Abstract
Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) is an established minimally invasive method for the diagnosis of benign and malignant conditions. Continuous efforts are underway to improve the material adequacy of EBUS-TBNA, including the introduction of a new technique called EBUS-guided transbronchial nodal cryobiopsy (EBUS-TBNC). This method allows for the retrieval of larger and well-preserved histologic samples from the mediastinum. We present a case series of four patients who underwent combined EBUS-TBNA and EBUS-TBNC procedures in our centre. All procedures were performed under general anaesthesia using a convex probe EBUS scope (Pentax EB-1970UK). Two patients were diagnosed with malignancy and two with benign disorders (silicosis and tuberculosis). In the malignant cases, both EBUS-TBNA/cell block and cryobiopsy provided a diagnosis but cryobiopsy yielded more material for ancillary tests in one patient. However, in the benign cases, there was discordance between EBUS-TBNA/cell block and cryobiopsy. Only cryobiopsy detected granuloma in the patient with TB (tuberculosis), and in the patient with silicosis, TBNC provided a better overall histological evaluation, leading to a definitive diagnosis. No complications were observed. This case series supports the potential diagnostic value of combining EBUS-TBNA and EBUS-TBNC, particularly in benign mediastinal lesions (granulomatous diseases), and in cases requiring additional molecular tests in cancer diagnosis.
Plain language summary
We explored a new technique for lung diagnosis called EBUS-guided transbronchial nodal cryobiopsy (EBUS-TBNC). This method helps get larger and well-preserved tissue samples from the chest area. In our study, we used this technique on four patients alongside the established method called EBUS-guided transbronchial needle aspiration (EBUS-TBNA). All procedures were done with the patient under general anesthesia using a specific type of scope. Two patients were found to have cancer, and two had non-cancerous conditions (silicosis and tuberculosis). In the cancer cases, both methods provided a diagnosis, but the cryobiopsy gave more material for additional tests in one patient. However, in non-cancer cases, there were differences between the two methods. Only the cryobiopsy detected granulomas in the tuberculosis patient, and in the silicosis patient, cryobiopsy gave a better overall tissue evaluation, leading to a clear diagnosis. No complications were seen in any of the cases. This study suggests that combining EBUS-TBNA and EBUS-TBNC can be valuable, especially for non-cancerous chest lesions (like granulomatous diseases) and when extra tests are needed for cancer diagnosis.
Introduction
Endobronchial ultrasound-guided transbronchial nodal aspiration (EBUS-TBNA) is crucial in the assessment of mediastinal lymphadenopathy. While reported diagnostic yields are high in primary pulmonary malignancy (90%), 1 the role of EBUS-TBNA in lymphoma remains controversial, 2 carrying a dismal sensitivity of 65%. 3 Similarly, sensitivity remains suboptimal in benign lesions such as sarcoidosis (84%) and tuberculosis (80%).4,5 Efforts to optimize EBUS-TBNA yields have focussed on the use of varying needle sizes, core biopsy needles and intranodal forceps biopsy6–8 but the lack of histological information remains an issue. Endobronchial ultrasound-guided transbronchial nodal cryobiopsy (EBUS-TBNC) is a novel method which demonstrated feasibility for histological diagnosis in both primary pulmonary malignancies and lymphoma 9 while also demonstrating superiority in benign lesions. 10 In this case series, we describe our preliminary experience utilizing EBUS-TBNC for diagnosing mediastinal lesions in a tuberculosis endemic region. The reporting of this study adheres to the CARE (CAse REport) checklist (available as Supplemental Material).
