Abstract
Background:
Prior pulmonary tuberculosis (PTB) might be associated with the development of chronic obstructive pulmonary disease (COPD). However, the impact of prior PTB on the risk of incident COPD has not been studied in a large prospective cohort study of the European population.
Objectives:
This study aimed to investigate the association of prior PTB with the risk of COPD.
Design:
Prospective cohort study.
Methods:
A multivariable Cox proportional model was used to estimate the hazard ratio (HR) and 95% confidence interval (95% CI) for the association of prior PTB with COPD. Subgroup analyses were further conducted among individuals stratified by age, sex, body mass index, smoking status, drinking status, physical activity, and polygenic risk score (PRS).
Results:
The study involved a total of 216,130 participants, with a median follow-up period of 12.6 years and 2788 incident cases of COPD. Individuals with a prior history of PTB at baseline had an 87% higher risk of developing incident COPD compared to those without such history [adjusted hazard ratio (aHR) = 1.87; 95% confidence interval (CI): 1.26–2.77;
Conclusion:
A history of PTB is an important independent risk factor for COPD. Clinical staff should be aware of this risk factor in patients with prior PTB, particularly in countries or regions with high burdens of PTB.
Plain language summary
Prior pulmonary tuberculosis (PTB) indicates that an individual has a history of PTB. The impact of prior PTB on the risk of incident chronic obstructive pulmonary disease (COPD) has not been studied in a large prospective cohort study of European population. Here, we investigated the association between prior PTB and risk of COPD in 216,130 participants from the UK biobank (a large biomedical database). After a median follow up of more than 12 years, 2,788 incident COPD cases were recorded. Individuals with prior PTB at baseline had an 87% higher risk of developing incident COPD compared to those without history of PTB. Specifically, individuals with prior PTB presented with a higher risk of incident COPD among those who were older than 50 years, male, obese, had a previous history of smoking, are currently drinking, have low physical activity, and have a low and high genetic predicted lung function. This study suggested prior PTB as an important and independent risk factor for COPD. Clinical staff should be aware of this risk factor in patients with prior PTB, particularly in countries or regions with high burdens of PTB.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a highly prevalent and heterogeneous chronic respiratory disease, which is characterized by a persistent, often progressive, and not fully reversible airflow limitation due to abnormalities of airways and/or alveoli.1,2 It ranks as the third leading cause of mortality worldwide, with ~3.2 million deaths annually. 3 Meanwhile, COPD is invariably accompanied by a relatively high degree of morbidity, disability, and healthcare expenditure, contributing to an enormous socioeconomic burden.4,5 This heavy burden of COPD can be attributed in part to greater exposure to risk factors as well as disparities in diagnosis and treatment.2,5 Therefore, it is imperative that we identify and modify potential risk factors to effectively control and alleviate the burden of COPD.
As of now, tobacco smoking remains the predominant etiological factor for COPD, but only ~25% of smokers will suffer from COPD and almost half of COPD patients are due to non-tobacco-related risk factors.6,7 These non-smoking-related risk factors encompass air pollution, occupational exposures, poorly controlled asthma, infectious diseases, body weight fluctuations, low socioeconomic status, lower educational attainment levels, and genetic predisposition.6,8,9 Tuberculosis (TB) is responsible for more than 10 million new cases and over 1 million deaths annually.
10
Prior pulmonary TB (PTB) can lead to perpetual lung damage and has shown association with reduced lung function.
10
Over the past two decades, several case–control or cross-sectional studies have demonstrated an association between PTB and airflow obstruction or COPD.9,11
–15 Most of these studies had small sample sizes and were not population based. Besides, Wang
To date, no prospective cohort study has been conducted to evaluate the association of incident COPD with prior PTB, as defined by bacteriological or histological tests. Although GOLD 2023 recommends that TB was a risk factor for COPD based on a meta-analysis of aforementioned studies, a well-designed prospective study is necessary to validate or refute these findings. Due to lacking bacteriological or histological confirmation, early studies primarily relied on self-reported history or chest X-ray to define prior PTB, which might potentially include some non-PTB participants in the PTB group. Therefore, we aimed to investigate the association of prior PTB, as defined by bacteriological or histological tests, with the risk of COPD in a large population-based prospective cohort.
