Abstract
Mucormycosis is an invasive fungal infection that can result in severe lung infections, with pulmonary mucormycosis (PM) being one of the most prevalent manifestations. Prompt diagnosis is crucial for patient survival, as PM often exhibits rapid clinical progression and carries a high fatality rate. Broncho-alveolar lavage fluid or endobronchial biopsy (EBB) has been commonly employed for diagnosing PM, although there is limited mention of endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) in the literature. In this report, we present a case of PM in a patient with diabetes. While EBB did not yield evidence of Rhizopus infection, a definitive diagnosis was obtained through EBUS-TBNA. The patient underwent combination therapy, including oral medication, nebulization, and EBUS-guided intrafocal amphotericin B injection, which resulted in significant improvement following the failure of initial therapy with amphotericin B injection cholesterol sulfate complex. Our case highlights the potential of EBUS-TBNA not only for mediastinal lymphadenopathy but also for obtaining extraluminal lesion specimens. Furthermore, for patients with an inadequate response to mono-therapy and no access to surgical therapy, the addition of EBUS-guided intralesional amphotericin B injection to systemic intravenous therapy may yield unexpected effects.
Introduction
PM commonly occurs in patients with diabetes especially diabetic ketoacidosis (DKA). 1 It is a highly invasive infection with a high mortality rate. Therefore, a patient needs to get a definite diagnosis and access to effective treatment early. In this case, we provide a novel use of endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) and hope to be helpful for clinical work. This clinical case report adheres to the CARE (Consensus-based Clinical Case Reporting Guideline) guidelines for reporting clinical cases from the EQUATOR Network. 2
Case report
A 37-year-old female patient presented with a history of fatigue for 1 month, and a nonproductive cough associated with low-grade fever, worsening dyspnea, and generalized weakness for 1 week. The consolidation of the right lower lobe (RLL) could be seen through chest computerized tomography (CT) (Figure 1). Also, DKA was found in the patient. She was treated with broad-spectrum antibiotics for 4 days before she was referred to our hospital for further management, but the symptoms of cough and dyspnea were worse than before. Chest CT (Figure 2) in our hospital showed that the lesions in the RLL aggravated. In addition, there were a few inflammatory lesions in the left lower lobe. On physical examination, her vital signs included a body temperature of 38.6°C, heart rate of 110 beats/min, blood pressure of 89/62 mmHg, and respiratory rate of 31 breaths/min. Moist rales can be heard in the RLL. The patient had no health complaints or particular medical history.

Chest CT showed the consolidation of the right lower lobe.
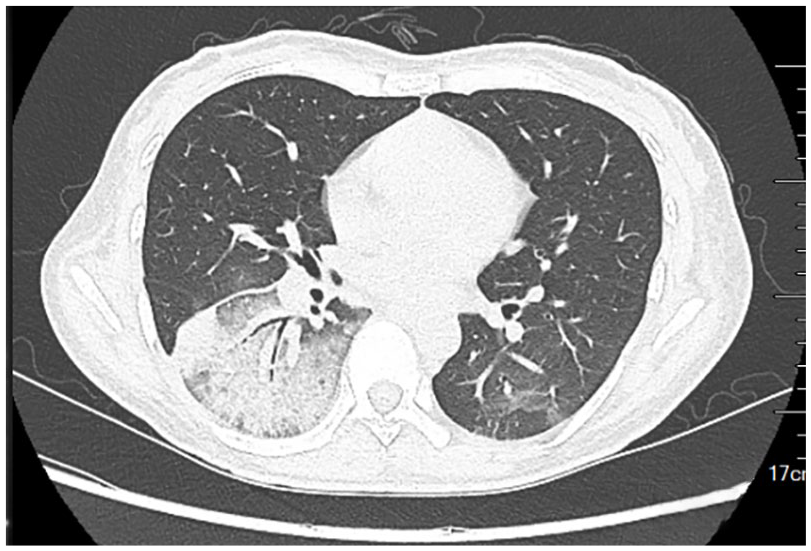
Chest CT showed rapid progression within 3 days, with consolidation and surrounding interstitial and ground glass lesions.
The patient underwent a fiberoptic bronchoscopy immediately after admission. An endobronchial growth was found at the opening of the basal segment of the RLL, resulting in lumen stenosis (Figure 3). Extrabronchial abnormal hypoechoic signals of the basal segment of the RLL could be seen under the ultrasonic bronchoscope (Figure 4). Endobronchial biopsy (EBB), bronchoalveolar lavage, and EBUS-guided extra luminal lesion needle aspiration biopsy were performed.