Procedural steps
The procedures in this case series were conducted on electively admitted inpatients. A convex probe EBUS scope (Pentax EB-1970UK Linear-Array Ultrasound Bronchoscope, Japan) was used (Figure 1). After systematic inspection of lymph node stations, EBUS-TBNA was first performed using a 22-gauge EBUS needle (Cook Medical, EchoTip ProCore HD Ultrasound Biopsy Needle). Aspirate samples underwent immediate rapid on-site evaluation (ROSE) to confirm the sample adequacy. Histologic cores were fixed in formalin. A total of 3–4 passes were made at each nodal station. A 1.1-mm cryoprobe (ERBE 20402-401, Medizintechnik, Tϋbingen, Germany) was then passed through the working channel of the EBUS scope. The tip of the cryoprobe was inserted through the puncture site created by the EBUS-TBNA needle. In cases where the cryoprobe could not be inserted, a Needlecut 3 V (Olympus needle knife) was employed to enlarge the tract, facilitating cryoprobe insertion. The position of the cryoprobe within the node was confirmed using EBUS imaging. Doppler imaging was used to ensure that there were no intervening vessels. The cryoprobe was activated for a duration of 3–10 s. The entire EBUS scope along with the cryoprobe was then removed, and the specimen was thawed in saline and placed in a specimen pot containing formalin. The EBUS scope was then used to promptly examine the puncture site to ensure there was no bleeding.
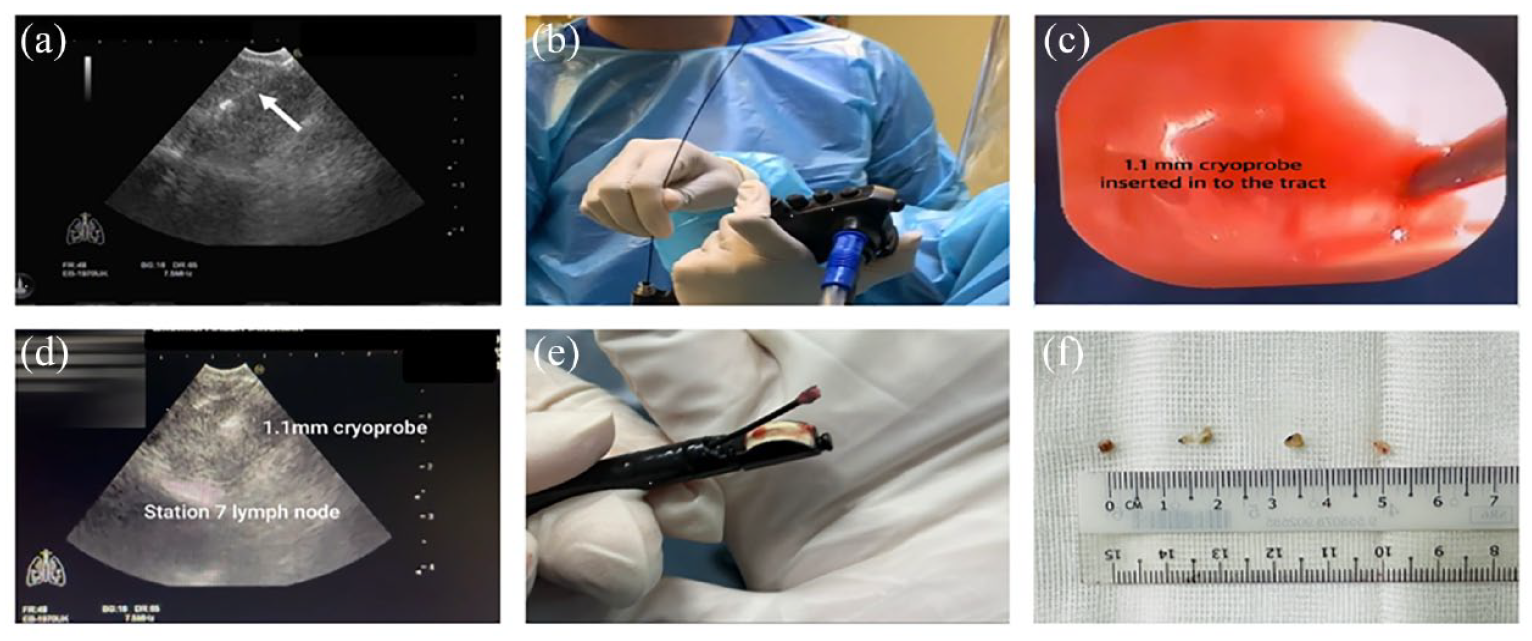
Procedure (a) EBUS-TBNA is performed using a 22-gauge EBUS needle (white arrow). (b) A 1.1-mm cryoprobe is passed through the working channel of the EBUS scope. (c) The tip of the cryoprobe is inserted through the puncture site created by the EBUS-TBNA needle. (d) The position of the cryoprobe within the node is confirmed using EBUS imaging and the cryoprobe is activated for a duration of 3–10 s. (e) The EBUS scope along with the cryoprobe is then removed, and the specimen can be seen at the tip of the cryoprobe. (f) Retrieved cryobiopsy specimens.
