Abstract
Background:
Low oxygen saturation (LOS) is a frequent occurrence for patients with post-tuberculosis tracheobronchial stenosis (PTTS) during bronchoscopic procedures. However, there are currently no systematic assessment tools to predict LOS risk in PTTS patients during bronchoscopy.
Objectives:
This study aimed to develop an effective preoperative predictive model to guide clinical practice.
Design:
Retrospective cohort study.
Methods:
Data was retrospectively collected from PTTS patients who underwent bronchoscopic interventions between January 2017 and December 2022. Among all patients included in this study, patients between January 2017 and December 2021 were used as training cohort for the logistic regression model, and patients between January 2022 and December 2022 were utilized as validation cohort for internal validation. We used consistency index (C-index), goodness-of-fit test and calibration plot to evaluate the model performance.
Results:
A total of 465 patients who met the inclusion criteria were enrolled in the study. The overall incidence of LOS was 26.0% (121/465). Comorbidity, degree of stenosis, bronchoscopist level, thermal ablation therapy, balloon dilation, and airway stenting, as independent risk factors for the presence of LOS, were used to construct the nomogram prediction model. The C-index of training cohort was 0.827 (95% CI, 0.786–0.869), whereas that of validation cohort was 0.836 (95% CI, 0.757–0.916), combining with the results of the calibration plot and goodness-of-fit test, demonstrating that this model had good predictive ability.
Conclusion:
The predictive model and derived nomogram with good predictive ability has been developed to preoperatively predict the risk of LOS in PTTS patients during bronchoscopy, allowing for individualized interventions for high-risk patients.
Introduction
Tuberculosis (TB) is a communicable disease that is caused by the bacillus Mycobacterium tuberculosis and one of the most common causes of death globally. Among HIV-negative people, there were 1.4 million TB deaths in 2021, according to the global tuberculosis report 2022. 1 Of these patients with active pulmonary TB, 10–50% are reported to have tracheobronchial TB (TBTB).2–4 Further, TBTB is aggravated as tracheobronchial stenosis and if left untreated, can lead to the irreversible damage of the lung.5,6 In high TB burden areas, such as many Asian countries, post-tuberculosis tracheobronchial stenosis (PTTS) is the leading cause of benign tracheobronchial stenosis.2,7 Whilst open surgical procedure previously served as the treatment of choice for PTTS, there has been concern regarding the considerable complication rate, which has limited further application of this technique.8,9 In contrast, minimally invasive bronchoscopic interventions, such as thermal ablation therapy, cryotherapy, balloon dilation and airway stenting, have opened up a new way to deal with PTTS and gained popularity, benefiting a considerable number of selected patients who failed to perform surgical correction due to poor pulmonary reserves, inferior health conditions, technical difficulties and significant medical comorbidities.10–14
For patients with PTTS, restenosis is a frequent occurrence, despite efficacious bronchoscopic management. Therefore, they will undergo bronchoscopic procedures as many times as needed. 12 During bronchoscopic interventions, low oxygen saturation (LOS) is common and the reported incidence of LOS has been as high as 19.5–38.2% in different study settings,15–18 even though multiple oxygen supplementation measures have been developed to prevent LOS. 19 Recent publication undertaken by Kim et al. 18 has demonstrated that LOS during bronchoscopy is linked to the increased risk of post-bronchoscopy respiratory adverse events. It seems essential for bronchoscopists to identify PTTS patients who may experience LOS during bronchoscopy, by which early and preventive clinical interventions can be applied to reduce the risk of LOS.
Previous studies have identified age, sex, comorbidity, remaining lung function, basal saturation of percutaneous oxygen (SpO2), specific procedure types and duration of procedure as some factors related to LOS.17,20–24 Where no previous research has reported the systematic assessment tools to predict the risk of LOS in patients with PTTS during bronchoscopy, the present study reported the clinical characteristics of patients with LOS and aimed to develop a predictive clinical model to stratify LOS risk in PTTS patients during bronchoscopy. The resulting nomogram has the potential to allow the risk for an individual patient to be identified early, thereby assisting bronchoscopists in applying preoperative interventions for individuals with different levels of risk.