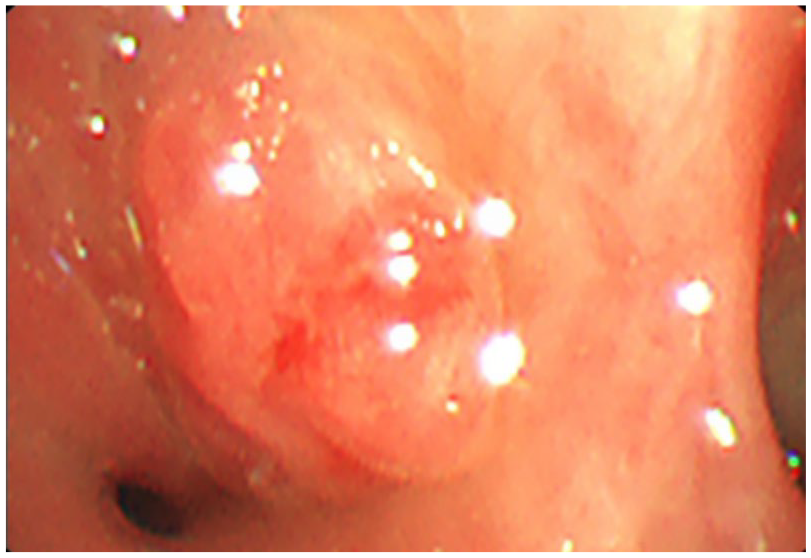
A neoplasm was found at the opening of the basal segment of the RLL.
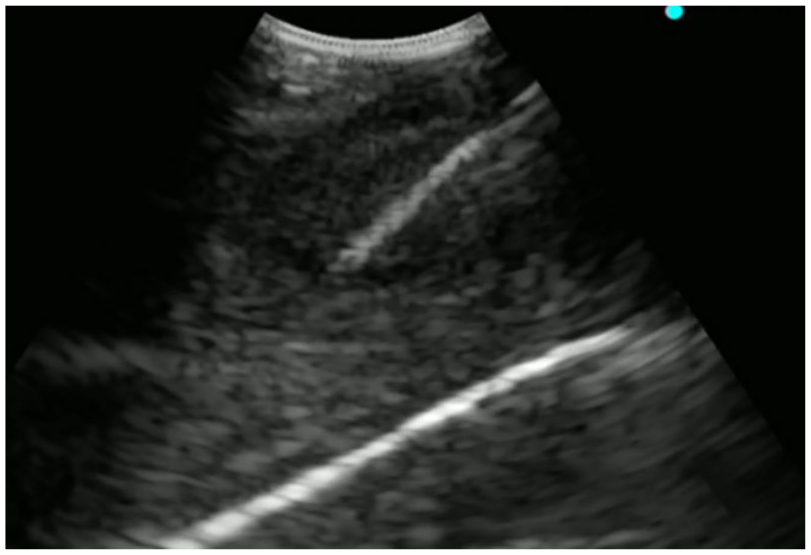
Abnormal hypoechoic signals outside the lumen of the basal segment of the RLL could be seen and a biopsy was performed through EBUS-TBNA.
At admission, blood investigations revealed a white blood cell count of 12.76 × 109/L, and a lymphocyte count of 0.64 × 109/L. Hemoglobin A1c was increased to 13.4%. In addition, procalcitonin, C-reactive protein, and erythrocyte sedimentation rates were higher than normal, while the count of CD4+ cells and CD8+ cells decreased. Her renal and liver function parameters were within normal limits. Viral test is negative.
Rhizopus oryzae (four reads) was detected by clinical metagenomic next-generation sequencing (mNGS) in broncho-alveolar lavage fluid (BALF) 36 h after the tracheoscopy. Three days later, the endobronchial growth obtained by EBB was considered nothing special except necrosis. However, the histopathology of extraluminal lesion specimens obtained by EBUS-TBNA revealed broad aseptate ribbon-like hyphae consistent with Rhizopus. The hyphae were identified on hematoxylin and eosin stains and confirmed with Periodic Acid-Schiff (PAS) and Grocott–Gomori’s silver (GMS) special stains (Figures 5–7). Ten days after the tracheoscopy, the culture result of extraluminal lesion specimens also suggested Rhizopus (Figure 8).

Pathology showed typical Rhizopus mycelia with positive staining for PAS and GMS.

Pathology showed typical Rhizopus mycelia with positive staining for PAS and GMS.

Pathology showed typical Rhizopus mycelia with positive staining for PAS and GMS.