Case series
Case 1
A 57-year-old lady with underlying diabetes mellitus presented with 2 months of persistent cough and constitutional symptoms. The chest radiograph showed a right hilar lesion. Computed tomography (CT) thorax revealed a right hilar mass measuring 3.8 × 3.8 × 2.8 cm and mediastinal lymphadenopathy [Figure 2(a) and (b)]. The patient underwent EBUS-TBNA and EBUS-TBNC under general anaesthesia via laryngeal mask airway (LMA). Systematic inspection of lymph node stations revealed N3 nodes (11L, 10L and 4 L) measuring less than 5 mm, and an enlarged station 7 node (N2) measuring 40 mm with a well-defined, irregular margin with heterogeneous echogenicity and no central hilar structure. Four EBUS-TBNA passes were performed at the station 7 node, and ROSE revealed clusters of atypical cells suspicious of malignancy. Consequently, four EBUS-TBNC passes were performed with activation times ranging from 3 to 5 s, with preceding tract enlargement by the Olympus Needlecut 3 v knife. Procedural time was 20 min for EBUS-TBNA and 43 min for EBUS-TBNC. Both the EBUS-TBNA cell block and cryobiopsy sample confirmed adenocarcinoma and were sufficient for mutational analysis, confirming an EGFR (Exon19 deletion) mutation [Figure 2(c)–(g)] Following lung cancer multidisciplinary discussion, she was commenced on gefitinib and responded well to the treatment.

Case 1: (a) Computed tomography image shows a right hilar mass associated with adjacent ground glass opacity. (b) The subcarinal node is enlarged with a short axis of 16.9 mm (c) Cell block shows aggregates of tumour cells entrapped within fibrin clot (40× magnification). (d) Solid sheets of tumour cells with increased nuclear: cytoplasmic ratio, vesicular chromatin texture, prominent nucleoli and a modest amount of eosinophilic cytoplasm with indistinct border, in keeping with poorly differentiated carcinoma (400× magnification) seen on cell block. (e) Cryobiopsy specimen sheets of tumour cells (40× magnification). (f and g) Immunohistochemical staining of cryobiopsy specimens shows positive TTF-1 and Napsin A.
Case 2
A 52-year-old lady with underlying right breast cancer presented with a 1-year history of dry cough and weight loss. The chest radiograph revealed a left pleural effusion. Follow-up CT thorax revealed mediastinal lymphadenopathy, left lower lobe segmental collapse and left-sided pleural effusion [Figure 3(a) and (b)]. Thoracentesis revealed an exudative pleural effusion with a pleural adenosine deaminase (ADA) level of 10.8 IU/l and non-significant cytology results. Thoracoscopic biopsy demonstrated benign fibroadipose tissue with chronic inflammation. Due to concerns that the medical thoracoscopy findings were non-representative of the underlying disease, EBUS-TBNA was performed to sample enlarged mediastinal nodes. The procedure was performed under general anaesthesia using a laryngeal mask airway (LMA). On inspection, the angle of the carina was widened and deviated towards the left. The left main bronchus was slightly narrowed. EBUS-TBNA was performed with four passes made at station 7 node which was enlarged at 33 mm. ROSE revealed the presence of malignant cells. Subsequently, an EBUS-TBNC was conducted, and the Olympus Needlecut 3 v knife was used to enlarge the tract created by the EBUS-TBNA needle. Four cryobiopsy samples were obtained, with activation times ranging from 3 to 5 s, respectively. The overall procedure duration was 75 min, consisting of 25 min for EBUS-TBNA and 55 min for EBUS-TBNC. Both EBUS-TBNA cell block [Figure 3(c) and (d)] and EBUS-TBNC sample showed adenocarcinoma of the breast in origin but only the EBUS-TBNC sample was sufficient for hormonal biomarkers analysis, confirming a HER2 negative, ER and PR-positive tumour [Figure 3(e)–(g)]. She was subsequently referred to the oncology team for further treatment.
