Abstract
Background:
Coronavirus disease 2019 (COVID-19) is a global outbreak disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Cytomegalovirus (CMV) infection can occur in critical COVID-19 patients and is associated with adverse clinical outcomes.
Objective:
The aim of this study was to explore the clinical characteristics and outcome of CMV infection in critical COVID-19 patients.
Design:
This was a retrospective cohort study.
Methods:
From May to September 2021, SARS-CoV-2 reverse transcription polymerase chain reaction (RT–PCR)-confirmed COVID-19 patients with intensive care unit (ICU) admission were enrolled. CMV infection was confirmed by PCR. Baseline characteristics, critical illness data and clinical outcomes were recorded and analyzed.
Results:
Seventy-two RT–PCR-confirmed COVID-19 patients with ICU admission were included during the study period and 48 (66.7%) patients required mechanical ventilation (MV). Overall, in-hospital mortality was 19.4%. Twenty-one (29.2%) patients developed CMV infection. Patients with CMV infection had a higher likelihood of diabetes, higher lactate dehydrogenase and lactate levels, and higher proportions of MV, anticoagulant, and steroid use. Patients with CMV infection were associated with longer duration of SARS-CoV-2 shedding, longer ICU and hospital stay, and fewer ventilator-free days. The independent risk factor for development of CMV infection was a higher accumulative steroid dose.
Conclusion:
CMV infection adversely impacted the outcomes of critical COVID-19 patients, resulting in longer ICU stays, longer mechanical ventilation uses and prolonged shedding of SARS-CoV-2.
Introduction
Coronavirus disease 2019 (COVID-19) was declared a pandemic by the World Health Organization on 11 March 2020. The disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus has spread globally, leading to the deaths of millions of people. Roughly half of SARS-CoV-2 infections are asymptomatic, while about 15% develop severe pneumonia needing hospitalization and oxygen support. Around 5% experience acute respiratory distress syndrome, septic shock, and multiorgan failure, leading to high mortality rates during China’s initial pandemic wave.1–3 The severity has changed over time, which may be due to different SARS-COV-2 variants, vaccination, and advances in treatment.4,5 The main risk factors for severe COVID-19 are hypertension, obesity, type II diabetes, cardiovascular diseases, male sex and old age.1–3 The overall estimated death rates range from 0.3% to 1.0% but are strikingly higher in older people. 6 The reason for this is not known, but it is plausible that immune function declines with age.7–11 Recent studies have concluded that some potential factors could be associated with prolonged SARS-CoV-2 shedding (older individuals, tocilizumab use, and corticosteroid treatment, etc.), and these factors may be related to immune system impairment.12,13
Cytomegalovirus (CMV), recognized as an important viral pathogen in immunocompromised hosts also frequently affects critically ill but immunocompetent patients, and often correlates with adverse clinical outcomes.14–16 The use of corticosteroids during a patient’s intensive care unit (ICU) stay has been described as an independent risk factor for CMV infection in immunocompetent patients. 17
CMV infections have also been observed in individuals with severe COVID-19.18–23 Several studies have demonstrated a correlation between CMV infection and adverse outcomes in critically ill COVID-19 patients, such as the need for mechanical ventilation (MV) and increased mortality rates.21–23 However, one study indicated that CMV infection did not independently associate with mortality. 20 To our knowledge, the association between SARS-CoV-2 shedding and CMV infection remained unknown. Because CMV infection may induce immune dysfunction,24,25 we hypothesized that CMV infection may be related to disease severity and prolonged SARS-CoV-2 shedding of COVID-19. The aim of this study was to describe the risk factors, clinical course, and outcomes in COVID-19-related severe pneumonia patients who developed CMV infection.
Materials and methods
Study design and participants
This retrospective observational study was conducted in Taipei Veteran General Hospital, a 2800-bed tertiary medical center in northern Taiwan. During the initial COVID-19 outbreak in Taiwan from May 2021 to September 2021, patients admitted to the ICU with suspected COVID-19 infection were screened for eligibility. The inclusion criterion was patients with confirmed SARS-CoV-2 infection by reverse transcription polymerase chain reaction (RT–PCR). The exclusion criteria included patients with negative SARS-CoV-2 RT–PCR results or patients without SARS-CoV-2 RT–PCR examination, patients <18 years old, or patients with human immunodeficiency virus infection.