Materials and methods
Study design and participants
We used data from UK Biobank (Application No. 88159), a large population-based prospective cohort study with over 500,000 adults aged 40–69 years, collected in 2006–2010 from across the United Kingdom. The follow-up was conducted until December 2022, with a median of 12.6 years. The design of the UK Biobank has been described previously. 16 Study population, exclusions, and missing data are outlined in Figure 1. Briefly, for the 502,396 UK Biobank participants, 72,823 participants with prevalent respiratory and cardiovascular diseases, 397 participants with prevalent HIV infection, and an additional 165,705 participants were excluded due to the ratio of forced expiratory volume in 1 s (FEV1) to forced vital capacity (FVC) less than 0.7 at baseline (39,524) or missing lung function data (126,181). In all, 130 participants were further excluded due to prevalent malignant neoplasms of respiratory and intrathoracic organs. In total, 47,211 participants had missing data for missing smoking status (786) and physical activity status (46,425), leaving 216,130 participants in the main analysis. For the genetic study, we further excluded participants of non-Caucasian or without genotyping data (27,485). The study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline. 17
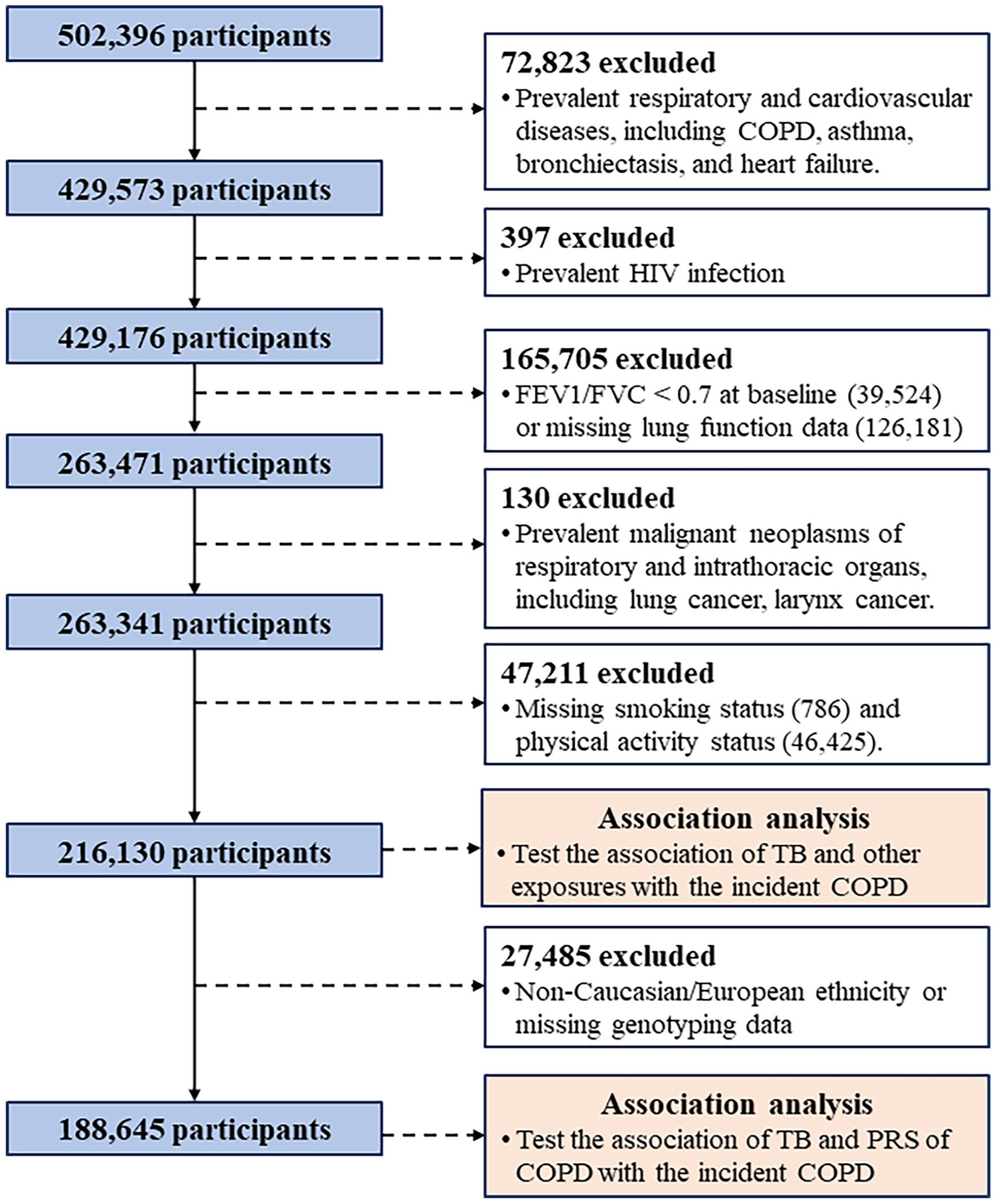
Participant inclusion flow chart.