Case 2: (a and b) Computed tomography images show mediastinal lymphadenopathy (enlarged station 7 node), left lower lobe segmental collapse and left-sided pleural effusion (c) Cell block (40× magnification) shows cohesive clusters of pleomorphic cells. (d) CK 7 positive from the cell block. (e) Moderately pleomorphic cells with mitosis seen from cryobiopsy specimens (40× magnification). (f and g) ER and PR positivity were seen on cryobiopsy specimens.
Case 3
A 65-year-old ex-quarryman, never a smoker, presented with a chronic cough since 2013, which had worsened recently. In his 20-year history of work, he had only used a simple cloth mask for protection. Chest radiograph showed multiple small nodules bilaterally. Sputum tests for acid-fast bacilli (AFB) and gene expert were negative but sputum culture revealed Klebsiella pneumonia, for which he received intravenous antibiotics. A repeat chest radiograph showed persistent lung nodules. Follow-up CT thorax revealed bilateral numerous small lung nodules, along with a conglomerating mass (3.1 × 2.6 × 3.0 cm) in the right upper lobe and mediastinal lymphadenopathy, suggestive of silicosis. Under general anaesthesia, with the airway secured using a rigid bronchoscope, he underwent EBUS-TBNA of the enlarged station 7 node (measuring 40 mm). The node showed ill-defined margins, specks of calcification and homogeneous echogenicity. Four passes were made and ROSE showed benign lymphoid cells. A 1.1-mm cryoprobe was gently passed through the tract created by the EBUS-TBNA needle, obtaining five specimens from TBNC of the station 7 node with activation times ranging from 3 to 10 s. The procedure lasted a total of 65 min, with EBUS-TBNA taking 25 min and EBUS-TBNC taking 40 min. Cell block analysis from EBUS-TBNA revealed pigmented alveolar macrophages, benign lymphocytes and inflammatory cells. EBUS-guided TBNC showed multiple micronodular lesions with central lamellar hyalinization consistent with silicosis.
Case 4
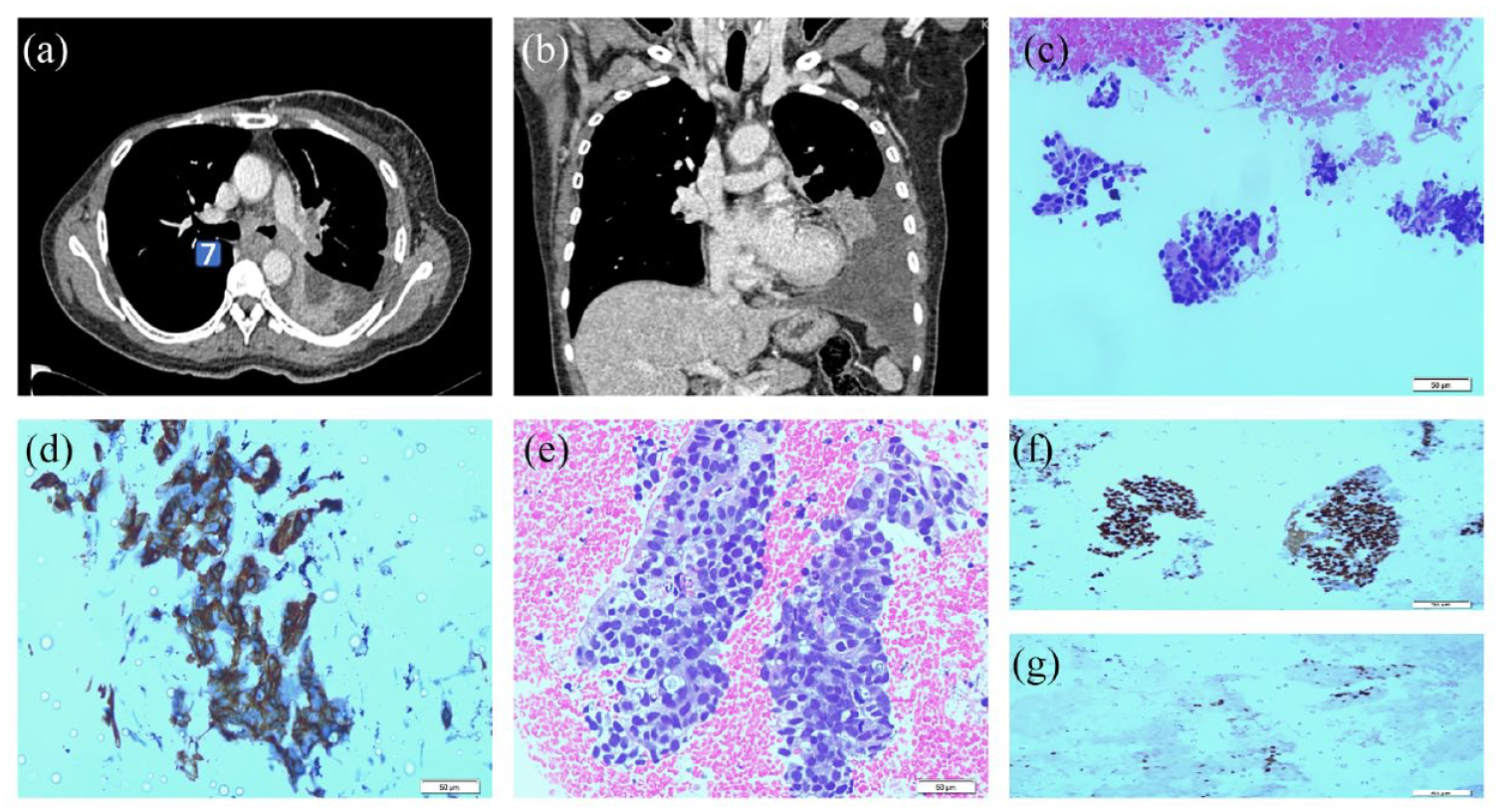