SARS-CoV-2 RT–PCR and shedding evaluation
SARS-CoV-2 RT–PCR was performed in a Cobas® 6800 system (Roche, Pleasanton, CA, USA). Selective amplification of target nucleic acids from the sample was achieved by the use of target-specific forward and reverse primers for the ORF1ab nonstructural region that is unique to SARS-CoV-2. Another conserved region in the structural protein envelope E-gene was chosen for pan-Sarbecovirus detection. The pan-Sarbecovirus detection sets will also detect SARS-CoV-2 virus. In this study, we reviewed Ct values for both gene targets for all RT–PCR tests that were performed on nasopharyngeal swabs or on endotracheal aspirates if patients were mechanically ventilated, as in a previous study. 26 RT–PCR results were expressed in terms of the cycle threshold value, and samples were considered positive if the cycle threshold was ⩽40. Viral dynamics were calculated according to the cycle threshold value (Ct value) of the SARS-CoV-2-specific target (ORF1ab). The duration of viral shedding was defined as days from symptom onset to the first day of Ct value above 30, considering that previous studies showed that no virus was isolated from samples where the Ct value was >30, and this represented a reasonable cutoff with a low risk of secondary transmission.27–29 Patients without Ct values >30 during their hospital stay were right-censored at the time of their last Ct value of the SARS-CoV-2 viral RT–PCR report.
Diagnosis and definition of CMV infection
CMV infection was defined as virus isolation or detection of viral proteins (antigens) or nucleic acids in any body fluid or tissue specimen, according to previous publications. 30 The nucleic acid amplification techniques were calibrated to a standard calibrator, such as the World Health Organization International Standard for human CMV. 31 In our study, CMV infection was represented by the detection of DNA in peripheral blood by real-time quantitative PCR. The cobas® CMV test (cobas® 6800, Roche Molecular Systems for Roche Diagnostics GmbH, Germany) was used to quantify the CMV load in the sample (detection range: 34.5 to 1.1 × 107 IU/mL, 1 IU = 1.1 copies). Serum CMV immunoglobulin (IgG) data was recorded to differentiate CMV reactivation from new infection. CMV IgG were measured by a chemiluminescent immuno-assay on the ARCHITECT i2000SR immunoassay analyzer (Abbott Diagnostics, Abbott Park, IL, USA), results ⩾ 1.0 U/mL were considered reactive.
Data collection and measurements
Information regarding the demographic characteristics and underlying comorbidities were obtained from a hospital chart review. The results of laboratory tests on ICU admission were recorded. Disease severity was evaluated by the Acute Physiology and Chronic Health Evaluation (APACHE) II score 32 and Sequential Organ Failure Assessment scores. 33 The application of MV, renal replacement therapy, prone positioning, and extracorporeal membrane oxygenation (ECMO) were also recorded. Medications related to COVID-19, including corticosteroids, tocilizumab, remdesivir, and enoxaparin, were obtained during ICU admission. Various types of corticosteroids, such as dexamethasone, methylprednisolone, and prednisolone, were employed in our study population. Corticosteroids exposure was quantified as dexamethasone equivalents. It was measured by the accumulative dosage of dexamethasone in milligrams (mg) from admission to CMV infection diagnosis in the CMV infection cohort, and from admission to ICU discharge or death in the cohort without CMV infection.
Outcome evaluation
The outcomes evaluated in the present study included ICU stay, hospital stay, 28-day ventilatory-free days, and all-cause mortality rate on day 30, during ICU admission, and upon hospital discharge. All patients were followed up from admission to death or discharge, with the last patient discharged on 21 October 2021.
Vaccination
The COVID-19 outbreak in Taiwan at the study period was caused by the alpha variant. 34 Owing to a vaccine shortage during the study period in Taiwan, none of the study population had received a COVID-19 vaccine.