Ascertainment of prior PTB
Prior PTB was ascertained at baseline according to the ‘first occurrence’ of any code mapped to three-character ICD-10 (category ID 1712), which was linked with the death register, primary care, hospital inpatient records, and self-reported medical conditions reported at the baseline or subsequent visit to UK Biobank assessment center. The prior PTB cases were defined as the bacteriologically and histologically confirmed PTB (field ID 130020, ICD-10 A15). In our sensitivity analysis, the prior PTB cases also included the PTB which was not confirmed bacteriologically or histologically (field ID 130022, ICD-10 A16) and the miliary TB (field ID 130028, ICD-10 A19).
Ascertainment of incident COPD
The outcome, incident COPD (field ID 42016) was extracted from the ‘algorithmically defined outcomes’ (category ID 42). The diagnosis of COPD was obtained through algorithmic combinations of coded information from UK Biobank’s baseline assessment data collection (i.e. self-reported medical conditions, operations, and medications), along with linked data from hospital admissions (diagnoses and procedures) and death registries. Detailed information regarding the linkage procedure is available online (https://biobank.ndph.ox.ac.uk/showcase/refer.cgi?id=460). We calculated the follow-up time from the date of attendance until the date of first diagnosis, date of death, date lost to follow-up, or 31 December 2021, whichever occurred first.
Assessment of covariates
Potential sociodemographic characteristics and lifestyles were selected as covariates based on prior literature. 2 The covariates included sex, age, body mass index (BMI), smoking status (never, previous, or current smoking), drinking status (never, previous, or current drinking), Townsend deprivation index, physical activity, ethnicity, and land use regression (LUR)-based estimates of fine particles with diameter <2.5 μm (PM2.5).
BMI was calculated as body weight in kilograms divided by the square of height in meters and then categorized into four groups: <18.5, 18.5–24.9, 25–29.9, or ⩾30 kg/m2. The Townsend deprivation index was used as a measure of socioeconomic status (with a greater value corresponding to greater deprivation). Physical activity was based on self-report, using the International Physical Activity Questionnaire short form including walking, moderate, and vigorous activity, measured as metabolic equivalent minutes per week (MET-min/week). 18 The participants were further categorized into three groups (i.e. low, moderate, and high) according to the time and intensity of physical activity (https://biobank.ndph.ox.ac.uk/showcase/refer.cgi?id=540).
Polygenic risk score
The single-nucleotide polymorphisms (SNPs) information regarding genotyping, imputation, and quality control has been conducted by the UK Biobank. Polygenic risk score (PRS) for the lung function was constructed based on 279 lung-function-associated genetic variants weighted by the effect size of decreasing FEV1/FVC, which were selected from populations of European ancestry.
19
The weighted PRS was calculated for

where
Statistical analyses
R software version 4.13 was used for data analysis. The baseline characteristics of the study population were reported as median [interquartile range (IQR)] for continuous variables, and number (%) for categorical variables. The comparison of baseline characteristics was conducted by the chi-square test, Fisher’s exact test, or Mann–Whitney
The multivariable Cox proportional hazards regression models were used to estimate the HR and 95% confidence interval (CI) for the association between prior PTB and risk of COPD. In Model 1, Cox proportional hazards regression was not adjusted for any covariable. In Model 2, we adjusted for the following potential confounders: age, sex, ethnicity, Townsend deprivation index, BMI, physical activity, drinking status, smoking status, and ambient particles with a median diameter less than 2.5 (PM2.5).