A 63-year-old farmer, an active smoker, with underlying hypertension presented with a 2-week history of dry cough and breathlessness. The chest radiograph revealed a moderate right-sided pleural effusion. Sputum AFB direct smears were negative and thoracocentesis revealed an exudative, lymphocyte-predominant effusion with negative culture, cytology, AFB smear, and ADA of 4.18 IU/L. A CT thorax showed right upper lobe nodules, right segmental middle lobe collapse, mediastinal lymphadenopathy and a new left-sided pleural effusion [Figure 4(a) and (b)]. EBUS was performed under general anaesthesia via a laryngeal mask airway. Systematic examination of lymph node stations revealed an oval-shaped, 7 mm node at station 11 L with well-defined borders and a 21 mm node at station 7 with an ill-defined border. Four passes of EBUS-TBNA were performed at the 11L node, with ROSE revealing benign lymphoid cells. Four passes of EBUS-TBNA were performed at the station 7 node, with ROSE revealing a hypocellular smear with few lymphocytes. In view of the inconclusive ROSE, we proceeded with EBUS-TBNC of the station 7 node, obtaining five specimens with activation times ranging from 3 to 10 s. The procedure lasted a total of 100 min, with EBUS-TBNA taking 25 min and EBUS-TBNC taking 75 min. Cell block analysis from TBNA showed benign lymphocytes from station 11 L and station 7 [Figure 4(c)]. No malignant cells or granulomas were detected from either nodal station. However, EBUS-TBNC showed extensive caseous necrosis with aggregates of epithelioid histiocytes, multinucleated giant cells of Langerhans type mixed with necrotic debris and degenerated inflammatory cells [Figure 4(d) and (e)]. Furthermore, Ziehl-Neelsen staining confirmed the presence of acid-fast bacilli [Figure 4(f)], leading to the initiation of anti-tuberculous therapy for the patient. During the 2-month follow-up, the patient remained well with no new symptoms or re-accumulation of pleural fluid.

Case 4: (a and b) Computed tomography images show mediastinal lymphadenopathy (enlarged lymph node stations 4 R, 4 L and 7). (c) Cell block from station 7 lymph node shows benign lymphocytes. No granuloma seen (10× magnification). (d and e) Cryobiopsy specimens from the station 7 node show multinucleated giant cells of Langerhans type, necrotic debris and granulomas (20× magnification). (f) Acid-fast bacilli visualized on Ziehl-Neelsen’s staining confirming tuberculosis (40× magnification).
