 6MWD was associated with increased risk for PH in the multivariate analysis (odds ratio: 1.009, 95% CI: 1.003–1.015, p = 0.003; Table 4).
Risk factors for PH in patients with IPF.

Data are presented as mean ± standard deviation, or number (%).
Excluded in the multivariate analysis because of significant correlation with Minimum SpO2 (r = 0.649, p < 0.001).
Continuous variables.
CT, computed tomography; DLco, diffusing capacity of the lung for carbon monoxide; FVC, forced vital capacity; IPF, idiopathic pulmonary fibrosis; 6MWD, six-minute walk distance; PH, pulmonary hypertension; SpO2, saturation of peripheral oxygen.
Analysis of serial six-minute walk test
Of the 198 patients, 133 patients (67.2%) had 6-month follow-up 6MWT data, and 144 patients (72.7%) had 12-month follow-up data. A comparison of changes in the 6MWT at 6 and 12 months after IPF diagnosis between non-survivors and survivors is shown in e-Figure 1. The non-survivors showed significantly greater deterioration in 6MWD at 12 months after the IPF diagnosis than in survivors (mean absolute changes: −43.3 m vs 12.0 m, p < 0.001) (Supplemental e-Figure 1-A and e-Table 3). There were no statistical differences in the minimum SpO2 during the 6MWT between the non-survivors and survivors at 12 months following IPF diagnosis (mean absolute changes: −1.9% vs −1.0%, p = 0.225) (Supplemental e-Figure 1-B and e-Table 3). The changes in the 6MWT at 6 months were not statistically significant between the two groups.
Notably, patients who showed a decline in 6MWD of more than 50 m over 12 months following IPF diagnosis demonstrated poorer overall survival than those who did not (median survival: 45.0 months vs 58.0 months, p < 0.001; Figure 3(a)). Not only those with initial minimum SpO2 during 6MWT below 88% but also patients with worsening desaturation at 12 months after the IPF diagnosis, demonstrated a poorer prognosis compared to those who did not (median survival: 29.0 months (initial SpO2 < 88%) vs 48.0 months (SpO2 < 88% at 12 months) vs 58.0 months (SpO2 ⩾ 88%), p < 0.001; Figure 3(b).

Comparison of overall survival curves according to follow-up 6MWT results: (a) Comparison of overall survival curves between patients with a decline equal to or more than 50 m over 12 months and those with a decline less than 50 m. (b) Comparison of overall survival curves according to the desaturation in the 6MWT during the 12 months.
We evaluated the changes in the 6MWD at 1 year following diagnosis, based on the pulmonary function changes observed during the same period (Supplemental e-Figures 2-A and 2-B). Our findings indicate a trend toward greater declines in 6MWD as lung function declines.
Discussion
In this study, the baseline lower minimum SpO2 during the 6MWT was a significant prognostic factor in patients with IPF, and those with minimum SpO2 below 88% showed poorer prognosis than those with minimum SpO2 at 88% or higher. Furthermore, non-survivors showed a more significant decline in 6MWD over the 1-year follow-up than survivors. Notably, shorter 6MWD was an independent risk factor for PH which is related to poor prognosis.