Statistical analysis
The results are presented as the median with the interquartile range (IQR) or number (%) whenever appropriate. Continuous variables were compared by the Mann–Whitney U test, while categorical variables were compared using Pearson’s chi-square test, as appropriate. Odds ratios and 95% confidence intervals (CIs) were calculated via logistic regression. The proportion of weaning from MV, proportion of ICU discharge, proportion of CMV infection, and viral shedding of SARS-CoV-2, which was defined as days from symptom onset to a Ct value >30, were evaluated using a Kaplan–Meier survival analysis and the log-rank test. Hazard ratios and 95% CIs were calculated using Cox proportional hazard models and used to measure the associations between CMV infection and viral shedding of SARS-CoV-2, controlling for potential confounding factors in the multivariate model. The cutoffs of white blood cells and accumulative steroid dose were the median. The clinical variables with significant differences were subjected to univariate regression, and variables with p values <0.1 in the univariate model were included in the multivariate analysis. Post hoc power analysis was conducted to evaluate the effect of sample size. All statistical analyses were performed using IBM SPSS Statistics for Windows/Macintosh, Version 25.0 (IBM Corp., Armonk, NY, USA) and MedCalc, Version 20.215.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplementary STROBE checklist).
Results
Patient characteristics
In the study period, a total of 296 patients with suspected COVID-19 and ICU admission were eligible for enrollment. After excluding 224 patients with negative SARS-CoV-2 RT–PCR reports for at least two samplings, 72 RT–PCR-confirmed COVID-19 patients were included in the analysis (Figure 1). The demographic characteristics and clinical features of the enrolled patients are shown in Tables 1 and 2. Prior to the study period, three patients underwent CMV IgG testing, and all of them exhibited positive results. Twenty-one (29.2%) patients developed CMV infection during ICU admission. Dexamethasone was prescribed in 93% (67/72) of the enrolled cases. Fourteen (66.7%) patients with CMV infection received ganciclovir as an anti-CMV treatment, subgroup analysis of patients with CMV infection showed ganciclovir treatment is not significantly related to clinical outcomes. The median time from ICU admission to CMV infection diagnosis was 22 (IQR 12.5–31.5) days. Kaplan–Meier survival analysis of CMV infection after ICU admission is shown in Supplemental Figure S1.

Flow chart of the study.
Clinical characteristics of patients with COVID-19 categorized according to CMV infection status.

Continuous data are expressed as the median with the interquartile range [IQR], and categorical data are expressed as the number of patients (%). The p value was analyzed by Chi-square test or Mann–Whitney U test.
APACHE II, Acute Physiology and Chronic Health Evaluation II; FiO2, fraction of inspiration oxygen; LDH, lactate dehydrogenase; PaO2, partial arterial pressure of oxygen; SOFA, Sequential Organ Failure Assessment score.
Treatment and outcomes of patients with COVID-19 categorized according to CMV infection status.

Accumulative dose of dexamethasone was counted from admission to CMV infection diagnosis in CMV infection cohort, and from admission to ICU discharge or death in cohort without CMV infection. Data are presented as median (IQR) and n (%) unless otherwise indicated.
Continuous data are expressed as median with interquartile range [IQR], and categorical data are expressed as number of patients (%). p value is analyzed by Chi-square test or Mann–Whitney U test.
ICU, intensive care medicine unit. N/A, not applicable.
Compared to patients without CMV infection, patients with CMV infection were older, had higher rates of diabetes mellitus, were more likely to require MV, and had higher C-reactive protein, lactate dehydrogenase, lactate, and D-dimer levels. Patients with CMV infection were exposed to a higher dosage of corticosteroids and more enoxaparin use than those without CMV infection.
Risk factors for developing CMV infection
Univariate and multivariate analyses of clinical factors associated with CMV infection are shown in Table 3. In univariate analysis, clinical factors associated with CMV infection included diabetes mellitus, a higher initial serum white blood cell count, a higher initial lactate dehydrogenase level, a higher initial D-dimer level, mechanical ventilator use, and higher accumulative dexamethasone dose. In multivariate analysis, the independent factor associated with CMV infection was a higher accumulative dexamethasone dose (aOR, 1.013, 95% CI, 1.002–1.024).
Factors associated with developing CMV infection by multivariate and univariate logistic regression analysis.