Complications
All procedures were completed without any complications, and instances of minimal bleeding were effectively managed through the use of cold saline instillation. In case 1, we conducted a follow-up bronchoscopy on the following day to evaluate the puncture site, which revealed a smaller puncture site with no visible bleeding. Post-procedure chest radiographs were performed for all patients, and no pneumomediastinum or pneumothorax was detected. Patients were advised to report any fever, shortness of breath or other concerning symptoms upon discharge and they were subsequently followed up 2 weeks later.
Discussion
In our case series, EBUS-TBNC demonstrated superiority compared to EBUS-TBNA supplemented by ROSE for the diagnosis of tuberculosis (TB) and silicosis (Case 3 and Case 4). In our centre, where TB is prevalent, this technique proves highly valuable in diagnosing mediastinal TB lymphadenitis. It is particularly useful for patients with negative results from other TB tests and when granulomas are not detected through EBUS TBNA (Table 1).
Patient demographics and procedural outcomes.
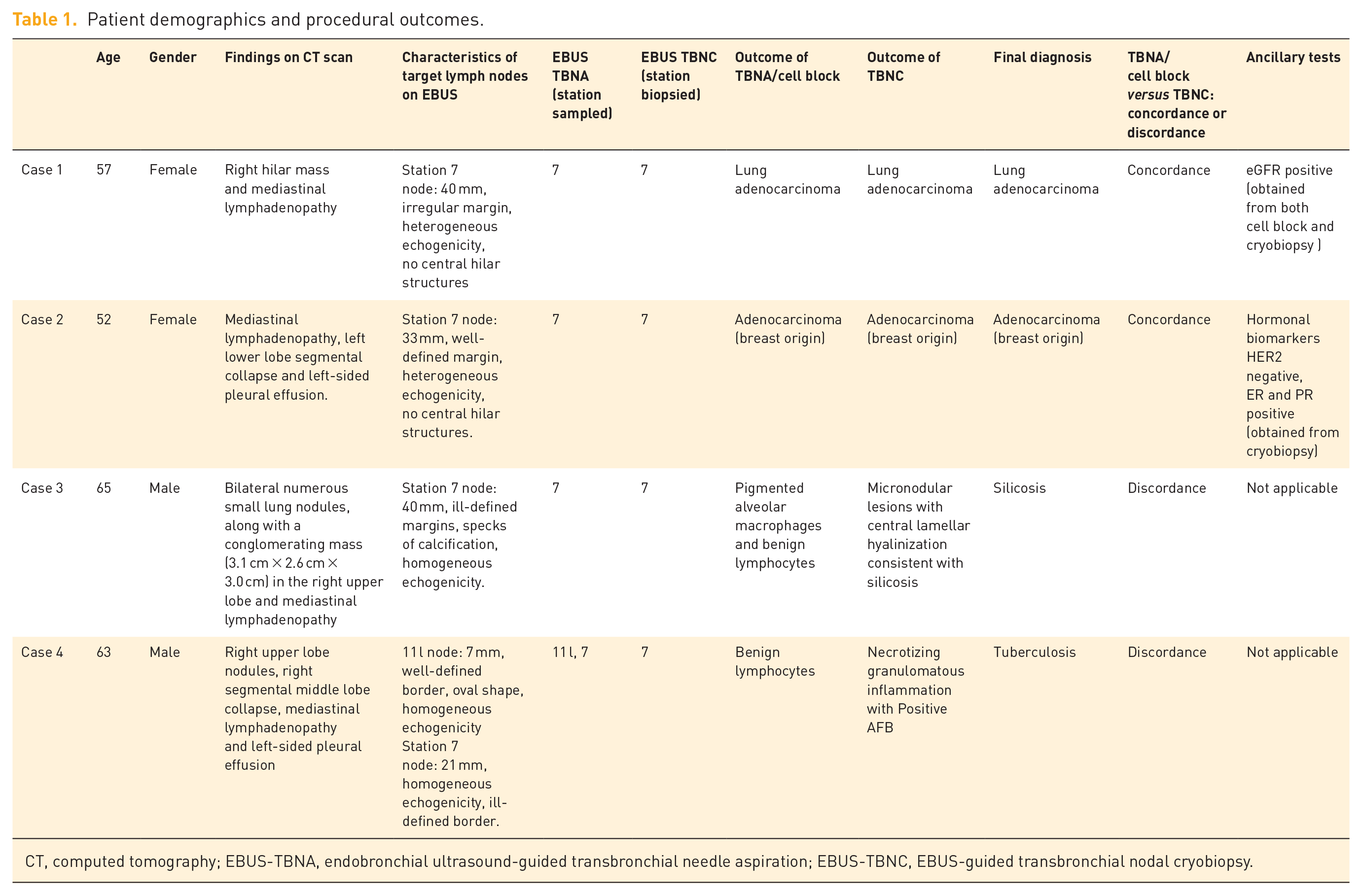
CT, computed tomography; EBUS-TBNA, endobronchial ultrasound-guided transbronchial needle aspiration; EBUS-TBNC, EBUS-guided transbronchial nodal cryobiopsy.