Subgroup analyses were further conducted among individuals stratified by age, sex, BMI, smoking status, drinking status, physical activity, and PRS. We also conducted the following sensitivity analyses to test the robustness of our results by including the PTB which was not confirmed bacteriologically or histologically (field ID 130022, ICD-10 A16) and the miliary TB (field ID 130028, ICD-10 A19) as the prior PTB cases.
Results
Characteristics of the study population
The baseline characteristics of all participants are presented in Table 1. Throughout a median follow-up period of 12.6 years, a total of 2788 incident cases of COPD were documented among the cohort consisting of 216,130 participants. Among those who developed incident COPD, the mean age and BMI were 62 years (IQR: 57–66) and 28.4 kg/m2 (25.4–31.8), respectively. Participants who developed COPD were characterized by advanced age, male gender, smokers, previous alcohol consumption, overweight status, higher Townsend deprivation index score, lower physical activity levels, and greater exposure to PM2.5 compared to those without incident COPD. No significant differences in terms of race or PRS were observed. A total of 25 individuals were suffering from incident COPD among the individuals with prior PTB history at baseline and had no death reported during the follow-up. The time between TB and COPD ranged from 5 to 51 years, with a median of 38 years.
The characteristics of participants at the baseline.

Continuous variables were expressed as median (IQR), and categorical variables were expressed as number (%).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; IQR, interquartile range; PM2.5, land use regression (LUR)-based estimates of fine particles with diameter <2.5 μm PRS, polygenic risk score; PTB, pulmonary tuberculosis.
Associations of prior PTB with risk of COPD
An association was observed between prior PTB (confirmed bacteriologically or histologically) and incident COPD. The pooled analysis revealed that individuals with a history of prior PTB were at a higher risk of developing COPD compared to those without such a history, with an HR of 2.45 (95% CI: 1.65–3.63) (Table 2, Model 1). After adjusting for potential covariates, the adjusted hazard ratio (aHR) for incident COPD in individuals with prior PTB was 1.87 (95% CI: 1.26–2.77,
Risk of incident COPD according to prior PTB1.

In this table, participants with no evidence of PTB were as a reference group. The prior PTB1 cases were defined as the bacteriologically and histologically confirmed PTB. Model 1: Cox proportional hazards regression did not adjust for any covariable. Model 2: Cox proportional hazards regression adjusted for age group, sex, race, Townsend deprivation index, BMI group, physical activity, drinking status, smoking status, and PM2.5.
CI, confidence interval; COPD, chronic obstructive pulmonary disease; HR, hazard ratio; PM2.5, land use regression (LUR)-based estimates of fine particles with diameter <2.5 μm; PTB, pulmonary tuberculosis.
Subgroup analysis
We conducted stratified analyses based on several potential risk factors, including age, sex, BMI, drinking status, smoking status, physical activity, and PRS. The associations between prior PTB and the risk of incident COPD were significant among participants aged 50–59 years (aHR: 2.47, 95% CI: 1.02–5.95), over the age of 60 (aHR: 1.81, 95% CI: 1.16–2.81), male (aHR: 2.37, 95% CI: 1.47–3.83), obesity (aHR: 3.55, 95% CI: 2.16–5.82), with previous history of smoking (aHR: 2.27, 95% CI: 1.39–3.72), current drinkers (aHR: 1.98, 95% CI:1.32–2.96), with low physical activity (aHR: 2.62, 95% CI: 1.3–5.26), and with low (aHR: 3.24, 95% CI: 1.61–6.53) and high PRS (aHR: 2.43, 95% CI: 1.15–5.14; Table 3).
Risk of COPD according to different factors.

The prior PTB1 cases were defined as the bacteriologically and histologically confirmed PTB. The stratification analyses in the subgroups of age, sex, BMI, drinking status, smoking status, and physical activity were conducted in the retained individuals (
CI, confidence interval; COPD, chronic obstructive pulmonary disease; HR, hazard ratio; PM2.5, land use regression (LUR)-based estimates of fine particles with diameter <2.5 μm.