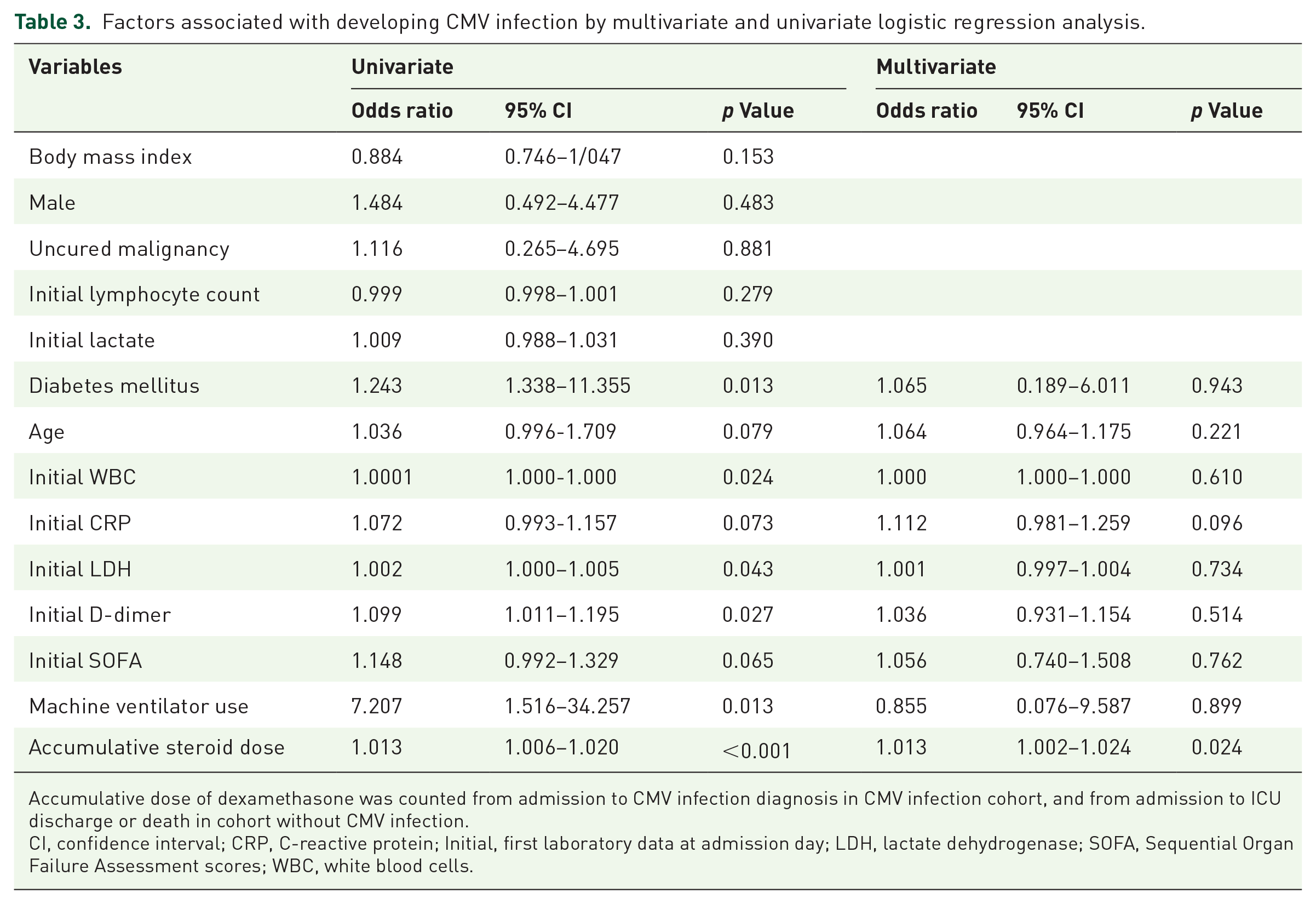
Accumulative dose of dexamethasone was counted from admission to CMV infection diagnosis in CMV infection cohort, and from admission to ICU discharge or death in cohort without CMV infection.
CI, confidence interval; CRP, C-reactive protein; Initial, first laboratory data at admission day; LDH, lactate dehydrogenase; SOFA, Sequential Organ Failure Assessment scores; WBC, white blood cells.