Our study demonstrated that patients with minimum saturation of 88% or less during their initial 6MWT showed poorer prognosis than those with SpO2 above 88%, and desaturation during the baseline 6MWT was a significant predictive factor of mortality in patients with IPF, which is in line with previous studies.22,23,25,26 In a study involving 83 patients with biopsy-proven usual interstitial pneumonia and 22 patients with biopsy-proven nonspecific interstitial pneumonia, Lama et al. reported that desaturation to 88% or less was a strong predictor of mortality in patients with usual interstitial pneumonia, even after adjusting for age, sex, baseline DLco, FVC, and the amount of fibrosis on high-resolution computed tomography. 23 Furthermore, Flaherty et al., in a study including 197 patients with IPF, showed that patients with desaturation to 88% or less during the baseline 6MWT had worse survival than those with baseline SpO2 > 88% (median survival: 3.21 years vs 7.83 years, p = 0.006). 26 They also reported that desaturation area, defined as the total area above the curve created by desaturation percentage values and each subsequent minute of the 6MWT, was a powerful predictor for mortality. Also, in a study carried out of 93 patients with IPF, Nishiyama et al., reported that patients with minimum SpO2 < 88% during the 6MWT showed worse survival than those with SpO2 ⩾ 88% (median survival 41.0 months vs 62.9 months, p = 0.023), and minimum SpO2 was a significant prognostic factor in the multivariable Cox analysis (adjusted HR: 0.952, 95% CI: 0.924–0.981, P = 0.0014). 25 Taking into account these previous studies and our findings, a 6MWT, which is simple, cost-effective, and reproducible to perform, is a useful test for predicting the prognosis at the time of diagnosis in IPF patients.
In our study, non-survivors showed more decline in 6MWD than the survivors at 12 months after the IPF diagnosis, and patients with a decline in 6MWD ⩾50 m over 12 months showed worse overall survival than those with a decline of less than 50 m. Furthermore, patients who exhibited worsened desaturation at 12 months of follow-up also demonstrated poorer survival than those who did not. Very similarly, a previous study including 826 patients with IPF demonstrated that patients who experienced a decline in 6MWD greater than 50 m over 24 weeks had a higher risk of mortality (HR: 4.27, 95% CI: 2.57–7.10, p < 0.001) than those with a decline in 6MWD less than or equal to 25 m. 9 This study reported that a decline in 6MWD of 25–45 m represents a minimal clinically important difference. Similarly, in a study by Nathan et al. involving 338 patients with IPF, patients with a >50 m decline over the first 6 months tended to have greater 1-year mortality risk than patients with a ⩽50 m decline (HR: 2.53, 95% CI: 0.94–6.79, p = 0.066). 24 In summary, not only does the baseline 6MWT, but also serial change in the 6MWT appears to have clinical significance in patients with IPF. While the disease progression criteria for physiologic lung parameters such as FVC and DLco are relatively well established, as seen in recent international guidelines for progressive pulmonary fibrosis 27 and the INBUILD trial, 28 there has been no consensus definition of disease progression criteria for 6MWT, highlighting the need for further research.
Our study demonstrated that echocardiographic PH was a significant prognostic factor. Patients with PH showed reduced 6MWD and had a poorer prognosis than those without. Furthermore, 6MWD was an independent factor for PH, consistent with previous studies.18–21 In a clinical trial with IPF patients, those with PH (n = 68) had significantly lower 6MWD along with minimum SpO2, than those with no PH (n = 374). 29 Alkukhun et al., in a study including 235 patients with IPF, reported that shorter 6MWD was associated with the presence of PH in IPF patients (unadjusted OR: 0.95, 95% CI: 0.92–0.98, p = 0.001), consistent with our study. 30 Minai et al., in a study of 124 patients with IPF who underwent right heart catheterization (RHC) and 6MWT, also reported that patients with precapillary PH had lower 6MWD compared to those without. 31 These studies as well as our current report highlight the importance of considering the presence of PH when interpreting the 6MWT results in the management of patients with IPF. Minai et al. also demonstrated that increasing mean pulmonary arterial pressure was the best predictor of 6MWD; specifically, a multivariate analysis (p = 0.0006) demonstrated that each increase of 1 mmHg was associated with a decrease of 2.6 m in 6MWD. 31 Although we could not confirm the diagnosis of PH via RHC, we observed that the 6MWT was significantly associated with echocardiographic PH, which might explain the possible mechanism of its role as a predictor of prognosis in patients with IPF.