Materials and methods
Patients
Data was retrospectively collected from PTTS patients who underwent bronchoscopic interventions between January 2017 and December 2022 in the Department of Respiratory and Critical Care Medicine, Shanghai Changhai Hospital, The First Affiliated Hospital of Naval Medical University. Among all patients included in this study, patients between January 2017 and December 2021 were used as development data set for the logistic regression model, and patients between January 2022 and December 2022 were utilized as validation data set for internal validation. The exclusion criteria were as follows: (1) inadequate perioperative information, and (2) patients with basal SpO2 maintained ⩽94% regardless of whether oxygen supplementation was performed. All participants provided written informed consent for this retrospective analysis prior to inclusion in the study. This retrospective study was performed in accordance with the Declaration of Helsinki and the study protocol was approved by the Institutional Review Board of Shanghai Changhai Hospital (IRB No. CHEC2021-049). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 25
Data collection
Patient demographics including age, sex, height, weight, smoking history, and comorbidity (chronic obstructive pulmonary disease, asthma, bronchiectasis, coronary heart disease, arrhythmia, obesity hypoventilation syndrome, obstructive sleep apnea syndrome, interstitial lung disease, thyroid disease, and history of other malignancies) were collected from electronic medical records. Information of pre-bronchoscopy evaluations including basal SpO2, American Society of Anesthesiologists (ASA) grade, 26 bronchoscope type (flexible or rigid), location, type and degree of stenosis, length of the stenotic segment, anesthesiologist and bronchoscopist level were also recorded. Operation-related data were collected including LOS, duration of procedure, anesthesia method (local anesthesia, sedation and general anesthesia), thermal ablation therapy, cryotherapy, balloon dilation and airway stenting.
In combination with the definition of Kim et al., 18 LOS was defined to be present when SpO2 ⩽ 94% with the non-transient property. Duration of procedure, that is, time from bronchoscope insertion to bronchoscope removal. Location, type, degree of stenosis, and length of the stenotic segment were determined based on the classification of Freitag et al. 27 combined with an expert consensus. 28 Given the clinical practice of our institution, we divided levels of anesthesiologist into three categories: none (local anesthesia without the need for anesthesiologist), specialty training, and attending or higher level; whereas those of bronchoscopist were divided into two categories: advanced training and attending or higher level.
Statistical analysis
Continuous variables were presented as the mean ± standard deviation (SD) and compared by t-tests or Wilcoxon rank-sum tests. Chi-square (χ2) tests were applied to test differences for categorical variables which were expressed as frequencies. Univariate logistic regression analysis was used to identify risk factors of LOS. All variables with p values <0.15 in univariate analysis were included in the forward stepwise multivariable logistic regression analysis which was utilized to ascertain the independent predictors of LOS. All independent risk factors (p < 0.05 in multivariable logistic regression) were remained and used to establish the nomogram via Stata program nomolog. 29 The total score predicting the probability of LOS could be yielded by summing the score of each variable obtained on the basis of the regression coefficient values.
The model performance was further evaluated using the accuracy and discrimination. The accuracy of the nomogram, as a measure that discriminates between two states, quantified by the receiver operating characteristic (ROC) curve and commonly summarized by the consistency index (C-index), which could be formed by calculating the area under the ROC curve (AUC). 30 The discrimination of the nomogram measuring the consistency between the predicted probabilities and the actual results was verified using the Hosmer-Lemeshow (H-L) goodness-of-fit test and the calibration plot. 31
All aforementioned statistical analyses were conducted using Stata 15.1 (StataCorp., College Station, TX, USA) and SPSS 26.0 (IBM, Armonk, NY, USA). A p value of <0.05 (bilateral) was considered significant unless specified otherwise.
Results
Patient characteristics
The study flow of the detailed selection process is illustrated in Figure 1. Clinical information of 581 patients with PTTS who received bronchoscopic interventions between January 2017 and December 2022 in our institution was screened. One hundred sixteen patients were excluded because of inability to obtain complete perioperative data (n = 97) and basal SpO2 ⩽ 94% despite oxygen supplementation (n = 19). Finally, a total of 465 patients who met the inclusion criteria were enrolled in the study. Among them, 362 patients were used as training cohort, and the remaining 68 cases served as internal validation cohort. The overall incidence of LOS was 26.0% (121/465). According to the absence or presence of LOS, all eligible patients were classified into either the normal group (n = 344) or the LOS group (n = 121).

Flow diagram showing patient enrolment.
Demographical and clinical data of both groups are presented in Table 1. It was found that patients with PTTS were more likely to be female with a rate of 78.1% (363/465). In addition, significant differences were detected between the LOS and non-LOS patients in basal SpO2, duration of procedure, comorbidity, bronchoscope type, anesthesia method, degree of stenosis, length of the stenotic segment, thermal ablation therapy, balloon dilation and airway stenting. Of note, for patients who underwent thermal ablation therapy and balloon dilation, the incidences of LOS were 47.3% (86/182) and 34.1% (75/220), respectively, which were higher than in patients who had not undergone thermal ablation therapy (12.4%, 35/283) and balloon dilation (18.8%, 46/245).