Sensitivity analysis
The results remained robust in the sensitivity analysis, which included prior cases of PTB with or without bacteriological or histological evidence. Participants with prior PTB had an HR of 2.32 (95% CI: 1.59–3.39) for incident COPD compared to those without prior PTB (Supplemental Table 1, Model 1). In addition, after adjusting for a wide range of covariates in Model 2, the same pattern of results was observed (Supplemental Table 1). Subgroup analyses yielded similar findings with only minor variations (Supplemental Table 2).
Discussion
COPD results not only from tobacco smoking but also from other causes, such as biomass exposure, poverty, infections such as TB, or even asthma.6,8,9 The GOLD 2023 has proposed a new taxonomy for COPD, which includes genetic COPD (COPD-G), cigarette smoking COPD (COPD-C), biomass and pollution exposure COPD (COPD-P), COPD due to infections (COPD-I), and so on.1,21 COPD from different causes evolves in different patterns compared with that of the classic COPD (COPD-C). Therefore, updating the taxonomy of COPD is meaningful to study its natural course and enable the development of specific therapies to improve outcomes. The interaction between infectious processes and COPD-I is unclear and few studies have addressed the therapeutic implications of COPD-I. Identifying and modifying potential risk factors for COPD is of great value in reducing the disease burden. Increasing evidence suggests that PTB is a risk factor for COPD. 11 In our current study, with a follow-up period of 12.6 years, we found the risk of incident COPD was 87% higher among individuals with prior PTB compared with those without. In the subgroup analysis, individuals with prior PTB exhibited an elevated risk for COPD in participants who were older than 50 years, male, obese, previous smoking, current drinkers, low physical activity, and possessed either low or high PRS. Our finding validated that prior PTB is an important risk factor for COPD-I in prospective cohort studies.
During and after treatment with PTB, the lung undergoes a reparative process that results in scarring and ultimately leads to loss of parenchymal tissue. 22 Consequently, patients commonly experience sequelae associated with active TB infection, which significantly impacts their quality of life. However, the precise mechanism by which PTB leads to airflow obstruction or COPD remains largely unknown and requires further investigation. It has been suggested that mycobacteria infection can result in bronchiectasis and bronchial stenosis. 23 Some studies have proposed that latent intracellular mycobacteria infection can raise a dysfunction of macrophages, resulting in persistent and excessive activation of inflammatory immunity.24,25 The aforementioned processes are pivotal in the airway remodeling process that leads to chronic airflow obstruction. 26
Age is often listed as a risk factor for COPD because there is a physiologic decline in lung function with age. Many of the extrapulmonary manifestations related to COPD are also related to the aging process. The presence of these extra-pulmonary manifestations of COPD contributes to the overall morbidity and mortality of COPD patients.27 –29 In agreement with the existing surveys, the incident trend of COPD showed a direct proportional relationship with age in our study.13,30 Our results showed that the prior PTB had a risk effect just among individuals who were older than 50. A history of PTB might have a synergistic effect with aging on the incidence of COPD.
A systematic review demonstrated that female smokers had a faster annual decline in FEV1 than male smokers, even when they smoked fewer cigarettes. 31 Moreover, women with severe COPD have a higher risk of hospitalization and death. 32 The reasons for the gender discrepancies in COPD susceptibility and progression of COPD are largely unknown. Sex hormones may partially account for it. In our study, we exhibited that positive association between prior PTB and the risk of COPD in males, instead of females. So far, no study has investigated whether sex hormones interact with prior PTB in the development of COPD. In a large population study, severe and early-onset COPD was predominantly prevalent in females. 33 A higher risk of COPD was observed in the older ones with prior PTB in our study. Thus, the onset age of COPD might account for those disparities.
Cigarette smoking is the major risk factor for COPD. 2 Stopping smoking benefited FEV1 trajectories with a persisting decade in undiagnosed-COPD smokers. 34 Plenty of evidence has shown that smoking cessation was the most effective strategy for slowing down the disease progression and reducing mortality in COPD patients regardless of age or lung function.35,36 We detected no difference in incident COPD between participants with prior PTB in never and current smokers. However, the positive associations were significant among previous smokers. The exact pathogenesis is still unclear. It is possible that the effect of smoking on COPD was so powerful that it masked the effect of prior PTB. We hypothesized that prior PTB combined with ex-smoking influence might even have an additive effect on the risk of COPD.