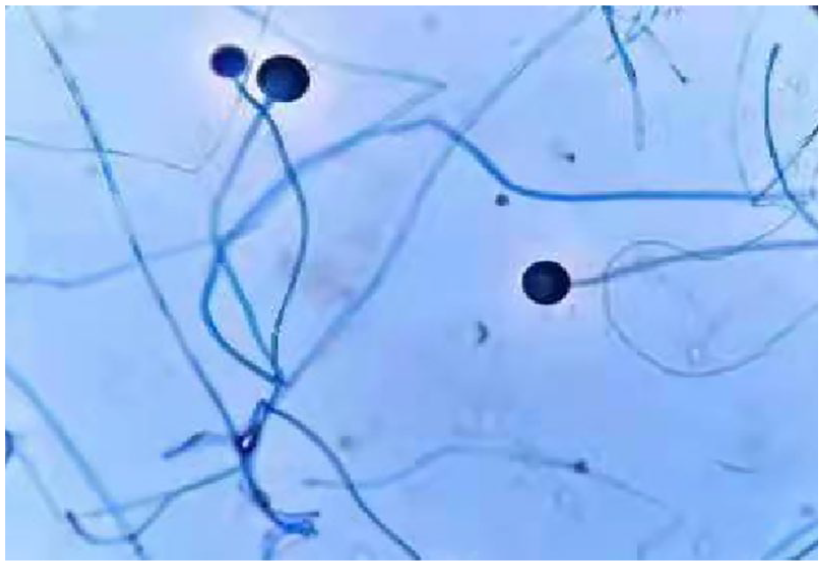
The culture result of purulent puncture fluid obtained by EBUS at the lesion suggested Rhizopus.
The patient was therefore diagnosed with severe community-acquired pneumonia (pulmonary mucormycosis, PM) and DKA. The patient had DKA, which was considered a risk factor for PM, so we used insulin to decrease blood glucose. In addition to controlling hyperglycemia, amphotericin B cholesterol sulfate complex was given (1 mg/kg on days 1–2; 3 mg/kg on day 3; and 4 mg/kg/day on days 4–16). Despite antifungal treatment, the patient did not improve. The symptoms of fever, cough, and dyspnea were worse than before. The sequential organ failure assessment (SOFA) score also raised from 2 to 3, and CT examination after 2 weeks of treatment showed that the lesions advanced. Combination therapy was given then, including posaconazole oral tablet (600 mg on day 6, then 300 mg/day on days 7–16), inhalation treatment of amphotericin B (5 mg every 12 h on days 6–16), EBUS-guided intralesional amphotericin B injection (10 mg diluted with 5 ml sterile water for injection; one position each time; on day 6, day 10, and day 13) (Figure 9). Twelve days after combination therapy, the clinical improvement of the woman was observed, and no adverse or unanticipated events happened. And the lesions shown on chest CT were significantly absorbed compared with the previous ones (Figure 10). The woman was discharged and is still being closely followed up.

Amphotericin B was injected into the lesion through EBUS.

Obvious absorption can be seen after combination therapy.
Discussion
Mucormycosis is an invasive fungal infection. The pathogenesis of diabetes-related mucormycosis may lie in acidosis, neutrophil dysfunction secondary to hyperglycemia, decreased phagocytosis of macrophages, and their promotion of iron uptake by Mucorales, which makes the patient particularly susceptible to mucormycosis.3–5 Therefore, diabetic patients (especially those with ketoacidosis) are highly susceptible to PM when Mucorales microspores are inhaled.
Obtaining ideal samples is essential for the definitive diagnosis of PM. Bronchoscopy is a useful and safe procedure. EBUS-TBNA is a minimally invasive intervention that is increasingly used to obtain diagnostic materials. Some studies suggest that in cases where fungal infection is clinically suspected, the fungal culture of EBUS-TBNA core tissue and needle rinse fluid can be considered. 6 It is usually an important means of obtaining samples of mediastinal lymphadenopathy. 7 However, Li et al. considered that EBUS-TBNA is not limited to aspiration of mediastinal masses and lymph nodes but is also suitable for exploration of other tissues external to the central airway. Moreover, therapeutically, it can be used for cyst drainage and drug injections. 8 We extended this technique to extraluminal lesions by needle aspiration biopsy and made a definitive diagnosis, although no mediastinal lymphadenopathy was found in this patient. Parikh et al. 9 have successfully treated pulmonary aspergillosis by EBUS-guided Transbronchial Needle Injection of liposomal amphotericin B. The real-time puncture can control the depth and position of needle insertion which may avoid massive bleeding caused by the damage to vessels. And ensure that the drug can be distributed in the lesions, improving the therapeutic efficacy. After the failure of monotherapy, we chose EBUS-guided intrafocal drug injection in combination with intravenous drug therapy as a remedial measure, and our patient benefited from it. So far, many cases of PM have been diagnosed by bronchoscopy, but mostly by BALF or transbronchial lung biopsy(TBLB). Few mentioned EBUS-guided extraluminal lesion needle aspiration biopsy or EBUS-guided intralesional drug injection for the treatment of PM. We have proposed a new application here.