Demographic and clinical characteristics between patients with and without LOS.

Due to local anesthesia without the need for anesthesiologist.
ASA, American Society of Anesthesiologists; BMI, body mass index; LOS, low oxygen saturation; SpO2, saturation of percutaneous oxygen.
Risk factors of LOS
In the training cohort, we conducted the univariable logistic regression analysis to identify all related risk factors of LOS. In view of the preoperative characteristic of this predictive model, duration of procedure was excluded from the univariable logistic regression analysis and further multivariable analysis. As shown in Table 2, the results determined 12 LOS-related variables, including basal SpO2, ASA grade, comorbidity, bronchoscope type, anesthesia method, type and degree of stenosis, length of the stenotic segment, bronchoscopist level, thermal ablation therapy, balloon dilation and airway stenting. All variables significantly associated with LOS were included in the multivariable logistic regression analysis, and the results revealed that comorbidity, degree of stenosis, bronchoscopist level, thermal ablation therapy, balloon dilation, and airway stenting were independent risk factors for the presence of LOS (Table 3).
Univariate logistic regression of LOS presence.

Due to local anesthesia without the need for anesthesiologist.
ASA, American Society of Anesthesiologists; BMI, body mass index; CI, confidence interval; LOS, low oxygen saturation; OR, odds ratio; SpO2, saturation of percutaneous oxygen.
Multivariable logistic regression of LOS presence.

CI, confidence interval; LOS, low oxygen saturation; OR: odds ratio; SE, standard error.
Construction and validation of the prediction model
Based on the coefficients of independent predictors presented in multivariate logistic regression analysis, we established the nomogram as a visual prediction model to predict LOS risk in PTTS patients during bronchoscopy (Figure 2). Degree of stenosis and thermal ablation therapy were the most important factors related to the presence of LOS, followed by airway stenting, comorbidity, bronchoscopist level, and balloon dilation. This nomogram provides an easy way for predicting the probability of LOS. For example, for a PTTS patient with a 60% degree of stenosis who will undergo thermal ablation therapy performed by an attending bronchoscopist, the scores for the respective variables are as follows: Score (degree of stenosis) ≈ 4; and Score (thermal ablation therapy) ≈ 7.5; and Score (bronchoscopist level) = 0. The total score will be approximately 11.5, which is equivalent to an estimated probability of LOS of approximately 25% for this patient.
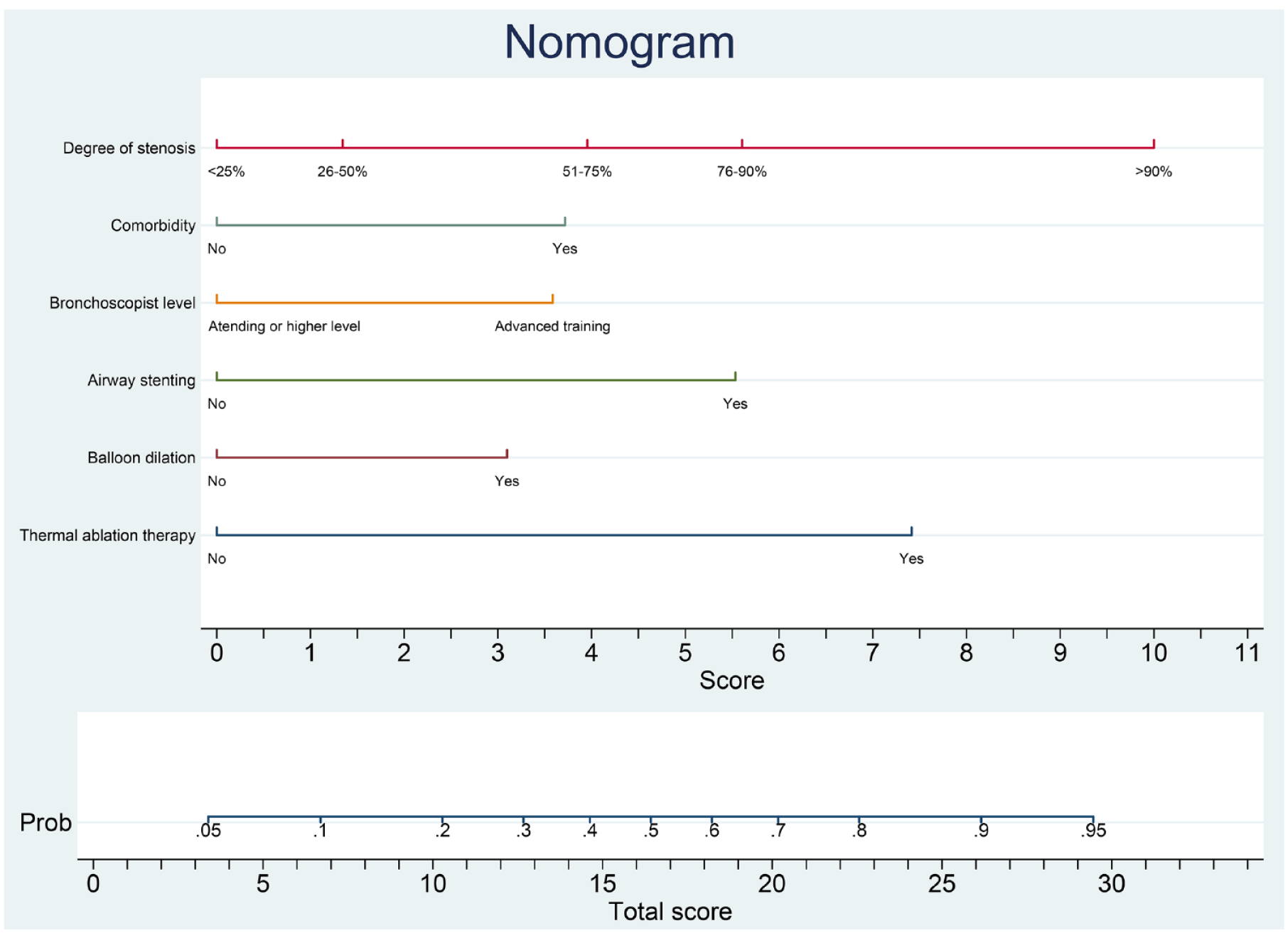
Nomogram visualizing the predictive model to preoperatively estimate the risk of LOS in patients with PTTS during bronchoscopy.