Impact of CMV infection on clinical outcomes
Comparisons of treatment outcomes between patients with and without CMV infection are shown in Table 2. Compared to patients without CMV infection, patients with CMV infection had longer hospital stays, longer ICU stays and fewer 28-days ventilator-free days. Survival analysis demonstrated a significantly longer time to weaning from MV and a significantly longer time to be discharged from the ICU in patients with CMV infection [Figure 2(a) and (b)].
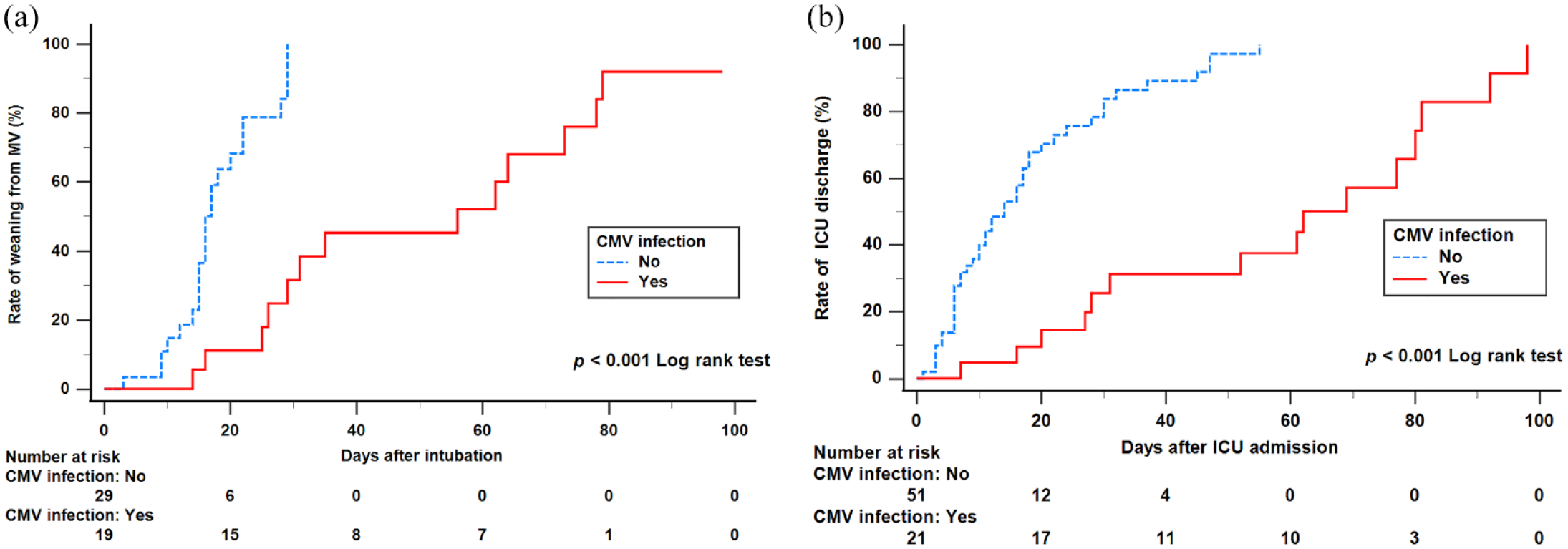
Kaplan–Meier curves for the cumulative portion of events. (a) Patients in the CMV infection group had a significantly longer time to weaning from MV (log-rank test: p < 0.001). (b) Patients in the CMV infection group had a significantly longer time to discharge from the ICU (log-rank test: p < 0.001).
CMV infection is associated with prolonged SARS-CoV-2 shedding
We further investigated the association between CMV infection and viral shedding of SARS-CoV-2. As shown in Supplemental Table S1, the median time from symptom onset to Ct >30 was 16.5 days (IQR 12–21 days). The distribution of time intervals (days) from symptom onset to Ct > 30 between patients with and without CMV infection is shown in Figure 3(a). Patients with CMV infection had a longer duration of viral shedding than patients without CMV (22 days versus 15 days, p < 0.001). In Kaplan–Meier survival analysis, the time intervals from symptom onset to Ct >30 were significantly prolonged in patients with CMV infection compared to those without CMV infection (log-rank p < 0.001) (Figure 3(b)). Comparisons of demographic characteristics and clinical features between patients with longer and shorter SARS-CoV-2 shedding days (median cut-off) are shown in Supplemental Table S2. The patients with longer viral shedding days had a high proportion of CMV infection (18 versus 3, p < 0.001) and were exposed to a higher dosage of corticosteroids (177.5 versus 97, p = 0.049).