PH is known to be a common comorbidity in patients with interstitial lung disease (ILD) that has been reported in up to 86% of ILD patients 32 and is associated with poor prognosis with a 3-year mortality rate of up to 70%. 33 There have been many clinical trials of pulmonary vasoactive therapy targeting PH in ILD; however, their effectiveness has not been proven until recently 32 ; and treatment options have been limited to supplemental therapies such as long-term oxygen treatment. 34 Notably, a recent randomized clinical trial (INCREASE trial) has demonstrated that inhaled treprostinil improved the functional exercise capacity in patients with PH in ILD, 35 and its effectiveness has been reaffirmed in numerous post hoc analyses.36–38 As the 6MWT has been employed as an end-point in ILD clinical trials, 22 but further research is needed to determine the optimal ways to analyze 6MWT data.
Our study had several limitations. First, it was difficult to calculate the sample size because this study was a retrospective cohort study conducted at a single center; however, we included as many patients as possible to maximize the sample size. Additionally, the retrospective nature of the study may limit generalizability and introduce potential biases such as selection biases. Second, the diagnosis of PH was established based on echocardiography, rather than RHC, which might have compromised the accuracy of the diagnosis, as well as the severity of the PH could not be determined. While echocardiography can provide information about the probability of PH, additional studies based on RHC are needed for a precise assessment of its impact on the 6MWT. Third, there may have been a selection bias as the interval between serial 6MWT was not constant and the test was conducted depending on the patient’s condition. Nevertheless, we believe that it is a strength of our study to present the relation between the baseline 6MWT and echocardiographic PH, and the impact of 6MWT changes reported in a relatively large number of patients the follow-up 6MWT. Fourth, since this is a retrospective study in nature, we were unable to evaluate all variables associated with PH, such as the ratio of tricuspid annular plane systolic excursion to systolic pulmonary artery pressure, right ventricular outflow tract acceleration time, pulmonary artery diameter, or aortic root diameter. Consequently, there may be selective bias as we defined echocardiographic PH by tricuspid regurgitation velocity. Nonetheless, it is noteworthy that among patients with TR Vmax ⩽2.8, none exhibited more than two additional echocardiographic signs suggestive of PH. Fifth, there are several factors that can impact the reproducibility of the 6MWT, including learning effects, environmental factors, medication effects, or subjective effort, which can lead to variability in test results. Therefore, serial follow-up tests may not accurately reflect the actual exercise capacity, and caution is needed in interpreting the serial follow-up test results.
Conclusion
In conclusion, desaturation during 6MWT was a significant predictive factor of mortality. Moreover, a decline in 6MWD during follow-up 6MWT was also associated with the prognosis of IPF patients, highlighting the importance of both baseline and serial 6MWT.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241275329 – Supplemental material for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study
Supplemental material, sj-docx-1-tar-10.1177_17534666241275329 for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study by Min Jee Kim, Steven D. Nathan, Hyeon Hwa Kim and Ho Cheol Kim in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-5-tar-10.1177_17534666241275329 – Supplemental material for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study
Supplemental material, sj-docx-5-tar-10.1177_17534666241275329 for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study by Min Jee Kim, Steven D. Nathan, Hyeon Hwa Kim and Ho Cheol Kim in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-jpeg-4-tar-10.1177_17534666241275329 – Supplemental material for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study
Supplemental material, sj-jpeg-4-tar-10.1177_17534666241275329 for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study by Min Jee Kim, Steven D. Nathan, Hyeon Hwa Kim and Ho Cheol Kim in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-jpg-2-tar-10.1177_17534666241275329 – Supplemental material for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study
Supplemental material, sj-jpg-2-tar-10.1177_17534666241275329 for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study by Min Jee Kim, Steven D. Nathan, Hyeon Hwa Kim and Ho Cheol Kim in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-jpg-3-tar-10.1177_17534666241275329 – Supplemental material for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study
Supplemental material, sj-jpg-3-tar-10.1177_17534666241275329 for Clinical implications of six-minute walk test in patients with idiopathic pulmonary fibrosis: a retrospective cohort study by Min Jee Kim, Steven D. Nathan, Hyeon Hwa Kim and Ho Cheol Kim in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