A C-index of the prediction model of 0.827 (95% CI, 0.786–0.869) was achieved in the training cohort, 0.836 (95% CI, 0.757–0.916) in the validation cohort, and 0.821 (95% CI 0.784–0.858) in the overall sample, which demonstrated good accuracy in estimating the risk of LOS (Table 4). H-L goodness-of-fit tests were insignificant for the training and validation cohort, indicating that the model had good calibration. Furthermore, the nomogram calibration plot for the entire cohort showed good concordance between the predicted and actual risk of LOS, signifying that the model could be utilized to predict LOS risk in PTTS patients during bronchoscopy (Figure 3).
C-index of the nomogram prediction model.

C-index: consistency index; CI: confidence interval.

Calibration plot of the nomogram for preoperative prediction of LOS risk in patients with PTTS during bronchoscopy. The closer combination of the solid and dashed lines indicates better predictive power.
Discussion
It is well established that bronchoscopic interventions play an important role in the management of various pulmonary diseases because of its safety and utility32,33; however, LOS is a frequent occurrence during bronchoscopy. 34 In this study, the overall incidence of LOS was 26.0% (121/465), which was consistent with that of previous published reports.15–18 Previous studies have identified LOS as an independent risk factor for post-bronchoscopy respiratory adverse events that may worsen clinical outcomes or lead to fatal events.18,35 Additionally, given the considerable restenosis rates even after bronchoscopy, PTTS patients tend to undergo bronchoscopic procedures as many times as needed. Research is, therefore, necessary for preoperatively predicting LOS risk in PTTS patients during bronchoscopy. Nomogram providing the presentation format for clinical prediction models have been currently widely used for estimating the risk of existing disease and future outcome for an individual, greatly making it convenient for bronchoscopists in their decision-making in clinical settings. 36
In the present study, univariate and multivariate logistic regression analysis were applied to identify the most dominant factors associated with LOS development, and the results demonstrated that comorbidity, degree of stenosis, bronchoscopist level, thermal ablation therapy, balloon dilation, and airway stenting were independent risk factors for LOS. Based on these predictors, a predictive model and the derived nomogram was developed to predict LOS occurrence for individual patients with PTTS during bronchoscopy. The validation cohort was used to further evaluate the established model, and it was revealed to possess excellent predictive power according to the C-index, H-L goodness-of-fit test and calibration plot. Therefore, this tool can estimate the individual risk of LOS at an early stage, which enables early preventive measures, especially for the high-risk patients, thereby reducing post-bronchoscopy respiratory adverse events.