SARS-CoV-2 shedding and CMV infection. (a) SARS-CoV-2 shedding in patients categorized according to CMV infection status. We defined SARS-CoV-2 shedding as RNA quantification by a cycle threshold value (Ct value) less than 30 cycles. Data are expressed as the median with interquartile range [IQR], and the p value was analyzed by the Mann–Whitney U test. (b) Cumulative proportion of patients with SARS-CoV-2 RNA Ct values less than 30 by day after illness onset between patients who had CMV infection (log-rank p < 0.01).
In Table 4, univariate and multivariate Cox regressions were performed on the possible risk factors for a longer duration of SARS-CoV-2 shedding. CMV infection was associated with longer duration of SARS-CoV-2 shedding. In univariate analysis, clinical factors associated with a longer duration of SARS-CoV-2 shedding included CMV infection and white blood cell counts over 5980 cells/µL. Multivariate analysis revealed that only CMV infection was independently associated with a longer duration of SARS-CoV-2 shedding (p = 0.018).
Univariate and multivariate Cox proportional hazard regression analysis for prediction of SARS-CoV-2 shedding days.

Cox regression with multivariate adduction. Accumulative dose of dexamethasone was counted from admission to CMV infection diagnosis in CMV infection cohort, and from admission to ICU discharge or death in cohort without CMV infection. The cutoffs of WBC and accumulative steroid dose were median.
CI, confidence interval; WBC: white blood cell count.
The results derived from the accumulative dose of dexamethasone divided by body weight (mg/kg) are displayed in Supplemental Table S3. These findings parallel those calculated from the accumulative corticosteroid dose independently of body weight. We did not find significant association between ganciclovir treatment for CMV infection and clinical outcomes (Supplemental Table S4).
Discussion
Our study demonstrated that higher dexamethasone exposure is an independent factor associated with CMV infection. We also found an adverse impact of CMV infection on the treatment outcomes of COVID-19 patients with ICU admission, including longer ICU stay, longer MV use, and prolonged shedding of SARS- CoV-2. In this retrospective cohort study that enrolled critically ill COVID-19 patients, CMV infection was diagnosed in 29.2% of the enrolled patients. Our findings were in line with a recent meta-analysis and observational study, which reported an incidence rate of 27% in immunocompetent patients in the ICU 35 and an incidence rate of 20.4% in COVID-19 critically ill patients. 20 However, inconsistency in several epidemiologic studies was noted,20,35–37 with variation in the incidence of infection ranging from 0% to as high as 98%. This inconsistency could be explained by many factors, including the types of patient populations studied and their serologic status on admission, as well as the microbiological tools used to diagnose CMV and the type and frequencies of sampling.
Accumulative dosage of dexamethasone was an independent factor associated with CMV infection in our analysis. Low dose dexamethasone is now considered the standard of care in critically ill COVID-19 patients, owing to its benefit in reducing mortality, which was demonstrated by The RECOVERY Collaborative Group 38 and in increasing the number of ventilator-free days which was demonstrated by the CoDEX randomized clinical Trial. 39 However, corticosteroids exert immunosuppressive effects mainly by inhibiting the activity of crucial transcriptional regulators of proinflammatory genes and reducing lymphocyte levels. 40 Therefore, exposure to systemic corticosteroids is a risk factor for CMV infection in patients with severe COVID-19.15,41 In our study, we observed an increased incidence of CMV infection in patients with exposure to higher corticosteroid doses. Although dexamethasone may reduce mortality in severe COVID-19 patients, clinicians should be aware of the potential risk of corticosteroids in developing CMV infection and avoid their unnecessary use. Higher initial CRP level had a trend of increased risk of CMV infection in our analysis. In previous studies, sepsis was a risk factor for CMV infection. The propensity of sepsis to promote CMV infection may result from its pleiotropic effects on the host immune system.42,43 CMV infection is caused by immune dysregulation and inflammation.44,45 The association of CMV infection and sepsis patients suggests that an inflammatory reaction may induce CMV infection. 46 Another in vitro study also demonstrated the strong relationship between CMV infection and human immune response. 47 Thus, the immune dysregulation caused by SARS-CoV-211,48 may trigger CMV infection. Regardless of corticosteroid use, SARS-CoV-2 infection itself can also be a risk factor of CMV infection.