In recent years, mNGS has developed rapidly. The suspected pathogen in the sample can be detected quickly and without bias, enabling early diagnosis. Nevertheless, some hurdles still need to be overcome. Many experts have reservations about the use of NGS in the diagnosis of infectious diseases due to limitations such as high costs, lack of standardization, false-positive results, and contamination. But for patients with difficult or complicated infectious diseases, such as immunodeficiencies in critically ill patients or outbreaks of infectious diseases of unknown origin, this technique is recommended as the first method of detection.10,11 In this case, we obtained evidence of Rhizopus infection by mNGS within 48 h of fiberoptic bronchoscopy, and it took 5 days for pathology and 10 days for culture, respectively. However, given the possibility of false-positive results, we waited with antifungal therapy until the pathology and culture results were available.
PM is a highly vascular, invasive infection associated with extensive thrombosis and tissue necrosis. 12 The mortality rate is approximately 55% and can be reduced to 25% with a combination of timely diagnosis, appropriate medical treatment, and early aggressive surgical intervention.13,14 Surgical debridement with clean margins should be performed for disease control, histopathology, and microbiologic diagnosis. Combined systematic drug therapy is also required. 15 But in multifocal infection, thoracic adhesion, and vascular invasion, the clinical benefit of surgical treatment may be modest, and the difficulty of surgery and bleeding risk are significantly higher than in other lung surgery. 16 In this case, CT showed lesions in both lungs which may lead to repeated surgical intervention to achieve surgical control. The patient was generally in poor condition and extremely frail with a Body Mass Index (BMI) of 13.6 kg/m2. Considering the high surgical risk, we decided to postpone surgery after multi-disciplinary treatment (MDT) and drug treatment was finally chosen. The maximum dose of liposomal amphotericin B initially is recommended by current guidelines, followed by isavuconazole, posaconazole sustained-release tablets, or intravenous preparations. 15 But the guiding principle of the drug available specifies a gradual increase in dosage, so finally we decided to follow the instructions. Limited data support combinations of polyenes and azoles or polyenes plus echinocandins, but some studies find that combination therapy can improve cure and survival rates without increasing toxicity.17,18
Some researchers have found that aerosolized inhalation and local bronchoscopic infusion of amphotericin B or liposomal amphotericin B also may achieve good results,19–21 some literature, on the other hand, suggested that nebulized amphotericin B (NAB) cannot improve overall response. 22 However, it is notable that NAB could be useful in patients with tracheobronchial disease.22,23 The symptoms of our patient became worse after antifungal treatment. The SOFA score raised, and the lesions advanced. All above suggested that initial intravenous therapy was ineffective, so we added local therapy based on combined drug therapy. The patient had both tracheobronchial disease and extraluminal consolidation. In consequence, NAB was received, and amphotericin B was injected into the extraluminal lesion under the guidance of EBUS which could achieve a higher drug concentration than local bronchial perfusion. After 1 month of treatment, the patient’s condition improved significantly and was discharged. She received diabetes control, systemic intravenous therapy, and NAB, all of which may have contributed to her improvement, but local interventional therapy may also have played an important role. This approach needs to be given sufficient attention. Currently, the patient is still being closely followed up.
Unfortunately, one limitation of this study is the lack of amphotericin B concentration detection in lung tissue after interventional therapy. Another deficiency is that we have only one such case. Additional cases are expected to be collected in the future, which will provide more clinical evidence to resolve this challenging clinical problem.
Conclusion
For patients who are unable to undergo surgery or have a poor response to monotherapy, EBUS-guided intrafocal drug injection combined with systemic chemotherapy may provide a potential alternative for PM treatment. But more evidence is needed.
The utilization of NGS in a clinical setting remains a topic of debate; nevertheless, it may be worth considering in immunodeficient critically ill patients or those with complex conditions.
Footnotes
Acknowledgements
We thank Dr Yong Wang from the Department of Clinical Laboratory of the Affiliated Hospital of Soochow University for providing the fungal culture picture of the patient. And thank Pranathi Vittal improved the grammar, spelling, and punctuation of the manuscript.