For cases 1 and 2, we found that EBUS-TBNA supplemented by ROSE was sufficient to provide a diagnosis of adenocarcinoma. In Case 1, the cell block obtained during the procedure showed good cellularity and was deemed adequate for further molecular analysis. On the other hand, in Case 2, although the cell block provided a diagnosis, testing for hormonal markers was only feasible using the samples obtained through EBUS-TBNC.
This was echoed in larger studies that were recently published. In a study of 197 patients comparing EBUS-TBNA and EBUS-MCB, EBUS-MCB had a higher diagnostic yield compared with EBUS-TBNA (91.8% versus 79.9%), while cryobiopsy was more sensitive than TBNA in rare tumours (91.7% versus 25%) and benign disorders (80.9% versus 53.2%). 10 Subsequently, the same group published a randomized controlled trial involving 297 patients comparing combined use of EBUS-TBNA and EBUS TBNC or EBUS-TBNA alone. 11 The study found that adding cryobiopsy to standard sampling significantly increased the overall diagnostic yield for mediastinal lesions [93% versus 81%, risk ratio (RR) 1·15 (95% CI 1.04–1.26); p = 0.0039]. Although subgroup analysis showed no significant difference in diagnostic yields for mediastinal metastasis between both groups, the combined approach (EBUS TBNA plus EBUS TBNC) was more sensitive than standard needle aspiration in diagnosing benign disorders. The combined approach also improved the suitability of tissue samples for molecular and immunological analyses of non-small-cell lung cancer. 11 Another prospective study of 50 patients comparing EBUS-TBNA and EBUS-TBNC found that EBUS-TBNC demonstrated a higher yield of 96% compared to the diagnostic yield of EBUS-TBNA of 82%; meanwhile, diagnostic yields were similar for cases of sarcoidosis. Of note, EBUS-TBNC demonstrated higher sensitivity than EBUS-TBNA in detecting lymphomas and metastatic lymph nodes. 12
The key advantage of EBUS-TBNC is the ability to obtain larger tissue with preserved tissue architecture. This proved particularly valuable in lymphoproliferative disorders, benign disorders and uncommon tumours. These situations often require histologic samples to assess the overall tissue architecture, making EBUS-TBNC a more valuable approach. While this may not be essential for primary pulmonary malignancies where EBUS-TBNA is excellent at obtaining a diagnosis, EBUS-TBNC nevertheless provides increased levels of confidence to bronchoscopists.