In our study, COVID-19 patients with CMV infection were associated with longer hospital stay, longer ICU stay, and longer days on MV than those without CMV infection. These findings were consistent with previous studies of CMV infection in critically ill patients15,16,21–23,35,36 and a recent observational study of critically ill patients with CMV infection. 20 Patients with CMV infection had higher serum levels of CRP, lactate dehydrogenase, lactate, and D-dimer and were more likely to require MV, which may indicate greater disease severity. It remains uncertain whether protocolized screening of CMV infection is helpful in severe COVID-19 patients for the early diagnosis of CMV infection and whether it improves treatment outcomes. The current method used to detect CMV, CMV PCR, is highly accurate regarding sensitivity and specificity, but the challenge is that the detection of CMV is highly sensitive to the timing and frequency of testing, which is related to the window of emergence of CMV infection within the first 2 weeks of critical illness but not earlier than 3 days.43,49 Second, it is unclear whether some cases represent innocent viral DNA shedding not necessarily related to tissue invasiveness, and thus not requiring therapy, which is an argument against routine screening for CMV. 36
The cycle threshold (Ct) values of SARS-CoV-2 RT–PCR were inversely correlated with viral load and work as a semiquantitative measurement. A higher viral load of SARS-CoV-2 was reported to be an independent factor associated with higher severity and worse prognosis.50,51 Prolonged viral shedding time has been reported in ICU patients and associated with survival status. 26 However, the characteristics of viral shedding of SARS-CoV-2 in patients with CMV infection have rarely been evaluated. In our study, viral shedding time was defined as days from symptom onset to the first day of Ct > 30, considering previous studies showed that no virus was isolated from samples and there was a significantly lower risk of secondary transmission with a Ct value >30.27–29,52 Although they tended to have a comparable Ct value of SARS-CoV-2 RT–PCR upon diagnosis, patients with CMV infection in our study had a longer viral shedding time than those without CMV infection. To the best of our knowledge, this is the first article to describe the association between CMV infection and prolonged SARS-CoV-2 shedding.
Several hypotheses may explain our findings. First, T cells have a crucial role in controlling viral infections. CMV causes clonal T-cell proliferation and a reduction in naïve T-cell diversity, which in turn may lead to a reduced capacity for immune responses to viral infections such as SARS-CoV-2. 25 Previous studies have concluded that potential factors associated with prolonged SARS-CoV-2 shedding may be related to impairment of the immune system.12,13,28,29 Thus, CMV infection may contribute to prolonged SARS-CoV-2 shedding via immune dysfunction. Second, increased concentrations of interleukin-6 have been observed in CMV-infected individuals in previous studies.53,54 CMV infection may trigger hyperinflammation, as mentioned in previous publications.25,55 Exaggerated inflammation could lead to immunity dysregulation, which might partially explain the phenomenon of prolonged SARS-CoV-2 shedding we observed in our study.
Our study suggests an association between CMV infection and adverse clinical outcomes. Initiating CMV prophylactic treatment is generally recommended across most guidelines for patients who have undergone organ transplants.56,57 However, the question of whether to treat CMV infection in immunocompetent critically ill patients remains contentious.36,58 Some research supports the curative treatment of clinically confirmed CMV disease, where microbiological evidence aligns with clinical conditions attributed to CMV. 59 Yet, the advantage of preemptive treatment of subclinical viral replication or prophylactic treatment in patients at risk of developing CMV disease is still uncertain.60,61 A review article proposes that treatment for evident CMV replication is not warranted unless associated with lung infiltrates and at least two other factors. 37 In the context of critical COVID-19 patients, a recent study found no independent correlation between CMV pneumonia and mortality following ganciclovir therapy. 20 Our research suggests that treating CMV infection appears to have no impact on clinical outcomes. Nonetheless, given the small sample size of patients who received anti-viral treatment for CMV, we cannot accurately assess the treatment’s effectiveness in this study. Additional clinical trials are required to determine whether CMV treatment or prophylactic intervention for high-risk patients (like patients with higher corticosteroid exposure) can improve patient outcomes.