The relationship between body mass and pulmonary function was complex and influenced by several factors. 37 It was widely accepted that underweight was a risk factor for COPD.9,38,39 However, previous literature was somewhat inconsistent in the association of adiposity with COPD. A meta-analysis showed that lung function decreased among obese adults. 40 However, some studies exhibited high BMI did not increase COPD risk. 41 On the contrary, overweight and/or obesity were associated with a decreased risk of COPD in two large cross-sectional studies in China.9,42 Intriguingly, Argentina researchers showed that mild to moderate COPD was associated with being overweight while lower BMI was related to more severe stages. 43 We found that prior PTB had a risk effect among obese participants, other than among underweight and overweight ones. New-onset COPD in our study was usual with mild to moderate COPD which might be responsible for associations between prior PTB and the risk for COPD in the obese participants. Another probability was that the number of prior PTB was relatively small in underweight and overweight participants, which led to the failure to reach statistical significance.
There have been conflicting studies on the effects of drinking on lung function. Some studies have shown no effect at all of alcohol on lung function or COPD mortality.44
–46 Tabak
In COPD patients, reduced physical activity was an important contributor to cardiovascular morbidity, recurrent hospital admissions, and disease progression. 53 Physical inactivity was also the strongest predictor for all-cause mortality in COPD. 54 Regular physical activity in COPD patients reduced the risk of morbidity, rate of hospitalization, and mortality. 55 Furthermore, several epidemiological analyses have found longitudinal associations between higher physical activity levels and lower COPD incidence.56 –58 We detected that a history of PTB was positively associated with COPD risk only in participants with low physical activity, but not in those with moderate or high physical activity. The role of physical activity in preventing respiratory diseases is overlooked in reference documents. 59 Thus, appropriate improving physical activity levels in populations with additional risk factors for COPD might be imperative and meaningful.
To date, studies have identified hundreds of genetic variants that are associated with lung function. 60 Genetic risk explains a substantial proportion of the phenotypic variability of COPD. 61 Yet, their individual effect size was small and it remained uncertain whether these genes were directly responsible for COPD.1,62 Aggregating multiple SNPs with small effects to generate a composite PRS may elucidate the genetic risk of complex diseases. The PRS, comprising many genetic variants, can enable the identification of a subset of individuals at high risk for COPD. 63 Interestingly, prior PTB was associated with incident COPD in participants with low or high PRS. It needs more study to explain this association.
To the best of our knowledge, our study is the first prospective cohort to confirm the association between prior PTB and the risk for COPD. However, several limitations exist in this study. The definition and diagnostic criteria of COPD have evolved over time. The diagnosis of COPD was obtained through algorithmic combinations of coded information from UK Biobank beyond the GOLD criteria. The impact of prior PTB on the risk of COPD may be overestimated due to the proportion of patients diagnosed with COPD without spirometry. We did not consider whether the TB was in active or non-active status, although most of PTB cases could be non-active at baseline. In addition, it was possible that some participants had suffered from PTB and healed without any treatment or trace; thus, the PTB infection may be underestimated.
Conclusion
Prior PTB is an important independent risk factor for COPD. Clinical physicians should be aware of this risk factor in patients who suffer from dyspnea, chronic cough or sputum production, and/or a history of exposure to the other risk factors, especially in countries or regions with high PTB burdens. Early diagnosis and interventions can be achieved under dynamic detection of pulmonary function in people with a history of PTB. Further studies are needed to understand the mechanism underlying the association between prior PTB with COPD.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241239455 – Supplemental material for Associations of prior pulmonary tuberculosis with the incident COPD: a prospective cohort study
Supplemental material, sj-docx-1-tar-10.1177_17534666241239455 for Associations of prior pulmonary tuberculosis with the incident COPD: a prospective cohort study by Zhilin Zeng, Huilong Chen, Zhonghe Shao, Yunlong Guan, Yuan Zhan, Xi Cao, Si Li and Xingjie Hao in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