There are a few limitations worth noting when combining EBUS-TBNC with EBUS-TBNA. Firstly, EBUS-TBNC requires increased cost due to the need for a cryomachine and the reusable 1.1-mm cryoprobe. Secondly, EBUS-TBNC will increase the total procedure time. The addition of EBUS-TBNC to EBUS-TBNA necessitates the repeated retrieval of the scope after cryobiopsy, followed by reinsertion of the EBUS scope and re-identification of nodes to obtain specimens. In some cases, inserting the cryoprobe through the tract created by the TBNA needle may be challenging, particularly when the bronchus is rigid and cartilaginous. To facilitate insertion, additional tools may be utilized to expand the tract. As the reusable electrosurgical knife (KD-31C-1, Olympus) capable of passing through the 2.0 mm working channel of the EBUS scope is no longer in production, alternative accessories such as Needlecut 3 V (Olympus needle knife), laser or larger TBNA needles are required. This necessitates switching from the EBUS scope to a therapeutic bronchoscope with a larger working channel to accommodate the insertion of these accessories. Consequently, this further lengthens the procedural time. Thirdly, there is a concern regarding the potential for missing diseased areas within the lymph node due to the introduction of the cryoprobe at the same puncture site. Unlike in EBUS-TBNA, where multidirectional sampling can be performed through a technique called ‘fanning’, it is challenging to reach different parts of the node during TBNC. Fourthly, while attempts can be made to freeze different portions of the lymph node by adjusting the penetration depth, this approach may prove challenging, particularly in smaller nodes or lesions. Finally, the sampling of larger tissue during transbronchial needle cryobiopsy (TBNC) carries potential complications that include bleeding, pneumothorax, pneumomediastinum and the risk of mediastinitis.12,13
Considering the potential limitations, it may not be justified for all patients undergoing EBUS-TBNA to also undergo EBUS-TBNC as the first-line diagnostic approach. To address this, Maturu et al. and team proposed a diagnostic algorithm. 14 According to their study, EBUS-TBNC was selectively utilized in cases where ROSE yielded inconclusive results. Among the 196 patients who underwent EBUS-TBNA, 46 patients received EBUS-TBNC due to inadequate or non-diagnostic ROSE. The inclusion of EBUS-TBNC in these cases resulted in an additional diagnostic yield of 43.7% compared to EBUS-TBNA alone. This is a pragmatic approach. ROSE has indeed been widely utilized for onsite evaluation of sample adequacy and guiding sampling during EBUS-TBNA not only for diagnosis but also for further immunohistochemistry or next-generation sequencing. A high concordance rate has been reported in cytologic results between EBUS-TBNA with ROSE and referenced histologic diagnosis, encompassing both malignant and benign cases, including granulomatous diseases.15–18 The use of ROSE was associated with fewer number of needle passes during EBUS-TBNA and an overall lower requirement for additional bronchoscopy procedures during TBNA to make a final diagnosis. Additionally, it reduces procedural time and proves to be cost-effective.19–22 The same concept can be used when deciding on the need for TBNC.
Maturu’s study also highlights the potential superiority of EBUS-TBNC in obtaining suitable samples for further ancillary investigations. In the subset of 14 patients diagnosed with malignancy through both EBUS-TBNA and EBUS-TBNC, the cell block samples obtained from EBUS-TBNA provided adequate material for ancillary studies in only 5 out of 14 cases (35.7%). By contrast, EBUS-TBNC yielded sufficient material for ancillary studies in all 14 cases (100%).
Future research should focus on investigating the optimal cryoprobe activation time and determining the optimal number of samples necessary to ensure material adequacy for accurate diagnosis and ancillary tests. Conducting larger-scale studies is crucial to thoroughly assess the justification for incorporating EBUS-TBNC alongside EBUS-TBNA and to determine the specific cases where this technique is most beneficial. It is imperative to design randomized controlled trials that compare various techniques for obtaining specimens from mediastinal nodes. These trials should assess not only the diagnostic yield of the techniques but also the adequacy of the obtained material for conducting ancillary tests.
Conclusion
EBUS-TBNC is a promising technique to acquire sufficient diagnostic tissue in both malignant and benign conditions; however, questions surrounding case selection and technical considerations such as optimal activation times remain to be investigated in more rigorous studies.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666241231122 – Supplemental material for Early experience of endobronchial ultrasound-guided transbronchial nodal cryobiopsy: a case series from Sabah, Malaysia
Supplemental material, sj-pdf-1-tar-10.1177_17534666241231122 for Early experience of endobronchial ultrasound-guided transbronchial nodal cryobiopsy: a case series from Sabah, Malaysia by Hema Yamini Ramarmuty, Nai-Chien Huan, Larry Ellee Nyanti, Teng Shin Khoo, Tamilarasi Renganathan, Ahmad Zaki Manoh, Nusaibah Azman and Kunji Kannan Sivaraman Kannan in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We extend our sincere gratitude to the dedicated and skilled bronchoscopy staff of the Respiratory Department at Queen Elizabeth Hospital, as well as the General Anaesthesia Department and Pathology Department of Queen Elizabeth Hospital.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