Our study had several limitations. First, this was a single center, retrospective cohort study; only 21 CMV infection cases and 51 non-CMV infection cases were included for analysis. We did not calculate the sample size. However, the post hoc power analysis was performed, even though this was a small sample size study, the post hoc power analysis showed the power reached 0.997. Second, there was no protocolized screening strategy for CMV infection in our institution during the study period. Therefore, the diagnosis of CMV infection may have been delayed or underestimated in our COVID-19 patients. Third, as in other observational study, we included all the ICU admitted patients, not merely the CMV-IgG seropositive population because CMV serological status was not examined routinely in clinical practice. Besides, owing to the extremely limited number of patients tested for CMV IgG, we could not determine if the CMV infection was reactivated or a new infection in this study. Fourth, due to limited bronchoscopy use, the lack of histological and autopsy specimens made it difficult to differentiate CMV disease from CMV infection. 30 Additionally, given the restricted collection of specimens, excluding blood, the scope of organ involvement by CMV remained indeterminate. A treatment recommendation for patients with CMV infection could not be derived from our study. Fifth, repeat testing of SARS-CoV-2 RT–PCR was based on the clinical judgment of the physician in-charge rather than a protocolized procedure. The difference in respiratory specimens may have also led to misestimation of the number of viral shedding days of SARS-CoV-2. Finally, none of the enrolled patients received vaccination against SARS-CoV-2. Therefore, the findings of the present study cannot be applied to vaccinated patients. To better understand the relationship between CMV infection and SARS-CoV-2 shedding time, a well-designed prospective clinical study is needed.
Conclusion
In conclusion, a large proportion of critically ill COVID-19 patients in our study developed CMV infection. Higher cumulative corticosteroid dose was found to be an independent factor for the development of CMV infection. Patients with CMV infection tended to have worse clinical outcomes and prolonged SARS-CoV-2 shedding.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231209150 – Supplemental material for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia
Supplemental material, sj-docx-1-tar-10.1177_17534666231209150 for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia by Hsiao-Chin Shen, Jia-Yih Feng, Chuan-Yen Sun, Jhong-Ru Huang, Yuh-Min Chen, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666231209150 – Supplemental material for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia
Supplemental material, sj-docx-2-tar-10.1177_17534666231209150 for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia by Hsiao-Chin Shen, Jia-Yih Feng, Chuan-Yen Sun, Jhong-Ru Huang, Yuh-Min Chen, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-3-tar-10.1177_17534666231209150 – Supplemental material for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia
Supplemental material, sj-docx-3-tar-10.1177_17534666231209150 for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia by Hsiao-Chin Shen, Jia-Yih Feng, Chuan-Yen Sun, Jhong-Ru Huang, Yuh-Min Chen, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-4-tar-10.1177_17534666231209150 – Supplemental material for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia
Supplemental material, sj-docx-4-tar-10.1177_17534666231209150 for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia by Hsiao-Chin Shen, Jia-Yih Feng, Chuan-Yen Sun, Jhong-Ru Huang, Yuh-Min Chen, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-png-5-tar-10.1177_17534666231209150 – Supplemental material for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia
Supplemental material, sj-png-5-tar-10.1177_17534666231209150 for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia by Hsiao-Chin Shen, Jia-Yih Feng, Chuan-Yen Sun, Jhong-Ru Huang, Yuh-Min Chen, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-doc-6-tar-10.1177_17534666231209150 – Supplemental material for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia
Supplemental material, sj-doc-6-tar-10.1177_17534666231209150 for Analysis of the effect of cytomegalovirus infection in clinical outcomes and prolonged duration of SARS-CoV-2 shedding in intensive care unit patients with COVID-19 pneumonia by Hsiao-Chin Shen, Jia-Yih Feng, Chuan-Yen Sun, Jhong-Ru Huang, Yuh-Min Chen, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