Thermal ablation therapies used for PTTS patients include argon plasma coagulation, laser, and contact electrocautery. There is a significant correlation between thermal ablation therapy and LOS occurrence, and possible reasons are listed as follows: a strict limitation of the inspired oxygen concentration to ⩽40% will be implemented to mitigate against the risk of airway fire at the time of ablation, which may increase the risk of LOS during bronchoscopy. 37 Plus, surgical smoke-induced bronchospasm may result in ventilatory disorder. Manual vacuum aspiration evacuating surgical smoke in time is, consequently, essential during thermal ablation therapy. Hopefully, CoreCath2.7S, a novel monopolar radiofrequency electrosurgical device that allows for smoke evacuation, coagulum, and suctioning of blood simultaneously when performing thermal ablation therapy, has been lately developed by Benn and coauthors. 38 During the period of their study, 87% patients with benign airway stenosis treated with CoreCath2.7S did not require additional bronchoscopic interventions such as balloon dilation and airway stenting associated with LOS occurrence. With these superiorities in mind, CoreCath2.7S has the potential to reduce the risk of LOS for PTTS patients who will undergo thermal ablation therapy. Balloon dilation has also been identified as an important risk factor for LOS. The presence of tracheal mucosal congestive edema and bronchospasm caused by the high-pressure balloon dilatation or the balloon per se may worsen the stenosis of the tracheal lumen. Thus, necessary preparations including thorough assessment for the location and degree of stenosis, the distal lung, and the choice of appropriate balloon catheter determined by the expected size of the airway must be made prior to balloon dilatation. Airway stenting has been demonstrated to be an effective and safe treatment option for PTTS patients. 13 A rigid bronchoscope providing a larger working channel is usually used for airway stenting and requires general anesthesia. 39 It was concluded with the previous studies that administration of muscle relaxants to PTTS patients may exacerbate airway obstruction, leading to LOS occurrence. 40 However, a recent prospective randomized interventional trial undertaken by Okamoto et al. 41 showed that general anesthesia with muscle relaxants during airway stenting decreased the incidence of LOS, which could be attributed to a greater volume of ventilation under this situation. Further research is clearly needed to evaluate aforementioned results.
Our findings that indicated the relation of comorbidity, degree of stenosis and the risk of LOS prompted that in anticipation of the procedure, perfecting relevant examinations to identify the absence or presence of comorbidity and degree of stenosis is essential. Furthermore, the levels of bronchoscopists could also influence the incidence of LOS. Undoubtedly, there is a learning curve for dealing with complex benign airway stenosis such as PTTS that improves with case experience 42 . Attending or higher-level bronchoscopist has greater experience as compared with advanced training bronchoscopist, which can avoid the unnecessary procedures and prolongation of the interventions. In the study of May et al., 24 it was shown that duration of procedure was associated with LOS. In spite of several studies to the contrary,15,17,23 we demonstrated that patients with LOS tended to have a longer procedure time. Therefore, for patients with higher risk of LOS, attending or higher-level bronchoscopist is warranted to perform bronchoscopic procedures.
The outcomes regarding the association between basal SpO2 and the risk of LOS are limited and variable.17,22,23 In our study, it was presented that basal SpO2 was not an independent indicator for LOS occurrence; however, the assessment of basal SpO2 was necessary and recommended by British Thoracic Society before bronchoscopy. 43
Indeed, our study carried certain limitations that should be addressed. First of all, this nomogram prediction model is based on retrospective study with inherent risk of bias. Second, generalization of this model remains to be corroborated due to the small sample size in a single center. Third, the absence of external validation may limit the popularity of this model. Thus, the multimodal database with available data associated with PTTS patients who receive bronchoscopic interventions should be built for future research, requiring more multicenter prospective clinical trials with larger patient cohort. With larger data accrual, this nomogram prediction model will be further optimized with a higher accuracy and stability. When considered together, this preoperative predictive model may have a role in improving the ability of respiratory intervention team to identify patients at risk of LOS. Preventive measures aimed at reducing LOS risk can be implemented. For example, preventive oxygen supplementation of 2–3 L/min is recommended for patients with higher risk of LOS.
Conclusion
In summary, we develop a nomogram prediction model with good predictive ability to preoperatively predict the risk of LOS in PTTS patients during bronchoscopy. Using this simple and validated model, interventions may be applied in advance by estimating individuals’ LOS risk.
